Abstract
Cultural and religious beliefs in certain regions, including many Muslim-majority countries, often lead families to request the withholding of medical information from patients. Despite the availability of validated symptom assessment tools for chronic conditions such as cancer, chronic obstructive pulmonary disease, neurological disorders, heart disease, and renal failure, the effect of nondisclosure on symptom burden remains insufficiently explored. This scoping review examines the role of symptom assessment tools in evaluating the outcomes of nondisclosure in advanced cancer patients, particularly in the United Arab Emirates (UAE). The review assesses five key instruments: the Edmonton Symptom Assessment Scale (ESAS), its revised version (ESAS-r), the Memorial Symptom Assessment Scale (MSAS), the short form (MSAS-SF), and the Palliative Care Outcome Scale (POS). An analysis of the psychometric properties, strengths, and limitations of these tools highlights their utility in understanding symptom burden and psychological well-being. The findings suggest that comprehensive tools like the MSAS provide valuable assessments but require further validation to confirm their effectiveness across diverse clinical and cultural settings. Future research should prioritize adapting these tools for wider application and ensuring their reliability and validity in measuring the impact of nondisclosure on symptom burden across various diagnoses, including both cancer and noncancer conditions.
Introduction
Palliative care plays a crucial role in improving the quality of life for individuals facing serious, life-limiting illnesses by addressing their physical, emotional, and spiritual needs (NCP-guidelines, Clark, 1999; Field & Cassel, 1997; Ganz, 2014; National Coalition for Hospice and Palliative Care, 2018; Saunders, 1998; Tark et al., 2023). With the global aging population expected to reach 2.1 billion by 2050, there is a projected 77% increase in cancer rates and associated geriatric syndromes, underscoring the escalating burden of symptoms in these populations (Johnston et al., 2024; World Health Organization, n.d.). Despite the benefits of palliative care, globally only 14% of patients actually receive it, leading to significant disparities in care provision (World Health Organization, n.d.). Common symptoms such as pain and shortness of breath are prevalent among 80% of oncology patients, highlighting the critical need for effective symptom assessment and management (Abu-Saad Huijer et al., 2015; Battat et al., 2023; Hui & Bruera, 2017; Shamieh et al., 2017; Teunissen et al., 2007; World Health Organization, n.d.). In Muslim-majority regions like the United Arab Emirates (UAE), nondisclosure of terminal diagnoses due to cultural and religious beliefs is common, influencing how patients cope with their illnesses and impacting the quality of end-of-life care (Abdullah et al., 2020; Malas et al., 2020). Research indicates that nondisclosure prolongs the dying process and exacerbates symptom burden, complicating care goals and patient outcomes (Abdullah et al., 2020; Alsirafy et al., 2017; Chittem et al., 2021; Verhoef et al., 2022). Subsequently, in Muslim-majority countries, there is evidence to suggest nondisclosure results in a lower quality of end-of-life care in those approaching the end of life (Nayfeh, 2023).
Furthermore, within the United Arab Emirates (UAE) approximately 4,500 new cancer cases are reported each year among a population of nearly 10.2 million (UAE Government, n.d). While the specific rates of nondisclosure requests among Muslim patients in the UAE and the broader Middle East are unknown, the prevalence of nondisclosure of cancer diagnoses among patients in Asia ranges from 16.9% to 63% (Chittem et al., 2021). It is well documented that nondisclosure significantly impacts how patients cope with their illnesses and increases total pain in Muslim populations (Abdullah et al., 2020; Degi, 2009).
As a result, efforts to improve care practices, such as the use of validated transcultural assessment tools like the Edmonton Symptom Assessment Scale-Revised (ESAS-r) and the Palliative Care Outcome Scale (POS), are essential. These tools enable healthcare providers to monitor symptom distress effectively and tailor interventions to meet the unique needs of Muslim patients facing nondisclosure (Henson et al., 2020). These tools not only facilitate comprehensive symptom evaluation but also promote better communication and understanding between healthcare providers and patients, particularly in regions with diverse cultural norms regarding illness disclosure (Portenoy, 1994).
The aim of this article is to examine the psychometric properties of various symptom assessment scales that have been adopted transcultural and in Arabic to measure outcomes of nondisclosure on the Muslim patients’ symptom burden.
Literature Review
Nondisclosure
Nondisclosure, the practice of withholding medical information from patients, is often driven by cultural and religious beliefs that prioritize family-centered decision-making over patient autonomy (Abdullah et al., 2020; Malas et al., 2020). This practice can lead to ethical dilemmas and challenges in patient care. Symptom assessment tools are vital in palliative care to monitor and manage patients’ physical and psychological symptoms (Abu-Saad Huijer et al., 2015; Battat et al., 2023; Hui & Bruera, 2017; Shamieh et al., 2017; Teunissen et al., 2007; World Health Organization, n.d.). Understanding these tools’ reliability and validity, particularly in diverse cultural settings, is crucial for ensuring effective patient care (Henson et al., 2020; Portenoy, 1994). This review addresses the need for conceptual clarity by discussing how the selected tools measure symptom burden and how they are adapted to cultural contexts.
Ethical Considerations
The American Society of Clinical Oncology (ASCO) in 2023 discussed the need to report patient-related outcomes measures (PROMs) to help identify early palliative care interventions, improve symptom management and patients’ quality of life (Prichett et al., 2023; Sanders et al., 2024). However, symptom scales that assess well-being are not clearly defined and therefore need further clarification. Inadequate results may result in suboptimal treatment and may therefore add to the patients’ burden (Busch et al., 2010; Holen et al., 2006; Hui & Bruera, 2017). Further research is needed to ensure that well-being and depression measures accurately reflect their intended constructs, known as construct validity. Moreover, ESAS and MSAS have not been adapted to the geriatric population but have shown validity in recent studies (Browner & Smith, 2013; Hui & Bruera, 2017). In addition to the above, multiple ethical issues related to nondisclosure and symptom burden arise.
Firstly, those with life-limiting illnesses such as advanced cancer are more vulnerable (Bruera, 1994). This is a population that is already overwhelmed by their illness and may have fear of repercussions or rehospitalization if they do not participate in the study which can result in the participants being unintentionally coerced into taking part in the research study (Addington-Hall, 2002).In this way, there must be a balance between nonmaleficence “do no harm” and beneficence, in other words, to do good by the patient and for the patient (Akdeniz et al., 2021). In palliative care, especially with the ethical principle of beneficence, managing symptoms in order to reduce the suffering to those with limiting illness and improve quality of life improves palliative care delivery (Akdeniz et al., 2021). Moreover, to avoid coercion there should be a “silent opt out” option for participants giving the participants an option to withdraw from the study (Carpenter et al., 2021).
Secondly, during consent, it is important to determine if the participants are cognitively intact as they may decline over the course of the study (Bruera, 1994; Pereira et al., 1997; Wohleber et al., 2012). It is recommended to have a proxy “dual consent” should withdrawal from the study be requested (Shepard et al., 2020; Wohleber et al., 2012).
Thirdly, nondisclosure of medical information to a patient without patient consent may be an ethical violation if participant or patient consent is not obtained directly thereby potentially violating the Health Insurance Portability and Accountability Act (Tariq & Hackert, 2023). This can be addressed directly if the participant, who has decisional capacity, defers to a family member directly around shared decision-making and respects the patient's autonomy (Kuosmanen et al., 2021). It is recommended to ask this question during the initial or routine patient visits with the healthcare provider (HCP).
Lastly, online surveys are considered to be cost-effective and simple, ethical issues arise around access, security of data, storage and the need for informed consent. The data should be kept secure at the hospital's electronic health record (EHR) site as symptom burden is being assessed (Buchanan & Hvizdak, 2008). A recent study suggests the use of electronic PROMs (ePROMs) with patient-informed consent to help address symptoms as some EHRs have the ability to integrate patient-related outcomes (PRO) functionality (Crockett et al., 2023; Prichett et al., 2023).
Consequently, screening for symptoms can be complex and burdensome for patients. Simple scales must be used that are considered reliable and valid. Overall, by justifying the need for better symptom management and end-of-life care in addition to the “psychosocial” impact of disease on stakeholders and participants, the researcher is able to improve the quality of life in those with advanced illness (Jubb, 2002).
Importance of Reliable and Valid Tools
There are different types of validity that generally fall under construct validity, except face and content validity which are classified as translational validity (Trochim, 1999). While construct validity measures what it was intended to measure, face validity is considered generally to be weak. In addition, content validity evaluates how the tool measures all aspects of the construct and the intended measure (DeVon, 2007). Identifying and selecting an instrument that has good validity and reliability in addition to good recall is essential. The selected tool ideally should have fewer, items although not always necessary. It has been suggested for weekly assessments that a 10–20-item scale would be preferred and should be electronically administered (Basch et al., 2016; Di Maio et al., 2022). In order to increase the validity of an instrument, content validity should be assessed in order to understand the research findings (DeVon et al., 2007; Lynn, 1986). There is a need to ensure that the consistency of the tool lasts over time to guarantee adequate measurements and therefore show test-retest reliability (Granda-Cameron et al., 2008). This ultimately assesses the symptoms of the patient accurately and improves their quality of life. It is important to use easy-to-use instruments that allow patients to complete them without being laborious or burdensome. One such scale, the ESAS, has a completion time of under 1 min in the clinic with a “multidimensional” symptom assessment (Wong et al., 2020). In addition, the ESAS showed prolonged survival in those who were administered ESAS and earlier management of symptoms (Barbera et al., 2020). Through the selection of the tool and the identification of the psychometric properties, the final tool can be implemented in the study.
Methods
On June 11, 2024, a search was conducted in CINHAL, Scopus, and PubMed from the years 2019 to 2024 using the terms Medical Subject Heading (MeSH)/Index headings in the selected database. The search was conducted with MeSH with keywords “non-disclosure” AND “end-of-life” OR “palliative care” AND “instrument or tool” AND “symptoms” OR “symptom,” AND “cancer” References from the articles were utilized to further identify relevant literature. The search strategy is outlined in Figure 1. The literature inclusion criteria focus was on studies documenting those aged 18 and above, covering the period from 2019 to 2024 with a cancer diagnosis. Excluded from the review are articles reporting nonterminal studies, patients under 18 years old, those conducted before 2019, and articles not in English (NEL). Refer to Table 1 for instruments selected for review.
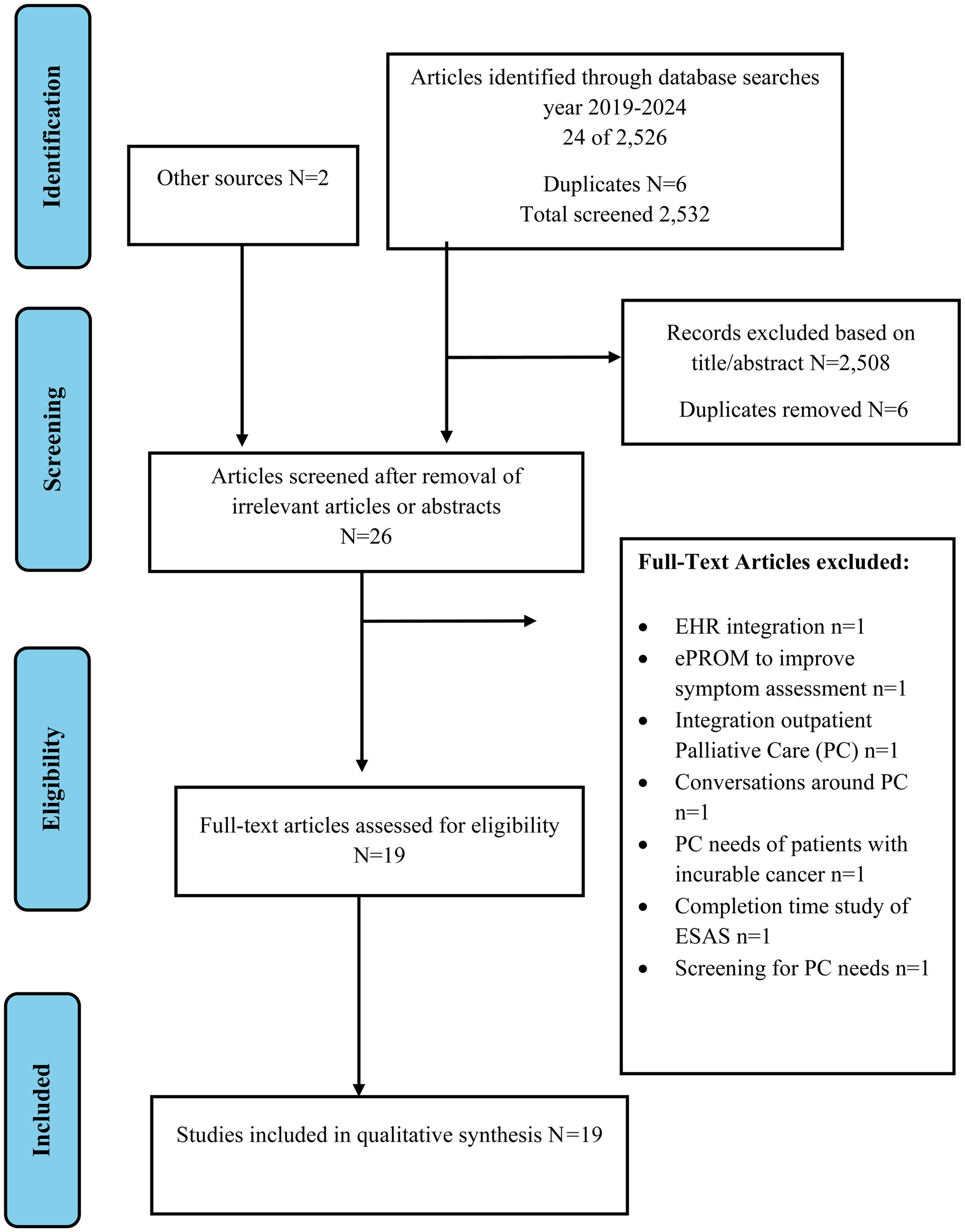
PRISM flow diagram chart of identification and selection of articles.
Summary of Selected Instruments to Measure Symptom Burden.

Note: ESAS = Edmonton Symptom Assessment Scale; ESAS-r = Edmonton Symptom Assessment Scale-revised; POS = Palliative Care Outcome Scale; MSAS = Memorial Symptom Assessment Scale; ESAS-SF = Edmonton Symptom Assessment Scale-Short Form.
Findings
The symptom assessment scales will help monitor the outcome of nondisclosure on symptoms of distress, and direct treatment over time in addition to improving the patients’ quality of life. Subsequently, standardized patient-related outcomes measures (PROM) are essential in oncology. There is evidence to show improved symptom management, quality of life, and a decrease in hospitalizations and readmissions (Basch et al., 2022; Di Maio et al., 2022; Müller et al., 2024).
The following five scales were reviewed: the Edmonton Symptom Assessment Scale (ESAS), the Edmonton Symptom Assessment Scale-Revised (ESAS-r), the Memorial Symptom Assessment Scale (MSAS), the Memorial Symptom Assessment Scale Short Form (MSAS-SF), and the Palliative Care Outcome Scale (POS).
The Edmonton Symptom Assessment Scale (ESAS)
The Edmonton Symptom Assessment Scale (ESAS) was developed in 1991 and is a self-reported scale of 10 physical and psychological symptoms ranging in scores from 0, indicating the absence of a symptom, to 10 indicating the worst possible symptom score. ESAS, in its’ earlier versions, was an 8-item scale which eventually progressed to a 9-item scale to include “a less frequent symptom that might be important for the given patient” (Hui & Bruera, 2017). However, given that earlier tools took longer time to administer, it was felt that a simpler tool was needed (Bruera et al., 1991). Examples of the items used in ESAS are pain, nausea, depression, anxiety, activity, drowsiness, well-being, and appetite. The ESAS is based on an open prospective study of 101 patients in the inpatient palliative unit (IPU). The ESAS helped with patient reports on symptom intensity. In the initial study, validity and reliability were not mentioned and only the mean and standard deviation were available. Additional studies in 1993 showed test–retest reliability. Moreover, a follow-up study on 249 participants compared the ESAS total “distress” score to the Memorial Symptom Assess (MSAS), Karnofsky scale (KPS), and the FACT scale (Functional assessment of cancer treatment). The ESAS demonstrated a Cronbach's alpha of .79, indicating good reliability, where values above .70 are generally considered acceptable. This value suggests that the items on the ESAS consistently measure the underlying construct of symptom severity (Chang et al., 2000b). The ESAS items were found to be valid when compared to MSAS and FACT. The Spearman's correlations were .86 (p < .001) at 2 days and .45 (p < .05) at 1 week. The ESAS items correlated well on day 2 but were found to be only true for pain, shortness of breath, depression, and ESAS distress at week 1. Similarly, the VAS single item score on day 1 correlated significantly at .43 with p < .04 but not a week 1. It was concluded that the ESAS had good test–retest reliability on day 1 but was smaller at week 1, which was attributed to being secondary to the patient's symptoms, which may vacillate, or secondary to the perception of symptoms, which may change. This indicates that the test–retest has good reliability over the items in the short term. The ESAS also exhibited limited test-retest for psychological symptoms such as depression, anxiety, and well-being as compared to the remainder of the six items for physical symptoms. Overall, this indicates that physical symptoms demonstrated greater reliability when compared to psychological symptoms and while the ESAS is a reliable tool for assessing physical symptoms, its reliability for psychological symptoms may be limited.
Similarly, the Arabic version of the ESAS (ESAS-A) followed a backward translation protocol with an overall Cronbach's α of .84 exhibiting good consistency with good reliability and validity in patients with advanced cancer in Jordan (Shamieh et al., 2023). This is consistent with previous studies with a range of .71–.88. The ESAS-A correlated well with the European Organization for Research and Treatment of Cancer quality of life questionnaire for palliative patients (EORTC QLQ-15 PAL) tool in addition to the Hospital Anxiety and Depression Scale (HADS). However, the study raises the possibility of selection bias and does not look into test–retest reliability.
The French Version of ESAS followed the backward and forward translation protocol in accordance with the EORTC (Tricou et al., 2019). Here 45 patients were studied, all equally distributed among inpatient, outpatient, and the palliative care unit (PCU). In terms of content validity, 89% found it easy to understand and 98% felt the instructions were clear with a mean administrative time of 10 min and good overall face validity. However, some items like sleep and appetite required re-reading, indicating areas for further refinement.
The Edmonton Symptom Assessment Scale-Revised (ESAS-r)
The ESAS-revised was developed in 2011 as a part of a prospective multicenter study of n = 160 patients with advanced cancer and revised 9 core symptoms (Noel et al., 2021). It is an 11-item rating system and a multidimensional scale that was developed to replace the original ESAS. It was developed to help patients with improved understanding and clarity of symptoms being assessed (Watanabe et al., 2011; Watanabe et al., 2009). It allows patients to report self-symptoms of pain, nausea, tiredness, anxiety, depression, drowsiness, well-being, appetite, and shortness of breath in addition to adding a “patient-specific” symptom. (Henson et al., 2020). Each symptom is rated on a 0 to 10 scale similar to the original ESAS. The ESAS-r has a transcultural adaptation similar to the ESAS. In one study, ESAS and ESAS-r were compared and it was determined that the ESAS-r was easier to comprehend with a p = .008 (Watanabe et al., 2011). Overall, the interclass coefficient (ICC) was strong for pain, tiredness, and depression at .83 while for appetite, well-being, and drowsiness it ranges from .65 to .79 which is a moderate ICC. These findings imply that while the ESAS-r is generally reliable, some symptoms are more consistently reported than others, but future prospective validation studies are needed for well-being, appetite, and drowsiness (Watanabe et al., 2011).
A study involving 140 patients with advanced cancer in Egypt, with 11 patients declining participation, found that the ESAS-r had an inter-rater reliability of 83%. The physician coefficient was .877, and the patient coefficient was .863, indicating strong agreement between different raters. The study also found that the ESAS-r had good internal consistency, with a Cronbach's α of .88 in the Arabic version (Salem et al., 2016). Additionally, the ESAS-r has been validated in Arabic in Egypt for patients with advanced cancer, further supporting its reliability and utility in diverse patient populations (Hussenein et al., 2017). The ESAS-r's ability to adapt to different cultural contexts while maintaining reliability and validity highlights its versatility as a symptom assessment tool. This adaptability ensures that it can be used effectively in various healthcare settings, providing accurate and comprehensive symptom assessments for patients with advanced cancer and other serious illnesses. The ongoing validation and refinement of the ESAS-r continues to enhance its role in improving patient care and symptom management.
The Memorial Symptom Assessment Scale (MSAS)
The Memorial Symptom Assessment Scale (MSAS) is a comprehensive 33-item tool designed to measure the severity, frequency, and distress of physical and psychological symptoms, significantly impacting the quality of life. Developed through a study involving 246 cancer patients, the MSAS was created as an improvement over the Rotterdam Symptom Checklist (RSC), which did not fully address the severity or frequency of symptoms alongside symptom distress (Portenoy et al., 1994). This tool was utilized among inpatients and outpatients with prostate, colon, breast, or ovarian cancer. The Likert-type scale was created for each dimension and was studied over a 14-month period from 1990 to 1991 at Memorial Sloane-Kettering Cancer Center. Those that were excluded from the studies were those with encephalopathy or severe psychiatric disease. The scale has demonstrated high correlations with quality of life (QoL) and clinical status, thus supporting its validity. Internal consistency was notably high in the physical high symptom and psychological groups (Cronbach's α coefficients of .88 and .83, respectively) and moderate in the physical low symptom group (α = .58). The inpatient setting had the highest symptoms. It was determined there is a high correlation between the clinical status and quality of life measures that support the validity of MSAS in addition to a high internal consistency (Portenoy et al., 1994).
Further validation of the MSAS was conducted at the University of Beirut in Lebanon with 200 adult cancer patients at a tertiary center. The MSAS was translated into Arabic (MSAS-Leb) using the backtranslation method. The convergent validity for the psychological subscale and the global distress index (GDI) demonstrated a moderate correlation (r > −.50, p < .01). The correlation coefficient between the EORTC QLQ C30 and MSAS ranged from −.55 to .81 (p < .01), except for the subscale of nausea and vomiting. The MSAS-Leb exhibited satisfactory internal consistency, with Cronbach's α values between .71 and .83. However, limitations of this study included a small sample size and susceptibility to recall bias (Abu-Saad Huijer et al., 2015). The original MSAS study recommended future studies to ensure reliability and validity through repeated testing. Supporting this recommendation, a study in Brazil showed consistent responses over time, further establishing the tool's reliability (Menezes et al., 2017). The administration time for the MSAS scale is under 20–30 min, making it a practical tool for clinical settings.
The MSAS showed a strong correlation with quality of life (QoL) and clinical status, suggesting that higher symptom severity, frequency, and distress measured by the MSAS are associated with lower QoL and worse clinical status. High internal consistency, especially in the physical high-symptom and psychological groups, indicates that the items within these subscales reliably measure the same construct. The validation study in Lebanon demonstrated moderate convergent validity and satisfactory internal consistency, further supporting the MSAS's reliability.
Despite the small sample size and recall bias in the Lebanon study, the overall findings from various studies underscore the MSAS's utility in assessing symptom severity, frequency, and distress in cancer patients. The consistency over time observed in the Brazilian study adds to the evidence supporting the MSAS's reliability. Thus, the MSAS proves to be a comprehensive and reliable tool that significantly correlates with quality of life and clinical status, with practical application in both inpatient and outpatient settings.
The Memorial Symptom Assessment Scale-Short Form (MSAS-SF)
The Memorial Symptom Assessment Scale-Short Form (MSAS-SF) is a concise tool that measures 32 symptoms and consists of three subscales: psychological, global distress index, and physical subscale (Chang et al., 2000). Developed through a study involving 299 elderly cancer patients, the MSAS-SF comprises about one-third of the questions in the original MSAS, reducing the administration time to under 5 min. The MSAS-SF has shown strong validity through high correlation coefficients for physical symptoms (−.74, p < .001), psychological symptoms (−.64, p < .001), and global distress index (−.71, p < .001). It demonstrated convergent validity with the severity of disease, functional status, and inpatient status. The scale's reliability is supported by Cronbach's α for physical symptoms (.82), psychological symptoms (.76), and global distress index (.80), with test–retest correlation coefficients ranging from .86 to .94 at 1 day and from .40 to .84 at 1 week.
The MSAS-SF has been utilized for patients with primary cancer sites, including head and neck, lung, gastrointestinal, genitourinary, hematologic, and other cancers (Chang et al., 2000a). To determine the criterion validity of the MSAS-SF, it was compared with the Functional Assessment of Cancer Therapy (FACT-G), Karnofsky Performance Scale (KPS), and extent of disease (EOD) at the section of hematology and oncology. The MSAS-SF demonstrated convergent validity and high test-retest reliability on day 1, along with Cronbach's α values ranging from .82 for physical symptoms to .76 for psychological symptoms and .80 for the global distress index (GDI). This indicates a high level of consistency between the items measuring GDI and physical symptoms. Between the MSAS-SF subscales and FACT-G, the correlation coefficient was nearly identical, except for social/family well-being (SFWB), which showed either nonsignificant or weak correlation when compared to the MSAS-SF. This finding suggests that while the MSAS-SF is robust in measuring most aspects of symptom distress, it may have limitations in assessing social and family well-being as compared to the FACT-G.
Palliative Care Outcome Scale (POS)
This tool was developed in 1999 to determine the palliative care (PC) needs of advanced cancer patients’ quality of life and symptoms by the Palliative Care Core Audit Project Advisory Group in the United Kingdom as a simpler tool was needed in Scotland and the UK (Hearn & Higginson, 1999; Henson et al., 2020). The POS has 10 items of which physical symptoms are n = 2, social needs are n = 4 and spiritual needs are n = 1 with each item being scored using the Likert scale from 0 to 4 with an administrative time of under 10 min. In addition, there is a free text section that includes “if any, what have been your main problems in the last three days?” (Hearn & Higginson, 1999). 450 patients were recruited across both inpatient and outpatient centers with diagnoses of cancer and noncancer diagnoses such as neurological disease, respiratory, liver, cardiac, or renal failure. Only one-third completed the questionnaire. The measures used were the POS, EORTC QLQ-C30, and the Support Team Assessment Schedule (STAS). 29 patients completed the EORTC QLQ-C30 and POS and 43 patients completed STAS and POS. The POS is a 10-item scale that measures psychological, emotional, physical, and spiritual domains with a Spearman rho of .43–.80 indicating that the correlation ranges from moderate to strong indicating validity and a good correlation between the items and the underlying constructs they aim to measure. Reliability was acceptable for seven of the 10 items, with good internal consistency demonstrated by Cronbach's α values of .65 for patient-reported outcomes and .70 for staff-reported outcomes. These psychometric properties suggest that the POS is a reliable and valid tool for assessing the multidimensional needs of palliative care patients.
To ensure its applicability in different cultural contexts, the POS was translated into Arabic using a rigorous forward and backward translation process. A study conducted at the National Centre for Cancer Care and Research (NCCCR) in Doha, Qatar, involved a small sample size of five cancer patients and demonstrated “good overall comprehension” of the POS tool among both healthcare providers and patients (Al Bulushi et al., 2023). Although this study affirmed the tool's validity and reliability, it did not report Cronbach's alpha or inter-item reliability, with psychometric analysis delayed for future studies involving larger patient samples.
Despite its increased usage for symptom assessment, a systematic review identified barriers to POS implementation, including time constraints, lack of resources, and insufficient training, alongside patient-related barriers. However, the review also confirmed that the POS is a reliable tool with concurrent validity (Collins et al., 2015). These findings collectively suggest that the POS is an effective instrument for assessing the multidimensional needs of palliative care patients, though challenges remain in its practical application.
Overall Findings and Comparative Perspective on Assessment Tools
The review of the ESAS, ESAS-r, MSAS, MSAS-SF, and POS reveals their strengths and limitations. The ESAS and ESAS-r are widely used for their simplicity and quick administration, making them ideal for busy clinical environments. However, they may not fully capture psychological well-being, a critical factor in nondisclosure cases. The MSAS, with its comprehensive assessment of physical and psychological symptoms, is most suitable for understanding the impact of nondisclosure but may be burdensome due to its longer administration time. The POS offers a balanced assessment of multidimensional needs but may lack the specificity required for detailed symptom analysis.
The applicability of symptom assessment tools varies globally, influenced by cultural, linguistic, and healthcare system differences (Collins et al., 2015; Hui & Bruera, 2017). While tools like the ESAS and POS have been adapted for use in different languages and cultural settings, including Arabic, their use and acceptance may differ (Al Bulushi et al., 2023; Shamieh et al., 2023). For example, the Arabic version of the ESAS (ESAS-A) has demonstrated good reliability and validity in Jordanian patients, though potential biases have been noted, emphasizing the need for further research on test-retest reliability (Shamieh et al., 2023). Similarly, the POS has been translated and validated in various contexts, showcasing its adaptability across cultural and clinical settings (Collins et al., 2015; Hearn & Higginson, 1999). However, challenges such as time constraints, limited resources, and training needs can impact the practical application of these tools in clinical practice (Collins et al., 2015). Understanding these differences helps provide a global perspective and a comprehensive overview of the tools’ applicability in diverse settings (Henson et al., 2020).
Discussion
Assessing symptom burden in oncology is crucial for measuring patient-related outcomes (Basch et al., 2022; Di Maio et al., 2022; Müller et al., 2024). Utilizing symptom screening tools helps identify the impact and outcome of nondisclosure on end-of-life care. This affects various outcomes that need further assessment using symptom burden instruments. Patients’ families often have concerns about non-disclosure and the potential negative influence of medical information on diagnosis and prognosis (Abdel-hafeez et al., 2022). Outcomes requiring further assessment include timely palliative care delivery, effective symptom management, patient and family well-being, and transition to hospice. Tools such as the Edmonton Symptom Assessment System (ESAS), ESAS-Revised (ESAS-r), Memorial Symptom Assessment Scale (MSAS), MSAS Short Form (MSAS-SF), and Palliative Care Outcome Scale (POS) are effective in identifying complex symptoms in oncology. Each instrument has its strengths and limitations, which will now be summarized. Among these, the ESAS is one of the most commonly used instruments globally (Hui & Bruera, 2017).
The Edmonton Symptom Assessment System (ESAS) stands out for its widespread use in assessing symptom burden in palliative care patients, aligning well with addressing the impact of nondisclosure on symptom burden and management. Its simplicity and quick administration time make it ideal for busy clinical settings. The ESAS has shown good validity and reliability across various patient populations, including those with advanced cancer (Bush et al., 2010; Miyasaki et al., 2012; Murray et al., 2012; Noel et al., 2021; Richardson & Jones, 2009; Watson et al., 2024). Its sensitivity to changes in symptom severity over time aids in monitoring the impact of nondisclosure on symptom burden (Asano et al., 2022; Donlan et al., 2024; Hui & Bruera, 2017; Rantala et al., 2022). However, its narrow focus on physical symptoms and less emphasis on psychological well-being could limit its effectiveness in assessing the full impact of non-disclosure on psychological outcomes like anxiety and depression (Hui & Bruera, 2017). Moreover, anxiety and depression items on the ESAS have been shown to have a strong correlation with a Spearman's of .707 with >3 score items (Ripamonti et al., 2014). Furthermore, based on ESMO (European Society for Medical Oncology) guidelines states that the ESAS can be utilized as a short screening tool as the “first stage” to identify those with depression or anxiety (Grassi et al., 2023). Despite these strengths, the multiple versions of the ESAS might complicate comparisons across studies, and some items might be challenging for patients to interpret, potentially affecting assessment.
The Edmonton Symptom Assessment Scale-Revised (ESAS-r) is multidimensional and has a completion time of under 1 min with seven physical symptoms, one well-being, and two emotional symptoms (Hui & Bruera, 2017; Wong et al., 2021). Well-being is also impacted by symptoms of tiredness and other co-existing symptoms (McKenna et al., 2023). The ESAS-r's ability to adapt to different cultural contexts while maintaining reliability and validity highlights its versatility as a symptom assessment tool. This adaptability ensures that it can be used effectively in various healthcare settings, providing accurate and comprehensive symptom assessments for patients with advanced cancer and other serious illnesses (Bush et al., 2010; Miyasaki et al., 2012; Murray et al., 2012; Noel et al., 2021; Watson et al., 2024). Furthermore, a recent study showed that ESAS-r Cancer which recognizes symptoms assessed by the National Comprehensive Cancer Network (NCCN) and ESAS-r showed a strong correlation with MSAS-SF (Watson et al., 2024). It was concluded that the ESAS-r Cancer has a Cronbach's α .90 and Spearman-Brown of .891 (Watson et al., 2024). However, the ESAS-r Cancer does not have the free text option which can make translation of the data difficult. Additionally, the ESAS-r has been validated in French, German, and Korean (Hui & Bruera, 2017). The ongoing validation and refinement of the ESAS-r continues to enhance its role in improving patient care and symptom management.
In contrast, the Memorial Symptom Assessment Scale (MSAS) provides a comprehensive evaluation, in both the inpatient and outpatient setting in both cancer and non-cancer diagnosis, of physical and psychological symptoms, making it particularly suitable for assessing symptom burden and psychological well-being, key outcomes of the nondisclosure (Bower et al., 2020; Chang et al., 2004; Jablonski et al., 2007; Li et al., 2024; Portenoy et al., 1994). The MSAS has strong validity and reliability, with high internal consistency for physical and psychological symptom subscales, offering detailed symptom profiles that can give in-depth insights into how nondisclosure affects patients (Portenoy, 1994). However, its length, requiring 20–30 min to complete, can be burdensome for patients with advanced cancer, leading to incomplete or rushed responses. This longer administration time also reduces its practicality for routine clinical use compared to shorter instruments like the ESAS, ESAS-r, or the MSAS-SF. Additionally, the MSAS's complexity may challenge consistent and accurate patient responses, especially in diverse cultural contexts. Furthermore, answering the MSAS screening question on “problems with sexual interest or activity” may be challenging for Muslim patients given their religious views, modesty, and conservative nature (Alkhaldy et al., 2023; Madni et al., 2022; Portenoy, 1994).
The MSAS-SF is based on expert opinion and empirical research. The MSAS-SF is an abbreviated version of the MSAS and retains the advantages of the original scale while reducing administrative time to one-third. However, it has limitations in assessing social and family well-being compared to the FACT-G (Chang et al., 2000). The same challenges regarding the sexual activity question apply to the MSAS-SF (Lobchuk, 2003).
The Palliative Care Outcome Scale (POS) is designed to measure a broad range of needs, including psychological, emotional, physical, and spiritual dimensions, making it well suited for assessing the comprehensive impact of nondisclosure on patients’ well-being. Its brief administration time and ease of use enhance its feasibility in clinical settings. In addition, it has a self (patient) and proxy (healthcare provider) version that incorporates the Eastern Cooperative Oncology Group (ECOG) (Rugno & Carlo, 2016). The POS has demonstrated good validity and reliability and its versatility in both cancer and non-cancer diagnoses such as COPD, heart failure, and neurological conditions, along with its adaptation in multiple languages, supports its applicability in diverse cultural contexts (Hearn & Higginson, 1999; Pelayo-Alvarez et al., 2013; Rugno & Carlo, 2016; Saleem et al., 2013). However, while the POS is effective in measuring a wide range of patient needs, it may not offer the same level of detail as more specialized instruments like the MSAS in assessing specific symptom frequency and distress. The tool's broad focus might dilute the precision of assessing specific outcomes, such as the impact of non-disclosure on psychological well-being. Barriers to implementation, such as time constraints and resource limitations, need to be addressed to maximize the POS's clinical utility (Collins et al., 2015). The review, however, affirmed the POS's reliability and concurrent validity, reinforcing its value in clinical practice. Addressing these barriers through improved training, resource allocation, and patient engagement strategies could enhance the utilization and impact of the POS in palliative care settings. The Palliative Care Outcome Scale (POS) is a valuable tool for assessing the complex needs of palliative care patients.
Conclusion
Identifying the most suitable symptom assessment tools for evaluating symptom burden related to nondisclosure in advanced cancer patients poses significant challenges. The findings of this review indicate that while instruments such as the Memorial Symptom Assessment Scale (MSAS) provide comprehensive assessments, they require adaptation and validation for broader cultural and clinical contexts to maximize their utility.
The MSAS stands out as a robust tool, capable of assessing both physical and psychological symptoms with high validity and reliability. Its ability to evaluate symptom severity, frequency, and distress makes it especially valuable for understanding how nondisclosure impacts patient outcomes, including psychological well-being. Despite its average completion time of 20 min, which may be seen as a drawback in certain clinical settings, the detailed symptom profiles it offers justify the time investment. The feasibility of its use has been supported by studies involving caregivers, demonstrating that thorough and accurate assessments can be achieved with adequate support and training (Lobchuk, 2003). This highlights the importance of equipping healthcare providers with the necessary training to ensure consistent and accurate application in diverse settings.
While other tools, such as the Edmonton Symptom Assessment Scale-Revised (ESAS-r) and the Palliative Care Outcome Scale (POS), offer valuable assessments for both cancer and noncancer diagnoses, they exhibit limitations in capturing the comprehensive picture needed for nondisclosure cases. The ESAS-r, with its practicality and ease of administration, focuses mainly on physical symptoms and provides less insight into psychological well-being. This aspect is particularly critical when assessing the holistic impact of nondisclosure on patients. Further research is warranted to enhance the construct validity of well-being and depression measures within the ESAS-r (Hui & Bruera, 2017). The POS, known for its versatility in assessing multidimensional needs, faces challenges in consistent implementation and may lack the specificity required for detailed symptom analysis.
Future research should prioritize cross-cultural validation of these tools to ensure their applicability in various healthcare settings. This includes adapting the MSAS and other comprehensive instruments to be culturally sensitive while maintaining their psychometric integrity. Simplified versions that retain essential assessment capabilities could enhance their use in diverse clinical environments, making symptom assessment more accessible and less burdensome for patients and healthcare providers.
Moreover, additional studies are needed to evaluate the specific impact of nondisclosure on patient outcomes across different diagnoses and settings. Such research should encompass both cancer and non-cancer conditions and consider the unique cultural contexts of Muslim and non-Muslim populations. This will contribute to a deeper understanding of how nondisclosure influences symptom burden, management, and overall patient well-being.
Expanding the research scope to include longitudinal studies could provide insights into the long-term effects of nondisclosure on patient outcomes. Additionally, incorporating qualitative research methods may help capture the nuanced experiences of patients and families, shedding light on how nondisclosure affects the psychological, emotional, and social dimensions of their care. Collaborative efforts between researchers, healthcare providers, and policymakers will be essential for developing tailored strategies that address the complexities of nondisclosure in palliative care, ultimately improving patient-centered outcomes.
In summary, the MSAS remains the optimal tool for evaluating symptom burden related to nondisclosure due to its comprehensive nature and proven psychometric properties. However, ongoing validation, adaptation, and exploration of innovative methods to simplify its administration are crucial for broadening its applicability. Such advancements will pave the way for improved care practices that cater to the diverse needs of patients facing nondisclosure, enhancing both their quality of care and quality of life.
Footnotes
AI or AI-Assisted Technologies
The author did not use AI or AI-assisted technologies in the writing process.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Reported as not applicable.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Other Disclosures
None reported.
