Abstract
Background:
Thyroid nodules are common in the general population, and most are benign. Thyroidectomy remains the most common treatment for symptomatic benign thyroid nodular disease. The objective of this study is to determine if a novel, cell-specific, nonthermal modality called nanosecond pulsed field ablation (nsPFA) can provide a safe and effective treatment for symptomatic thyroid nodules.
Methods:
In this clinical feasibility trial (NCT06117085), an nsPFA percutaneous electrode was used to ablate benign thyroid nodules under ultrasound guidance. In Cohort 1 (5 patients), ablations were created during a thyroidectomy procedure (treat-and-resect), so that initial ablation zone characterization could be assessed histologically. In Cohort 2 (20 patients), up to 4 isolated ablations were created in the in situ thyroid for dose-ranging and to allow for estimation of ablation zone volume. In Cohort 3 (5 patients), the entire nodule was ablated with therapeutic intent using multiple, overlapping ablations for resolution of symptoms.
Results:
For Cohort 1, the mean ablation zone measured 1.7 cm long by 0.7 cm wide post-ablation. For Cohort 2, the mean ablation zone was estimated to be 2.7 cc in volume at 90 days post-ablation (based on nodule size reduction from baseline). Transient dysphonia (<24 hours) was seen in two patients treated at the highest ablation setting (93 mJ/mm2). For Cohort 3, treated nodules had a mean volume reduction of 48.2% as early as 2 weeks, and 71.1% at 1 month and 85.8% at 1 year. Patients could typically resume normal activities on the same day. There was no transient dysphonia in this group. Noticeable volume reduction and relief of symptoms were seen as early as 2 weeks post-treatment. No fibrosis or scars were seen on follow-up ultrasounds. No serious adverse events were reported for any cohorts.
Conclusions:
This first-in-human study supports the initial safety/efficacy profile of the nsPFA electrode system in treating benign thyroid nodules. The minimally invasive and nonthermal nature of nsPFA energy has the potential to reduce risk of major complications in treatment of benign thyroid nodules as compared with thyroidectomy or thermal ablation and to improve healing through rapid reduction ofablated areas and lack of postprocedural scarring.
Introduction
Thyroid nodules are common in the general population and the prevalence goes up with increasing age and female gender. They are increasingly detected by improved imaging modalities. 1 Ninety percent of these nodules are clinically insignificant, and most are benign. 2 A small percentage of these nodules become locally symptomatic. Symptoms can include hoarseness or change in voice, difficulty in swallowing or breathing, neck pain, a sensation of fullness in the neck, or cosmetic deformities. A benign thyroid nodule causing local symptoms may require surgery if it is so large that it makes it hard to breathe or swallow. 3 The risks of a total thyroidectomy include possible hoarseness, permanent hypocalcemia, a mid-cervical scar, permanent hypothyroidism 4 and post-operative bleeding. More importantly, patients that choose a total thyroidectomy will undergo a lifetime of hormone replacement and the associated yearly appointments and testing to support proper thyroid and endocrine function.
With the development of modern imaging and ablation technologies, laser ablation, radiofrequency ablation (RFA), and microwave ablation have emerged as promising thermal modalities with proven effectiveness in the percutaneous treatment of symptomatic benign thyroid nodules. 5 RFA is known for its high success rates in reducing nodule volume and improving compressive symptoms and thyroid hyperfunction with relatively low complication rates. Complications include nodule rupture, dysphonia due to thermal injury to the recurrent laryngeal nerve, and thermal injuries to the trachea, skin and esophagus but these are very rare. An additional concern is the long-lasting, stiff fibrotic ball that results from the heat-induced protein denaturation inherent in this therapy which can complicate subsequent ultrasound diagnoses of thyroid health. Several randomized trials for ablation of thyroid nodules by RFA have been conducted internationally with consistent results and a high level of safety. 6 –9 Published studies using RFA for the treatment of symptomatic benign thyroid nodules in the North American population are limited, but the exploration of this technique has been encouraged by prominent experts in the field. 10 –15
The nanosecond pulsed field ablation (nsPFA) system applies nonthermal, nanosecond-duration electric pulses to initiate the endogenous pathway of regulated cell death (e.g., apoptosis) in the treated cells, which is a similar pathway used by cells when they reach the end of their useful life. As the cells initiate regulated cell death, they send out signals that attract the immune system to phagocytose them to remove them from the treated area. 16 The nsPFA system has been used previously for the treatment of benign skin lesions including the face, 17 abdomen, 18 trunk, back, arms, legs, hands, 19 and feet. No device or procedure complications and no serious related adverse events have been reported. Only minor expected adverse skin effects have been reported to date, and they were resolved within days of the procedure. 20
Here we describe the first-in-human clinical trial of benign thyroid nodule ablation on 30 patients using the nsPFA Percutaneous Electrode (nsPFA PE) System as a nonthermal alternative approach to surgery or thermal ablation applying nsPFA energy. We report a very rapid rate of nodule shrinkage and symptomatic relief and without the stiff fibrotic ball formation observed with thermal therapies.
Materials and Methods
Study design
This is a prospective, non-randomized, clinical feasibility study using the nsPFA PE System for the treatment of benign thyroid nodules, which was completed under IRB approval of the protocol (NP-TR-024) with informed consent of the patients (NCT06117085). All research was approved by the Comitato etico Campania territoriale Campania 1 ethics committee and completed in accordance with the Declaration of Helsinki as adopted in 2024. The study was conducted at Ospedale del Mare, ASLNA1Centro, Naples, Italy.
Detailed study procedures
Patients were recruited from the practice of the Principal Investigator (S.S.), who also performed the nsPFA procedures and thyroidectomies. Laryngoscopy was performed in all patients before the procedure and transcutaneous laryngeal ultrasound was performed in all patients after the procedure. All ultrasound examinations were performed and interpreted by S.S.
The patients were treated in the operating room. Where necessary, light sedation was administered (intravenous midazolam) and local anesthesia was performed with 2% lidocaine and 5% mannitol in the pericapsular area. No eligible patients were excluded from the study.
Cohort I
Five patients were enrolled with a planned thyroidectomy. The nsPFA procedure was performed on benign thyroid nodules under general anesthesia after they were surgically exposed and before any ligation of the vascular pedicles. This was followed by ultrasound-guided insertion of and treatment with a 13G nsPFA electrode. The patients then underwent a thyroidectomy and tissue samples were sent to the pathology laboratory for gross histological evaluation.
Cohort 2
Twenty patients underwent ultrasound-guided insertion of a 13G nsPFA electrode under local anesthesia (without sedation). There were 2–4 ablation zones created in a dose-ranging study without overlapping of treatment zones. Ultrasound evaluations were performed weekly until 1 month, then at 3, 6, and 12 months post-nsPFA PE procedure. Some of these patients (10) were retreated at 12 months with the 18G electrode as a Cohort 2 extension and all exhibited additional shrinkage.
In the cohort 2 extension (10 patients) and cohort 3 (5 patients)
The 18G nsPFA electrode was inserted into the nodule with 8-second cycles slightly overlapping the created ablation zones in order to achieve the full treatment with a position-pulse-proceed technique. Ultrasound evaluation was performed monthly up to 1 year.
nsPFA PE System
The nsPFA Console contains a high-voltage pulse generator that produces nanosecond-range pulsed electric fields that are delivered to the patient tissue by the Percutaneous Electrode. The nsPFA console consists of all the electronics and controls required to generate a bipolar nanosecond-duration electrical pulse (Supplementary Fig. S1). A touchscreen display allows the user to interact with the system, and the housing is a mobile cart with casters designed for ease of relocation. The back of the console is equipped with a footswitch connector for hands-free control of pulse generation. The CellFX® nsPFA PE System (Pulse Biosciences, Inc., Hayward, CA) is cleared under two 510(k)s: K233705 and K240782 and is indicated for ablation of soft tissue in percutaneous and intraoperative procedures.
The nsPFA PE is a sterile, bipolar electrode designed to deliver nsPFA energy from the nsPFA console to ablate soft tissue. The Percutaneous Electrode consists of a 10 cm shaft, two conductive electrodes with a 5 mm spacer between them. It is connected to the Console by the attached 3 m cable (Supplementary Fig. S1).
The Position-Pulse-Proceed method used with nsPFA
The needle insertion to the target nodule was initiated with a transisthmal technique, through the middle line. Under ultrasound guidance, the needle was guided through the thyroid isthmus to the nodule, which was then punctured. The needle was then advanced through the nodule to the deep and lateral aspect of the capsule boundary. The nsPFA energy was delivered with a fixed-duration ablation cycle, which was automatically applied by the system while the needle remained stationary. After the ablation cycle was complete, as indicated by a visual and audible signal from the system console, the needle was withdrawn about 1 cm, and another cycle was administered. This was repeated until the ablations reached the proximal edge of the nodule capsule, at which point a new track was initiated in the surrounding, untreated areas, and the procedure was repeated with enough tracks and ablation cycles to treat the entire nodule. This “position-pulse-proceed” technique is distinct from the typical “moving shot” technique often used with thermal ablation technologies, which is required to mitigate the risk of thermal spread. Since nsPFA energy is nonthermal, there is no risk of thermal spread with this modality. Furthermore, the nature of the pulsed electric fields allows for consistent field strength across the ablation zone through a fixed-duration cycle, so energy can be reproducibly delivered by the System. Previous studies have not reported any adverse effects on tissue for overlapping the ablation zones, so the user can distribute the ablation zones confidently to treat the entire nodule.
Statistical methods
Descriptive data are reported as number and percentage or mean and standard deviation (SD) (using GraphPad Software).
Results
Study participants
Cohort 1: 80% (4/5) of the participants were females of mean age 50.6 ± 5.3 (SD) years. The nodules were predominantly solid, with volumes ranging from 3.0 to 21.9 mL, (mean, SD 13.8, 7.1 mL).
Cohort 2: 75% (15/20) of the participants were females of a mean age 53.1 ± 12.2 years. The nodules were predominantly solid, with volumes of 1.3 to 87.2 mL (mean, SD 22.1, 18.8 mL).
Cohort 3: 80% (4/5) of the participants were females of mean age 50.4 ± 9.0 years. The nodules were predominantly solid, with nodule volumes ranging from 10.5 to 20.7 mL, (mean, SD 15.8,3.7 mL).
To date, there have been no reported cases of serious adverse events in the study patients. Recovery has generally been rapid with reports of only minor pain, swelling and bruising, typically resolving within a few days without scar (Supplementary Fig. S2.).
Cohort 1
Five patients in Cohort 1 received a single ablation composed of 0.14–0.93 J/mm2 delivered over 8–24 seconds during a thyroidectomy procedure (treat-and-resect), so that initial safety and ablation zone characterization could be assessed under ultrasound guidance. No patients showed any effects on critical anatomical structures (recurrent laryngeal nerve, superior laryngeal nerve, vascular-nervous bundle of the neck, trachea and esophagus) during delivery of nsPFA energy. No adverse effects were observed (i.e., no abnormal heartbeat, bleeding, or hoarseness). Histological examination and ultrasound assessment showed cellular tissue selectivity with a very minimal inflammatory response. The initiation of regulated cell death was indicated by pyknotic nuclei, sharp ablation zone margins (red arrows), and the absence of thermally induced necrosis (Fig. 1).
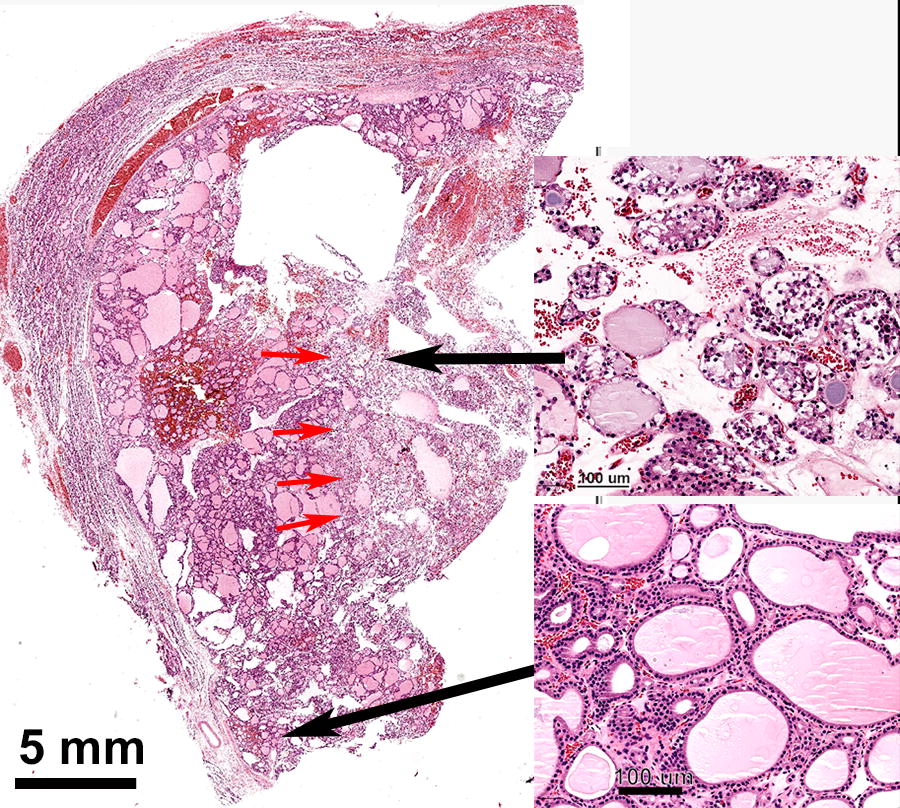
Hematoxylin and eosin (H&E)-stained histological section of treated thyroid nodule fixed about one hour after treatment. The clear demarcation between the viable tissue (magnified on lower right) and the treated area (magnified on upper right) is indicated by the red arrows in this H&E-stained histological section of treated thyroid nodule fixed about one hour after treatment. Treated tissue exhibits congestion and hemorrhagic extravasations with swollen connective tissue. Some destruction of the follicles was noted with leakage of colloid and presence of ghost cells.
Energy application was guided by ultrasound imaging and this permeabilized cells in the ablation zone, releasing fluid that generated a hyperechoic ablation zone in the ultrasound image (Fig. 2). The average volume of each ablation was 2.6 cc as measured by ultrasound.
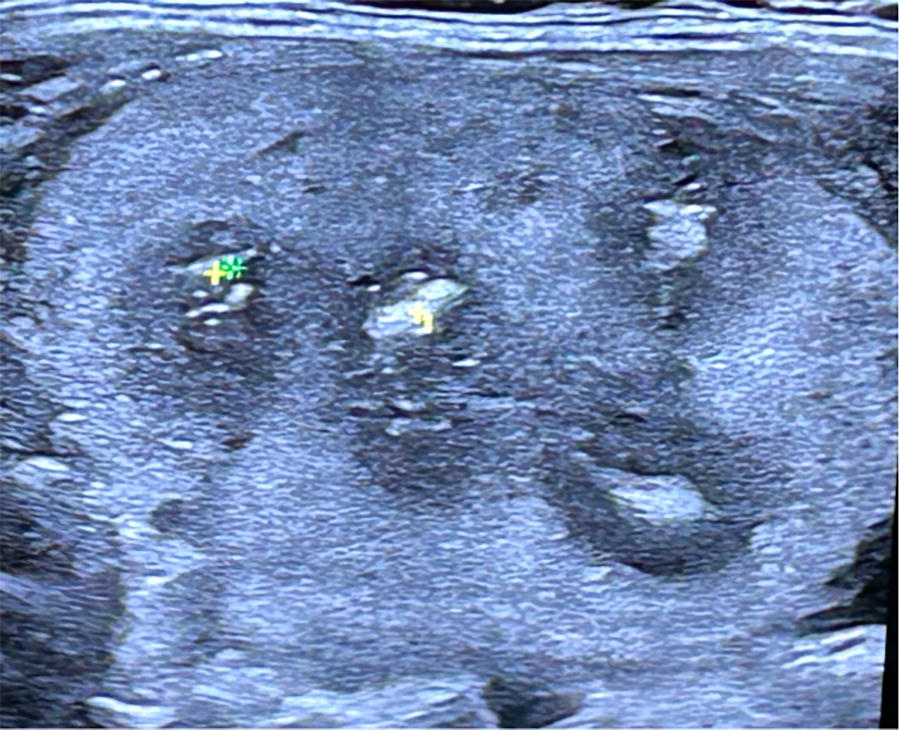
Ultrasound image which includes four hyperechoic ablation zones in a thyroid nodule. 0.3 J/mm2 were applied in each zone.
Cohort 2
Twenty patients in Cohort 2 received one to four ablations in a dose-ranging study (0.14–0.93 J/mm2) and were monitored at 1-, 3-, 6-. 12-month follow-up to measure the rate of thyroid volume reduction (Fig. 3). There were no serious or unanticipated adverse events associated with the procedure. However, two patients being treated with higher energies during dosing studies experienced a nonserious adverse event of transient dysphonia that was induced by the nerve stimulation, resolved spontaneously, and both patients were discharged from the hospital within 24 hours. These events were deemed mild in severity and transient. Reduction in thyroid nodule volume with no change in vascular patterns over time was shown at the 3-month follow-up as compared to baseline by color doppler ultrasonography (Fig. 4). Ultrasound assessment at 1 month and 3 months post procedure showed no scarring or fibrotic areas in the treatment zone typically observed during thermal ablation follow-ups (Fig. 4). There were no reported adverse events at 1-, 3- and 12-month follow-up assessments. Most of the volume reduction occurred during the first month after treatment (Fig. 3). In this series, the volume reduction rate was limited because the procedure was intentionally planned as dose-ranging, where a limited portion of the nodule was treated, and was not intended to fully treat the nodules. At 12 months, the 10 patients who opted for additional, full-nodule treatments, all exhibited a further volume reduction at 1 month following the re-treatment.
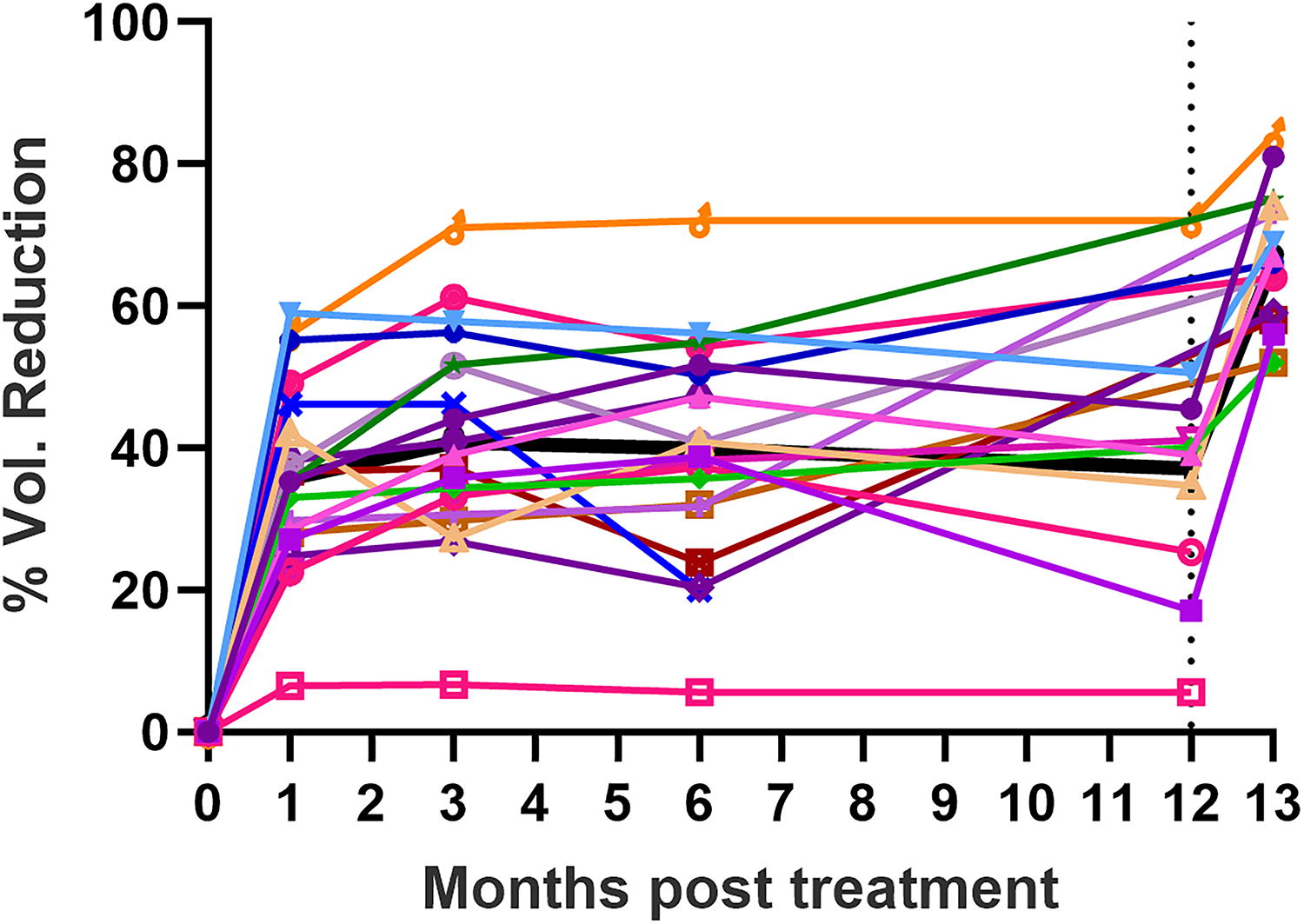
Time course of thyroid volume reduction in Cohort 2. At 12 months some of the patients (10) were retreated and all exhibited a volume reduction at 13 months. The mean value of these volume reductions over time is indicated by the black line.
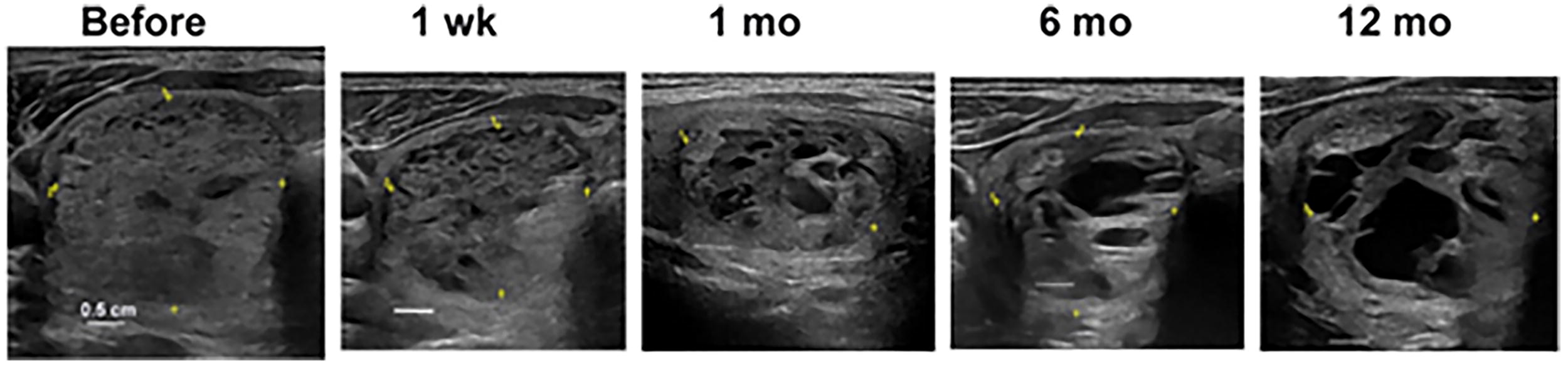
Ultrasound images of a treated thyroid nodule before and several times after treatment. All white scale bars are 0.5 cm.
Patients reported resolution of benign thyroid nodule (BTN)-associated symptoms as early as 2 weeks post nsPFA ablation. Patient satisfaction was very high, with scores of 4.84 ± 0.55 (mean ± SD) at 3 and 6 months post ablation (Cohorts 2 and 3, n = 25) and 4.9 ± 0.32 at 12 months (n = 10 who completed 12-month follow-up), on a Satisfaction Rating Scale of 1–5 (1 = Highly Dissatisfied, 2 = Dissatisfied, 3 = Neutral, 4 = Satisfied, and 5 = Highly Satisfied).
Cohort 3
For the five Cohort 3 patients, the mean treated nodule volume was 13 cc (SD_1.2), and the mean number of ablations per nodule was 55.4 (SD_16.99), completed in an average treatment time of 28 minutes. There were no serious or unanticipated adverse events associated with the procedure or the nsPFA Percutaneous Electrode System. The mean benign thyroid nodule volume reductions were 48.2% (SD12%) at 2 weeks, 71.1% (SD9.9%) at 4 weeks, and 85.8% (SD7%) at one year as measured by ultrasound (Fig. 5).
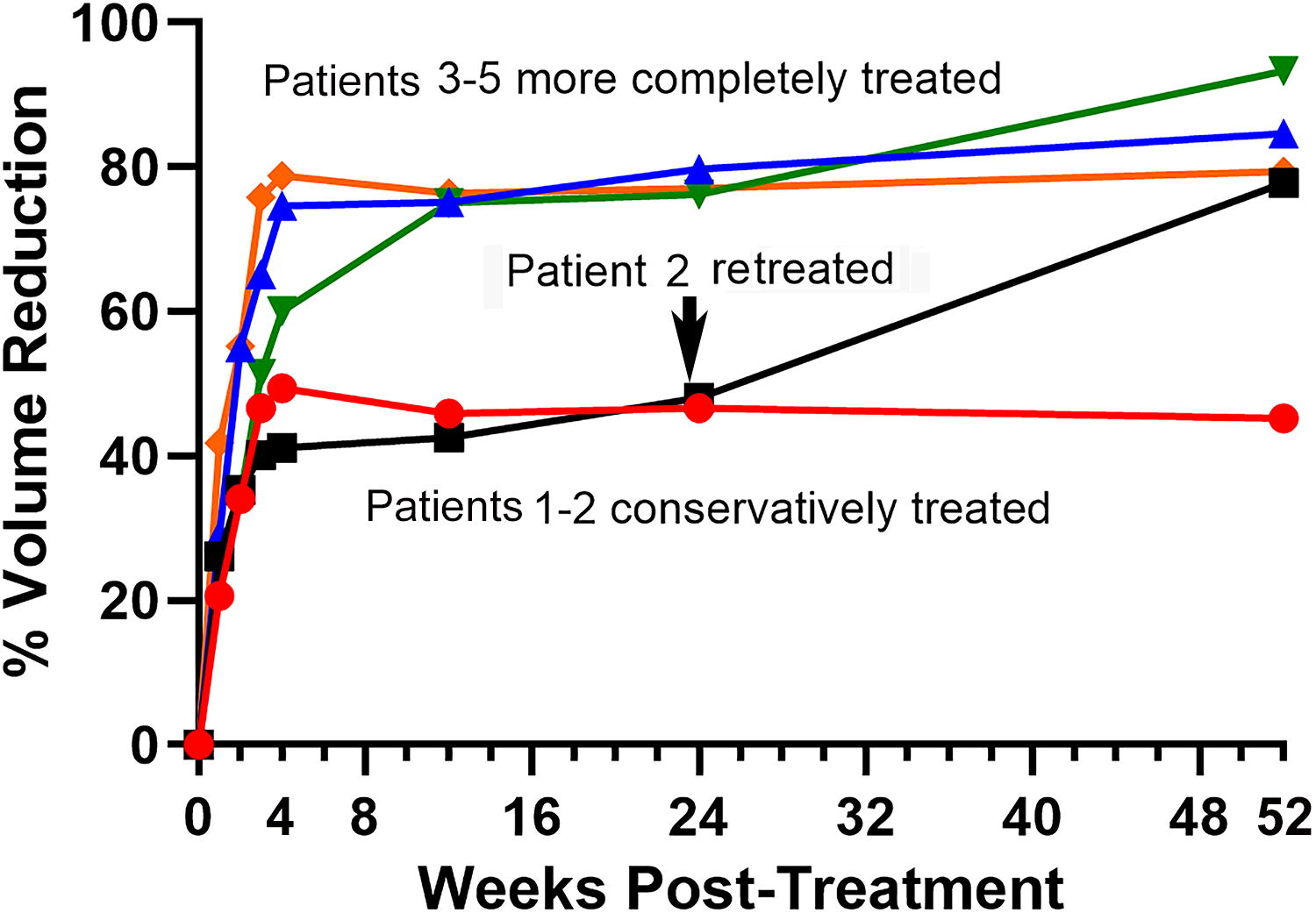
Time course of thyroid nodule volume reduction in 5 Cohort 3 patient in which the entire nodule was treated with therapeutic intent. Patient 2 was retreated (arrow).
Each ablation was visualized on ultrasound (Fig. 2) as hyperechoic ablation areas, and their mean volume was 2.6
Discussion
This first-in-human feasibility study supports the initial safety and effectiveness profile of the nsPFA percutaneous electrode in treating and reducing the volume of 25 benign thyroid nodules, leading to symptom reduction. In Cohort 1, the ablation zone was analyzed histologically, confirming the induction of regulated cell death as the primary mechanism of cellular destruction. The ablation zone boundary is sharp, with little transition zone, which supports the reproducibility of the electric field as opposed to traditional, thermal modalities. The absence of thermal necrosis or damage to acellular tissues highlights an important potential safety difference between nsPFA and thermal ablation modalities. Although the population size was too small to draw conclusions about safety, the absence of any significant, enduring adverse events or side effects is at least supportive of the potential safety profile of this new, nonthermal energy modality. This lack of thermal damage reduces the risk of severe complications and enhances the adaptability of the procedure to be performed in the office or in the OR under anesthesia/sedation. The lack of the fibrotic ball may potentially provide patients treated with nsPFA with a better long-term experience as the treated thyroid may feel soft to the touch.
In Cohort 2, individual, nonoverlapping ablations could be studied by ultrasound and subjective patient symptomatic relief. Although only a small portion of the thyroid nodule volume was treated, patients reported significant symptomatic relief. Ultrasound measurement of the nodules before and at various points after the procedure may also be used to estimate the size of the ablation zone. By dividing the volume reduction by the number of ablation zones, a measure of the volume reduction per ablation zone can be calculated. Since volume reduction peaked at 3 months in Cohort 2, the average ablation size was thereby calculated based on the volume reduction at 3 months, which computes to 2.9 mL. To effectively ablate an entire thyroid nodule, it may be appropriate to overlap the ablation zones by, for example, 25%. This would result in a treatment of 0.43 ablation zones per mL of nodule volume (1 mL of nodule divided by 2.9 mL per ablation zone X 1.25). Using this treatment algorithm, to treat a 20 mL nodule, one might deploy 9 ablation zones.
Cohort 3 offered the first opportunity to observe the efficacy and safety of employing nsPFA energy with the intent to ablate the full nodule. The first two patients were treated with a “conservative” method, not treating the portions of the thyroid nodule closest to the capsule. The next 3 patients were treated more completely, resulting in a more substantial volume reduction (Fig. 5). One patient was retreated at 24 weeks and exhibited an additional volume reduction. All 5 patients rated their satisfaction with the procedure, including symptom resolution, as 5 on a five-point scale. The ablation density averaged 3.5 ablation zones per milliliter of thyroid nodule. Assuming the average volume of each ablation is 2.9 cc, and using the procedure protocol proposed above, targeting 0.43 ablation zones per mL of nodule volume implies that patients in Cohort 3 were treated with about 8 times as many ablations as this hypothetical procedure protocol would suggest. Although there is apparently little or no downside to this type of overlap, it suggests that a more efficient procedure protocol may be effective.
A first advantage of nonthermal nsPFA technology is the enhanced safety profile of this ablation modality compared to thyroidectomy, as well as to thermal modalities like RFA. Unlike RFA, this novel energy spares nerves and blood vessels, making it easy to treat the entire nodule which is located in a sensitive region containing the laryngeal nerve, esophagus, trachea, and critical arteries.
The second potential advantage of nsPFA is that it theoretically may initiate the cell’s endogenous regulated cell death (RCD) pathway that is used by cells when they reach the end of their life. 21 One step in this RCD pathway involves generating danger-associated molecular patterns (DAMPs), several molecules released by the nsPFA-treated cells that attract the immune system. These DAMPs include ATP, mitochondrial cytochrome C, nuclear HMGB-1 and the translocation of calreticulin from the endoplasmic reticulum to their plasma membrane. 16 These DAMP molecules attract migrating dendritic cells of the immune system which then bind to the calreticulin on the plasma membrane, stimulating phagocytosis of the treated cells and removing them from the nodule so that no dead cells are left behind. This leaves the nsPFA-treated thyroid nodule soft to the touch without a fibrotic ball, which is often observed with RFA due to necrotic cell death and thermal denaturing of proteins and acellular structures.
A third characteristic that separates nsPFA from RFA is the rate of thyroid nodule volume reduction: PFA reduced the volume by 71% within one month while the RFA-treated nodules in four large clinical trials typically required 6 months to achieve that level of volume reduction. 22 –25
Study limitations
Further research is needed with a larger patient population as well as different types of benign thyroid nodules in order to more completely assess the effectiveness of this new treatment modality. There was no head-to-head comparison to thermal ablation technologies so any comparisons are speculative and require confirmation in further studies. Other limitations of this study include potential selection bias, small study size, lack of independent histopathological or ultrasound outcome assessment, variation in the extent of treatment in Cohort 3 (as the study progressed), and lack of follow-up data beyond 12 months.
Conclusions regarding feasibility
The rapid rate of shrinkage of the treated nodules and absence of any adverse effects of the treatment lead us to conclude that this new modality is feasible and holds promise to improve patient outcomes. The treated regions are soft to the touch and lack any residual indurated tissue. We believe the results of this feasibility study show that nsPFA offers a new approach that is feasible and promising for further study. This treatment is safe and provides a faster rate of thyroid nodule shrinkage than competing technologies.
Footnotes
Authors’ Contributions
S.S.: Methodology (lead), conceptualization (lead), review and editing (equal). C.O.: Methodology (supporting). C.M.: Methodology (supporting). G.A.: Methodology (supporting). R.N.: Writing—original draft (lead), formal analysis (equal), review and editing (equal). R.P.T.: Conceptualization (equal), review and editing (equal). W.A.K.: Conceptualization (equal), review and editing (equal).
Author Disclosure Statement
C.O., C.M., and G.A. declare that they have no conflicts of interest in conducting this work. S.S. and R.P.T. are consultants with Pulse Biosciences, Inc. R.N. and W.A.K. are employed by Pulse Biosciences, Inc., the company that sponsored this clinical trial and manufactures the nsPFA Percutaneous Electrode System.
Funding Information
Funding for this clinical trial came from Pulse Biosciences Inc. S.S. and R.P.T. have consulting agreements with Pulse Biosciences. C.O., C.M., and G.A. have no funding for this research project. R.N. and W.A.K. are employed by Pulse Biosciences but have no additional funding for this research project.
Supplementary Material
Supplementary Figures
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
