Abstract
Androgen deprivation therapy (ADT) is a hormone treatment for prostate cancer and is linked to altered cognition, including memory changes, which can have detrimental effects on quality of life (QOL). The aim of this research was to develop an intervention to support men with memory changes associated with ADT. The Intervention Mapping framework guided intervention development through a series of rigorous steps. A self-management booklet called MEMORY MANager was developed, which incorporated education, self-assessment, compensatory strategies and techniques to improve mood. It was found to be an acceptable means of supporting men. This booklet could be valuable to healthcare professionals and patients as a means of improving QOL among men who undergo ADT. This is the first reported self-management intervention developed for managing cognitive changes in men who undergo ADT. Although preliminary, findings appear promising. More research is needed to evaluate effectiveness of the intervention and its implementation.
Background
Cancer will affect approximately half the UK population during their lifetime (Ahmad et al., 2015). Earlier diagnosis and more effective treatments have paved the way for a growing population of ‘cancer survivors’ (Khan et al., 2012). However, delayed disease progression and prolonged survival are achieved at a cost. This cost includes side-effects, which can substantially undermine patient’s quality of life (QOL). One often poorly understood side-effect of cancer treatment is changes to cognitive function, typically associated with systemic (non-local) treatments, such as chemotherapy (Von Ah et al., 2013). A vast body of research has focused on the deleterious effects of chemotherapy on cancer patients, particularly women diagnosed with breast cancer (‘chemobrain’ or ‘chemofog’) (Raffa, 2010). Yet other systemic cancer treatments may also have detrimental effects on cognitive function e.g. hormone treatments (Nelson et al., 2008).
Androgen deprivation therapy (ADT) is a hormone treatment for prostate cancer, the most prevalent cancer diagnosed among men in the UK, with over 48,000 new cases every year (Cancer Research UK, 2020a, 2020b). Approximately 50% of patients with prostate cancer undergo hormone treatment (Nguyen et al., 2018). The treatment deprives the body of male hormones, which supresses growth of hormone-responsive prostate cancer cells (Huggins, 1967). Hormone treatment has been the cornerstone of managing advanced prostate cancer (Schröder et al., 2012), but is linked to numerous side-effects, such as cognitive changes. Up to 69% of patients who undergo this treatment, demonstrate a decline in one or more domains of cognitive function, including visuospatial abilities, executive function and memory (Gunlusoy et al., 2017; Nelson et al., 2008; Yang, Zhong, Qiu, & Wang, 2015; Yang, Zhong, Qiu, Cheng, et al., 2015). The cognitive changes faced by patients who undergo hormone treatment for prostate cancer, can adversely impact their lives (Grunfeld et al., 2012; Wu et al., 2013, 2016). Effects have been reported on daily activities (e.g. losing their way in familiar places), occupational functioning (e.g. difficulty completing tasks on time), social interactions (e.g. struggling to maintain focus on others) and emotional wellbeing (e.g. feeling distressed by cognitive changes) (Grunfeld et al., 2012; Wu et al., 2013, 2016). The impact on emotional wellbeing is particularly noteworthy since levels of anxiety and depression are often elevated in individuals diagnosed with cancer (Ando et al., 2006). Furthermore, improving the cancer survivorship experience is a priority area for the Department of Health and Social Care (2013).
Although interventions have been developed for cancer-treatment related cognitive changes (Chan et al., 2015), to date there are no effective interventions or guidelines for cognitive changes experienced by patients who undergo hormone treatment for prostate cancer (National Institute for health and Care Excellence, 2019; Nguyen et al., 2015). We aimed to develop an intervention to address cognitive changes, notably memory changes in this population of patients. Memory was the target domain, due to its importance in daily living (e.g. learning, planning and responding adaptively) (Badderly, 2014), the impact of memory impairment (e.g. on occupational and social functioning, leisure activities and self-perceptions) (Parikh et al., 2016) and the increasing salience of memory issues with age (e.g. age-related memory decline and Alzheimer’s disease) (Smits et al., 2015). Intervention development was guided by Intervention Mapping (Bartholomew et al., 1998, 2016), an established framework for developing interventions to improve health outcomes. To our knowledge this is the first reported application of Intervention Mapping to address cognitive changes experienced by cancer patients. We outline steps taken during intervention development and results from each step.
Methods
Intervention Mapping (Bartholomew et al., 2016) is concerned with developing individual-level interventions. The intervention development phase is detailed and provides rigor. It also incorporates theory, evidence and an assessment of the target population’s needs. Intervention Mapping encompasses six steps: (i) developing a model of the problem, (ii) defining intervention outcomes and objectives and developing a model of change, (iii) designing the intervention, (iv) producing the intervention, (v) forming an implementation plan and (vi) creating an evaluation plan. The first four steps guided the development of an intervention for men with memory changes associated with hormone treatment for prostate cancer.
Step One: Developing a Model of the Problem
In step one, previous relevant research was located to elucidate the problem and impact of memory changes for men who undergo hormone treatment for prostate cancer. This was supplemented with primary empirical research to generate further understanding of the support needs of men who undergo hormone treatment for prostate cancer. For this we carried out 17 qualitative, semi-structured interviews with men who had been prescribed hormone treatment (mean time on treatment: 46 months), to explore their behaviors, determinants of behavior (e.g., beliefs) and support needs. Interviews were audio-recorded, transcribed verbatim and then analysed using Framework Analysis (Ritchie & Spencer, 1994; Ritchie et al., 2003).
The specific behaviors and determinants linked to the impact of memory changes on patient’s QOL were identified. Theoretical concepts were also integrated, and the outcome of the intervention was determined. This resulted in the development of a model of the problem.
Step Two: Defining Intervention Outcomes and Objectives and Developing a Model of Change
In step two, determinants and behaviors linked to the impact of memory changes on QOL were selected based on whether they could be modified in an intervention to address the impact of memory changes on QOL. Outcomes and objectives of the intervention were defined. A model of change was developed.
Step Three: Designing the Intervention
In step three, interventions developed for the same or similar problem were reviewed. It was anticipated that there would be insufficient, if any, studies reporting interventions addressing memory changes in patients who undergo hormone treatment for prostate cancer (Von Ah et al., 2013). As a result, we searched broadly and widely to locate literature evaluating the effects of non-pharmacological (psychosocial) interventions on memory in patients who underwent systemic (non-local) cancer treatment. Nine online databases were searched with relevant keywords and subject headings. From the included studies, relevance of interventions for men who undergo hormone treatment for prostate cancer was ascertained, effectiveness of interventions was evaluated and intervention components were extracted and tabulated. Based on findings of the assessment of support needs in step one and findings from reviewing previous interventions, a novel intervention (a booklet) was designed.
Step Four: Producing the Intervention
In step four, a prototype of the intervention was produced with consideration of the preferred format and duration, as established through consultation with the target audience in step one. The acceptability of intervention content was evaluated with 30 individuals from the target population. A think-aloud procedure was adopted, where participants read through the booklet out loud while expressing their thoughts regarding the booklet (Charters, 2003). This was followed by an unstructured interview, to gain further detail and clarity around acceptability of the booklet.
A topic guide was developed for unstructured interviews. Areas of interest included components from a theoretical framework of acceptability (Sekhon et al., 2017). In addition, specific aspects of acceptability were considered e.g. suitability of language, appearance, format, etc. Data were analysed using Thematic Analysis to generate themes relating to the general perceptions of acceptability (Braun & Clarke, 2006). Intervention content was then revised based on findings of the acceptability study to produce the final version of the intervention.
Ethical Approval
Ethical approval to conduct primary and secondary research was obtained from Coventry University Ethics committee prior to recruitment of participants. For primary research, this included attaining written informed consent.
Results
Step One: Developing a Model of the Problem
Previous research was reviewed to gain an understanding of memory changes and impact on the QOL of patients who undergo hormone treatment for prostate cancer. Three qualitative studies were located from existing literature (Grunfeld et al., 2012; Wu et al., 2013, 2016). Findings from these studies suggest that memory changes during hormone treatment for prostate cancer, include forgetting names and words (e.g. difficulty completing crossword puzzles), plans, what was read, places (e.g. losing their way) and where things were placed (e.g. losing keys). There was evidence to indicate that cognitive changes, including memory changes affected QOL. There was an impact on daily tasks (e.g. difficulty retaining important information), relationships (e.g. others becoming annoyed when tasks are forgotten), occupational functioning (e.g. working fewer hours and switching jobs) and emotional wellbeing (e.g. frustration, concern about Alzheimer’s disease, lower confidence and embarrassment). Support needs were not reported in these studies, we therefore conducted additional research.
Of the 17 participants we interviewed, 12 reported experiencing memory changes during hormone treatment. A number of support needs for managing memory changes linked to hormone treatment were reported. The most frequently reported support need was information about memory changes being a potential side-effect of hormone treatment. There was also mention of the need to have memory changes assessed, monitored by others (e.g. during follow-up appointments) or self-monitored to enable awareness of the extent of memory issues. Appropriate skills and strategies to reduce forgetfulness (e.g., repeating a name), knowing how others were managing and techniques to improve mood (e.g. exercise) were also considered helpful.
The effects of pharmacological (medical) interventions, which aim to restore cognitive function in cancer patients remain unclear, however non-pharmacological (psychosocial) interventions for cognitive changes in cancer patients appear promising (Chan et al., 2015). Furthermore, the National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines recommend non-pharmacologic interventions for cognitive impairment (Denlinger et al., 2015). Given that hormone treatment is a cancer treatment that may last years (National Cancer Institute, 2019), we aimed to facilitate the self-management of memory changes resulting from hormone treatment, through compensating and adapting to reduce the QOL impact.
From the 17 interviews we conducted with patients who were prescribed hormone treatment, we explored and identified behaviors related to the impact of memory changes on QOL (self-management behaviors). Behaviors were (i) not seeking information regarding memory changes; (ii) not assessing own memory changes; (iii) not compensating for memory changes e.g. writing lists; and (iv) not coping with the emotional impact.
We searched the literature, but no studies investigating psychological determinants/causes of self-management of memory changes linked to hormone treatment for prostate cancer were located. Determinants were identified from the 17 interviews conducted with patients who were prescribed hormone treatment, after reviewing relevant theories and theoretical concepts (Ajzen, 1991; Bandura, 1998; Gibbons et al., 1998; Prochaska & DiClemente, 1982; Weinstein, 1988). Determinants were: level of (i) knowledge (the acquisition of facts, information or skills through experience, learning and education (lack of knowledge about memory changes and support available for memory changes)); (ii) self-awareness (being conscious of personal circumstances, qualities. weaknesses, thoughts, feelings, actions etc. (lack of awareness of own memory issues and need for support)); (iii) outcome expectancies (anticipated consequences of carrying out a behavior, these may be positive or negative (not perceiving or valuing benefits of self-management of memory changes)); (iv) perceived behavioral control (beliefs regarding the difficulty in enacting a behavior with consideration of personal experience, knowledge and skills as well as external barriers and facilitators (a lack of belief in the ability to self-manage memory changes and perceiving barriers)); and (v) willingness (readiness to undertake a behavior (lack of willingness to self-manage or be supported in self-managing memory changes)) (Figure 1).

A model of the problem depicting one possible interpretation of memory changes in patients who undergo hormone treatment for prostate cancer.
The primary goal of the intervention was to improve self-management of memory changes in men who undergo hormone treatment for prostate cancer. This would require changes to determinants of self-management behaviors and in turn changes to specific self-management behaviors. The secondary goal was to reduce self-reported memory complaints and improve mood and QOL.
Step Two: Defining Intervention Outcomes and Objectives and Developing a Model of Change
The expected outcomes for behavior were increased (i) knowledge; (ii) self-awareness; (iii) compensation; (iv) and coping. The behaviors and determinants of these outcomes were identified. The behaviors required for these outcomes were: to (i) read information; (ii) self-assess memory e.g. through answering some questions; (iii) learn and apply methods to compensate; (iv) learn and apply methods to cope with the emotional impact. Determinants required for these outcomes were: (i) knowledge, (ii) self-awareness, (iii) outcome expectancies (perceived benefits), (iv) perceived behavioral control (confidence in own ability and external barriers) and (v) willingness.
Determinants and behaviors were combined in a matrix to identify intervention objectives i.e. what the intervention needs to do (see Table 1 of the supplemental material for the full matrix). Intervention objectives were informed by previous interventions (step three). A model of change was produced to illustrate how the intervention would facilitate self-management of memory changes (Figure 2).
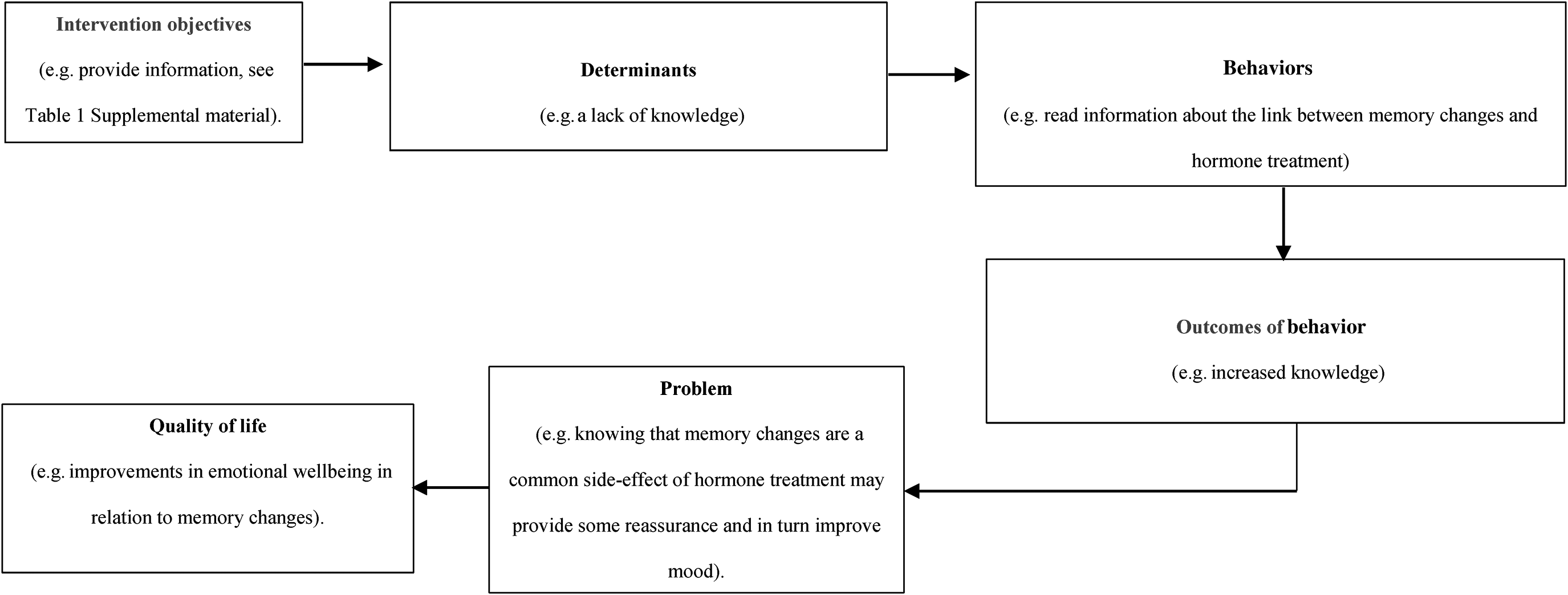
A model of change illustrating how the intervention could help patients manage memory changes linked to hormone treatment for prostate cancer.
Step Three: Designing the Intervention
It was crucial to review the literature to evaluate the effectiveness of previous interventions and to identify theory and evidence-based intervention methods i.e. how intervention objectives would be achieved. A total of 10,580 articles evaluating the effects of non-pharmacological (psychosocial) interventions on memory in patients who underwent systemic (non-local) cancer treatment, were identified from online databases. After removing duplicates, screening titles and abstracts and examining full-texts, 12 articles met inclusion criteria. A further article was located from searching reference lists of relevant reviews.
In total, the articles reported 14 interventions, including Cognitive Rehabilitation (Cherrier et al., 2013; Ercoli et al., 2013; King & Green, 2015; McDougall et al., 2011; Schuurs & Green, 2013; Von Ah et al., 2012), Cognitive Behavioral Therapy (Ferguson et al., 2007, 2012, 2016), Cognitive Training (Damholdt et al., 2016; Kesler et al., 2013; Von Ah et al., 2012), Tai Chi (Reid-Arndt et al., 2012) and Meditation (Milbury et al., 2013). Overall, interventions were found to be effective for improving memory performance and reducing memory complaints, with limited evidence of improvements in mood and QOL.
The findings suggested that efforts to develop non-pharmacological interventions for cancer treatment related cognitive changes may benefit cancer patients with memory changes. Many of the interventions reviewed, aimed to improve management of cognitive changes, including memory changes, as opposed to restoring cognitive function. We identified appropriate intervention methods from these interventions and from theories cited in these studies, theories from the wider self-management literature and health behavior theories (e.g. Austin & Vancouver, 1996; Beck, 2011; Gollwitzer, 1999; Rogers, 1983; Sniehotta et al., 2006). Intervention methods were psychoeducation, self-monitoring, self-assessment, feedback, skill training, stress management, goal setting, planning, commitment, social support and social comparison.
Suitable techniques i.e. practical applications of intervention methods were carefully selected from 137 techniques (Michie et al., 2008). In Michie et al. (2008) all techniques had been independently judged by four experts based on their effectiveness in targeting 11 theoretical constructs linked to behavior change. Recommended techniques for targeting each determinant were selected.
An intervention was designed for men who undergo hormone treatment for prostate cancer to promote changes in the relevant determinants (e.g., beliefs) and behaviors related to self-management of memory changes. Intervention content was informed by an assessment of support needs (step one) as well as previous interventions for cancer treatment related cognitive changes. The intervention had six parts (1) Support for memory changes, (2) Memory changes and hormone treatment, (3) Self-assessment, (4) Stress management, (5) Skill training, and (6) Goal setting and planning. Parts four, five and six focused on techniques, skills and strategies and their application, as information in the earlier parts set the context and provided a rationale for self-managing memory changes. This order was thought to be the most intuitive. The intervention was called ‘support for
Step Four: Producing the Intervention
The preliminary format of the intervention was based on preferences expressed during interviews with patients who underwent hormone treatment (see step one). Most participants articulated a preference for an individual format over a group format. Most participants stated they preferred a booklet over telephone, email, a website, a phone app and Skype. Information printed on paper was considered the most simple, familiar, and convenient option, as it could be accessed any time or place without a laptop/computer, smartphone, internet connection or skills in using these devices. Therefore, a self-guided printed booklet was selected as the format for the intervention.
It was intended that one section would be completed each week. It was expected that each section would take between one and two hours to complete. Duration of the intervention was six weeks, which is in line with previous interventions that were reviewed (range: four weeks (Schuurs & Green, 2013) to four months (Ferguson et al., 2007)).
The booklet was produced in draft form and its acceptability was evaluated with a sample of 30 men. The booklet was considered acceptable based on a seven-component theoretical framework of acceptability (Sekhon et al., 2017). However, some potential changes were suggested by participants. These included changes to language, such as removing technical terms and replacing them with simple, clear and appropriate language (e.g. “social network” was replaced with “social support”). Some content was also modified. For example, towards the end of section one, the task initially required listing the pros and cons of engaging with the booklet. However, this was later altered to identifying pros only, as many participants felt listing cons would deter people from engaging with the booklet (see Table 3 of the supplemental material for a brief outline of the intervention content following evaluation and improvement of acceptability).
Discussion
We aimed to develop an intervention to address memory changes in men who undergo hormone treatment for prostate cancer, using an established framework for intervention development (Bartholomew et al., 2016). We developed a self-management booklet (MEMORY MANager) comprising six sections: (1) Support for memory changes, (2) Memory changes and hormone treatment, (3) Self-assessment, (4) Stress management, (5) Skill training, and (6) Goal setting and planning.
The structure of MEMORY MANager is consistent with that of previous interventions developed for cancer treatment related cognitive changes (four to 48 sessions; 20 to 120 minutes per session, five times a week to monthly, intervention length: four weeks to four months). However, in line with the input of those with lived experience, the current intervention differs from the format of previous interventions, which took place ‘face-to-face’ (e.g. Cherrier et al., 2013; Ercoli et al., 2013) or virtually (e.g. online training (Damholdt et al., 2016; Kesler et al., 2013), videoconference (Ferguson et al., 2016)). It may be that patients with prostate cancer prefer a simpler, more traditional and autonomous format (i.e. a self-management booklet).
MEMORY MANager emphasizes the promotion of a positive mood through specific techniques (e.g. breathing exercises, muscle relaxation, managing unhelpful thoughts, acceptance, focusing on strengths, humor and hobbies). The need to improve emotional wellbeing in cancer patients and the challenge associated with this is well-established (Cousson-Géalie et al., 2011). Providing patients with tools to promote a positive mindset (e.g. focusing on strengths and positive psychology coping styles) has been found to enhance important outcomes, such as self-esteem, resilience, optimism, emotional intelligence, positive emotions, happiness and wellbeing (Cerezo et al., 2014). Interventions, which include acceptance demonstrate improvements in QOL and psychological flexibility and can reduce distress, emotional disturbances, traumatic responses and pain (Fashler et al., 2018).
Previous interventions developed for cancer treatment related cognitive changes in other cancer populations (see Results section, step 3), that included many of the elements incorporated in the current intervention have been found to be effective, which indicates that the current intervention is promising. For example, Ferguson et al. (2007) developed a non-pharmacological (psychosocial) intervention to manage cognitive changes (Memory and Attention Adaptation Training (MAAT)) among patients who had undergone breast cancer treatment. In their pilot study, improvements were noted for self-reported cognitive function, QOL and performance on neuropsychological tests post-treatment and at both follow-ups (two and six months post intervention). Participants also reported high levels of satisfaction with the intervention and rated it as helpful.
However, to date, one non-pharmacological intervention for cognitive changes in patients who undergo hormone treatment for prostate cancer has been reported in the literature (Wu et al., 2018). It was believed that this eight-week, home-based cognitive training programe may alter cortical representations through repetitive practice. However, in a randomized control trial (RCT), there were unexpected effects on memory post-intervention and no effects at eight weeks follow-up. The intervention resulted in temporary declines in visual memory and an absence of temporary improvement in verbal memory. Despite improvements in reaction time, there were no favorable effects on other domains of cognitive function, QOL or self-reports of cognitive function.
Potential limitations of the intervention reported by Wu et al. (2018) or explanations for its lack of beneficial effects on memory, include the aim to restore cognitive function, rather than to help patients adapt and compensate. There are several distinct domains of cognitive function, yet the intervention focused on cognitive function more generally. The computerized exercises centred on visual attention and information processing, with none targeting memory and mood changes. The intervention was not designed specifically for men who undergo hormone treatment for prostate cancer. It was also reliant on technology. However, our assessment of support needs revealed that interventions requiring technology may not be suitable for this population. Indeed, over half the participants in Wu et al. (2018) encountered technical issues (58%). In addition, an hour a day, five days a week may be excessive. Half the participants considered the intervention to be too time-consuming. The intervention was also repetitive and consequently a third of participants felt under-stimulated. The current self-management intervention addresses these potential limitations. It is hoped that this would translate into positive intervention effects.
Implications for Research
The results of primary research reported in this paper contribute to existing knowledge on the cognitive changes associated with hormone treatment for prostate cancer, including support needs and preferences and the determinants and behaviors linked to the impact of memory changes. It adds weight to the importance of developing interventions for cognitive changes in patients who undergo hormone treatment and suggests ways in which patients can be supported, including the determinants and behaviors to target in an intervention and what an intervention ought to encompass. The development of a self-guided booklet addresses a gap in the literature on self-management interventions for cancer treatment related cognitive changes, through a focus on male cancer patients (Von Ah et al., 2013). The evaluation and improvement of acceptability confirms and ensures that the intervention is both relevant and appealing to the target population, optimizing the likelihood of future engagement.
This report of intervention development provides a transparent and detailed outline of an application of the Intervention Mapping framework (Bartholomew et al., 2016). It allows the reader to see step-by-step how the intervention was developed. Researchers planning to adopt Intervention Mapping (Bartholomew et al., 2016) to develop an intervention can utilize this as a guide for how each step of the framework can be operationalized and what components a potential intervention could incorporate.
Implications for Practice
It is not appropriate to formulate recommendations for practice at this stage, as the intervention has yet to be evaluated (e.g. its effectiveness). However, a non-pharmacological (psychosocial) intervention is arguably low risk, when compared to pharmacological (medical) interventions, which can have unpleasant side-effects (Chan et al., 2015) and aspects of the booklet could be integrated into practice.
At present health care professionals may not be aware of memory changes linked to hormone treatment for prostate cancer or how best to support their patients with memory changes. The booklet could facilitate awareness among health care professionals and patients. With the help of this booklet, health care professionals could reassure patients that cognitive changes are a common side-effect of hormone treatment. The booklet could aid health care professionals in supporting patients, by encouraging patients to manage memory changes and address a QOL issue e.g. identify their specific memory issues, ways of overcoming negative feelings and compensating for memory changes. The booklet could assist health care professionals in recognizing and monitoring memory changes in their male cancer patients e.g. by indicating the need to ask about and act on memory issues during appointments.
Limitations
The samples recruited for the assessment of support needs and evaluation of acceptability may not be representative of all patients who undergo hormone treatment for prostate cancer. Most participants were retired, not employed, White, married or with a partner and well educated. All participants were recruited via support groups in the UK. Yet, social support from support groups may help regulate mood, enhance communication skills and promote a sense of belonging (Lehardy & Fowers, 2020), resulting in better adjustment to memory changes and fewer support needs. At present it is unclear if the booklet is as acceptable and/or as appropriate for other groups.
A booklet format has several advantages e.g. low cost, time and effort, with no need for an implementer or travel. However, there are some drawbacks. It can be said that the individual, self-guided booklet format results in an intervention that lacks a ‘person’ or ‘social’ aspect, when compared with other types of intervention, such as face-to-face, group-based interventions facilitated by an implementer (e.g. King & Green, 2015). However, the booklet does contain social aspects (e.g. encouragement to speak with other men at local prostate cancer support groups and request assistance; each section ends with a task that involves speaking with others).
Steps five and six of Intervention Mapping (forming an implementation plan and creating an evaluation plan, respectively) have yet to be completed. These steps are crucial in ensuring that appropriate ways of implementing the intervention are identified beforehand as well as determining the best ways for assessing whether or not the intervention has the intended effects.
Recommendations
A plan for effective implementation is integral i.e. how the intervention would take place in a real-world setting (step five of Intervention Mapping; Bartholomew et al., 2016). This could involve undertaking new research e.g. interviews to explore views regarding implementation with patients and individuals who would be involved in implementation (e.g. health care professionals, organizers of cancer support groups etc.). A small-scale, pilot study e.g. at three hospitals and three support groups, could help determine acceptability of the implementation strategy and its feasibility (Bartholomew et al., 2016; Charters, 2003). Findings could function as feedback for implementation as well as for the intervention.
Although the current intervention was found to be acceptable, its effectiveness in reducing memory complaints and improving mood requires evaluation (step six of Intervention Mapping; Bartholomew et al. 2016). In future research, the booklet could be evaluated in a preliminary pre-post study (e.g. Ercoli et al., 2013), which could inform a larger RCT (e.g. Damholdt et al., 2016). For example, the preliminary study could be used to provide a signal of efficacy and help establish feasibility of conducting a full-scale RCT.
Conclusion
Men who undergo hormone treatment for prostate cancer may experience memory changes, which affect their QOL. This report outlines the development of a booklet to facilitate self-management of memory changes linked to hormone treatment. The intervention developed is the first reported self-management intervention for cognitive changes linked to hormone treatment. It is also the first reported application of Intervention Mapping (Bartholomew et al., 2016) in the development of an intervention to address cancer-treatment related cognitive changes. Use of an established framework optimizes the likelihood that the intervention will be effective and appropriate for the target population. The booklet was considered an acceptable means of being supported with memory changes. It may also be a valuable tool for health care professionals supporting patients who undergo hormone treatment for prostate cancer. Although further work is required to evaluate the intervention, the current research is a critical steppingstone towards addressing cognitive changes associated with cancer treatment among male cancer patients.
Supplemental Material
sj-docx-1-icl-10.1177_10541373221110461 - Supplemental material for Development of a Self-Management Intervention for Memory Changes Linked to Androgen Deprivation Therapy for Prostate Cancer
Supplemental material, sj-docx-1-icl-10.1177_10541373221110461 for Development of a Self-Management Intervention for Memory Changes Linked to Androgen Deprivation Therapy for Prostate Cancer by Marya Mobeen, Elizabeth A. Grunfeld, Katie Newby and Andrew Turner in Illness, Crisis & Loss
Footnotes
Acknowledgments
The authors would like to thank Dr. Carol Percy for her comments on the reporting of intervention development and organizers of prostate cancer support groups for facilitating recruitment of participants.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was carried out as part of a PhD studentship. The author(s) received no financial support for the authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
