Abstract
Youth impacted by homelessness experience diminished cognition due to a variety of reasons including mental health symptoms, alcohol and substance use, and adverse childhood experiences. However, the status of specific brain regions which could impact important cognitive functions in homeless youth remains unclear. In this pilot comparative and correlational study, a series of demographic, psychological, cognitive assessments, and brain magnetic resonance imaging were performed in 10 male youth experiencing homelessness and 9 age-matched healthy male controls (age range: 18–25 years). Participants experiencing homelessness had significantly decreased regional brain gray matter tissue in comparison to the controls. Moreover, there were strong inverse correlations between the brain regions classically associated with executive decision-making (prefrontal cortices), depression (insular lobes), and conflict resolution (anterior cingulate), and the level of the symptoms detected by their questionnaires.
Introduction
There are over two million youth experiencing homelessness (YEH) under the age of 25 years in the United States (Morton et al., 2018). YEH report high rates of adverse childhood experiences (ACEs) which have been linked to the development of risk behaviors such as alcohol and substance use and poor physical and mental health outcomes (Barnes et al., 2021; Choi et al., 2017; Nelson et al., 2020). Multiple studies also indicate that YEH experience diminished cognition (Barone et al., 2019; Fry et al., 2017; Slesnick et al., 2018; Waclawik et al., 2019; Wong et al., 2016), which can be due to mental health symptoms (i.e., depression, anxiety, and post-traumatic stress disorder), behavioral risk factors (i.e., early substance use), and/or brain tissue changes that lead to diminished executive thinking/planning/decision-making. The convergence of ACEs, risk behaviors, and poorer health outcomes in YEH may result in or precede changes in brain structure and function. For instance, YEH may use substances to cope with adversity and/or self-medicate mental health disorders.
During adolescence, risk-taking and impulsivity are linked to an underdeveloped pre-frontal cortex (Romer, 2010). However, some adolescents seem to be at higher risk for impulsivity and risk-taking behaviors; most likely due to an imbalance between increases in motivational circuits in the ventral striatum and lack of development in control circuits in the frontal cortex (Bjork & Pardini, 2015). While risk behaviors may further exacerbate these imbalances, another possibility is that these brain differences may lead to risky behavior.
For instance, substance use has been associated with brain tissue alteration and function (Squeglia et al., 2009). During early to middle adolescence, heavy alcohol use has been connected to poorer verbal and nonverbal retention (Brown et al., 2000). Furthermore, constructive thinking and executive functioning may be compromised among drug users (Giancola et al., 2001). ACEs have also been connected to structural and functional changes in the brain (Meldrum et al., 2020). More specifically, tissue changes associated with the prefrontal cortex are connected to poorer executive functioning, increased risk-taking behaviors, including higher number of sexual partners and substance use (Piche et al., 2018).
During risk-taking, smoking compared to non-smoking adolescents showed greater activation of the dorsolateral prefrontal cortex, a region implicated in risk assessment and future planning, and also showed involvement in self-reported nicotine dependence. Galvan and study-co-authors have shown that risk-taking assessments of real-life decision-making show remarkable associations with increased accumbens activity (Galvan et al., 2007). Smaller cortical volumes, particularly lower gray matter volume, have been connected to increased substance abuse risk (Weiland et al., 2014), attention deficit hyperactivity disorder (Li et al., 2022), risky driving (Mas-Cuesta et al., 2022), and disruptive behavior disorders (Waller et al., 2020). In healthy adults, ACEs were linked to higher reductions in gray matter volume over 2 years (Ringwald et al., 2022). However, the status of specific brain regions which could impact important cognitive functions in YEH remains unclear. Therefore, the specific aims of this study were to (a) compare brain-region-specific tissue changes which impact executive decision-making (prefrontal cortices), depression (insular lobes), and conflict resolution (anterior cingulate) between YEH and age- and sex-matched, non-YEH controls; and (b) examine the association between regional brain tissue which control executive decision-making, depression, and conflict resolution functions and their symptoms in YEH and controls.
Methods
Participants and Setting
In this pilot comparative and correlational study, a series of demographic, psychological, and cognitive assessments and functional magnetic resonance imaging (MRI) testing were performed with 10 homeless male youth (YEH) and 9 age-matched healthy male controls aged 18 to 25 years to assess the aims of the study. YEH with a history of incarceration and recent alcohol and substance use (i.e., cannabis, cocaine, amphetamines) were recruited from two YEH centers in Southern California and healthy controls were recruited via flyers from the Los Angeles area. As we were interested in comparing YEH to a control group without mental health and substance use, the control group was screened for the absence of these characteristics. Thus, control participants were without any history of chronic medical or psychiatric conditions, drug abuse, or any previous history of head injury. Exclusion criteria for all participants were claustrophobia, non-removable metal (such as braces, bullets, implantable pumps), inability to lay flat for 2 hours, any psychiatric medication, chronic immune-deficiency conditions, neurological (brain) disease, being left-handed, or body weight more than 285 pounds (scanner limitation).
YEH and control participants provided written informed consent before the study and the study protocol was approved by the Institutional Review Boards at UCLA and UCI (IRB# 16001326).
Data Collection Instruments and Procedures
Magnetic resonance imaging
All brain studies were performed in a 3.0-Tesla MRI scanner (Siemens, Magnetom Prisma, Erlangen, Germany) while participants were positioned supine. Foam pads were placed on both sides of the head to minimize head motion during data acquisition. Two high-resolution T1-weighted images were collected using a magnetization prepared rapid acquisition gradient-echo (MPRAGE) pulse sequence (repetition time [TR] = 2200 ms; echo-time [TE] = 2.41 ms; inversion time = 900 ms; flip angle [FA] = 9°; matrix size = 320 × 320; field of view [FOV] = 230 × 230 mm2; slice thickness = 0.9 mm; number of slices = 192). Proton density and T2-weighted imaging (TR = 10,000 ms; TE 1, 2 = 12, 124 ms; FA = 130°; matrix size = 256 × 256; FOV = 230 × 230 mm2; slice thickness = 3.5 mm; no inter-slice gap) were performed simultaneously, covering the entire brain in the axial plane using a dual-echo turbo spin-echo pulse sequence.
Executive decision-making function examination (trail-making test B)
Executive decision-making has been described as “those capacities that enable a person to successfully engage in independent, purposive, self-directed and self-serving behavior” (Lezak et al., 2012). The trail-making test B (TMT-B) requires an individual to draw lines sequentially connecting 25 randomly placed encircled numbers alternating with letters in ascending order (e.g., 1, A, 2, B, 3, C, etc.) on a sheet of paper. The TMT-B score represents the amount of time (in seconds) required to complete the task. It is one of the most commonly used cognitive tests because of its high sensitivity to evaluate executive decision-making (Brown et al., 2000; Giancola et al., 2001). Test–retest reliability is 0.89 and it has alpha coefficients of .60 to .72 (Arbuthnott & Frank, 2000; Bowie & Harvey, 2006; Gaudino et al., 1995; Lamberty et al., 1994; Tombaugh, 2004).
Depression evaluation
The Beck Depression Inventory version II (BDI2) is a 21-item, self-report questionnaire that measures symptoms of depression (Lezak et al., 2012). Each item is scored on a scale from 0 to 3, with a total BDI2 score range of 0 to 63 (Beck et al., 1988). The BDI2 is a widely used instrument for detection of depression in persons between the ages of 13 and 80 years. It has an internal consistency of 0.73 to 0.92 and a reported high internal consistency (alpha coefficients of .86 and .92) (Beck et al., 1988; Huang & Chen, 2015; Steer et al., 1999, 2001).
Conflict resolution assessment
The Stroop Test is one of the most commonly used cognitive tests, specifically to evaluate conflict resolution as well as executive function (Cabrera-Mino et al., 2021; Roy et al., 2021; Singh et al., 2018; Stroop, 1935; Taylor et al., 1997). Periáñez et al. (2021) reviewed the construct validity of the Stroop Test and clarified its ability to measure cognitive mechanisms including working memory, cognitive flexibility, and conflict monitoring. The Stroop is a timed color-naming test, in which participants are instructed to name the color of the letters when the word is the name of a different color (e.g., the text may say “RED” while the color of the word is green). Scores were calculated from the total time required to respond to all stimulus words in the trial, with additional time added for the total number of errors committed. Validation of the Stroop Test has included both factor analysis methods and its correlation with activation with specific sections of the brain (anterior cingulate—associated with conflict resolution; prefrontal cortices—linked to executive decision-making) in other patient populations (Cabrera-Mino et al., 2021; Graf et al., 1995; Milham et al., 2003; Roy et al., 2021; Singh et al., 2018; Strauss et al., 2005; Taylor et al., 1997). Reliability has been cited to be good (r ≥ 0.80) (Ashburner, 2007, 2010; Connor et al., 1988; Graf et al., 1995; Homack & Riccio, 2004).
Brain MRI Data Processing and Analyses
We used the statistical parametric mapping package SPM12 (Wellcome Department of Cognitive Neurology, UK), MRIcroN, and MATLAB-based (The MathWorks Inc., Natick, MA, USA) custom software for data processing and analyses. Voxel-based morphometry (VBM) analyses were performed as described in an earlier report (Roy et al., 2021; Singh et al., 2018). Two high-resolution T1-weighted images were realigned and averaged for each subject to remove any potential variations between the scans. The Diffeomorphic Anatomic Registration through Exponentiated Lie algebra algorithm (DARTEL) toolbox was used to improve inter-subject image registration (Ashburner, 2007). The averaged images were segmented into gray matter, white matter, and cerebrospinal fluid tissue types using DARTEL toolbox, and flow fields and a series of template images were generated. The flow fields and final template image were used to normalize the gray matter maps (unmodulated, re-sliced to 0.7 × 0.7 × 0.7 mm3) and smoothed using a Gaussian filter (10 mm full width at half maximum).
Statistical Analyses
The IBM statistical package for the social sciences (IBM SPSS, v 24, Chicago, IL, USA) was used for data analyses. Demographic and clinical characteristics were assessed with independent samples t-tests, and categorical characteristics with the chi square. A p value of <.05 was considered statistically significant.
The smoothed whole-brain gray matter maps of YEH participants and controls were compared using analysis of covariance (ANCOVA), with age as a covariate (SPM12; p < .001, uncorrected) to assess gray matter density differences between two groups. The statistical parametric map showing brain sites with significant gray matter density differences between groups were overlaid onto the mean anatomical image for structural identification.
Brain masks of individual brain regions, which showed significant differences between groups based on whole-brain VBM analyses, were created and regional gray matter density values were calculated using the normalized and smoothed gray matter maps with MATLAB-based custom-written software. Regional gray matter density values were compared between groups using ANCOVA (SPSS software), with age included as a covariate to examine significant differences between groups on those sites. A p value of <.05 was chosen to establish statistical significance.
Partial correlations were examined between gray matter density and executive decision-making scores, depression levels, conflict resolution performance, and risky decision-making using smoothed gray matter maps, correcting for age (SPM12; p < .001, uncorrected) of YEH participants and controls. The clusters showing significant correlation were overlaid onto the mean anatomical image for structural identification. Brain maps were created for each brain region showing significant correlation and regional gray matter density values were calculated from smoothed gray matter maps of each participant. Correlation coefficients were determined using the regional density values and TMT-B, BDI2, and Stroop Test scores with partial correlation procedures (SPSS software; covariates, age). A p value of <.05 was considered statistically significant.
Results
No statistically significant differences appeared between the groups for age or body mass (Table 1). However, the YEH group had significantly higher TMT-B scores (worse executive decision-making), greater depression (higher BDI2 scores), and poorer conflict resolution (higher Stroop Test scores) in comparison to controls (Table 1).
Demographic and Symptom Questionnaire Measures.
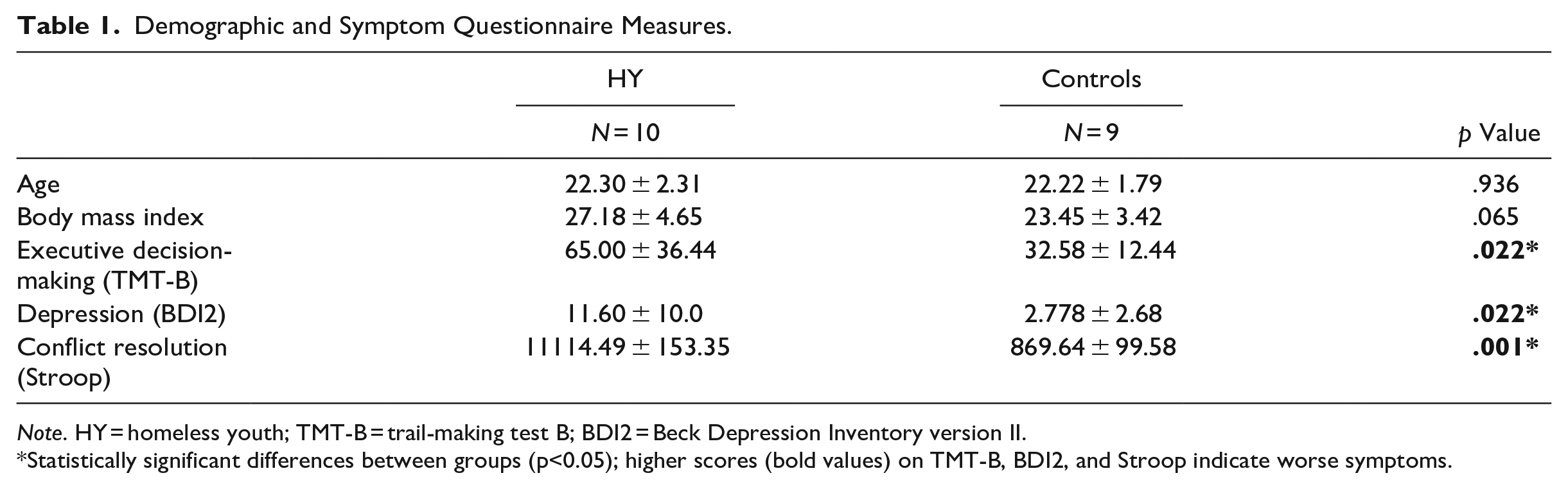
Note. HY = homeless youth; TMT-B = trail-making test B; BDI2 = Beck Depression Inventory version II.
Statistically significant differences between groups (p<0.05); higher scores (bold values) on TMT-B, BDI2, and Stroop indicate worse symptoms.
Regional Brain Gray Matter Density
The VBM analyses showed several brain regions with decreased gray matter density in YEH compared to healthy controls. Brain regions with decreased gray matter density in YEH emerged in the bilateral anterior, mid, and posterior cingulate, bilateral anterior and posterior insula, lingual gyrus, frontal and prefrontal cortex, left cerebellar cortex, inferior and mid occipital gyrus, middle temporal gyrus, postcentral gyrus, right amygdala, basal forebrain, and superior temporal gyrus (Figure 1). Regional gray matter density values of YEH and control participants are summarized in Table 2.
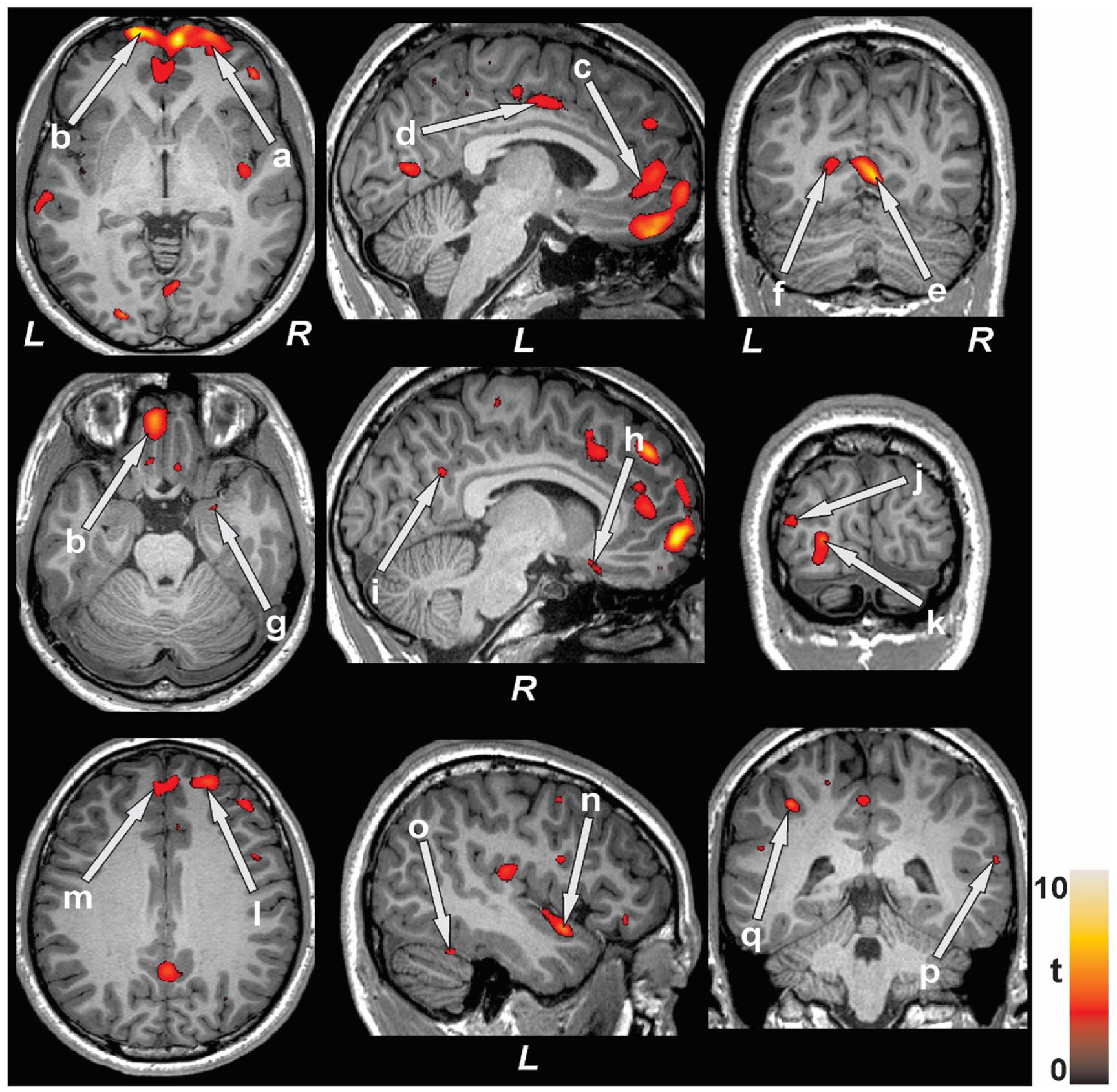
Brain sites with reduced gray matter density in homeless youth compared to control participants. Brain regions with lower gray matter density were observed in the bilateral prefrontal cortex (a, b), anterior (c), mid (d), and posterior (i) cingulate, bilateral lingual gyrus (e, f), right amygdala (g), right basal forebrain (h), left inferior (k) and middle (j) occipital gyrus, bilateral frontal cortex (l, m), left insula (n), left cerebellar cortex (o), right superior temporal gyrus (p) and left postcentral gyrus (q).
Regional Brain Gray Matter Density Values of YEH and Controls Corrected for Age.
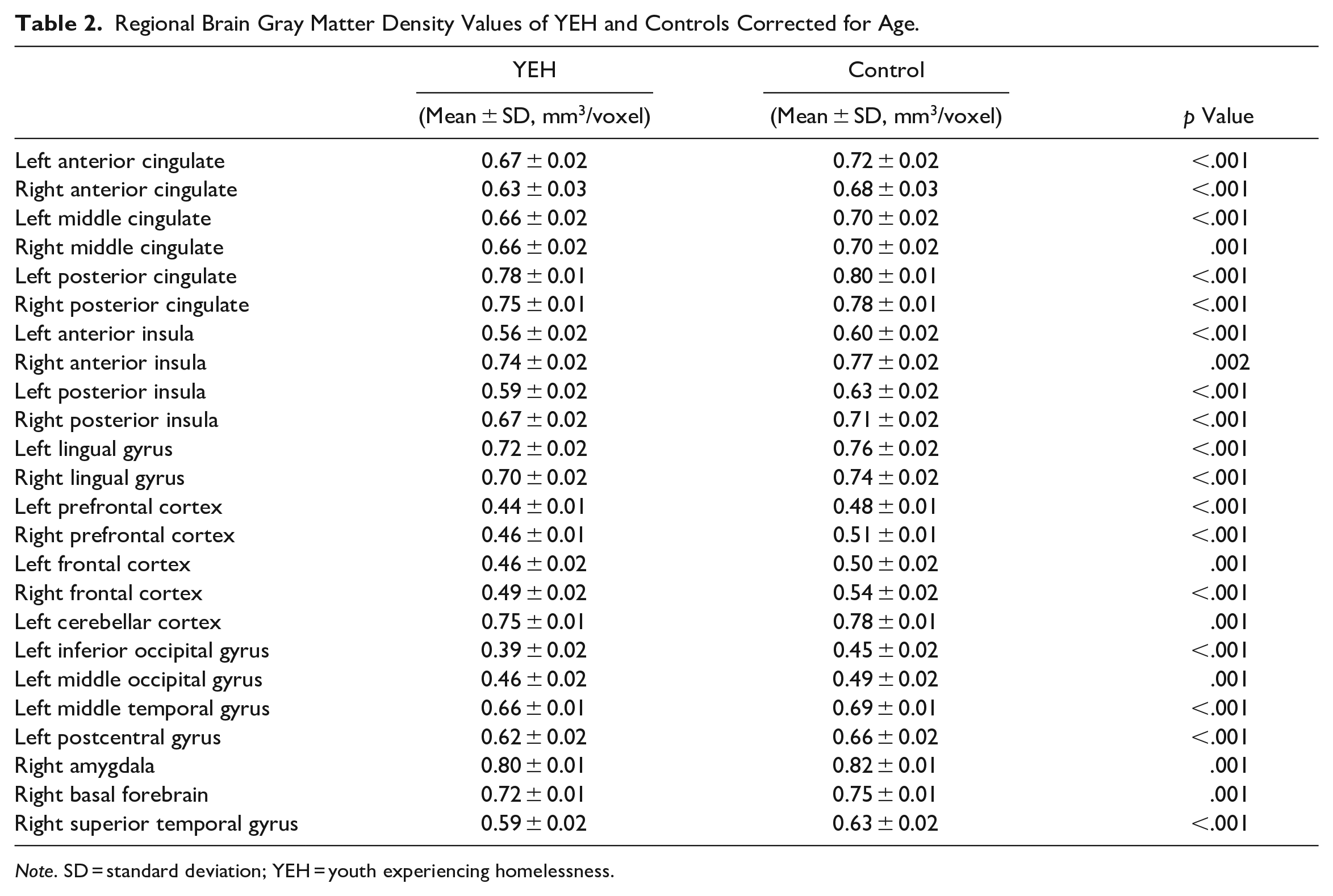
Note. SD = standard deviation; YEH = youth experiencing homelessness.
Relationships Between Brain Gray Matter Density and Symptoms
Multiple brain sites showed significant negative correlations between gray matter density and symptom measures in homeless youth and healthy controls. The gray matter density at bilateral prefrontal cortices showed negative association with executive decision-making performance, insular lobe gray matter density exhibited negative correlation with depression levels, and gray matter density at bilateral anterior cingulate showed inverse relationship with conflict resolution ability (Figure 2). The regional correlation coefficients between gray matter density and symptom measures in YEH participants and controls are listed in Table 3. Further graphs of correlations with data points plotted are provided in Figures 3 to 14. The negative correlations between the brain regions and symptoms remained significantly significant when examined in YEH participants alone (Table 3).
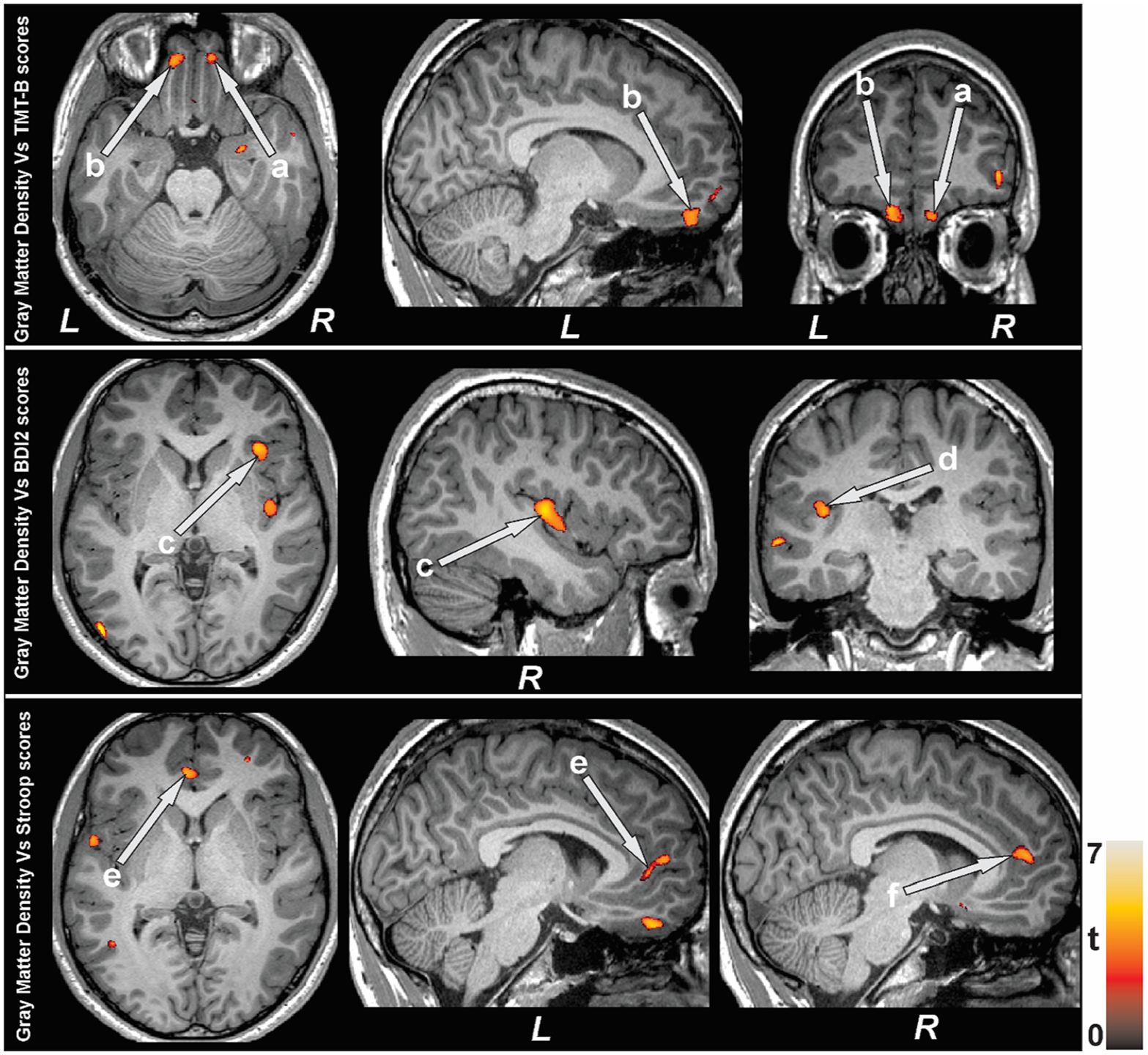
Negative association emerged between gray matter density and symptom measures in homeless youth and control participants. Negative correlation appeared between gray matter density and executive decision-making scores at bilateral prefrontal (a, b) cortices, depression level at bilateral insula (c, d), and conflict resolution measures at bilateral anterior cingulate (e, f).
Regional Correlation Values between Gray Matter Density and Symptom Measures in Both YEH and Control Participants and in Only YEH Participants Corrected for Age.
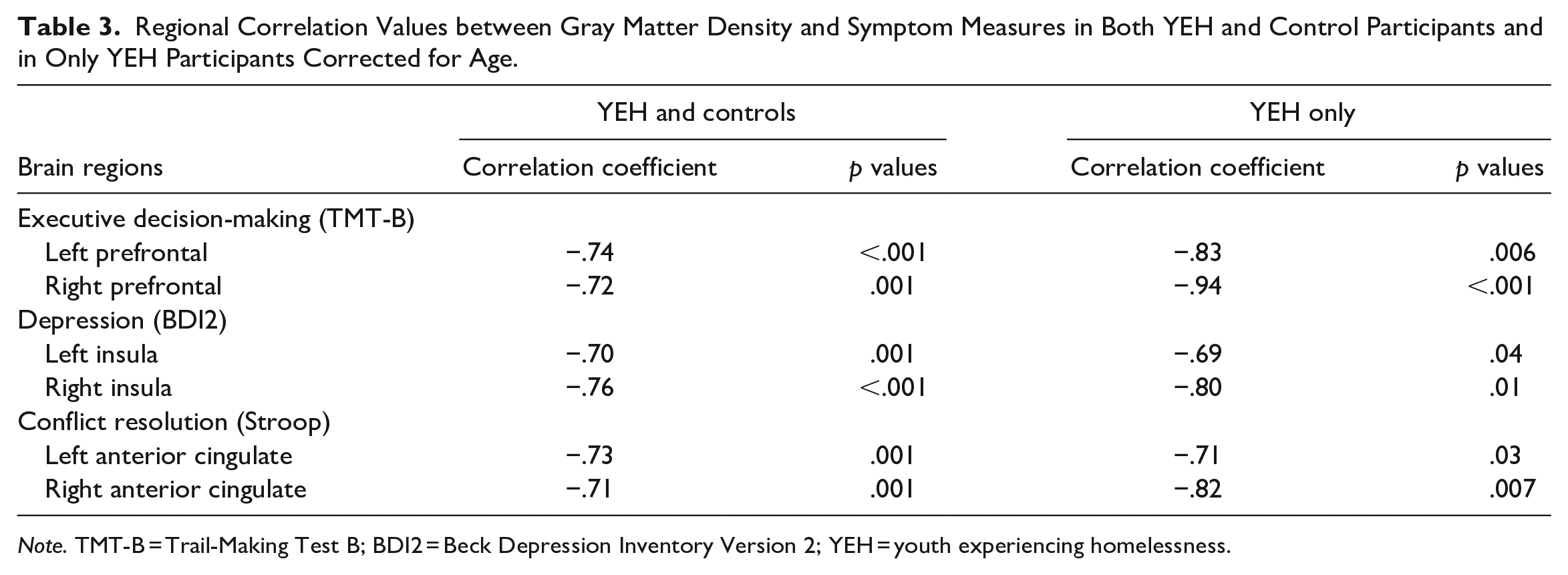
Note. TMT-B = Trail-Making Test B; BDI2 = Beck Depression Inventory Version 2; YEH = youth experiencing homelessness.
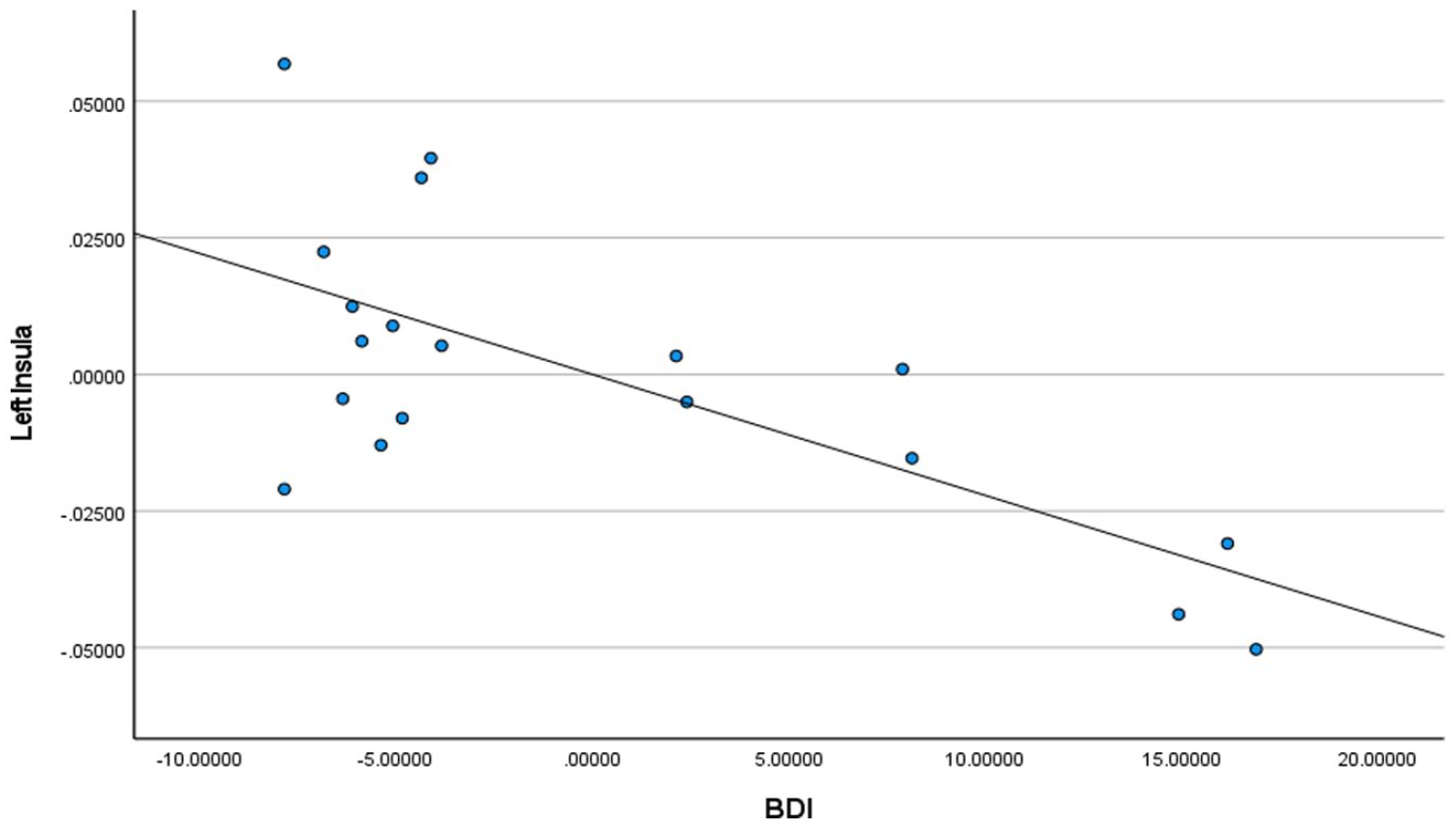
BDI2 scores for controls + homeless youth in left insula.
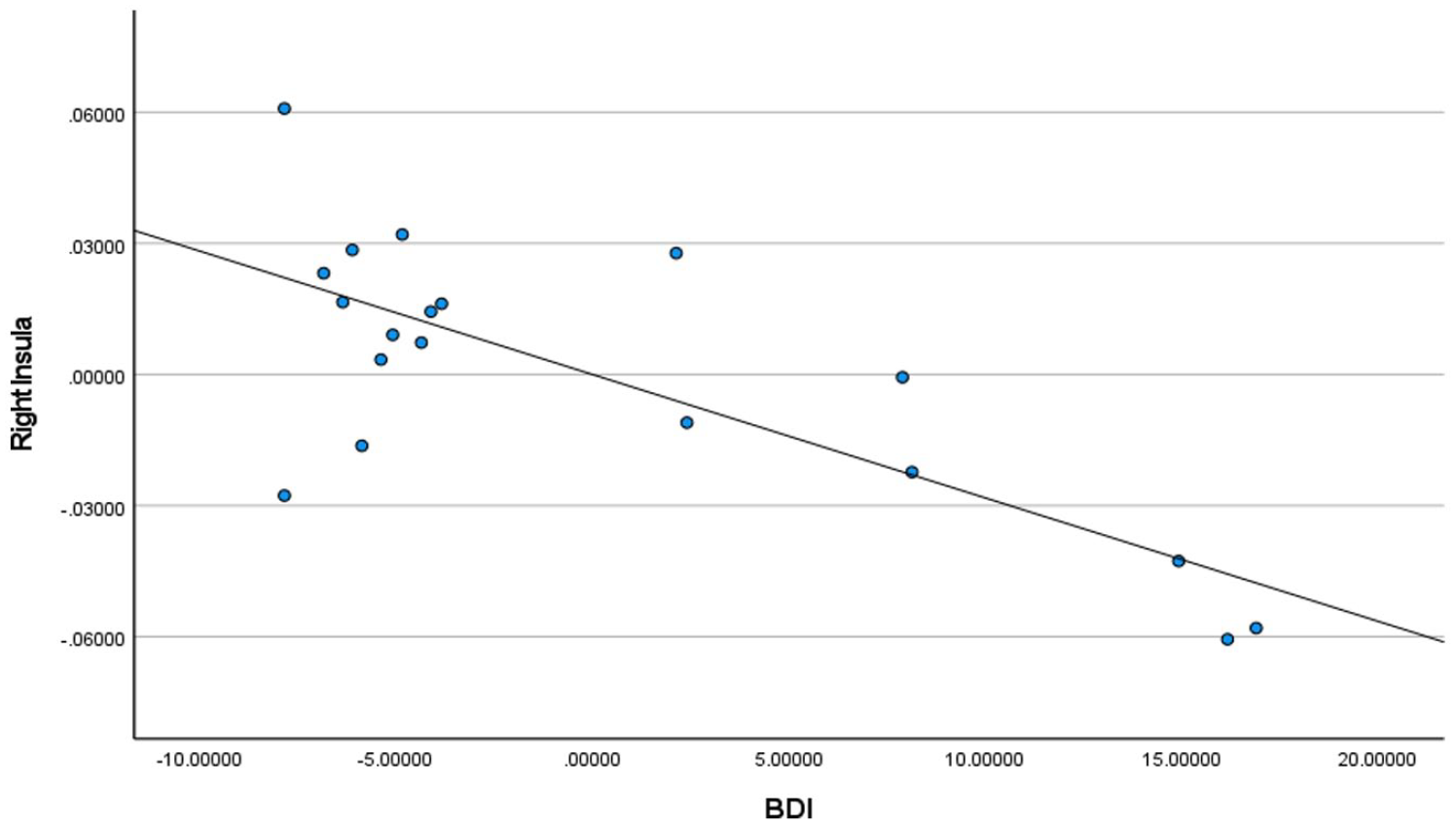
BDI2 scores for controls + homeless youth in right insula.
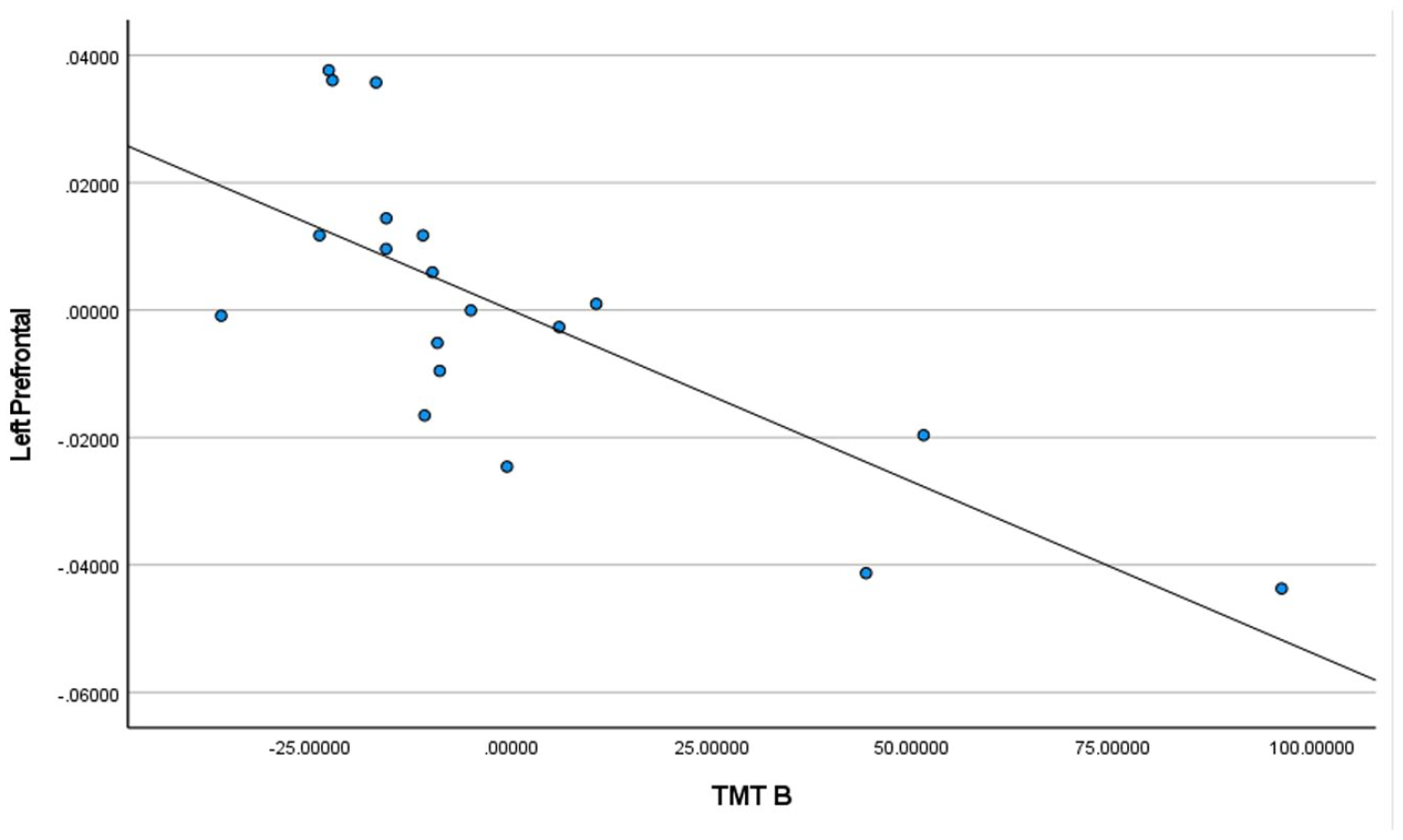
TMT-B scores for controls + homeless youth in left prefrontal cortex.
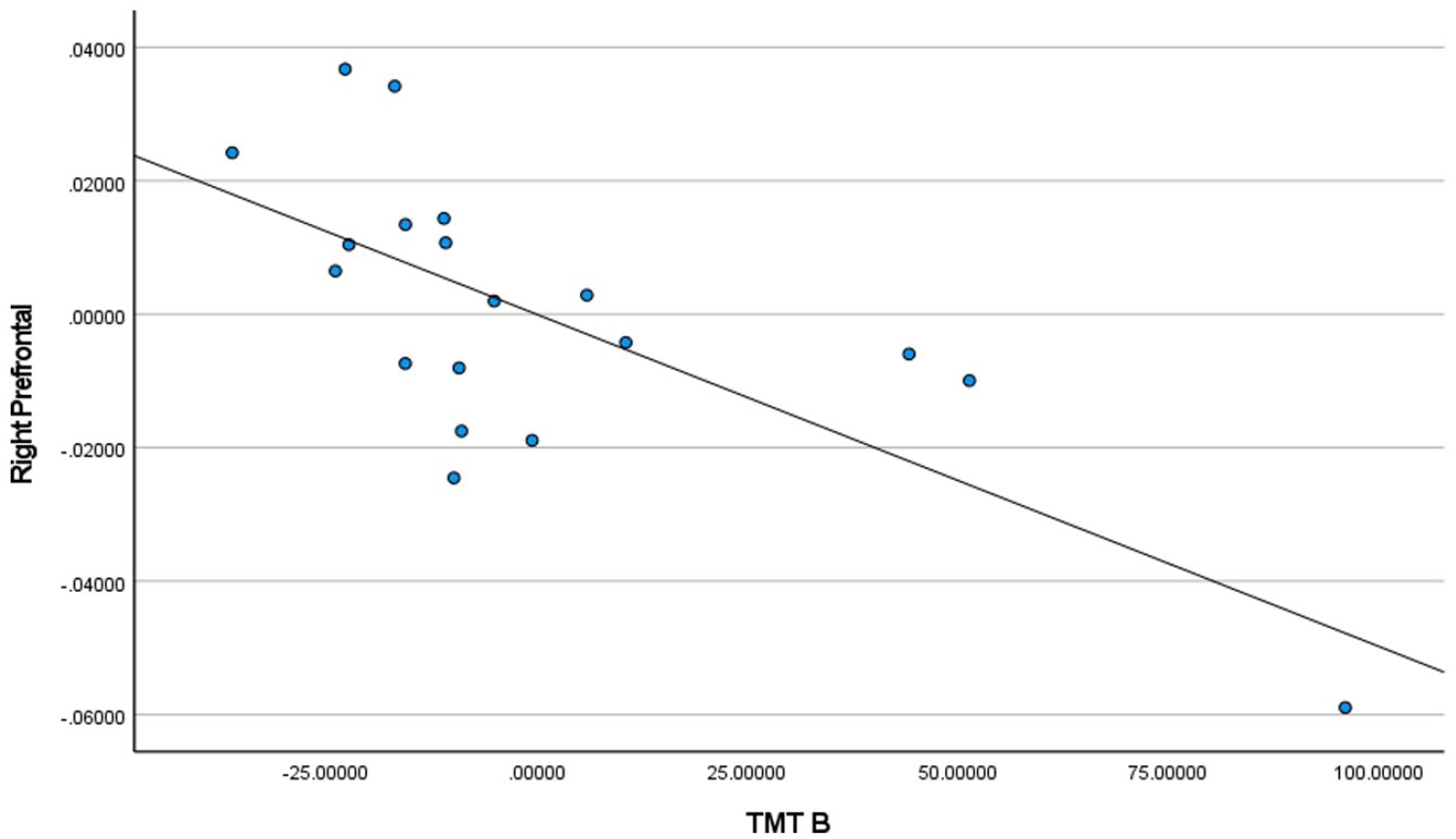
TMT-B scores for controls + homeless youth in the right prefrontal cortex.
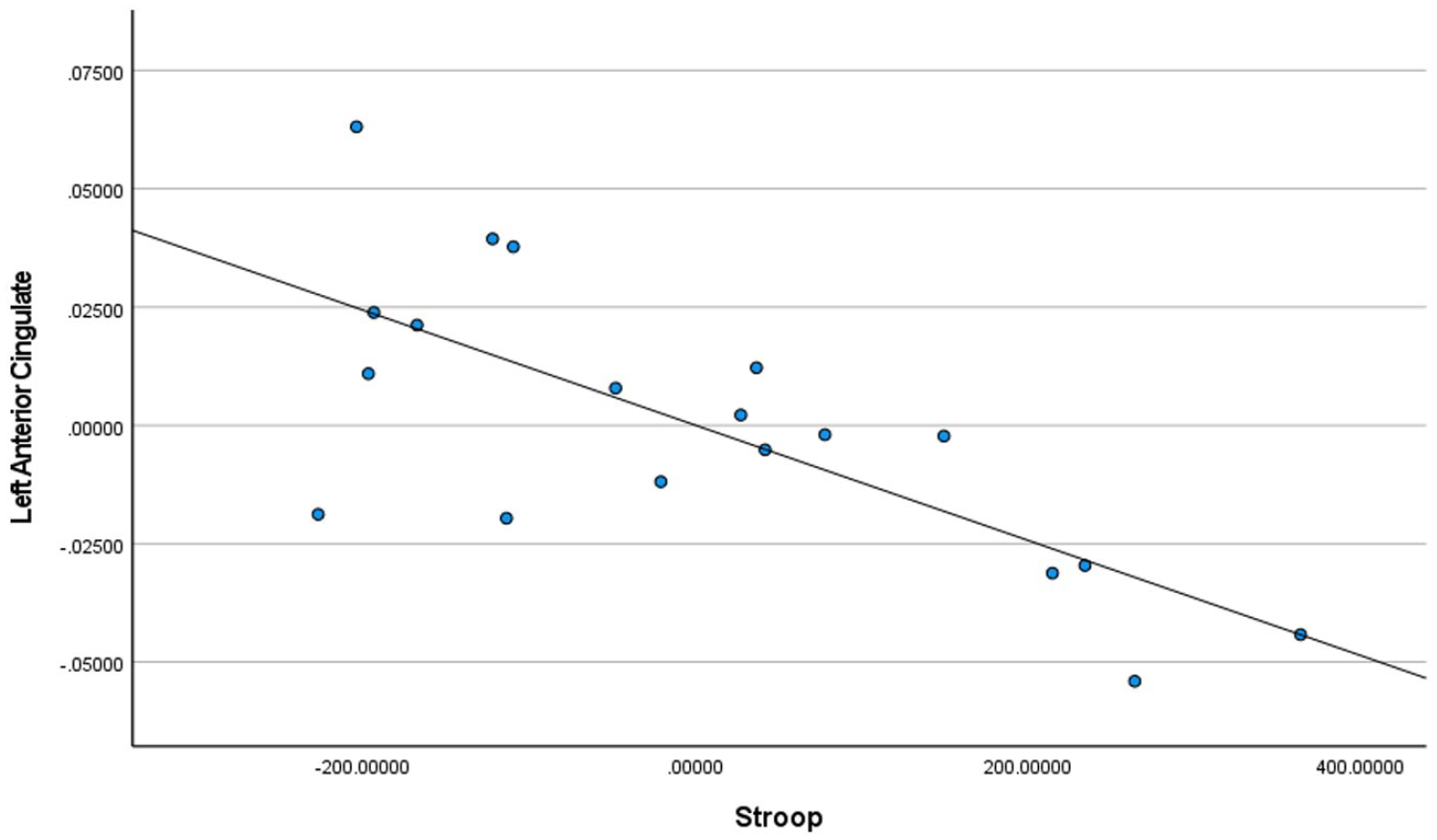
Stroop test scores for controls + homeless youth in left anterior cingulate.
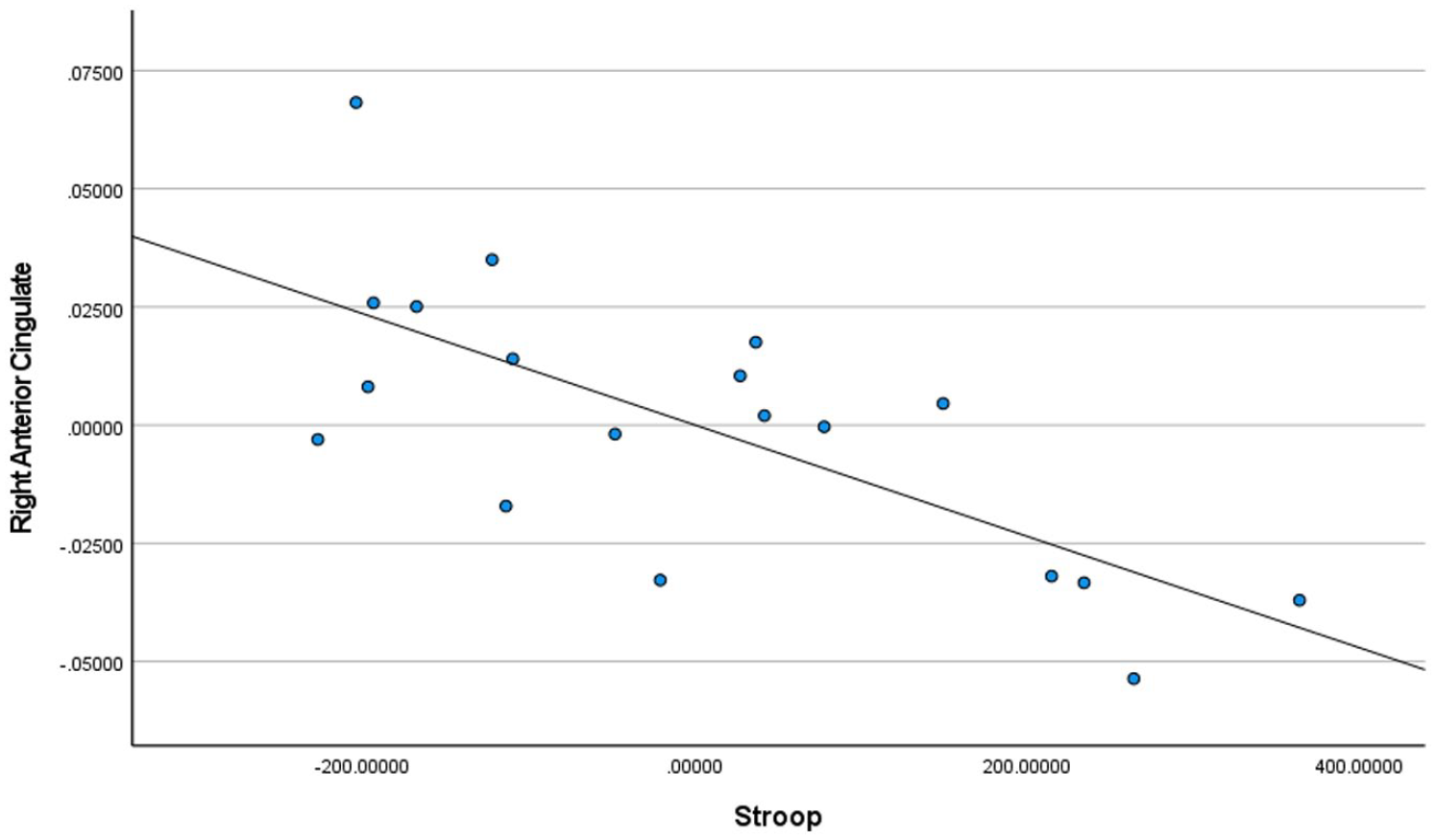
Stroop test scores for controls + homeless youth in the right anterior cingulate.
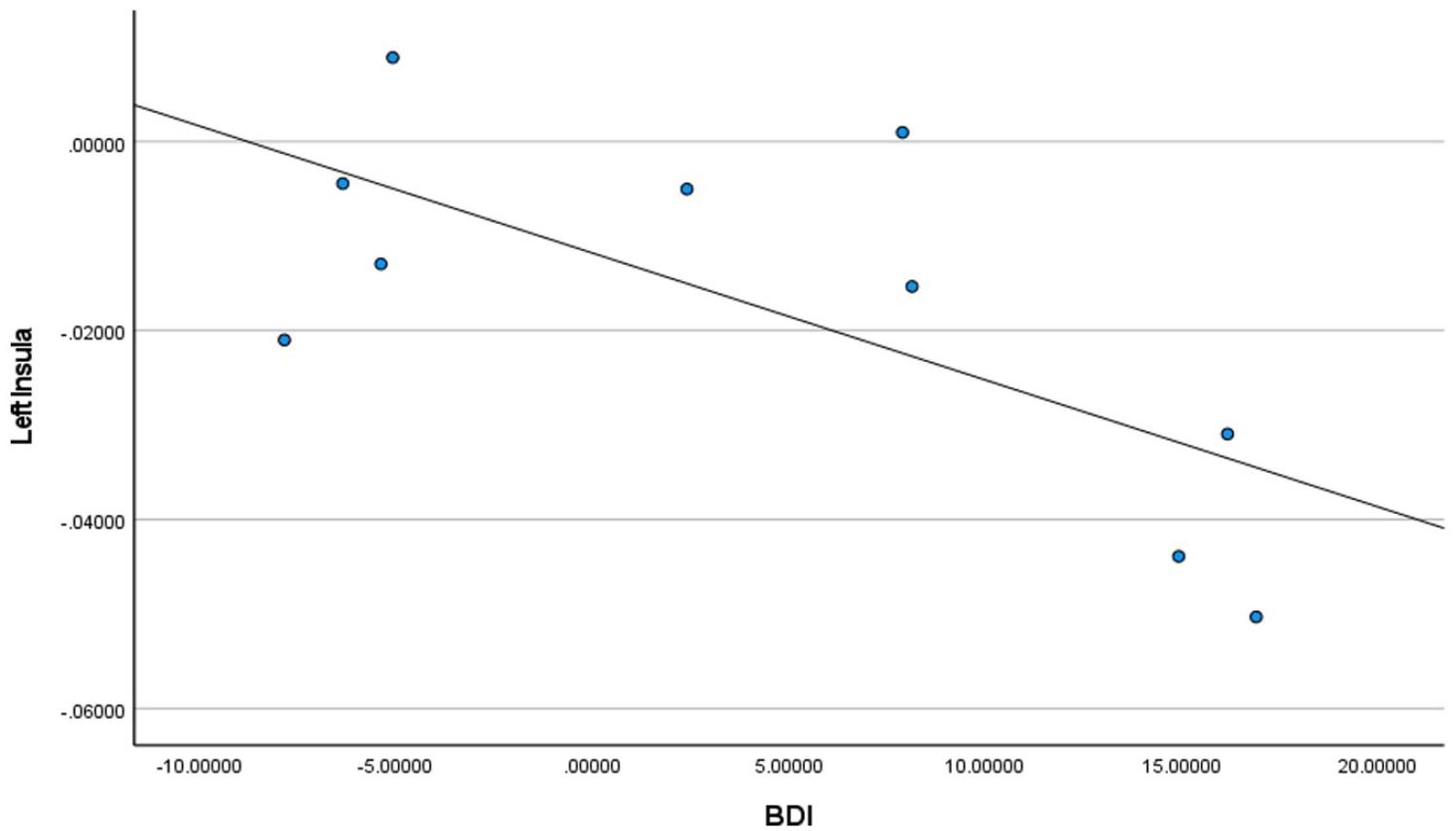
BDI2 scores for homeless youth only in the left insula.
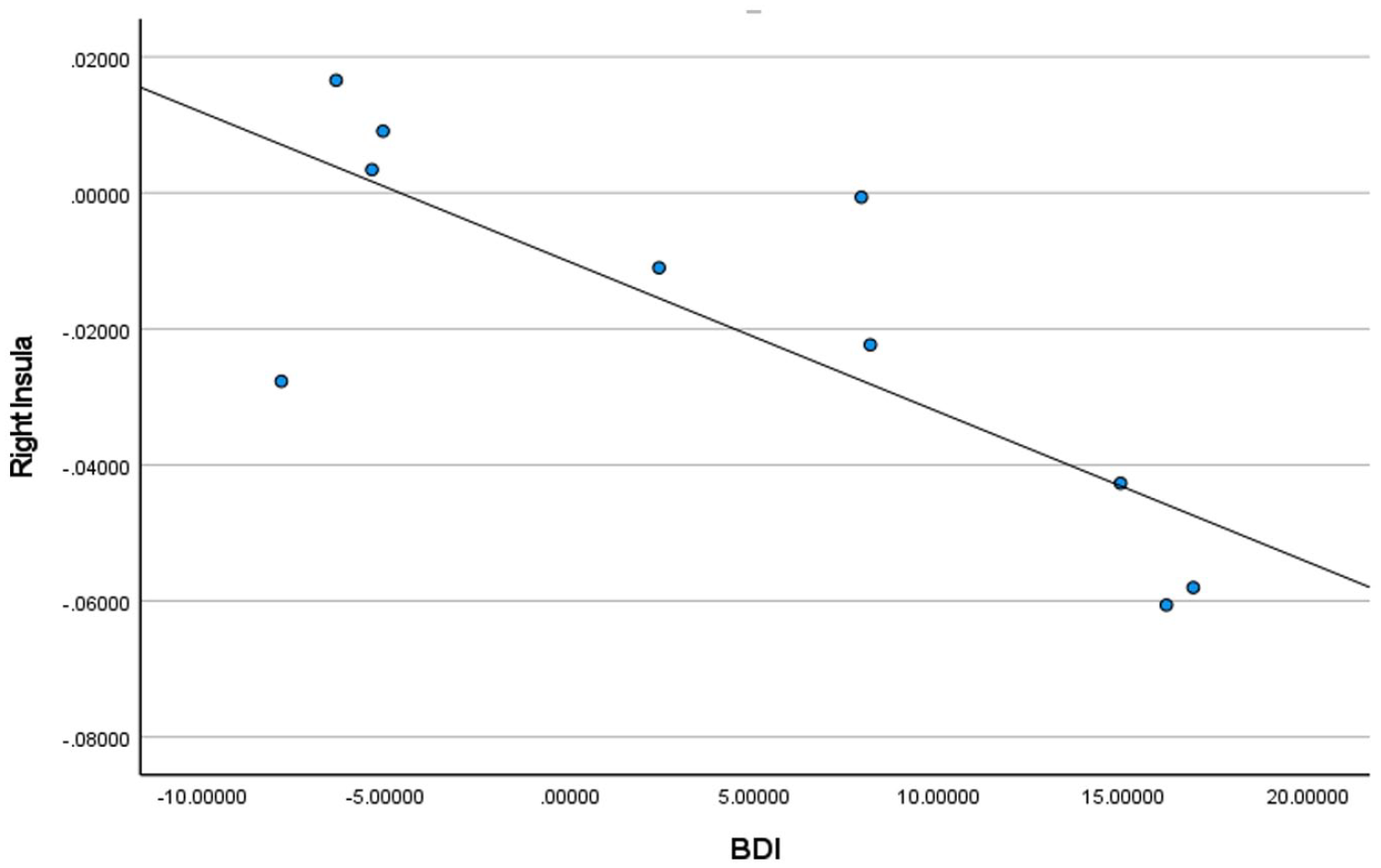
BDI2 scores for homeless youth only in the right insula.
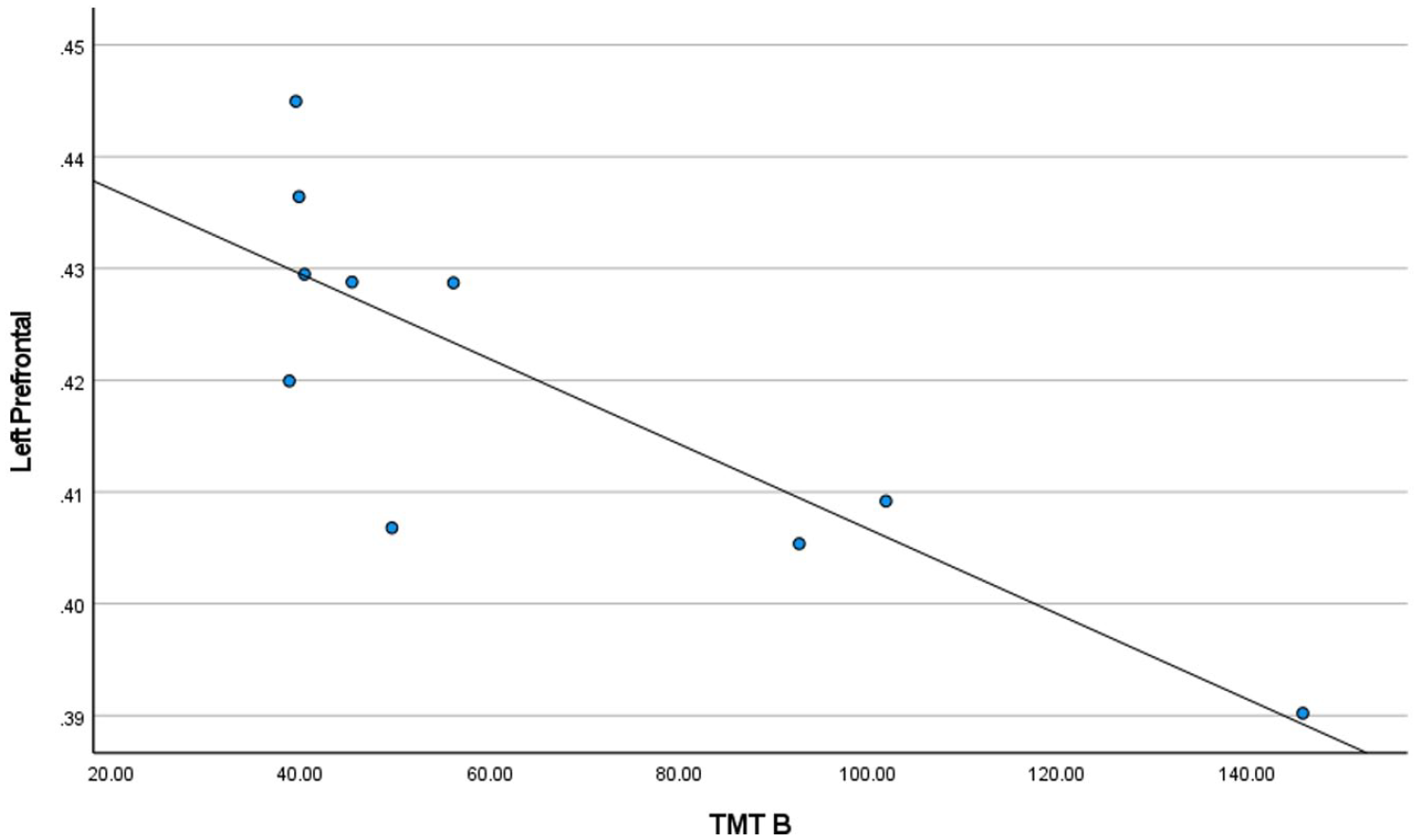
TMT-B scores for homeless youth only in the left prefrontal cortex.
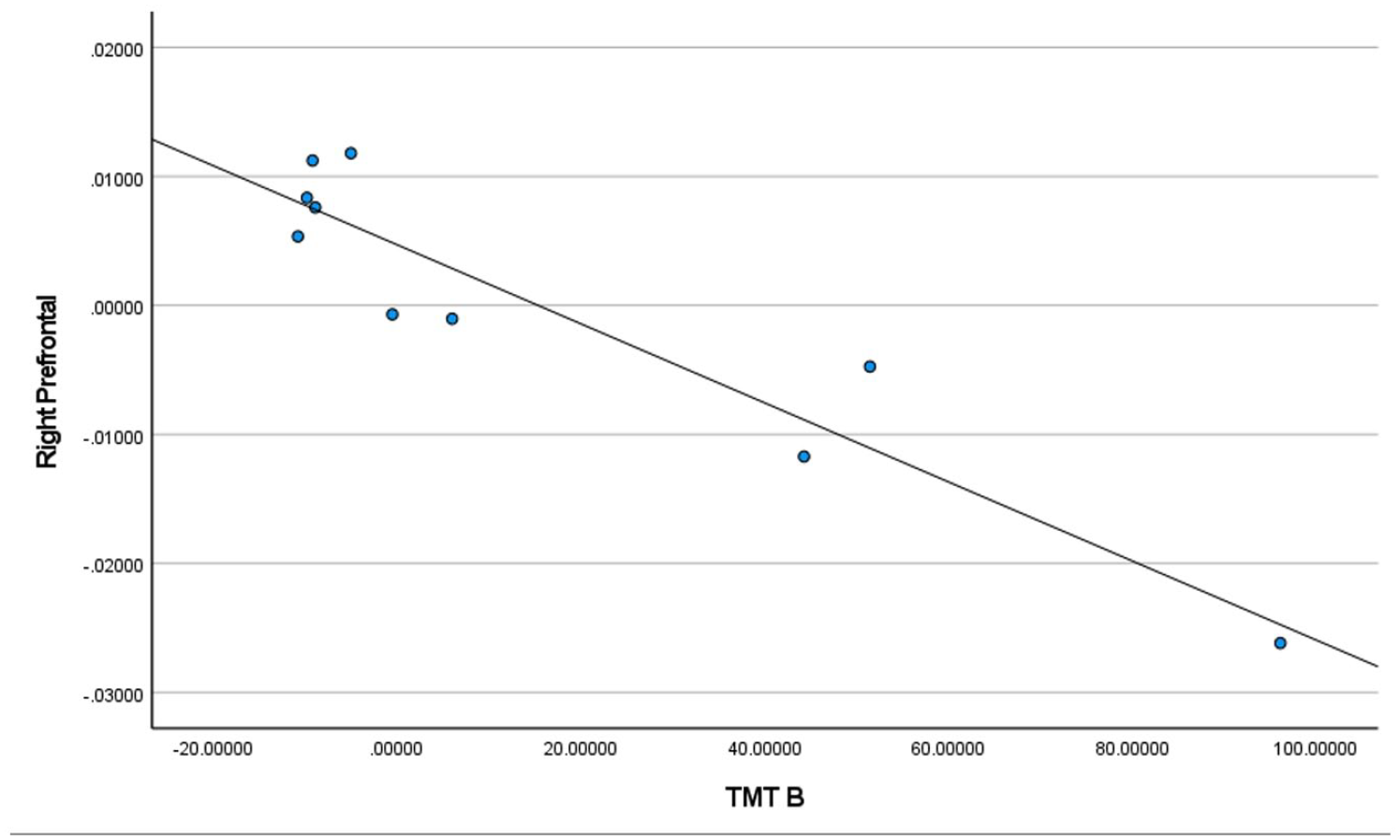
TMT-B scores for homeless youth only in the right prefrontal cortex.
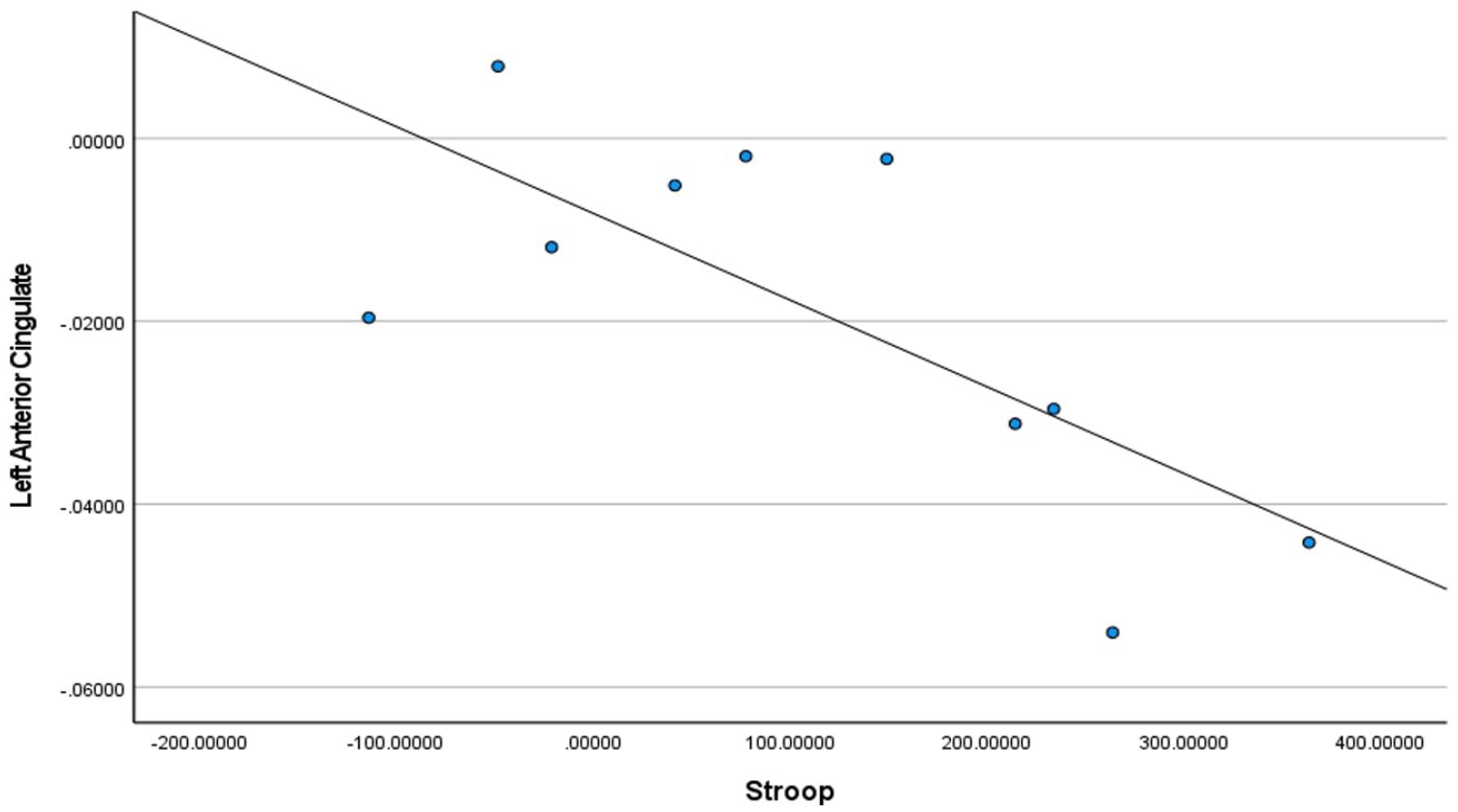
Stroop test scores for homeless youth only in the left anterior cingulate.
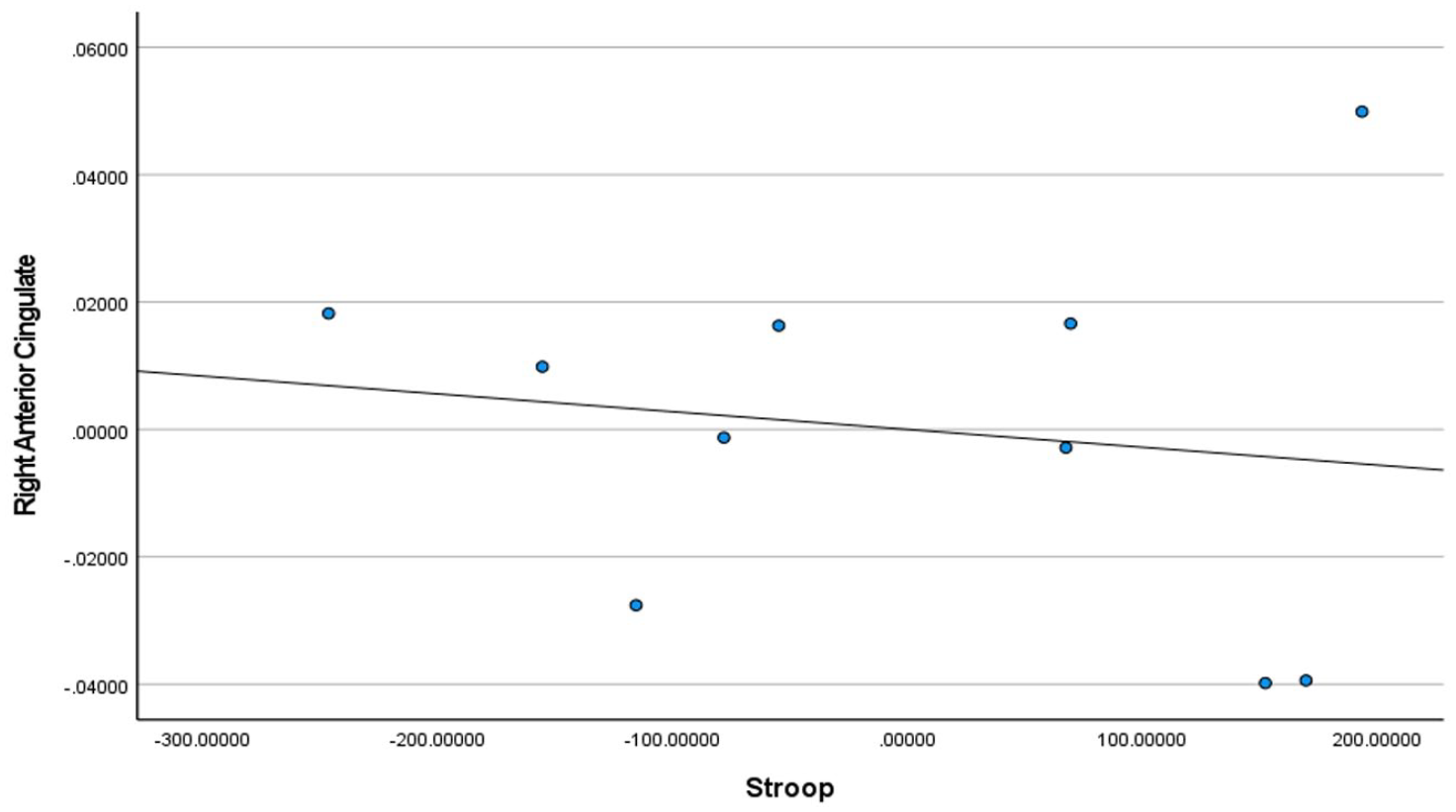
Stroop test scores for homeless youth only in the right anterior cingulate.
Discussion
The YEH participants had significantly decreased gray matter tissue in comparison to the controls. Moreover, there were strong inverse correlations between the brain regions classically associated with executive decision-making (prefrontal cortices), depression (insular lobes), and conflict resolution (anterior cingulate), and the level of the symptoms detected by their questionnaires. However, the underlying etiology and sequence (i.e., did symptoms or the brain changes occur first? Were the brain changes present before homelessness or after these neural alterations in YEH?) are unclear. Thus, questions remain on what could have caused such brain changes and on whether encouragement of neurogenesis (brain tissue regrowth), administration of neural protective measures, or resolution of homelessness can enhance outcomes (decreased symptoms) in the YEH population.
Potential Etiologies
It is known that YEH are at increased risk for a variety of factors which are known to increase the incidence of brain changes in the prefrontal cortices, insular lobes, and anterior cingulate of homeless, non-homeless, and animal studies (Marston, 2013). These factors include poverty, unstable housing, low social support, drug use, and traumatic brain injury (TBI; Bergen, 2008; Boyle et al., 2011; Marston, 2013). Many of these factors which are known to be associated with brain changes are frequently present in YEH.
Situational factors, such as poverty, unstable housing, and low social support, are multifactorial, and can have a combination of situational/chance, social, medical/psychological, and genetic causes. Nevertheless, all these factors can either be the cause of, or induced by, brain changes, particularly in the prefrontal cortices. For example, family incomes below the federal poverty level were associated with higher levels of developmental disabilities (Marston, 2013), learning disabilities and intellectual disabilities (Bergen, 2008; Boyle et al., 2011), increased prefrontal cortex abnormalities (Farah et al., 2006; Noble et al., 2012; Rinaldi et al., 2008), and lower brain volumes in the hippocampi and amygdala brain regions (Noble et al., 2012). In addition, low-income status is linked to the risk of malnutrition, which is associated with increases in neurobehavioral disorders, lower brain volumes, and decreased development in the prefrontal cortex (Farah et al., 2006). Adversity and trauma accumulate over time and can concomitantly impact physical and mental health (CDC, 2020). ACE research has convincingly established both the impact of chronic stress and the reinforcement of health inequities in marginalized populations (Anda et al., 2006; Dube et al., 2006; Felitti et al., 1998). Under-detected and untreated toxic stress as well as comorbidities such as mental illness, risk behaviors, and somatization is connected to lower functioning, productivity, and levels of quality of life, and poor self-management (Liu et al., 2015). Repeated and/or prolonged exposure to adversity during childhood triggers a stress response that without countering with buffering protective factors can lead to long-term structural and functional changes in brain, metabolic and immune systems, and DNA (Nelson et al., 2020; Shonkoff et al., 2012).
While it is acknowledged that genetic predispositions (i.e., the actual associations between genetics and YEH risk) are uncertain, there is increasing evidence that hereditary factors can be linked to homelessness, high-risk behaviors, and brain changes. Several reports indicate that genetic makeup can significantly increase smoking progression in adolescents (Audrain-McGovern et al., 2004) and epigenetic changes, such as an increase in methylation, result in an excessive release of stress hormones in times of stress, which can impact depression (Dunlop & Nemeroff, 2007; Fraga et al., 2005; Li et al., 2013; Pembrey et al., 2014; Radtke et al., 2011).
A physical factor known to induce brain changes is TBI, which can be defined as any self-reported head injury which leads to being dazed, confused, disoriented, or unconscious (Hwang et al., 2008). Few researchers have studied TBI among homeless populations (Hwang et al., 2008; Mackelprang et al., 2014). TBI can be defined as any self-reported head injury which leads to being dazed, confused, disoriented, or unconscious Mackelprang et al., 2014). In one study among homeless adolescents and young adults, over half of the participants (51%) reported sustaining their first TBI prior to becoming homeless or at the same age of their first homeless episode (Mackelprang et al., 2014). Furthermore, YEH with TBI became homeless at a younger age, and were more likely to report mental health diagnoses, substance use, suicidality, victimization, and difficulties with activities of daily living (Mackelprang et al., 2014).
While some studies have assessed TBI among homeless populations, none have focused on structural brain changes or how interventions may be impacted. It is known that TBI can increase the risk for major depression to two to five times above that of the general population and is associated with significantly increased incidence of anxiety, alcohol abuse, difficulty in remaining employed, maintaining social relationships, and fulfilling many other social roles (Brooks et al., 1986; Hibbard, et al., 1998; Kreutzer et al., 2003; Sander et al., 1996; Van Zomeren & Van den Burg, 1985). Although the participants’ nutritional status, trauma and TBI histories were unknown, this particular population reported a history of recent alcohol and substance use. Thus, the association between neurocognitive changes associated with alcohol and substance abuse as well as trauma and TBI cannot be underestimated (Hamidullah et al., 2020).
Implications
These are novel findings and imply that the high incidence of the adverse symptoms of poor executive decision-making, significant depression, and inadequate conflict resolution skills in YEH may be related to physiological changes in the brain. The study’s findings may imply that YEH interventions for these symptoms should consider the impact of these potential treatments on the brain such as cognitive behavioral therapy, exercise, social skill training, nutritional supplements (such as thiamine), medications (such as psychostimulants and antidepressants), and resolution of homelessness. It may be that without restoration and/or repair of the brain, standard YEH treatment of these symptoms may be ineffective or transient.
In terms of limitations, it is important to remember that this is a one-time, cross-sectional study with a very small sample, and that causal inferences cannot be made related to the results. Thus, additional research is needed to further elucidate the underlying causes of brain changes with larger samples of YEH, to gain greater understanding of the relationships between brain injury and symptoms in YEH. We also did not collect information on history of brain injury/trauma for YEH; which should be an area of attention in future research. Furthermore, research is also needed to identify and test potential interventions to minimize or induce neurogenesis in these areas of the brain and to determine the chronological sequence of these brain changes (before or after homelessness), which may impact adverse symptomatology and identify innovative interventions in YEH.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
