Abstract
Background
Spinal cord injury (SCI) disrupts neuronal pathways, alters spinal reflexes, and often leads to spasticity—a condition of increased muscle tone and involuntary contractions that interfere with daily function. Spasticity typically emerges progressively after injury; however, its mechanisms remain poorly understood, particularly regarding why it develops in some individuals but not in others. Increased spinal excitability has been implicated, but prospective evidence remains scarce.
Objective
This pilot, proof-of-concept study explored whether early electrophysiological markers of spinal excitability, specifically the soleus H-reflex and post-activation depression (PAD), could be linked to the subsequent development of spasticity during intensive functional rehabilitation (IFR).
Methods
Seventeen individuals with traumatic SCI were assessed. The Hmax/Mmax ratio and PAD were measured prior to or shortly after IFR admission. At IFR admission and discharge, spasticity (Modified Ashworth Scale and clonus) and motor function (Lower Extremity Motor Score, LEMS) were evaluated. Each lower limb was analyzed separately to account for asymmetry. Correlations between early electrophysiological measures and clinical outcomes were examined.
Results
Spasticity measures tended to increase from IFR admission to discharge, but only LEMS improved significantly, reflecting motor recovery. Several participants did not present spasticity at admission or discharge. A higher Hmax/Mmax ratio and reduced PAD at early IFR were associated with greater clonus at both admission and discharge, whereas no significant associations were found with Ashworth scores. Across injury levels and AIS grades, AIS A participants displayed the lowest Hmax/Mmax ratios and minimal spasticity, while AIS B or D participants with H/M ratios exceeding 0.2 in early IFR developed clonus.
Conclusion
These exploratory findings suggest that early signs of spinal hyperexcitability reflect spasticity development, but it does not show that they precedes spasticity development, which would be required to support its prognostic value. Assessment at earlier timepoints, such as in acute care, would be necessary to validate both the Hmax/Mmax ratio and the PAD as predictive measures of spasticity in SCI. While not clinically generalizable due to the small and heterogeneous sample, this proof-of-concept study highlights the feasibility and potential value of H-reflex as early markers of spasticity. Larger, adequately powered studies stratified by injury level and severity are needed to confirm utility and clinical applicability.
Keywords
Introduction
Traumatic spinal cord injury (SCI) is a severe neurological condition most often caused by falls, motor vehicle collisions, or acts of violence (Chen et al., 2013). Globally, more than 700,000 new cases occur annually (Kumar et al., 2018). Approximately 60% of individuals with traumatic SCI develop spasticity, characterized by increased muscle tone, exaggerated reflexes, and involuntary spasms that impair daily function and quality of life (Andresen et al., 2016; Rekand et al., 2012; Sköld et al., 1999; Vural et al., 2020; Westgren & Levi, 1998).
After SCI, patients initially experience spinal shock, a transient state of flaccid paralysis and areflexia that can last from days to weeks or, in some cases, months (Ko, 2018). As spinal shock resolves, reflexes return gradually, and abnormal increases in muscle tone can emerge, notably during the subacute rehabilitation phase (Kirshblum et al., 2021). The timing and severity of spasticity onset vary considerably between individuals, and predicting who will develop clinically significant spasticity remains a challenge (Pandyan et al., 2005; Sangari et al., 2023; Sheean, 2002).
Mechanistically, spasticity has been linked to heightened excitability of spinal neuronal circuits (Gong et al., 2025). The loss of descending supraspinal inhibition after SCI alters the function of spinal interneurons, notably reducing presynaptic inhibition which can contribute to both hypo- and hyperexcitability (Calancie et al., 1993). Additionally, changes in motoneuron ion channel properties enhance their intrinsic excitability. A key contributor is the emergence of persistent inward currents (PICs) long-lasting depolarizing currents in motoneurons (Heckman et al., 2008; Li et al., 2004; Li & Bennett, 2003). Under normal conditions, PICs amplify synaptic inputs to sustain voluntary muscle contractions. However, following SCI and loss of inhibitory control, PICs become pathologically enhanced, enabling motoneurons to fire repetitively in response to minimal inputs. This leads to hyperreflexia, increased muscle tone (spasticity), and involuntary rhythmic contractions (e.g., clonus; Li et al., 2004). Despite advances in understanding these mechanisms, clinical tools for early identification of individuals at risk of spasticity remain lacking.
Studies suggest that spinal reflex alterations, including changes in post-activation depression (PAD), may underlie the development of spasticity (Grey et al., 2008; J. Nielsen et al., 1995; J. B. Nielsen et al., 2007). PAD can be assessed using neurophysiological measures and is characterized by a decrease in H-reflex amplitude in response to repetitive stimulation at short interstimulus intervals, reflecting a reduced probability of neurotransmitter release (PAD; Crone & Nielsen, 1989). A smaller suppression of the H-reflex amplitude indicative of impaired PAD has been associated with spasticity (Grey et al., 2008; Schindler-Ivens & Shields, 2000), raising the question of whether early alterations in reflex modulation may serve as predictive markers. Therefore, the present study aims to determine whether early assessment of spinal reflexes at admission to a subacute intensive functional rehabilitation (IFR) unit can predict the development of spasticity by discharge from IFR.
The subacute rehabilitation period (in this paper, referred to as IFR) was selected for investigation, as early initiation of rehabilitation has been identified as a key determinant of long-term functional recovery after an injury to the central nervous system (Asano et al., 2022; Burns et al., 2017; Lewis et al., 2022; Norrie et al., 2005). Participants underwent IFR in an in-patient rehabilitation center immediately following stabilization of vital signs in the acute care hospital. IFR is defined as daily sessions of high repetition, task-oriented training targeting essential activities of daily living and mobility. In our rehabilitation center, patients typically arrive from the acute care about 1-month post-lesion, and the average length of stay is approximately 56–62 days for in-patient rehabilitation, providing time for task-specific training and the promotion of activity-dependent neuroplasticity. Patients are then discharged either to their homes, if appropriate, or to another phase of rehabilitation focused on social integration, aimed at supporting their successful return to community roles. Although spasticity is often absent at admission, it frequently develops during this period. Therefore, the ability to predict spasticity early during IFR could facilitate timely interventions and inform the development of personalized therapeutic strategies (Elbasiouny et al., 2010).
Accordingly, this pilot, proof-of-concept study investigated whether spinal excitability assessed by the soleus Hmax/Mmax ratio at IFR admission predicts the emergence of spasticity by IFR discharge, as measured by the Modified Ashworth Scale (MAS) and clonus, and whether these measures correlate with motor recovery, assessed by the Lower Extremity Motor Score (LEMS). A second objective aims at gaining mechanistic insight: in a subset of participants, PAD was also examined to explore its potential predictive value. We hypothesized that increased Hmax/Mmax ratio and reduced PAD early after IFR admission would identify individuals more likely to develop spasticity during the rehabilitation period.
Material and Method
Participants
All SCI patients provided written informed consent prior to participation. This pilot study received ethical approval from the Research Ethics Boards of the Comité d'éthique de la recherche CIUSSS du Nord-de-l'Île-de-Montréal, of the Comité d'éthique de la recherche en réadaptation et en déficience physique du CCSMTL, and was conducted in accordance with the principles of the Declaration of Helsinki.
A total of 21 individuals with traumatic spinal cord injury (SCI) were recruited. An initial feasibility phase included five participants enrolled between May 2015 and December 2016 (Huang et al., 2022). Subsequently, 16 additional participants were recruited for the pilot phase between July 2019 and June 2022. Four participants were excluded after recruitment due to: newly identified comorbidities (n = 2), technical issues preventing early H-reflex data acquisition (n =1), incomplete clinical evaluations upon admission and discharge from IFR (n = 1). In one participant the data from the left leg was excluded due to technical issues preventing H-reflex acquisition, but data could be collected from the right leg. Therefore, data from 17 participants are included.
Clinical and demographic characteristics of the participants are summarized in Table 1. The study included thirteen men and four women. Nine participants were classified as AIS A, two as AIS B, and six as AIS D. One participant was initially assessed as AIS C at admission but converted to AIS D by discharge; his data were pooled with the AIS D group for analysis. Eligible patients were approached either at the end of their acute care hospitalization or upon admission to the IFR unit. Inclusion criteria were: age ≥18 years, hemodynamic stability, capacity to provide informed consent, ability to understand instructions, and absence of significant cognitive impairments. Exclusion criteria included: history of skull fracture at the time of injury, or contraindications to electrical or magnetic stimulation (e.g., uncontrolled hypertension, presence of a pacemaker, metallic fragments, chronic illness, or epilepsy).
Participants’ Clinical and Demographic Data.
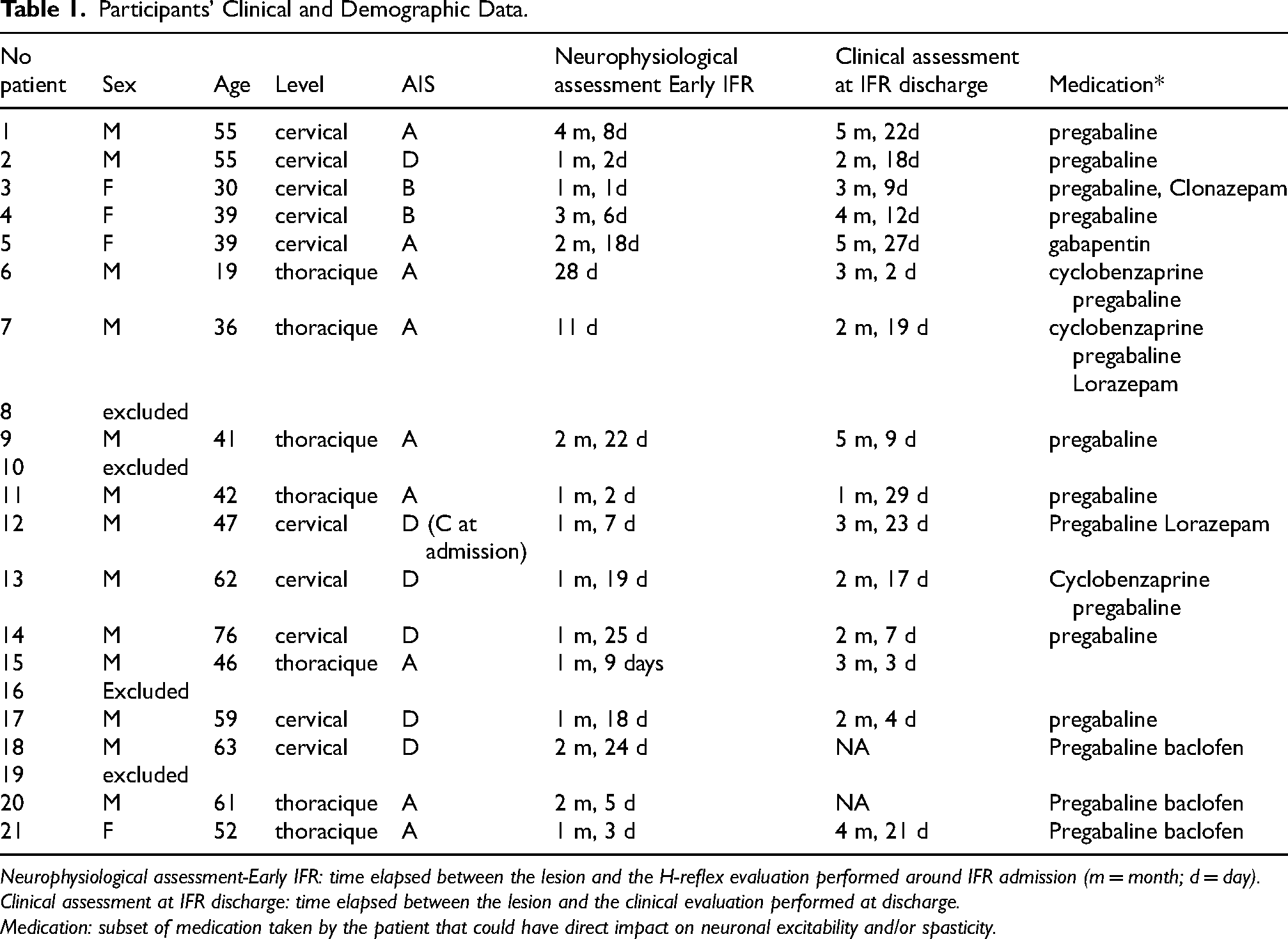
Neurophysiological assessment-Early IFR: time elapsed between the lesion and the H-reflex evaluation performed around IFR admission (m = month; d = day).
Clinical assessment at IFR discharge: time elapsed between the lesion and the clinical evaluation performed at discharge.
Medication: subset of medication taken by the patient that could have direct impact on neuronal excitability and/or spasticity.
Early Electrophysiological Assessments
The H-reflex of the soleus (SOL) muscle was assessed by the research team either prior to or shortly after admission to the IFR center. Electrical activity was recorded bilaterally from the SOL using surface electrodes. The skin was prepared using 3 M Red Dot Trace Prep, and two Ag/AgCl electrodes (AMBU BlueSensor M-00-S) were placed 1.5 cm apart on each muscle according to established guidelines (Hermens et al., 2000). A ground electrode was positioned over the tibial tuberosity. Signals were amplified (×1000; Grass Model 12, West Warwick, RI, USA), filtered using a bandpass of 10–1000 Hz, digitized at a 2 kHz sampling rate, and recorded using Signal software (Cambridge Electronic Design, Cambridge, UK) with a CED Micro 1401 acquisition system.
The tibial nerve was stimulated in the popliteal fossa using a 1-ms monophasic rectangular pulse delivered via a monopolar probe (cathode: 1 cm diameter hemisphere). The anode (8 × 15 cm) was positioned above the knee over the femoral condyles and secured with straps above and below the patella. After identifying the optimal stimulation site by locating the region that elicited the highest H-reflex amplitude with suprathreshold stimulation, a recruitment curve was generated for both the H-reflex and M-wave (Figure 1A).
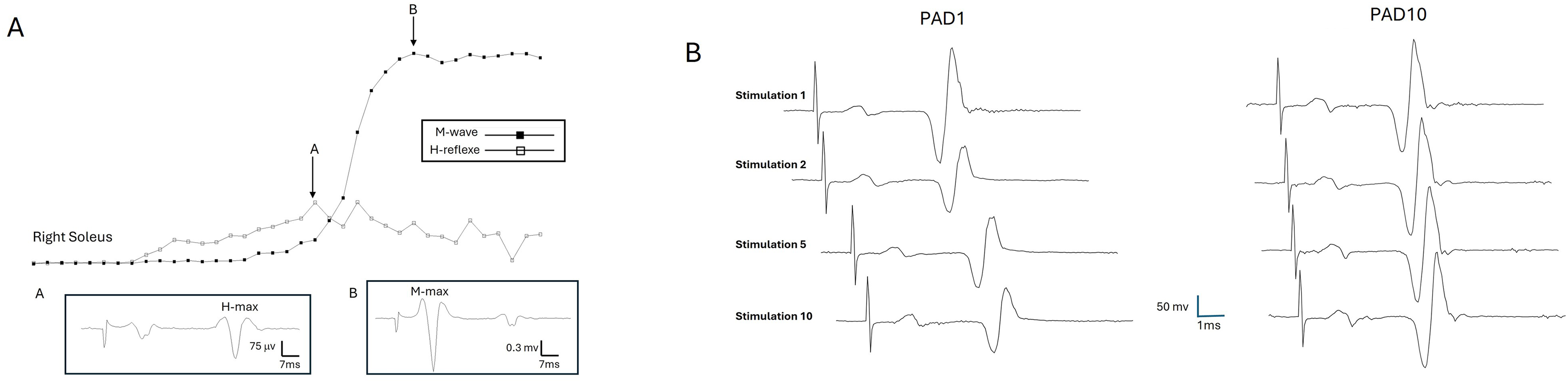
Electrophysiological Methodology. (A) Recruitment Curve of the Soleus H-Reflex in one Participant. Hmax and Mmax Values are Used to Determine the Hmax/Max Ratio. (B) Protocol for PAD1 (ISI = 1s) and PAD 10 (ISI = 10s). In PAD 1 (Left) ten Tibial Nerve Stimuli are Applied at ISI = 1s. Based on the Literature, the Amplitude of the H-Reflex Elicited by the 2nd to the 10th Stimulation Will be Decreased Compared to the Amplitude of the 1st H-Reflex, as Seen in the Figure. This Pattern is Expected in Individuals who do not Show Spasticity. In PAD10, 10 Tibial Nerve Stimuli are Applied at ISI = 10s. The Amplitude of H Reflexes Should Remain Similar from the 1st to the 10th Response.
Stimulation intensity was increased in 2 mA increments every 5 s until the maximal H-reflex amplitude (Hmax) was reached. Then, increments of 10 mA were applied until the maximal muscle response (Mmax) was obtained. The Hmax/Mmax ratio was calculated to quantify reflex excitability. To evaluate post-activation depression (PAD; Figure 1B) of the H-reflex, two stimulation protocols were applied: ten stimuli spaced 10 s apart (interstimulus interval (ISI) = 10; PAD10 protocol; baseline), followed by ten stimuli with 1-s ISI (PAD1 protocol; depression). PAD was quantified as the percentage ratio of the average amplitude of the 2nd to 10th evoked H-reflex to the amplitude of the first H-reflex multiplied by 100.
Clinical Measures Obtained at Admission and Discharge from IFR
Three clinical assessments were conducted at admission and discharge from IFR by trained physical therapists from the SCI unit. The experimental procedures did not interfere with clinical care throughout the rehabilitation process.
Analysis
Clinical evaluations included the LEMS, MAS, and clonus assessments, all reported as numerical scores. H-reflex data were collected and analyzed separately for each leg to account for side-to-side differences. Spinal reflex excitability was quantified using the Hmax/Mmax ratio from the SOL H-reflex, and PAD was also calculated (Ratio for PAD1 and PAD10).
All values are presented as mean ± standard deviation (SD). Changes in clinical measures between early IFR admission and discharge were assessed by paired t-tests for normally distributed data and Wilcoxon signed-rank tests for nonparametric data. Spearman's correlation analyses were performed to evaluate associations between the early Hmax/Mmax ratio and clinical outcomes at discharge, including motor recovery (LEMS) and spasticity (MAS and clonus), as the Shapiro-Wilk test indicated that the data were not normally distributed. A Kruskal Wallis test was performed to evaluate differences in H/M ratios between AIS grades, as the Shapiro–Wilk test indicated that the data were not normally distributed. Post-hoc comparisons were conducted using Dunn's test. Differences between PAD1 and PAD10 were assessed using paired t-test. If normality was not met, a Wilcoxon signed-rank test was used. Group differences in PAD between participants who developed spasticity and those who did not were analyzed using independent t-tests. If the normality assumption was violated, a Mann-Whitney U test was conducted. All statistical analyses were conducted using MATLAB (v.2024b) and GraphPad Prism (v10.2.3). Results were considered statistically significant at p < 0.05.
Results
Changes in Spasticity and Muscle Strength During IFR
Figure 2 illustrates changes in spasticity and lower limb muscle strength between admission and discharge from IFR. Both the MAS and clonus scores showed an increasing trend over the course of rehabilitation, but did not reach statistical significance (MAS: mean = 0.17 ± 0.5 (admission) vs 0.38 ± 0.6(discharge), p = 0.113; clonus: mean = 2.3 ± 4.6 (admission) vs 3 ± 4,1 (discharge) p = 0.906). In contrast, the LEMS significantly improved from admission to discharge (mean = 6,70 ± 9,2 vs 8,26 ± 10,3, p = 0.01), indicating functional motor recovery.
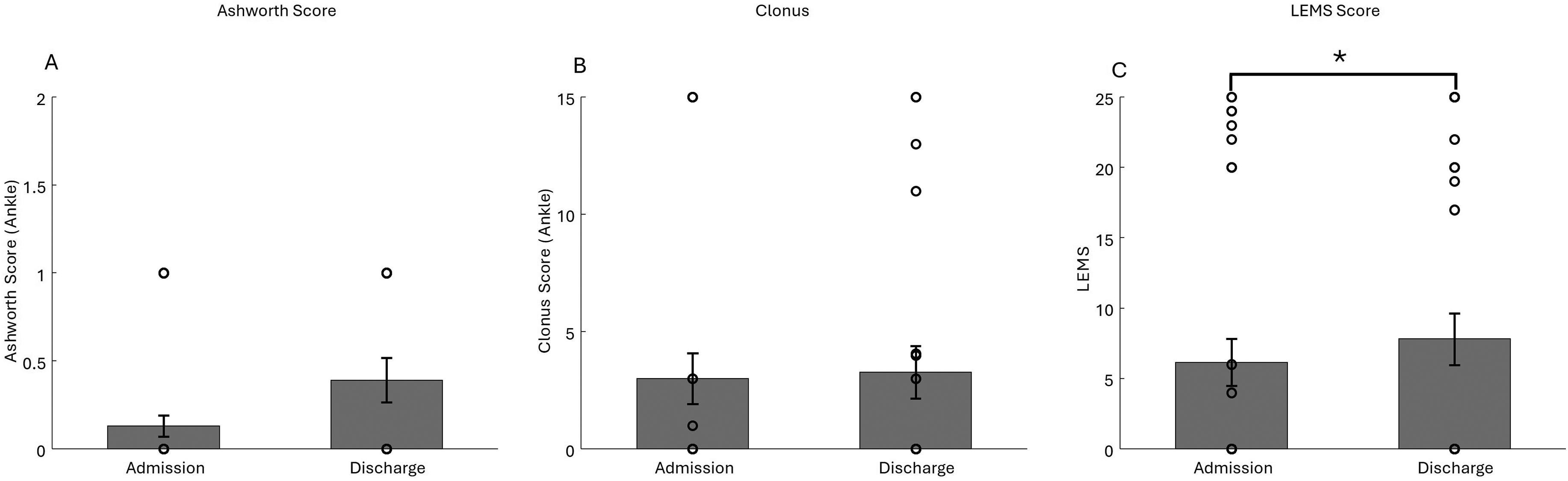
Spasticity and Strength Assessment. (A) Ashworth Score. Each circle Represents value for one leg of a SCI Participant at Early IFR (Admission) and Discharge. Error Bars Represent the Mean ± SD (B) Number of Beats During Clonus Assessment. (C) LEMS Score.
Correlation Between Early Spinal Excitability and Clinical Outcomes
The SOL Hmax/Mmax ratio (H/M ratio), used as an index of spinal excitability, was assessed at IFR admission. In six participants, the H-reflex could not be elicited. Spearman correlation analyses were performed to examine the relationship between the H/M ratio at admission and clinical measures of spasticity and muscle strength at admission and discharge). As shown in Figure 3A-C, there was no significant correlation between the H/M ratio and the MAS score at admission (r = 0.1759, p = 0.3275). In contrast, a moderate and statistically significant correlation was found between the H/M ratio and the clonus score at admission (r = 0.5310, p = 0.003). Similarly, a moderate and significant correlation was observed between the H/M ratio and the LEMS at admission (r = 0.4915, p = 0.0037). As shown in Figure 3 D-F, no significant correlation was observed between the H/M ratio and MAS score at discharge (r = −0.33, p = 0.09). A moderate, statistically significant correlation was observed between H/M ratio and clonus score at discharge (r = 0.55, p = 0.005). Similarly, a moderate and significant correlation was observed between H/M ratio and LEMS at discharge (r = 0.47, p = 0.008).
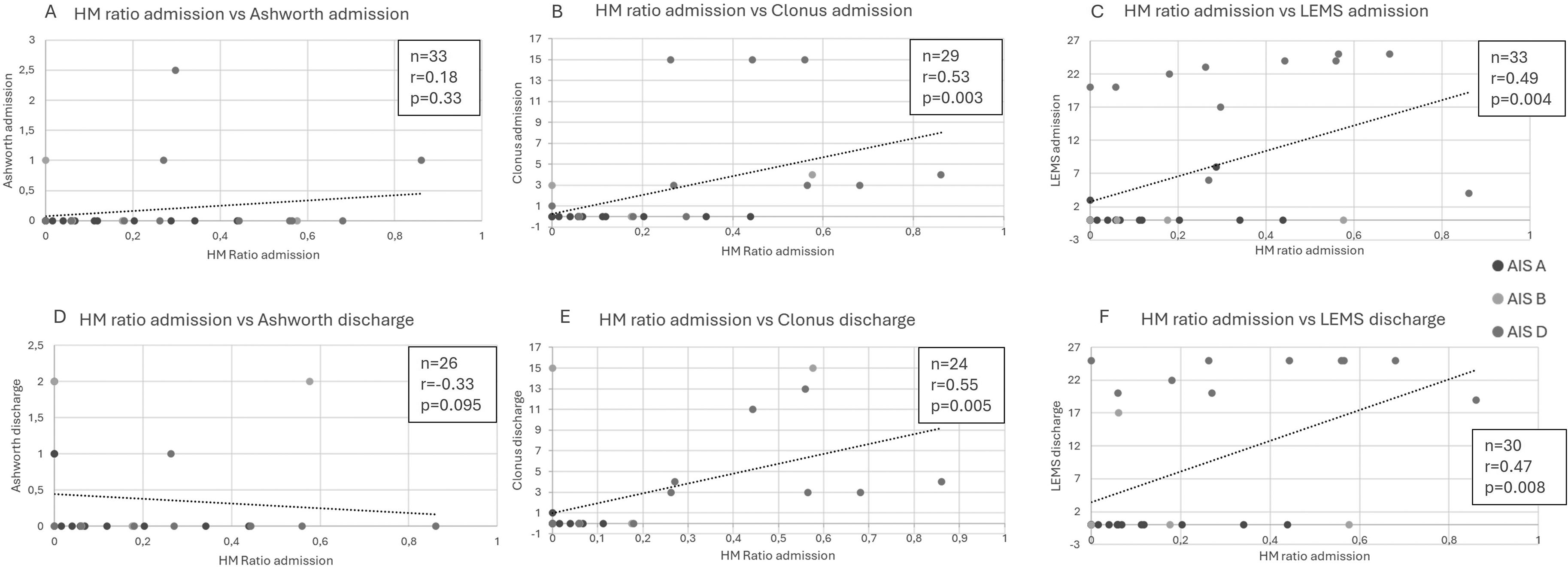
Correlation Between Hmax/Mmax Ratio at Admission and Clinical Measures at Discharge. (A) Lack of Correlation Between the Hmax/Mmax Ratio at Admission and Ashworth Score. (B-C) Significant Correlation Between the Hmax/Mmax Ratio and Clonus (B) and LEMS (C).
Subgroup analysis revealed distinct patterns based on injury severity (AIS grade). A Kruskal Walis test revealed a significant effect of AIS grade on H/M ratios (p = 0.0161). Mean H/M ratios were lower in participants with complete injuries (AIS A: 0.11 ± 0.13; black circles in Figure 3) compared to those with sensory and motor-incomplete injuries (AIS D; 0.38 ± 0.27; white circles in Figure 3; p = 0.01). No difference was observed between participants with motor-complete injuries (AIS B: 0.20 ± 0.26; grey circles in Figure 3) and those with AIS A (p = 0.99), or AIS D (p = 0.82). Participants with AIS A exhibited negligible clonus (mean = 0,1 ± 0,31) and LEMS scores of 0 at discharge. In contrast, those with AIS B and D had larger H/M ratios at admission, individuals with H/M ratios > 0.2 displayed clonus by discharge.
Post-Activation Depression (PAD) as a Potential Predictor of Spasticity
To evaluate whether early PAD measurements could predict the development of spasticity, we compared PAD responses obtained at admission between participants who developed spasticity at admission and at discharge (MAS > 0 and/or clonus > 0) and those who did not (MAS = 0 and clonus = 0). If either MAS or clonus were missing, we based our classification on the available test (clonus or MAS). Figure 4 illustrates this comparison, which was identical at admission and discharge, as the same participants exhibited spasticity at both time points. In the PAD10 protocol (ISI = 10 s), there was minimal change in H-reflex amplitude between the first and subsequent stimulations for both groups (145% and 90% of baseline for spasticity and non-spasticity groups, respectively, p = 0.91, Figure 4, right panel). However, in the PAD1 protocol (ISI = 1 s; Figure 4, left panel), a clear group difference emerged. In participants who did not develop spasticity (Figure 4, white bars), amplitude of the subsequent H-reflexes was significantly reduced (34% of the initial response). In contrast, in participants who did develop spasticity (Figure 4, grey bars), the amplitude of subsequent H-reflexes remained elevated at 89% of the first response, reflecting a reduction in PAD. The difference in PAD1 value between participants who did and those who did not develop spasticity was statistically significant (p = 0,019). Furthermore, a significant decrease in PAD1 ratio compared to PAD10 ratio was observed only in participants who did not develop spasticity (p = 0,031), consistent with patterns reported in healthy individuals. No significant reduction in PAD1 compared to PAD 10 ratio was seen in those who developed spasticity (p = 0,63), suggesting that synaptic depression was impaired already at admission to IFR in this group.
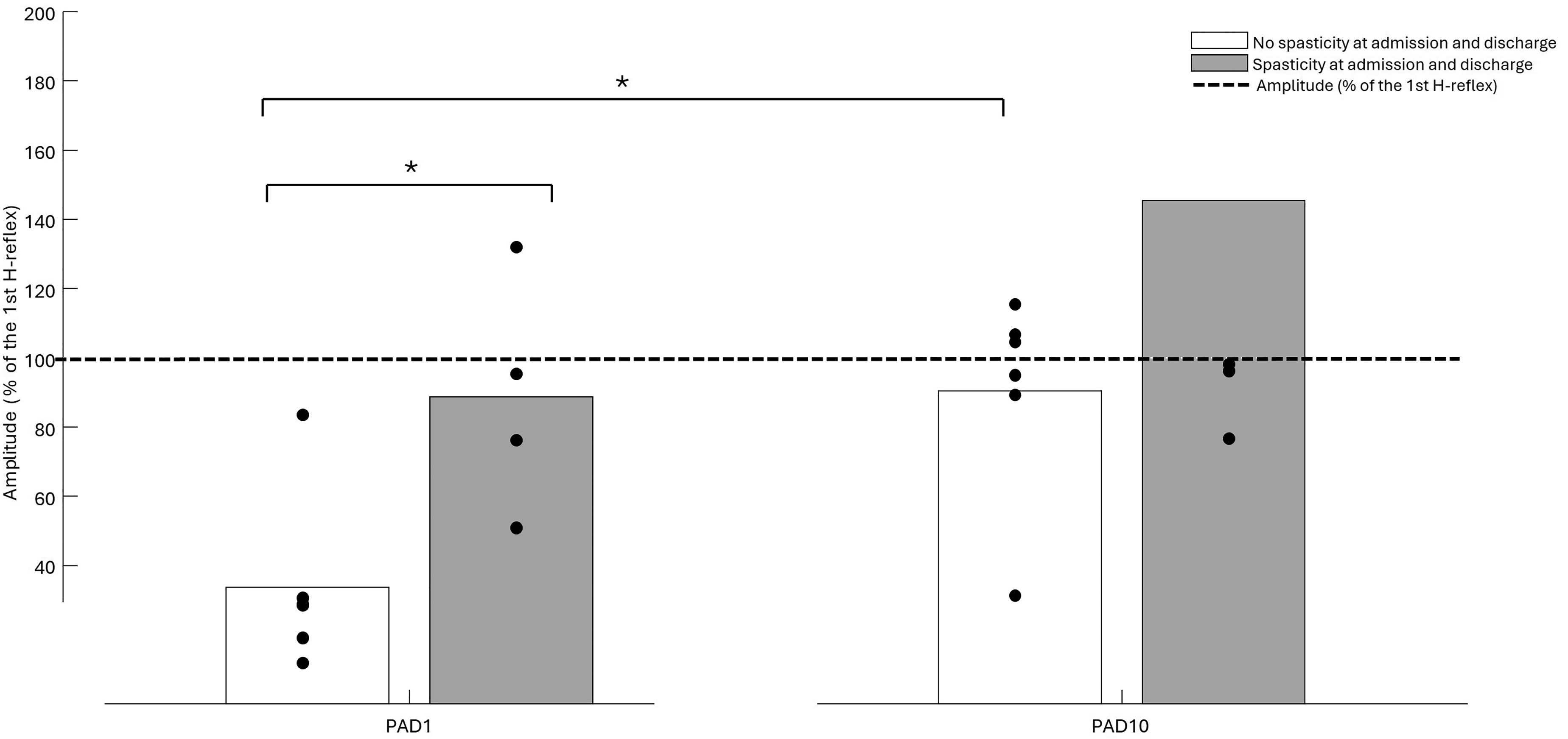
The Relationship of PAD to the Development of Spasticity. PAD1 at Admission Showed a Significant Depression of the H-Reflex Amplitude in Patients who did not Developed Spasticity at Discharge (White Bars), but not in Participants who Developed Spasticity (Grey Bars). When Assessing PAD10, no Depression was Observed in Either Group. Significant Differences Between Groups are Indicated by * (p < 0.005).
Discussion
This pilot, proof-of-concept study examined whether early assessment of spinal reflex excitability—specifically the SOL H-reflex and PAD—could help predict the development of spasticity in individuals with SCI during IFR. Although the small and heterogeneous sample precludes clinically generalizable conclusions, the findings provide preliminary evidence that neurophysiological changes are a marker of spasticiy onset. However, the predictive value could not be directly assessed.
Spasticity, measured by the MAS and ankle clonus, tended to increase between IFR admission and discharge, consistent with the progressive emergence of spasticity in the subacute phase. These changes did not reach statistical significance, likely reflecting the limited sample size and variability in both timing and expression of spasticity across participants (Calancie et al., 2005). Nonetheless, several noteworthy patterns emerged: (1) the Hmax/Mmax ratio at admission correlated positively with clonus severity at admission and discharge; (2) the Hmax/Mmax ratio also correlated with greater lower limb strength at admission and discharge, as reflected by LEMS; and (3) reduced PAD suppression during the PAD1 stimulation protocol was associated with development of spasticity (MAS > 0 and/or clonus > 0). Taken together, these data show that the H-reflex measures are correlated to spasticity at discharge but also at admission. This suggests that these measures could become biomarkers of spasticity. However, their predictive nature is not supported. Assessing H-reflex earlier after SCI within the acute care stay and before any emergence of spasticity could help determine the predictive nature of H-reflex measures.
Spinal Excitability Measures as Biomarkers for Spasticity and Strength
A significant positive correlation was observed between the early Hmax/Mmax ratio and the number of beats in clonus both at admission and discharge. This relationship was evident across the level and severity of injuries (AIS grade), suggesting that early increased spinal excitability may underlie the development of spasticity in SCI (Katz & Rymer, 1989; Pandyan et al., 2005; Trompetto et al., 2014). Although the Hmax/Mmax ratio was significantly correlated with clonus it was only weakly correlated with MAS, underscoring differences in what these clinical scales capture.
Spasticity is a complex, multifaceted condition that cannot be fully characterized by a single outcome measure. While the MAS is widely used in clinical practice and has been shown to be correlated with electrophysiological measures in previous studies (Pizzi et al., 2005; Shen et al., 2022; Tekgül et al., 2013), it has known limitations due to its subjective nature and poor sensitivity to subtle changes in excitability (Fleuren et al., 2010; Pandyan et al., 2005). In contrast, ankle clonus—defined by rhythmic, involuntary muscle contractions—offers an objective, quantifiable index of segmental hyperexcitability and may better reflect the neurophysiological processes underlying spasticity at the ankle. Clonus has been included in validated spasticity assessment tools such as the Spinal Cord Injury Spasticity Evaluation Tool and the Spinal Cord Assessment Tool for Spastic reflex (Adams et al., 2007; Akpinar et al., 2017). These preliminary results highlight clonus as a potentially reliable clinical marker of emerging spasticity in SCI.
The observed positive correlation between the Hmax/Mmax ratio and LEMS suggests that reflex excitability is not solely pathological. In some cases, elevated spinal reflex excitability may coexist with retained voluntary control. This is consistent with prior studies showing that individuals with incomplete SCI may display increased reflex amplitudes alongside meaningful motor function (Norton et al., 2008; Sangari et al., 2023). Thus, while elevated Hmax/Mmax ratio can indicate hyperexcitability, it may also reflect spared or reorganized spinal networks that support motor recovery. However, the Hmax/Mmax ratio did not clearly discriminate LEMS scores in our sample (see Figure 3). This echoes previous studies showing that H/M ratios were comparable between complete and incomplete SCI in the chronic condition (more than 1 year post-lesion), but generally higher than in neurologically intact individuals (Nakazawa et al., 2006). This finding contrasts with mechanically elicited reflexes that were able to predict a motor-complete status with 100% accuracy and a motor-incomplete status with 91% accuracy (Calancie et al., 2004).
A meaningful finding of this study is the association between reduced PAD and the emergence of spasticity. Participants who developed spasticity (defined by MAS > 0 and/or clonus > 0) demonstrated significantly less PAD following the PAD1 stimulation protocol compared to those who did not. PAD reflects presynaptic inhibitory mechanisms in Ia afferent–motor neuron transmission, and its reduction corresponds to spastic spinal circuits both post-SCI and post-stroke (Hultborn & Malmsten, 1983; Lamy et al., 2009; Pierrot-Deseilligny & Burke, 2005). The fact that PAD was impaired early in patients who developed spasticity suggests that presynaptic inhibition deficits contributes to the pathophysiology of spasticity in SCI.
Mechanisms Underlying Altered Reflex Excitability
SCI leads to profound disruption of both segmental and descending control circuits, initially manifesting as spinal shock characterized by flaccid paralysis and areflexia. In many individuals, this phase is gradually followed by the development of spasticity. Several neuroplastic mechanisms underlie this shift in spinal excitability, including the sprouting of excitatory afferents, the loss or dysfunction of inhibitory interneurons, changes in neurotransmitter release, and alterations in ion channel properties (Bareyre et al., 2004; Bennett et al., 2001; Koerber et al., 1994; Li & Bennett, 2003). One critical component of this process is the emergence of PICs in motoneurons, driven by sodium and calcium channels, which amplify synaptic inputs and enable motoneurons to continue firing after the initial stimulus has ceased (Heckman et al., 2008; Powers et al., 2001). This contributes to reflex hyperexcitability and symptoms such as clonus and increased muscle tone (Li et al., 2004; Norton et al., 2008; Murray et al., 2010). PAD reflects an additional of control, limiting reflex responsiveness during repeated stimulation. Impaired PAD among participants who developed spasticity, likely reflects maladaptive neuroplasticity in segmental inhibitory pathways. Taken together, the increased Hmax/Mmax ratios and reduced PAD may represent early physiological signatures of maladaptive plasticity. These findings underscore the potential of spinal reflex assessments to monitor evolving neurophysiological changes after SCI and to provide mechanistic insight into the processes that drive the transition from flaccidity to spasticity.
Clinical Implications
Efforts to objectively predict motor recovery after SCI or stroke are not new. Previous studies have employed modalities such as transcranial magnetic stimulation (TMS; Stinear et al., 2017) or magnetic resonance imaging (MRI; Martineau et al., 2019; Wilson et al., 2012) to forecast outcomes. In the present study, we explored spinal reflex measures as a potential spasticity-specific predictive tool. We found that these measures were reflecting spasticity already at admission to IFR and we could not state on their predictive nature. Nonetheless, earlier assessment of neurophysiological markers (in acute care for example) could show predictive capacities. If this was validated, Hmax/Mmax ratio and PAD could be combined with other clinical indicators. When combined with other clinical indicators, such as injury level, AIS grade, or structural imaging, to enable the timely initiation of personalized rehabilitation strategies aimed at minimizing spasticity and optimizing functional recovery.
Limitations
As a proof-of-concept study, several limitations restrict the generalizability and immediate clinical applicability of our findings. First, the sample size was small, and although exploratory statistical analyses were conducted, a larger cohort is needed to consolidate these preliminary observations. In particular, our sample included individuals with a wide range of lesion levels and severities (e.g., cervical vs. thoracic vs. lumbar; AIS A vs. AIS B or D). Future studies should recruit more homogeneous clinical groups or enroll sufficient participants to enable stratified analyses, thereby determining the specific utility of spinal excitability measures within different subpopulations of SCI.
Second, the absence of long-term follow-up limits interpretation. Several participants included in this study did not show spasticity at discharge. Spasticity evolves dynamically and can continue to progress well beyond the subacute rehabilitation period. Accordingly, future studies should incorporate longitudinal assessments at 6–12 months post-injury to capture the trajectory of spasticity and validate the predictive value of early reflex measures.
Third, H-reflexes could not be elicited in 6 of the 17 participants, potentially due to ongoing spinal shock or other early post-injury changes. Although H-reflexes can sometimes be recorded during spinal shock, interlimb reflexes often require several months to reappear (Calancie et al., 2004). Determining the end of spinal shock remains challenging, as there is no single definitive test. Clinically, the return of the bulbocavernosus reflex is widely used as a marker, though recovery of deep tendon reflexes or reflexive detrusor activity may also indicate resolution (Ko, 2018). In this study, the bulbocavernosus reflex was not systematically assessed during rehabilitation, preventing the determination of the precise timing of spinal shock resolution in each participant. This limitation may partially explain our observation that individuals with AIS A injuries exhibited low H/M ratios and minimal spasticity at discharge. Implementing electrophysiological assessments after confirmation of bulbocavernosus reflex recovery could help standardize timing and improve predictive accuracy in future studies.
Fourth, clinical measures of spasticity were obtained from routine physiotherapy admission and discharge assessments. While these were performed by experienced clinicians, variability in personnel and documentation may have reduced measurement consistency and sensitivity. Moreover, assessments occurred at fixed time points without systematically accounting for medication effects. Since pharmacological agents can directly or indirectly modulate spinal excitability and spasticity, future trials should document medication timing and dosage to allow these factors to be included as covariates in the analysis.
Overall, these limitations underscore the exploratory nature of this pilot work. While not clinically generalizable in its current form, the study provides a foundation to guide the design of larger, sufficiently powered trials needed to determine the predictive value and clinical utility of spinal reflex assessments after SCI.
Conclusion
In summary, this study highlights several important contributions. It addresses a critical gap in SCI research by focusing on the early identification of individuals at risk of developing spasticity—a complication that profoundly affects quality of life and functional recovery. By leveraging objective neurophysiological measures (Hmax/Mmax ratio and PAD), the study provides mechanistic insight beyond traditional clinical scales and points to potential biomarkers of maladaptive spinal plasticity. Conducted in the subacute rehabilitation phase, it also emphasizes a clinically meaningful window in the continuum of care, although assessments at earlier time-points would be warranted to determine if these biomarkers could also become predictive of spasticity. Together, these contributions offer a conceptual framework for future investigation aimed at improving the timely prediction and management of spasticity after SCI.
Footnotes
ORCID iDs
Funding
This work was funded as I wrote on the website during the submission process. Here are the information:
Consortium pour le développement de la recherche en traumatologie, Fonds de recherche du Québec en Santé
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
