Abstract
Background/Objectives
Musculoskeletal pain limits function and quality of life worldwide. This study investigated whether Extracorporeal Magnetotransduction Therapy (EMTT) improves pain and physical function in patients with degenerative joint diseases and enthesopathies.
Methods
In this single-center, randomized, double-blind, placebo-controlled trial, 126 patients with >6 weeks of pain from knee osteoarthritis, rotator cuff enthesopathy, or lumbar spondyloarthrosis were randomized to EMTT (80 mT, 8 Hz, 10,000 pulses/session) or sham therapy once weekly for eight weeks. The primary outcome was quality of life measured by the SF-12 Physical (PCS) and Mental (MCS) Composite Scales. Secondary outcome was pain on the Visual Analog Scale (VAS). Differences were analyzed using the Wilcoxon-Mann-Whitney test and Mann-Whitney estimator.
Results
Baseline PCS, MCS, and VAS were comparable between groups. At six weeks, EMTT led to significantly higher PCS (42.9 vs. 38.6, p < 0.001) and lower VAS pain (2.8 vs. 4.3, p < 0.001), with effects persisting at 12 weeks (PCS 45.5 vs. 38.7; VAS 2.2 vs. 4.2; all p < 0.001). MCS showed no significant differences. Mild discomfort and skin redness occurred more often with EMTT.
Conclusions
EMTT appears effective in improving physical function and reducing pain in degenerative musculoskeletal conditions, with minor side effects.
Keywords
Introduction
Musculoskeletal pain is a widespread health issue, contributing to disability, reducing quality of life, and imposing a significant economic burden. 1 These conditions affect 33% of adults in low- and middle-income countries and is the leading cause of disability worldwide, affecting 1.71 billion people.1,2 Common forms of musculoskeletal pain result from knee osteoarthritis, which affect 250 million people globally.3Additionally, rotator cuff enthesopathy is a major cause of shoulder pain. 4 While less common, spondyloarthrosis affects up to 2.5% of the population and leads to chronic back pain and stiffness.5,6 The management of musculoskeletal conditions may include both non-operative and surgical management. A combination of options including nonsteroidal anti-inflammatory drugs (NSAIDs), physical therapy, corticosteroid injections, and long-term exercise programs are often offered to reduce pain and improve function. 7 While important to address impairments in function, physical therapy and exercise programs may be limited due to pain or long-term compliance. 8 The long-term use of NSAIDs and analgesics is often limited due to systemic side effects, and growing evidence suggests corticosteroids can have both systemic and local side effects, including long-term damage to soft tissue, joints, and bones.9,10 Surgical management is typically reserved for persistent symptoms due to risk of potential complications. 11 Despite these options of musculoskeletal care, a significant portion of patients with chronic pain receive inadequate relief. 12
The high burden of musculoskeletal conditions and current limitations in conventional treatments highlights the value of developing additional innovative treatment options.7,13 There has been ongoing interest in applications of electromagnetic fields to aid in musculoskeletal healing. Electromagnetic wave therapy was initially approved by the Food and Drug Administration (FDA) in 1979 for treating bone fractures and non-unions. More recent work has explored the applications of Pulsed Electromagnetic Field Therapy (PEMF) for other musculoskeletal conditions. A systematic review of PEMF therapy for osteoarthritis revealed significant reductions in pain and stiffness and improvements in physical function. 14 However, systematic reviews and meta-analyses have reported heterogeneous and often inconclusive results regarding the clinical effectiveness of PEMF therapy.15,16 The authors indicated that discrepancies likely arise from variability in treatment parameters, study designs, and the necessity for prolonged daily applications, which together limit consistency and reproducibility across trials. These inconsistent clinical results often lead to medical payment models not covering the cost of treatment with PEMF, creating financial barriers that further limit use.
An emerging technology that addresses some of these limitations of PEMF is Extracorporeal Magnetotransduction Therapy (EMTT). Most conventional PEMF devices generate magnetic field intensities between 1 and 10 mT at frequencies typically below 100 kHz. 17 In contrast, EMTT reaches a magnetic field strength up to 80 mT and operates within the 100–300 kHz range, potentially enhancing tissue penetration and biological effectiveness. 18 A randomized controlled study investigated the effects of EMTT application for management of adults with chronic back pain 18 and demonstrated significant reductions in pain and improved function, highlighting its effective integration into the management of complex orthopedic conditions. A separate randomized controlled trial demonstrated that a combination of EMTT and extracorporeal shockwave therapy (ESWT) reduced pain and improved function in patients with rotator cuff tendinopathy, highlighting the potential benefits of integrating these non-invasive treatments. 19 Other case reports suggest that EMTT may enhance healing of fractures.20,21 While the mechanisms for EMTT are still being explored, findings from in vitro studies suggest that electromagnetic fields may have cellular effects on blood flow and cellular differentiation. A study exposing human bone marrow mesenchymal stem cells (MSCs) to EMTT measured increased Vascular Endothelial Growth Factor (VEGF) expression, suggesting promotion of angiogenesis. 22 Additionally, EMTT may influence osteoblastogenesis and mineralization, resulting in accelerated bone healing of bone-related conditions. 23
Recognizing the limited studies evaluating EMTT in musculoskeletal conditions, the purpose of this study was to evaluate the influence of EMTT in management of knee osteoarthritis, rotator cuff enthesopathy, and lumbar spondyloarthrosis. Compared to placebo condition, we hypothesized that eight weekly sessions of EMTT would result in reduced pain using the Visual Analog Scale (VAS) and improved SF-12 physical and mental composite scores at 6 weeks and 12 weeks following completion of treatment.
Materials and methods
Study design
The study design was a single-center, randomized, double-blind, placebo-controlled study, reported in accordance with the CONSORT 2010 guidelines for randomized controlled trials. 24 The study was approved by the local ethical committee Schleswig-Holstein Germany (158/15) and was conducted in accordance with the ethical principles of the Declaration of Helsinki and relevant aspects of Good Clinical Practice (GCP) but was not prospectively registered in an international trial registry. Following screening and obtaining written informed consent, each participant initially completed a one-week washout phase. In the washout phase, no pharmacological treatments were allowed, including all prescription NSAIDs and pain medications. From the start of the washout phase until 12 weeks post-treatment, patients were instructed to avoid anti-inflammatory medications and limited to use of analgesics Paracetamol (maximum of 1500 mg daily) or Metamizole (maximum dose of 2000 mg daily).
After the washout phase, each participant was assigned to receive EMTT or sham and completed eight study visits lasting approximately 15 min at one-week intervals between visits. Patient-reported outcomes were obtained at baseline, 6 weeks after completing study visits, and 12 weeks following study visits. The study was conducted from February 2016 to March 2017.
Patients
A total of 126 patients were enrolled based on the inclusion criterion of experiencing pain for more than six weeks from knee osteoarthritis (n = 31), rotator cuff enthesopathy (n = 43), or lumbar spondyloarthrosis (n = 52). These three conditions were chosen to represent common degenerative and enthesopathic musculoskeletal disorders affecting the lower limb, shoulder, and spine. The required sample size was determined a priori using validated software (N and Nnpar, idv, Krailling, Germany) for a two-sided Wilcoxon–Mann–Whitney test. Assuming a stochastic superiority of MW = 0.64, representing a clinically relevant effect size for musculoskeletal interventions, a total of 126 participants (63 per group) were needed to achieve 85% power at a significance level of α = 0.10. This statistical assumption aligns with previous EMTT RCTs and represents a clinically relevant effect size for musculoskeletal interventions. 18
Recruitment
Research participants were recruited from a single orthopedic outpatient clinic. Patients treated in clinic were heterogeneous in epidemiological and biometric data, disease progression, previous treatments, and medical knowledge.
Inclusion criteria
Study indications: Knee osteoarthritis, rotator cuff enthesopathy, lumbar osteoarthritis
Symptom duration > 6 weeks
Previous physiotherapeutic and pharmacological treatments: Defined as a minimum of 6 weeks of structured physiotherapy (including manual therapy and exercise programs) and standard pharmacological interventions (such as NSAIDs, paracetamol, or corticosteroid injections).
Age 18 years or older
Legal capacity
VAS > 3
Ability and agreement to provide written informed consent
Exclusion criteria
Participation in other interventional trials
Contraindications to receive EMTT according to the product information or Investigator's Brochure
Diseases or findings that could significantly alter the target outcome and obscure or potentially prevent the therapeutic effect being studied, such as rheumatoid arthritis or neuropathic pain syndromes.
Pregnancy or breastfeeding
Individuals in a dependent/working relationship with the sponsor or investigator
Institutionalization due to court or administrative order
Randomization and blinding
Participants were assigned using block randomization of varying block sizes with 1:1 ratio across treatment and control conditions. Following a one-week washout, randomization occurred via telephone using a central number just prior to first treatment visit (sham or EMTT). The investigator documented the patient‘s initials, gender, age, duration of unsuccessful conservative therapy, and timing of the last conservative treatment. The randomization code and treatment allocation were documented, and the randomization list was securely maintained by an independent coordinator. A sham EMTT device was developed in collaboration with STORZ Medical for the control group and was engineered to be visually and acoustically indistinguishable from the active device. Both devices had identical appearance, interface, indicator lights, and emitted the same audible clicking sounds during operation. The sham unit, however, did not generate an electromagnetic field. All treatment procedures were performed identically in both groups. To assess the success of blinding, participants were asked after the final session whether they believed they had received active or placebo therapy. Blinding was preserved throughout the trial by strictly separating the roles of investigators responsible for treatment administration, outcome assessment, and data analysis. The randomization list remained inaccessible to investigators until the database was locked. Device settings were preprogrammed by a technical assistant not involved in patient interaction, ensuring that neither patients nor treating physicians could identify group allocation. These procedures maintained complete double blinding of both participants and assessors during the entire study.
Interventions
The participants assigned to EMTT received high-energy impulse therapy weekly for 8 sessions, with a 1-week break between treatments with the MAGNETOLITH device (STORZ Medical, Tägerwilen, Switzerland). Each session lasted 20 min and involved the following treatment parameters: 80 mT and 8 Hz, resulting in a total of 10000 pulses per session. Those assigned to placebo received the same treatment setups using the placebo device not generating electromagnetic impulses. For knee osteoarthritis, the EMTT applicator was positioned over the front of the knee while the knee was slightly flexed. For lumbar spondyloarthrosis, the applicator was placed over the lower back, specifically in the L4-L5 region, with the patient lying in a prone position. For rotator cuff enthesopathy, the applicator was positioned over the anterior-lateral part of the shoulder, with the arm slightly abducted. The applicator was placed directly on the skin, maintaining minimal distance (approximately 1–2 cm) to ensure comfortable contact.
EMTT physical background
The transmitter coil, which has high inductance (L) and low resistance (R), generates a magnetic field that can penetrate biological tissue with minimal disturbance. The spatial distribution of this magnetic field can be calculated using Biot-Savart's law, which is determined by the geometry of the coil and the flow of current. The mechanism of action for EMTT may involve several processes, including the induction of electrical fields, the generation of forces (such as Lorentz force and torque on magnetic structures), and the excitation or modulation of excited states (such as the Zeeman effect and radical pairs). Additionally, there may be potential thermal effects. However, the primary mechanism likely involves the rapid oscillation and high amplitude of the magnetic field, which generates a strong electrical field through induction. This transduction capability, exceeding 60,000 T/s in the case of EMTT, is thought to drive its therapeutic effects.
Outcome measures
Primary
The primary outcome measure was defined as change in the SF-12 score 12 weeks after the last treatment compared to the baseline value at the start of the study. 25 The SF-12 is a health survey that measures physical and mental health through 12 questions. It provides two summary scores: the Physical Component Summary (PCS) and the Mental Component Summary (MCS). These scores reflect overall health status, with higher scores indicating better health and well-being. The SF-12 is widely used in clinical studies to assess the impact of treatments on quality of life. 25 The target outcome is the difference in these changes between the treatment and control groups. The benchmarks for the Mann Whitney effect size measure (MW) were classified in accordance to Coheńs benchmark values
Secondary
The secondary endpoint was defined as change in pain using the VAS at 12 weeks after the last treatment compared to the baseline value. The target outcome was the difference in these changes between the treatment and control groups.
Adverse events
Adverse events and treatment-related discomfort were systematically monitored and documented at each treatment visit using standardized reporting forms completed by study staff. Outcome measures included local treatment-related effects such as transient pain and skin redness.
Statistical methods
The planned sample size was 63 participants per group based on a pre study power calculation to determine the target sample size. Nonparametric analysis was performed using the Wilcoxon-Mann-Whitney (WMW) test, a robust method developed by Wilcoxon (1945) and Mann and Whitney (1947). The pre-specified Mann-Whitney estimator (MW) was used as the effect size measure for the WMW test. It is a sensitive and robust measure that determines the magnitude and direction of treatment effects. The MW is recommended by many authors for its reliability across various data situations and is regarded as the ‘gold standard’ for ordinal or rating scales in non-inferiority trials. 26 Technically, the MW provides the probability that a randomly selected subject from the treatment group fares better than one from the comparison group, with probabilities ranging from 0 to 1 (where 0.5 indicates no difference between groups). It is statistically defined as P(X < Y) + 0.5P(X = Y).
The relevant benchmarks for the Mann-Whitney effect size measure (MW) are as follows: 0.29 large inferiority 0.36 medium-sized inferiority 0.44 small inferiority 0.50 equality 0.56 small superiority 0.64 medium-sized superiority 0.71 large superiority
All analyses were conducted according to the intention-to-treat principle, including all randomized participants with available outcome data. As missing data were <5% in both groups, no data imputation was necessary.
Results
Demographic characteristics
An equal number of participants were recruited to each group (n = 63) with similar age, gender, duration of symptoms and other characteristics (Table 1). By the end of the study, one participant was lost to follow-up in the EMTT cohort, and three in the placebo group 12 weeks after the final treatment (Figure 1). The percentage of missing observations from randomized subjects was less than 5% in both groups, complying with recommended standards and enhancing the reliability and validity of our findings. 27
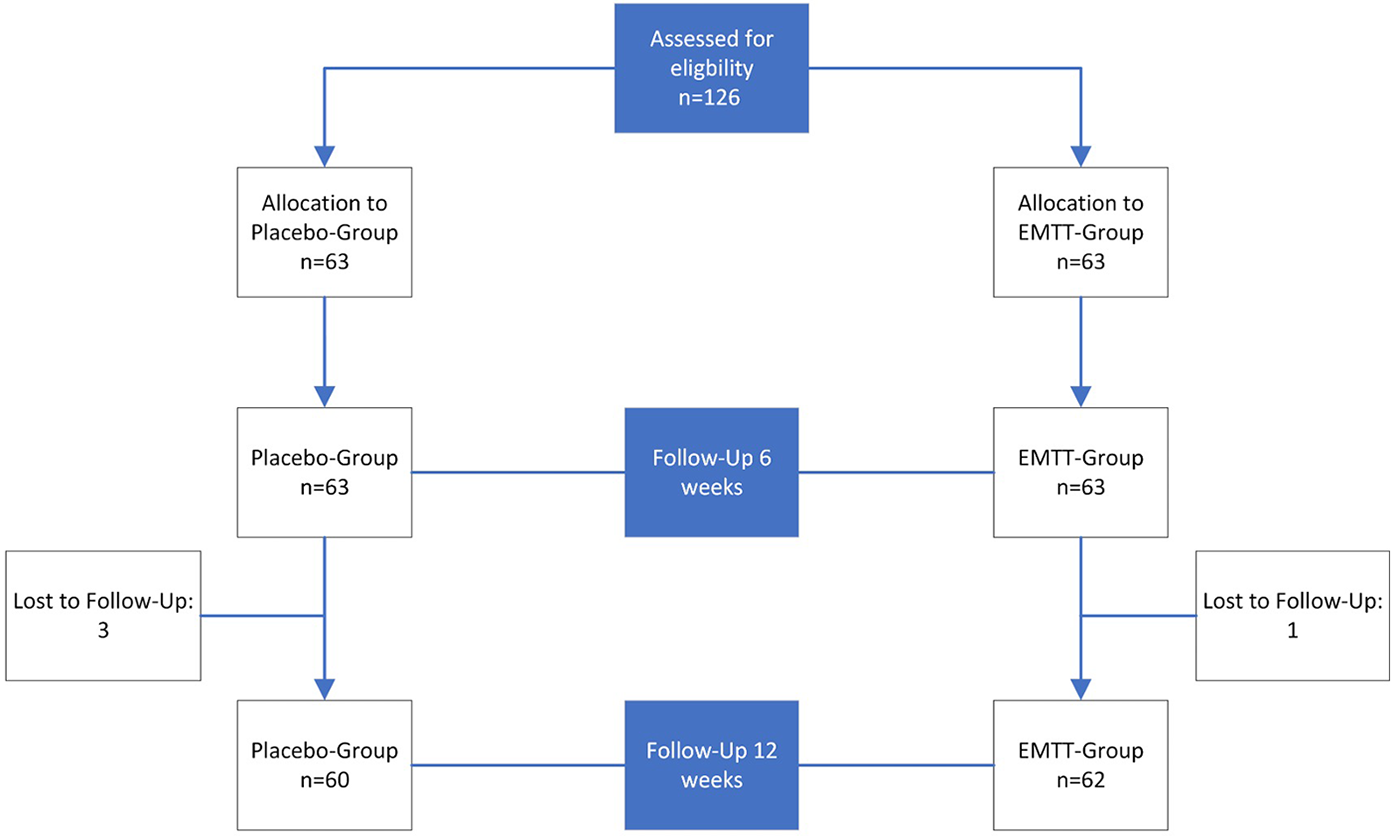
Flow chart of the randomized controlled trial in accordance with the CONSORT statement.
Demographic characteristics.
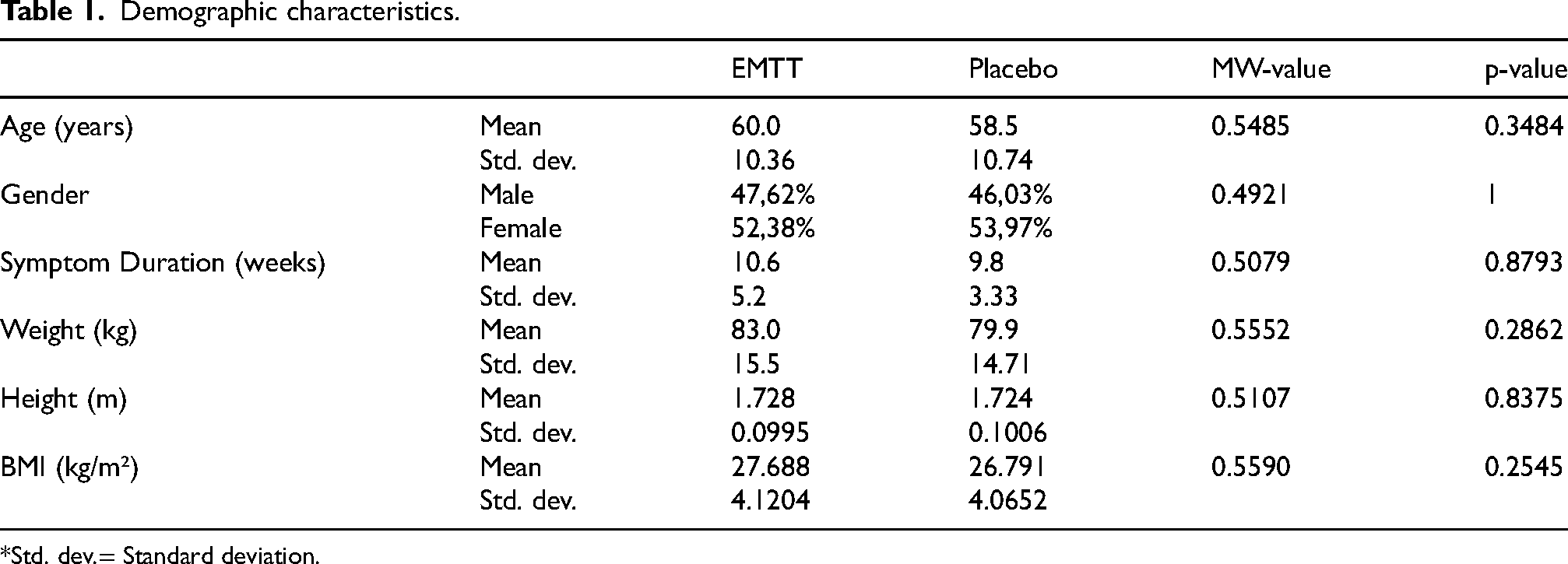
*Std. dev.= Standard deviation.
Outcomes
VAS
Baseline VAS was similar between groups (VAS: 5.1 vs. 5.2, Figure 2). Pain scores in the EMTT group were lower than in the placebo group at 6 (VAS: 2.8 vs. 4.3) and 12 weeks (VAS: 2.2 vs. 4.2) following treatment compared to placebo and was found to be statistically significant at Week 6 and 12 for absolute values (all P < 0.0001). The corresponding Mann–Whitney effect sizes indicated large superiority of EMTT at Week 6 (MW = 0.76, 95% CI 0.67–0.86) and Week 12 (MW = 0.87, 95% CI 0.78–0.96).
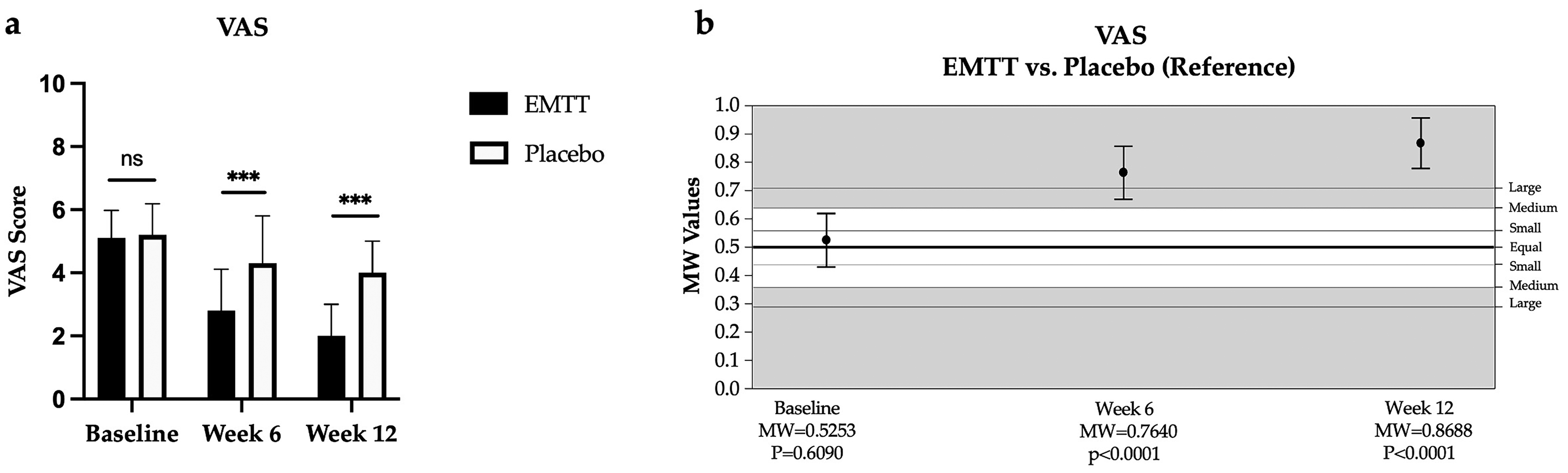
EMTT stimulation is associated with lower pain intensity scores, as measured by the VAS.
SF-12
SF12 measures for EMTT and placebo group were similar at baseline, including for PCS and MCS (PCS: 34.4 vs. 35.3; MCS: 46.2 vs. 48.1, Figure 3). The PCS scores were significantly higher in the EMTT group compared to the placebo group at 6 (PCS: 42.9 vs. 38.6) and 12 weeks (PCS: 45.5 vs. 38.7) following intervention. The superiority of the EMTT group at Weeks 6 and 12 reached the threshold for large superiority, with Mann–Whitney effect sizes of MW = 0.76 (95% CI 0.67–0.86) at Week 6 and MW = 0.87 (95% CI 0.78–0.96) at Week 12 (both p < 0.0001). The baseline MCS value for EMTT cohort was lower than placebo group, but similar values were observed at 6 and 12 weeks following intervention.
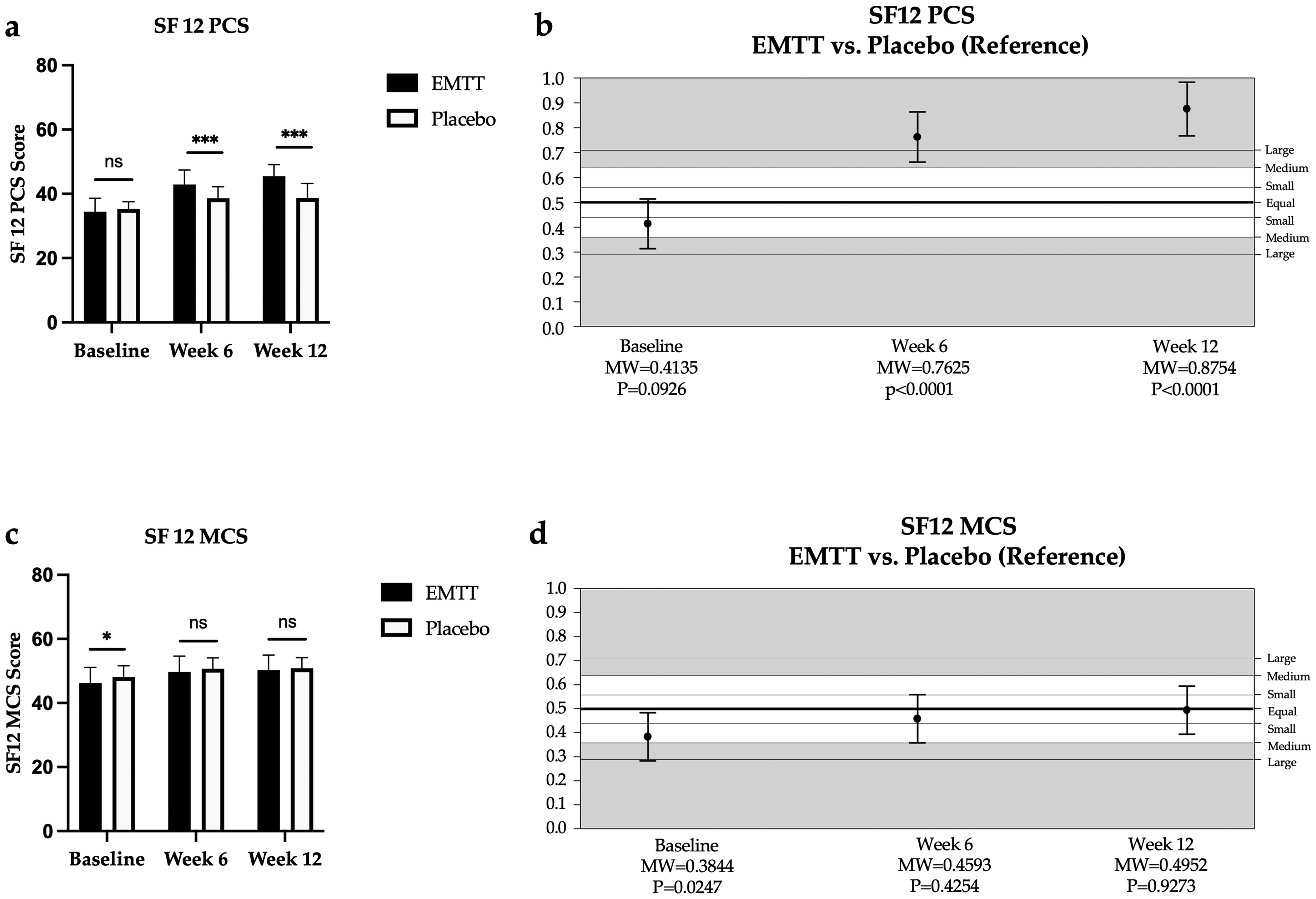
EMTT stimulation is associated with improvements in health-related quality of life, as measured by two scores of the sf-12: the physical component summary (PCS) and the mental component summary (MCS). The absolute values for PCS (a) and MCS (c) for EMTT and placebo cohorts at baseline, 6 and 12 weeks following intervention. The Mann-Whitney effect sizes (MW Values) for PCS (b) and MCS (d) of EMTT compared to placebo. 0.29 / 0.71 = large difference; MW benchmarks: 0.36 / 0.64 medium-sized difference; 0.44 / 0.56 small difference; 0.50 equality. Data are expressed as the mean ± SD. ns: non-significant. * p < 0.05, ** p < 0.01, and *** p < 0.001.
Adverse events and treatment guess
Adverse events and treatment-related discomfort were systematically recorded at each treatment visit (Figure 4(a)). No serious adverse events were reported. Redness was reported as an adverse event in 49.2% (n = 31) of the patients receiving EMTT compared to 9.5% (n = 6) in the sham condition (Odds Ratio OR = 9.20, 95% CI: 3.49–27.04, P < 0.0001). Pain during visit treatment was reported in 42.9% (n = 27)) of participants in the active treatment group compared to 17.5% (n = 11) in the sham condition (OR = 3.55, 95% CI: 1.53–8.21, P < 0.0001). Pain one day after each study visit was similar in 6.3% (n = 4) of the patients in the active treatment group and 9.5% (n = 6) in the sham group (OR = 0.64; 95% CI 0.17–2.40; P > 0.5 (ns)).
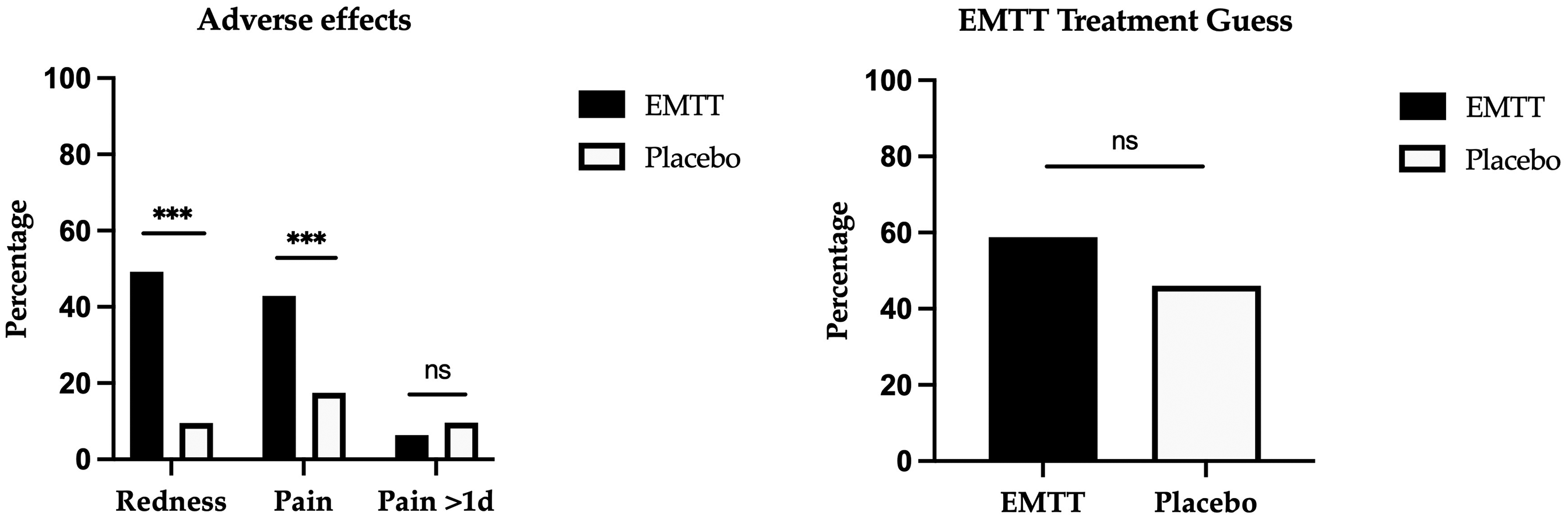
Stimulation with EMTT led to more frequent reports of redness and pain and patients encountered difficulty in accurately assigning themselves to their study group. Data are presented as percentage values. ns: non-significant. * p < 0.05, ** p < 0.01, and *** p < 0.001.
Patients were asked whether they believed they were assigned to the EMTT or placebo groups. While no significant difference was found, patients had a slight tendency to correctly guess their assignment (EMTT: 58.8% vs Placebo: 46.0%, Figure 4(b)).
Subgroups
To evaluate differences by condition, subgroup analysis was performed for each condition of Rotator Cuff Enthesopathy (n = 43), Knee Osteoarthritis (n = 31), and Lumbar Spondyloarthrosis (n = 52) (Figure 5). Except for VAS score at week 6 for rotator cuff enthesopathy, pain levels were significantly lower in the EMTT group compared to the sham condition at each time point. Additionally, the PCS was consistently higher for EMTT over sham for each time point. The MCS scores did not show any statistically significant differences.
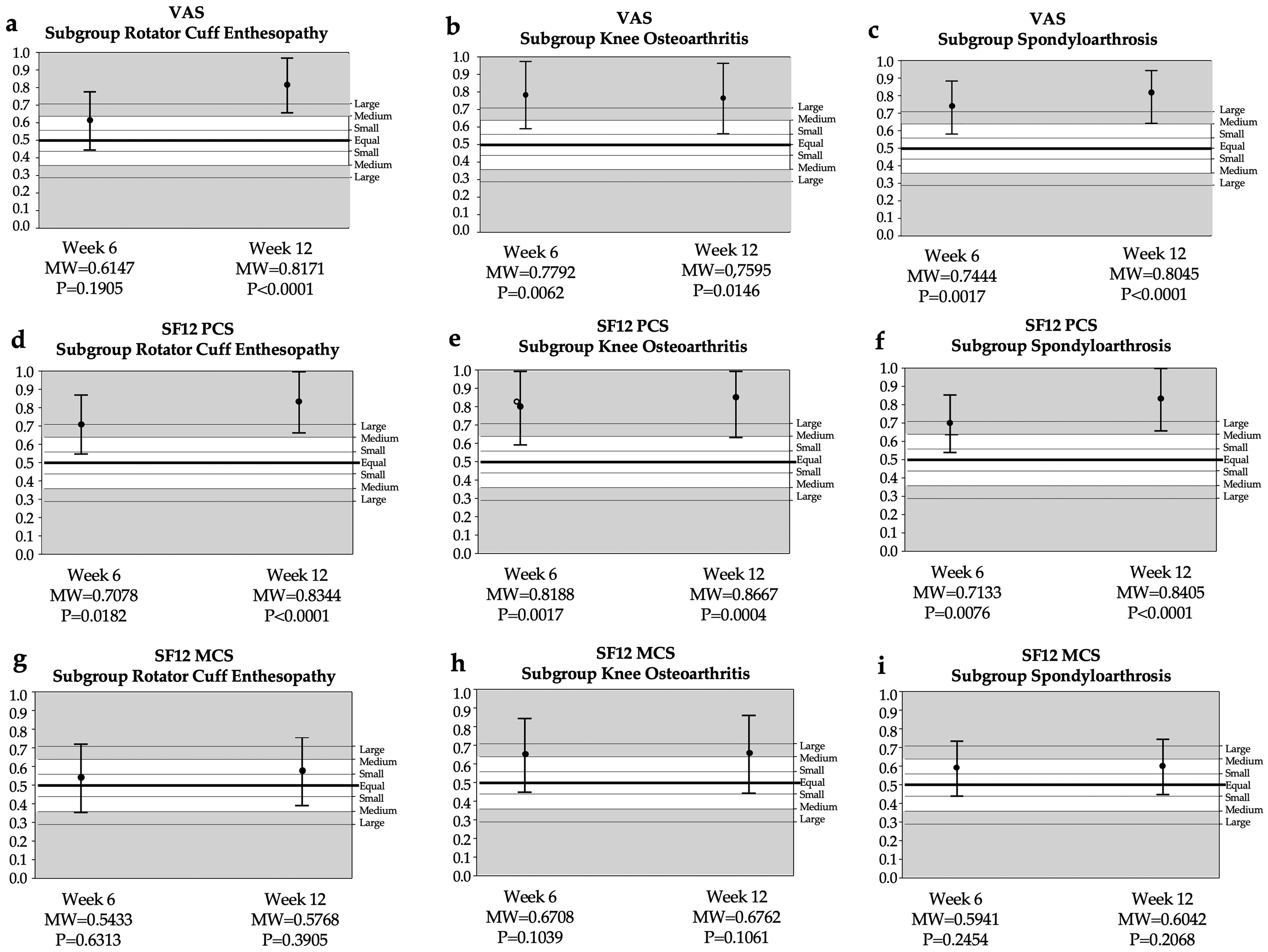
Subgroups a-c VAS score compared to placebo at week 6 and week 12.
Discussion
The purpose of this study was to evaluate the influence of EMTT on quality of life and pain in patients with musculoskeletal conditions. When compared to placebo, results of this study suggest that EMTT may reduce pain and improve physical function and have similar benefits across conditions of knee osteoarthritis, rotator cuff enthesopathy, and lumbar spondyloarthrosis. While recognizing greater rate of minor adverse events with EMTT, the results of this study are encouraging and suggest potential use of this technology for patients with these conditions.
The favorable results of EMTT in management of musculoskeletal conditions add to limited studies and are vital for enhancing patient outcomes and realizing economic benefits. Improved health outcomes enable patients to return to their daily activities and work more swiftly, thereby minimizing productivity losses and reducing sick leave costs for employers. Furthermore, effective management of chronic musculoskeletal conditions with EMTT can diminish the need for extended healthcare interventions, resulting in lower long-term healthcare costs. 28 Despite its potential, the implementation of electromagnetic wave therapy remains limited for several reasons. One significant criticism is the inconsistency in the clinical effects of magnetic wave therapy. 29 This inconsistency may arise from physical factors, such as the low achievable impulse frequency and magnetic field intensity of most pulsed electromagnetic field (PEMF) devices, which can result in diminished biological impact. 30 Furthermore, there is no consensus on the optimal duration of stimulation, with treatment times varying from 1 to 8 h over periods of 1 to 12 weeks. 16 However, these concerns primarily pertain to traditional forms of magnetic wave therapy like PEMF.
The EMTT device used in our study generates a magnetic field strength of 80 mT. It operates at an effective transduction power exceeding 60 kT/s, with an oscillating frequency of 100–300 kHz. For comparison, typical PEMF devices can generate magnetic field strengths ranging from 1 mT to 10 mT, with frequencies between 1 kHz to 10 kHz. 17 These enhanced physical parameters will likely provide a more substantial biological impact, potentially reducing the required treatment duration. To address concerns regarding the safety of these increased physical parameters, in vitro studies have shown no adverse effects on cell viability, indicating that EMTT may be safe for use.22,23 In our study, no serious adverse events were reported, however, two adverse events were observed: increased pain and localized redness. Patients described the pain as minimal and tolerable, with no need to interrupt stimulation due to discomfort. This mild pain may suggest that EMTT targets the injured and pain-sensitized area, which likely contributed to patients’ ability to guess their assigned study group. This experience of pain during treatment can be compared to that of extracorporeal shock wave therapy, where pain often increases during treatment but is typically followed by significant pain relief and improved function within a few days post-treatment. 31 Importantly, pain lasting more than one day after treatment (pain >1 day) occurred at similar rates in both groups, indicating that EMTT does not result in prolonged discomfort following therapy. The lower pain levels, as measured by the Visual Analog Scale (VAS), became statistically significant at weeks 6 and 12, with notable improvements across all three subgroups. The temporary redness observed during EMTT stimulation likely indicates increased local microcirculation, a commonly noted physiological response to electromagnetic stimulation. 32 This effect may be associated with the upregulation of the Vascular Endothelial Growth Factor (VEGF), a crucial molecule in promoting angiogenesis and enhancing blood circulation. 33 This clinical observation aligns with findings from an in vitro study that reported elevated VEGF levels. 22 In addition to its vascular effects, preclinical studies have demonstrated that electromagnetic field stimulation can modulate inflammatory cytokines and enhance regenerative cellular activity in musculoskeletal tissue.34,35 Enhanced circulation, often signaled by redness, supports oxygen and nutrient delivery, playing a vital role in tissue regeneration and healing processes. 36 Together, these factors create a favorable environment that supports tissue healing and regeneration. 37
The conventional treatment for pain often requires medications and other interventions with less favorable risk profiles. Many patients who have chronic musculoskeletal disorders do not receive adequate therapeutic interventions. 38 Current guidelines recommend pharmacological treatments, including paracetamol and non-steroidal anti-inflammatory drugs (NSAIDs), which have demonstrated effectiveness in providing clinically relevant improvements in pain and function. 39 However, given that the incidence of chronic musculoskeletal conditions is primarily associated with advancing age, the rising prevalence of comorbidities, such as gastrointestinal and cardiovascular issues, must be carefully considered. Due to the potential gastrointestinal and cardiovascular side effects, the use of oral NSAIDs is typically limited to short-term treatment at the lowest effective dose. 40 Intra-articular corticosteroids are frequently recommended for chronic joint conditions, such as hip and knee osteoarthritis. 7 Nevertheless, a Cochrane review has indicated that the evidence supporting clinically significant benefits of intra-articular corticosteroids for knee osteoarthritis remains inconclusive, primarily due to the overall low quality of the available trials. 41 Furthermore, a randomized study conducted in 2017 revealed that patients with knee osteoarthritis who received intra-articular corticosteroid injections every three months experienced a slightly greater cartilage volume loss over two years than those receiving placebo treatment. 42 While the differences were minimal, the potential impact on long-term clinical outcomes remains uncertain. Ultimately, surgery should be considered only when all suitable, less invasive treatment options have been attempted over a reasonable period without achieving adequate symptom relief. 3
While this study demonstrated favorable effects of EMTT, there are limitations. One crucial point that needs to be discussed is the potential placebo effect observed in our study. Notably, 46.03% of patients in the placebo group believed they were receiving EMTT treatment, likely due to the device's sound and appearance. This belief may have contributed to the reduced pain levels (VAS) and improved SF-12 scores in the placebo group. However, pain during therapy was still significantly higher in the active group (42.9%) compared to the placebo group (17.5%). While the placebo effect likely contributed to some improvements, the significant differences between the EMTT and placebo groups indicate that the therapeutic effects of EMTT cannot be explained by placebo alone. The overall magnitude of the change was moderate and should be interpreted in the context of a heterogeneous patient population. Additionally, no imaging or biomarker endpoints were included in the study, and the follow-up period was limited to 12 weeks, leaving long-term effects unknown. These limitations should be addressed in future multicenter studies that have longer observation periods and include objective outcome measures. Most clinical and preclinical data regarding EMTT were obtained from a small number of research groups, including ours. As EMTT is a novel therapeutic approach, there is currently a lack of systematic reviews. We strongly advocate for the initiation of more investigator-led and multicenter studies to validate these preliminary findings.
EMTT presents a promising alternative to PEMF, offering higher physical parameters and enhanced biological effects. These advantages may enable EMTT to provide efficient pain relief and functional improvement in patients with chronic musculoskeletal conditions. The observed improvements are comparable to that of other non-invasive interventions, like extracorporeal shockwave therapy (ESWT). Meta-analyses of ESWT trials have demonstrated small-to-moderate reductions in pain and improvements in function across various musculoskeletal disorders.43,44
Conclusions
This study highlights EMTT as a promising treatment for chronic pain associated with degenerative articular disease and enthesopathy, significantly improving patients’ health-related quality of life (SF-12 scores) and pain levels (VAS). By enabling quicker recoveries, EMTT may enhance patients’ daily functioning and may reduce productivity losses and healthcare costs. Integrating EMTT into standard care may improve patient outcomes, making it a promising option for managing chronic musculoskeletal disorders. Additional research involving larger, multicenter groups, extended follow-up, and objective outcome measures is essential to confirm these findings and promote wider clinical use.
Footnotes
Acknowledgments
We thank Prof. Dr Ludger Gerdesmeyer and his team at MedBaltic GmbH, Mare Klinikum Kiel, Germany for the invaluable support in facilitating the study. We are grateful for the opportunity to conduct the research at his department. The study was supported by Storz Medical AG (Tägerwilen, Switzerland), which provided the treatment devices during the study period. However, the sponsor had no involvement in subject handling, data collection, analysis, or manuscript preparation. Additionally, all procedures followed a standardized, established protocol to ensure that the study remained objective and free from bias.
Informed consent statement
Informed consent was obtained from all subjects involved in the study.
Author contributions
Conceptualization, L.G.; methodology and software, L.G.; validation, L.G., and R.B..; formal analysis, J.V.; investigation, R.B. and R.v.E.-R.; resources; data curation, L.G.; writing—original draft preparation, L.G..; writing—review and editing, A.S.T., L.G., K.H. and R.v.E.-R.; visualization, L.G. and J.V.; supervision, R.v. and E.-R.; project administration, L.G.; funding acquisition, R.B. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by STORZ Medical by providing the placebo device within the study time. The sponsor did not have influence on handling of subjects, data collection, data analysis or preparation of the manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KH is editor of the German Journal of Sports Medicine and associate editor of British Medical Journal Sports and Exercise Medicine. He received a lecture fee from STORZ Medical AG and was a consultant for Angelini Pharma S.p.a., both in the context of conservative treatments of musculoskeletal pathologies. R.v.E.-R is editor of the German Journal Das Knie and Die Orthopädie. He is a paid consultant for Johnson & Johnson and Stryker. The remaining authors declare no conflicts of interest.
Data availability statement
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author.
Institutional review board statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board and Ethics Committee Schleswig Holstein Germany 158/15 (I) 09.02.2016.
