Abstract
Background:
Progression-free survival (PFS) may not fully capture the impact of treatment on patients, especially in cancers with longer natural histories and thus, could be complemented by robust measures of patient-reported tolerability (PRT). We report the use of a novel, quantifiable PRT metric as a multiplicity-controlled endpoint to support regulatory and clinical decision-making for selpercatinib use. Comparative PRT was assessed in LIBRETTO-531 (NCT04211337), a randomized phase 3 trial of selpercatinib versus vandetanib/cabozantinib (control) in advanced RET-mutant medullary thyroid cancer (MTC).
Patients and Methods:
Patients were self-administered the single Functional Assessment of Cancer Therapy item GP5: “I am bothered by side effects” weekly, and scores were dichotomized into “low” (0–2) and “high” (3–4) side-effect burden. PRT measured the proportion of time on treatment (PTT) with “high” side-effect burden for each patient. Comparative PRT was tested at a two-sided significance level of 0.05, conditional on achieving significance for efficacy endpoints. Complementary patient-reported outcomes included health-related quality of life (HRQoL) and symptomatic adverse events self-administered at baseline and at different intervals post-baseline during treatment period.
Results:
In the tolerability evaluable population (N = 242; selpercatinib n = 161 and control n = 81 [56 received cabozantinib, 25 received vandetanib]), patients on selpercatinib had significantly better PRT with lower PTT with “high side-effect burden” than control (8% vs. 24%, p < 0.0001). Post-baseline compliance rates for PRO questionnaires were generally greater than 80% in both treatment groups. Patients on selpercatinib reported significantly less PTT with HRQoL impairment across physical (36% vs. 52%), role (2% vs. 11%), cognitive (4% vs. 8%), emotional (6% vs. 11%), and social (2% vs. 8%) function (all p < 0.01); and significantly less PTT with severe diarrhea (5% vs. 38%), fatigue (6% vs. 21%), taste change (3% vs. 15%), decreased appetite (2% vs. 15%), and hand-foot syndrome (2% vs. 9%) (all p < 0.001).
Conclusion:
This study demonstrated superior PRT for selpercatinib compared with control in patients with RET-mutant MTC, further supporting selpercatinib use as the first-line treatment for patients with advanced RET-mutant MTC. Comparative PRT deserves further adoption as a complement to traditional endpoints in future randomized-controlled trials.
Introduction
Progression-free survival (PFS) continues to be a key endpoint upon which regulatory decisions are made for approval of cancer therapies. 1 Some have questioned whether prolongation of PFS alone represents a meaningful clinical benefit, especially in cancers with longer natural histories like medullary thyroid cancer (MTC). There is a need to complement traditional efficacy endpoints like PFS, overall survival, and safety with outcomes that capture the overall burden of treatment-related side effects. While alternative endpoints such as treatment failure-free survival (TFFS) incorporate both efficacy and tolerability (as discontinuation due to treatment-related adverse events), these are not typically considered by regulatory authorities as acceptable and established outcomes.
Tolerability is typically measured by clinicians via Common Terminology Criteria for Adverse Events (CTCAE), while measuring patient-reported tolerability (PRT) has been challenging as it requires measurement of direct patient experience along with symptomatic adverse events and functional data. 2 The Functional Assessment of Cancer Therapy—Item GP5 (GP5) is a patient-reported outcome (PRO) measure that asks directly how patients feel about their overall burden of treatment side effects. Patients are asked to rate “I am bothered by side effects of treatment” using a 5-point Likert scale: 0 (“not at all”), 1 (“a little bit”), 2 (“somewhat”), 3 (“quite a bit”), or 4 (“very much”). GP5 has been validated 3 –6 and used in cancer clinical trials as a qualitative measure (i.e., descriptive report). 7 –9 We developed a novel endpoint, using GP5, to assess comparative PRT as a quantifiable and prespecified measure of treatment impact. PRT captures the time the individual patient spent with high level of side-effect burden.
Here we report the use of comparative PRT, along with supportive PRO data to complement the efficacy and safety outcomes in LIBRETTO-531 (NCT04211337). This randomized-controlled trial demonstrated the PFS and TFFS benefits of selpercatinib over vandetanib/cabozantinib in advanced, RET-mutant MTC (HR = 0.28 and 0.25, respectively; both p < 0.0001). It also reported that dry mouth, peripheral edema, and erectile dysfunction occurred at a higher incidence with selpercatinib. 10 Comparative PRT and the supportive PRO data were also intended to support regulatory reviews and inform clinical decision-making in first-line MTC treatment.
Patients and Methods
Study design and participants
LIBRETTO-531 (NCT04211337) is a global, open-label, randomized, phase 3 study comparing selpercatinib versus physician’s choice of cabozantinib or vandetanib in advanced RET-mutant MTC patients. Detailed methods of the study, and efficacy and safety results have been published. 10,11 Briefly, eligible patients were 12 years of age and older (if permitted by local regulatory authorities and institutional review boards, otherwise ≥18 years of age) who had pathologically confirmed, unresectable, locally advanced or metastatic MTC and no history of treatment with kinase inhibitors. Patients were also required to have radiological progressive disease per Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1, within 14 months of screening (confirmed by blinded independent central review). An identified pathogenic RET mutation (somatic or germline) was required for enrollment. A full list of inclusion and exclusion criteria can be found in the protocol. 10
Patients were randomly assigned in a 2:1 ratio to receive selpercatinib (160 mg twice daily) or the treating physician’s choice of control treatment: cabozantinib (140 mg once daily) or vandetanib (300 mg once daily). Patients were stratified according to RET mutation (M918T vs. other) and, if assigned to the control group, the intended treatment (cabozantinib vs. vandetanib). Treatment continued until the occurrence of disease progression, discontinuation of treatment due to a treatment-related adverse event, withdrawal of consent, or death. Patients randomly assigned to receive control treatment could cross over to selpercatinib after confirmed disease progression by blinded independent central review.
The study was conducted in accordance with Good Clinical Practice guidelines and the Declaration of Helsinki, and with all applicable country and local regulations. The protocol and its amendments were approved by the institutional review board or independent ethics committee at each site. All the patients, or legal representatives of patients younger than 18 years of age, provided written, informed consent or assent.
PRO measures
Six validated and commonly used PRO instruments were self-administered at baseline (cycle 1, day 1) and then daily for the first year (Worst Pain Numeric Rating Scale, Bowel Movement Count, and Bristol Stool Form Scale) or weekly (GP5 and Patient-Reported Outcomes version of the CTCAE [PRO-CTCAE]) or once every treatment cycle (European Organisation for Research and Treatment of Cancer core Quality of Life Questionnaire [EORTC QLQ-30]) during the treatment period. The use of all PRO instruments was covered under an agreement between the study’s sponsor and appropriate copyright holders of each instrument. The definitions of these PRO measures are presented in the Supplementary Appendix A1. Patients completed their PRO assessments electronically using a provisioned handheld electronic device.
Outcomes
In this analysis, we report the alpha-controlled secondary endpoint of comparative PRT. We also report other PROs that were analyzed as prespecified exploratory endpoints.
Tolerability was patient-reported and defined in this study as the overall burden from side effects of the cancer treatment and assessed using GP5. Post-baseline GP5 scores were dichotomized as “low side-effect burden” (scores 0–2) or “high side-effect burden” (scores 3 or 4). Evidence supporting the use of GP5 to measure treatment tolerability and the definition of “high side-effect burden” was generated in prespecified analyses: a qualitative research sub-study conducted in 40 patients from LIBRETTO-531; 12 and in a psychometric validation using blinded, pooled data from LIBRETTO-531. 13 Comparative PRT was defined as the comparison of the mean PTT with a “high side-effect burden” between treatment groups. This proportion was calculated for each patient by dividing the cumulative weeks of reported “high side-effect burden” by the total duration of therapy (in weeks). Baseline GP5 data were excluded from the PRT analysis as patients were assessed before their first dose of study treatment.
Patients’ health-related quality of life (HRQoL) and symptoms were assessed using the EORTC QLQ-C30. We compared the mean PTT with “clinically meaningful impairment” between selpercatinib and control group using prespecified cutoff values (Supplementary Appendix A2). 14 The mean PTT that patients experienced severe to very severe symptomatic adverse events was assessed using the PRO-CTCAE score 3 or 4 and compared between the two treatment groups. The proportion of patients with worsening of worst pain from baseline was assessed using the Worst Pain Numeric Rating Scale. Lastly, stool consistency and bowel movement frequency were assessed using the Bristol Stool Form Scale, and the Bowel Movement Count measures.
Statistical analysis
All analyses were prespecified and conducted in accordance with the statistical analysis plan (SAP) and PRO SAP. Comparative PRT was an alpha-controlled secondary endpoint to be tested against a two-sided significance level of 0.05, conditionally on achieving statistical significance for PFS and TFFS as assessed by blinded independent central review. Both have been reported previously. 10
The tolerability evaluable population included all patients who received the first dose of study treatment prior to the interim efficacy analysis and at least 6 months prior to the data cutoff date. PRO evaluable population was defined as all randomized patients who took at least one dose of study treatment. The primary PRT analysis (i.e., comparison of the mean PTT that the patient reported “high side-effect burden” [GP5 score of 3 or 4]) was compared between treatment groups using the van Elteren test, 15 a stratified Wilcoxon rank sum test stratified by the two randomization strata: RET mutation (M918T vs. other) and intended control treatment (cabozantinib vs. vandetanib). Details of the estimand framework, missing data handling, and sensitivity analysis are in the Supplementary Appendix A3.
The PTT that patients’ HRQoL and symptoms experienced “clinically meaningful impairment” for the EORTC QLQ-C30 subscales and patient experienced severe to very severe symptomatic adverse events (severity scores 3 or 4) for the PRO-CTCAE items was compared between treatment groups using the same tests as above. The change from baseline on the functional, global health status/QoL and symptom subscales of the EORTC QLQ-C30 were descriptively reported at prespecified time points. The proportions of patients with improved, stable, or worsened EORTC QLQ-C30 domain scores and the worst pain from baseline to prespecified timepoints were descriptively reported. Descriptive statistics of stool consistency and bowel movement frequency, assessed using the Bristol Stool Form Scale and the Bowel Movement Count measures, were reported by weekly average. Statistical analysis was performed using SAS software version 9.4.
Results
Participants
From February 2020 through March 2023, 291 patients were randomly assigned to receive selpercatinib (193 patients) or control (98 patients). All patients who received the first dose of study treatment prior to the prespecified interim efficacy analysis and at least 6 months prior to the data cutoff date (22 May 2023) were included in the tolerability evaluable population (N = 242), 161 in the selpercatinib group and 81 in the control group (56 received cabozantinib, 25 received vandetanib). All patients who received a dose of study treatment were included in the PRO evaluable population (N = 290), 193 in the selpercatinib group and 97 in the control group (72 received cabozantinib, 25 received vandetanib) (Fig. 1). Median time on treatment was over 1 year (68 weeks for the tolerability evaluable population [Q1–Q3: 40–96]; and 56 weeks for the PRO evaluable population [Q1–Q3: 24–92]). Demographic characteristics of the patients in the overall tolerability and PRO evaluable populations were well balanced between treatment groups (Supplementary Table A1). In both populations, most of the patients were male, white, and younger than 65 years of age.
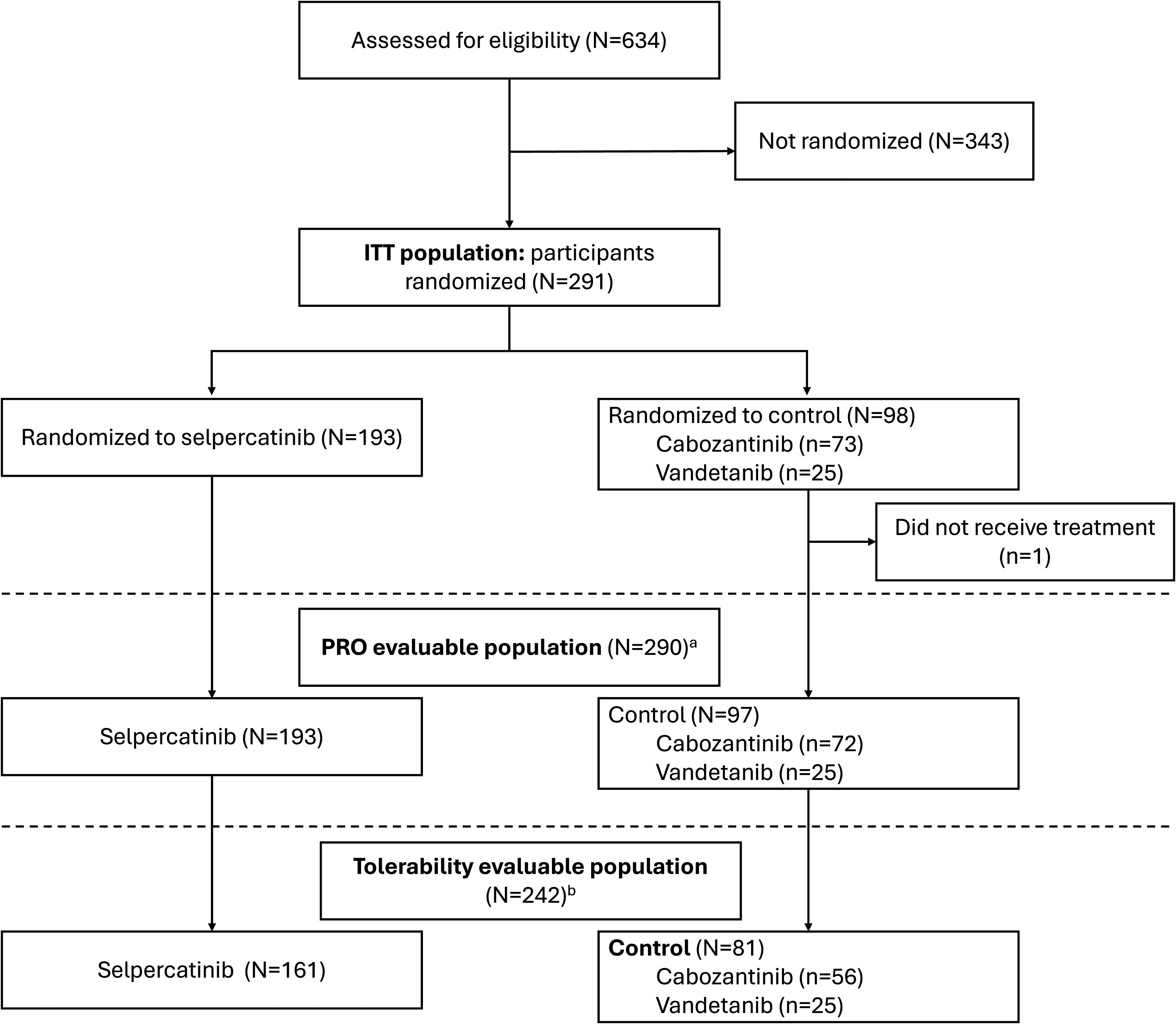
Participant flow diagram. Data cutoff date: March 22, 2023. aPRO evaluable population: patients who had received a dose of study treatment. bTolerability evaluable population: patients who had received the first dose of study treatment prior to the prespecified interim analysis and at least six months prior to the data cutoff date. ITT, intent-to-treat population; PRO, patient-reported outcomes.
Comparative PRT
In the tolerability evaluable population (N = 242), post-baseline compliance rates of the GP5 were mostly greater than 80% at each cycle in both treatment groups. The median duration on treatment was 73 weeks in the selpercatinib group (Q1–Q3: 50.6–108.4) and 44 weeks in the control group (Q1–Q3: 22.3–72.0).
At baseline, only one patient in each group reported “high-side effect burden” with GP5 score 3 or 4. A lower proportion of patients who reported “high-side effect burden” in the selpercatinib group, compared with the control group, was observed starting from Week 1 (3.7% vs. 8.8%), and consistently lower throughout the first year. Approximately 30% of patients in the selpercatinib arm reported GP5 score of 0 (not at all bothered), compared with ∼10% in the control arm, in the first year of treatment (Fig. 2).
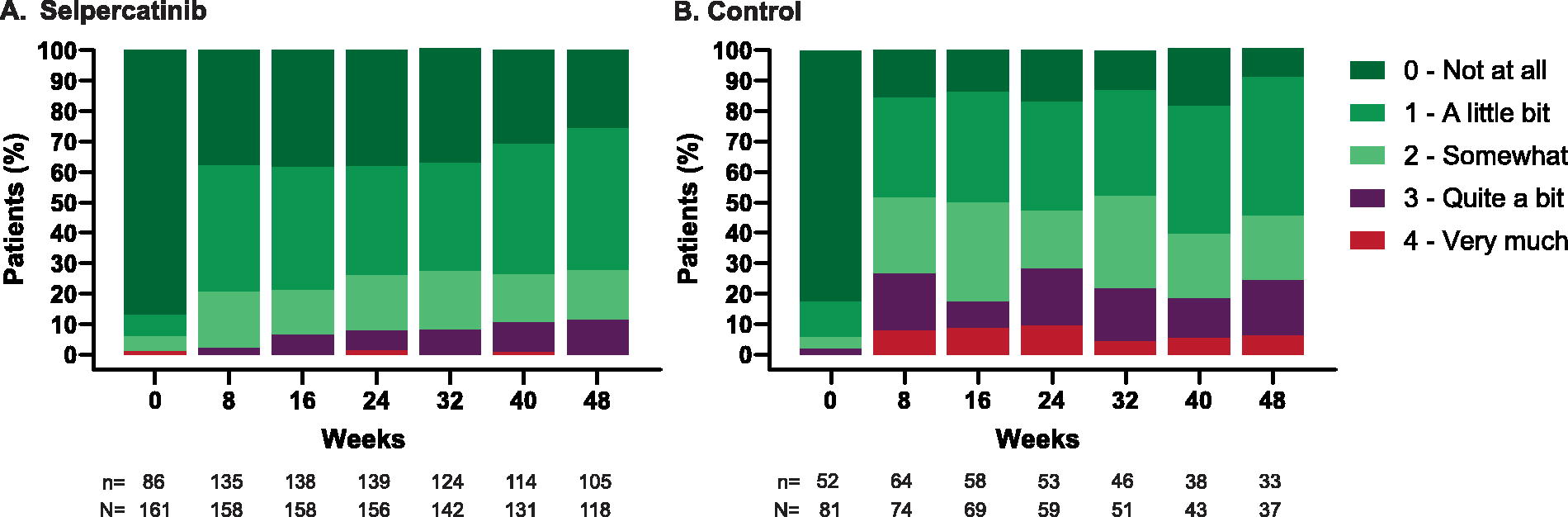
Patient-reported outcomes on GP5 “I am bothered by side effects of treatment” in
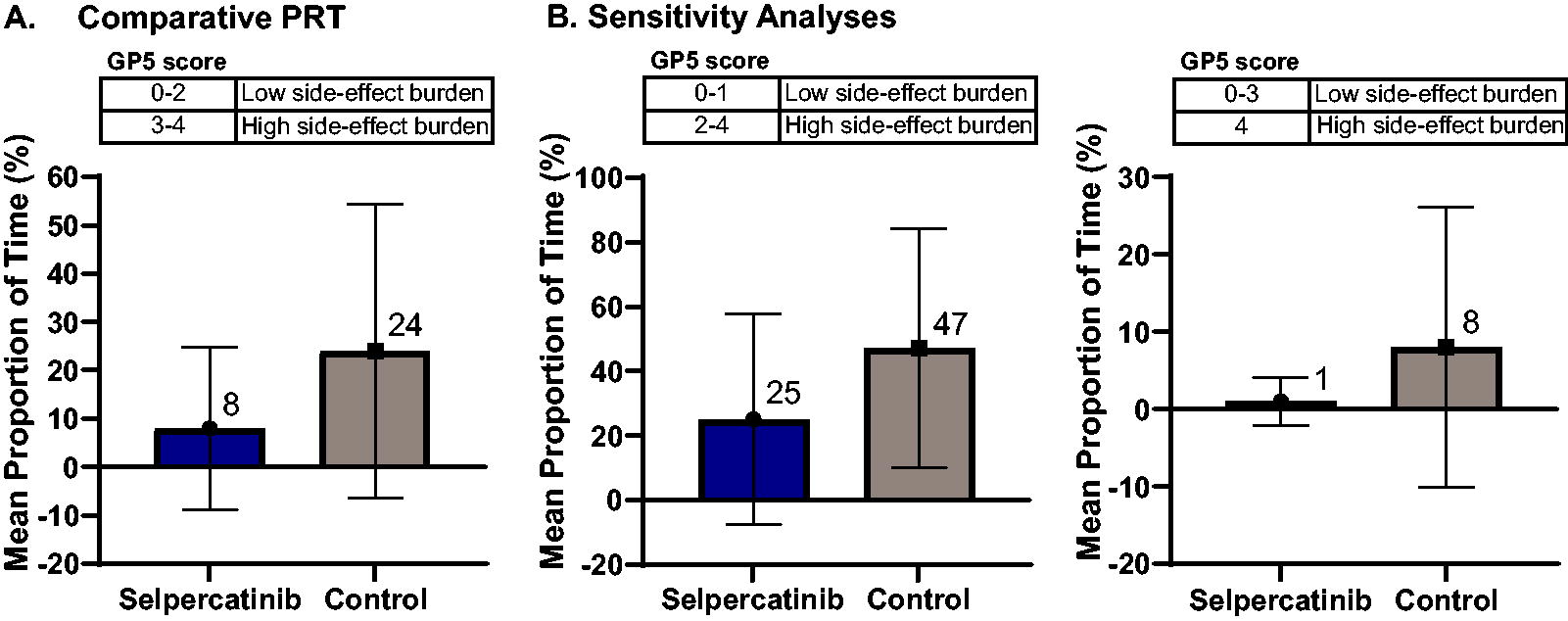
Patients in the selpercatinib group reported significantly less PTT with “high side-effect burden” than those in the control group (mean [SD] 8% [16.9] vs. 24% [30.4], p < 0.0001) (Fig. 3A). Sensitivity analyses using different cutoffs for high side-effect burden showed consistent results, using GP5 score of 4 (1% [3.1] vs. 8% [18.1], p < 0.0001) or using GP5 score of 2, 3, or 4 (25% [32.7] vs. 47% [37.1], p < 0.0001) (Fig. 3B, Supplementary Table A2 and Supplementary Appendix A3). Sensitivity analyses using different severity cut-off points showed consistent results of lower PTT with high side-effect burden in the selpercatinib group compared with the control group, which confirmed the robustness of the primary definition of high side-effect burden with the GP5 score of 3 and 4. The PRT main result was also robust to missing data handling on both imputation rules and prolonged periods of consecutive missing GP5 (p < 0.0001) (Supplementary Table A2).
Health-related quality of life
In the PRO evaluable population (N = 290), 94% completed a baseline EORTC QLQ-C30; 96% in the selpercatinib group and 90% in the control group. Post-baseline, compliance rates were mostly greater than 90% at each cycle in both treatment groups. Baseline scores of EORTC QLQ-C30 are presented in Supplementary Table A3.
Patients in the selpercatinib group reported significantly less PTT with clinically meaningful HRQoL impairment compared with those in the control group. This benefit was reported across EORTC QLQ-C30 function and symptom scales, including physical (36% vs. 52%), emotional (6% vs. 11%), role (2% vs. 11%), cognitive (4% vs. 8%), and social (2% vs. 8%), fatigue (5% vs. 13%), nausea and vomiting (4% vs. 11%), pain (7% vs. 16%), dyspnea (8% vs. 14%), insomnia (2% vs. 6%), appetite loss (1% vs. 9%), and diarrhea (7% vs. 18%) (all p < 0.01). Both treatments showed comparable PTT with constipation (2% and 3% respectively) and financial difficulties (8% vs. 13% respectively) (Fig. 4).
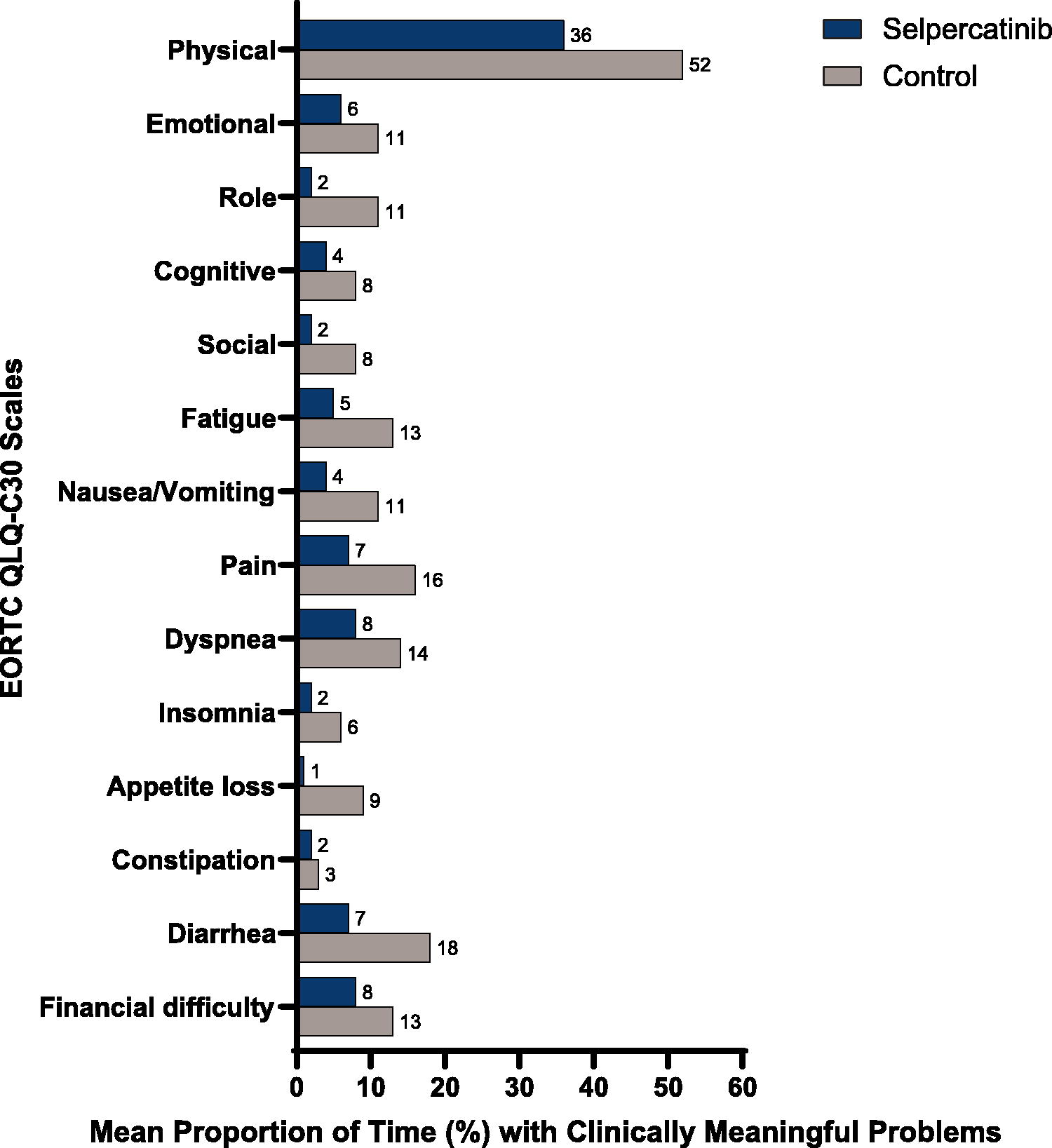
Mean proportion of time on treatment that patients reported “clinically meaningful impairment” on their functioning and symptom scores, as measured by the EORTC QLQ-C30, on each treatment group. The plot shows five functional scales (i.e., physical, emotional, role, cognitive, and social, functioning), three symptom scales (i.e., fatigue, nausea and vomiting, and pain), and six single items assessing additional symptoms commonly experienced by patients with cancer (i.e., dyspnea, insomnia, loss of appetite, constipation, diarrhea) and perceived financial difficulties, as measured by the EORTC QLQ-C30. Physical function domain was collected weekly, while all other domains were collected every four weeks to minimize data collection burden. Since the recall period for the EORTC QLQ-C30 is one week, and the calculation covered the entire treatment period, there were no data for the other three weeks in the cycle for the other domains. This could potentially underestimate the proportion of time with clinical meaningful impairment of symptoms for the other domains during the treatment. We compared the mean proportion of time on treatment with “clinically meaningful impairment” between selpercatinib and control group using prespecified cutoff values (Supplementary Appendix A2). Analyzed in the tolerability evaluable population, which required patients to be on treatment for at least six months. All p < 0.01, except constipation and financial difficulties. EORTC QLQ-C30, European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire—core 30 items.
Additional analyses across treatment cycles showed that the proportion of patients with clinically meaningful improvement or stable scores on the overall quality of life (global health status/QoL) and function (including physical, role, emotional, cognitive, and social function) were higher in the selpercatinib group compared with the control group (Fig. 5).
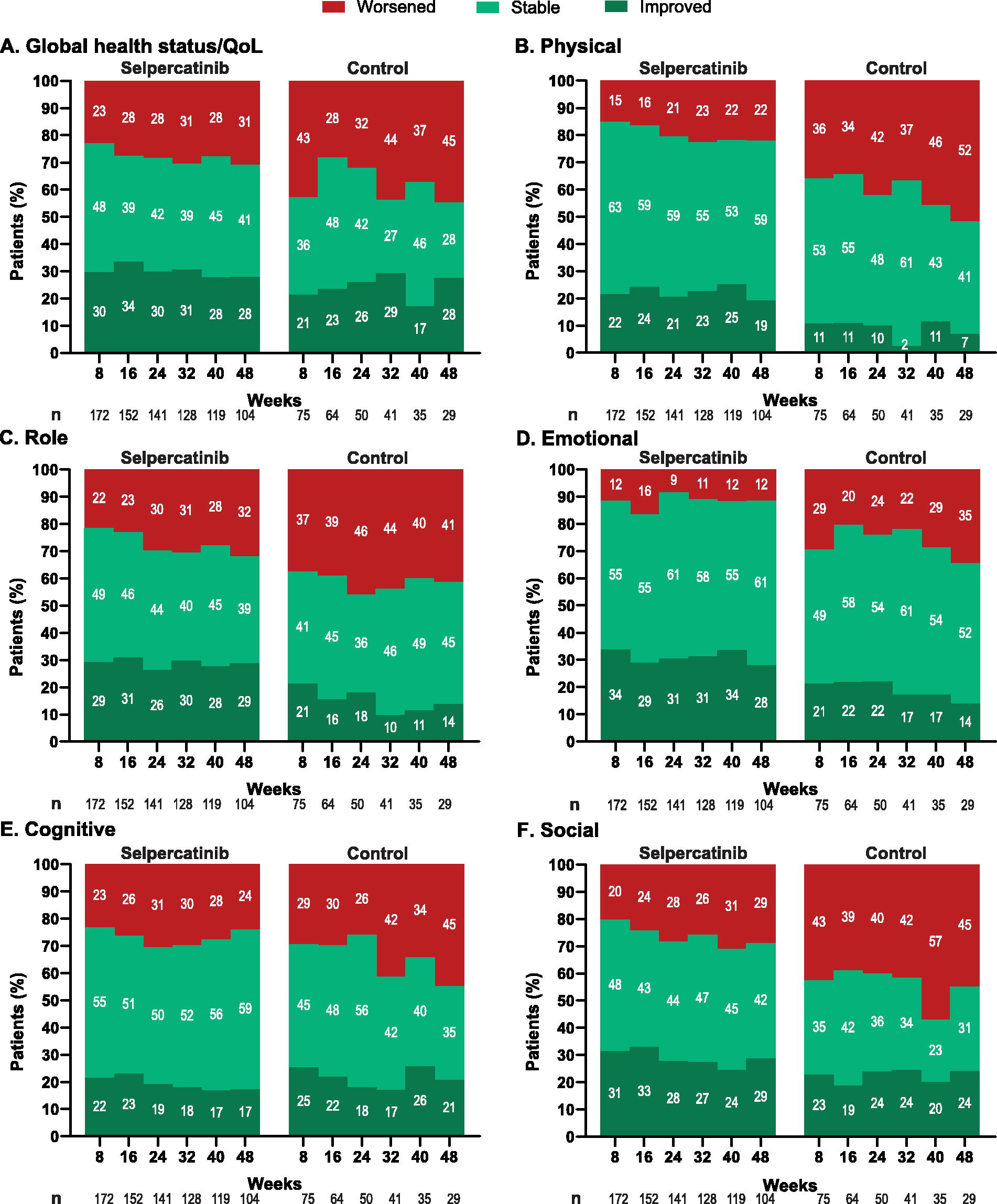
Proportion of patients who met the meaningful change threshold (≥10 points decrease) for improved, stable, or worsened overall quality of life
Severity of symptomatic adverse events
In the tolerability evaluable population, post-baseline compliance rates of the PRO-CTCAE during the first 6 months of treatment were mostly greater than 80% at each week in both treatment groups. Patients in the selpercatinib group reported a lower PTT with severe or very severe symptomatic adverse events, as measured by the PRO-CTCAE, compared with the control group. These included diarrhea (5% vs. 38%), hand-foot syndrome (2% vs. 9%), decreased appetite (2% vs. 15%), nausea (1% vs. 8%), fatigue (6% vs. 21%), taste change (3% vs. 15%), mouth/throat sores (3% vs. 10%), and acne (1% vs. 7%) (all p < 0.001). Both treatments showed comparable PTT with severe or very severe constipation (3% vs. 5%), vomiting (0% vs. 1%), headache (4% vs. 8%), and dry mouth (10% vs. 13%) (Fig. 6).
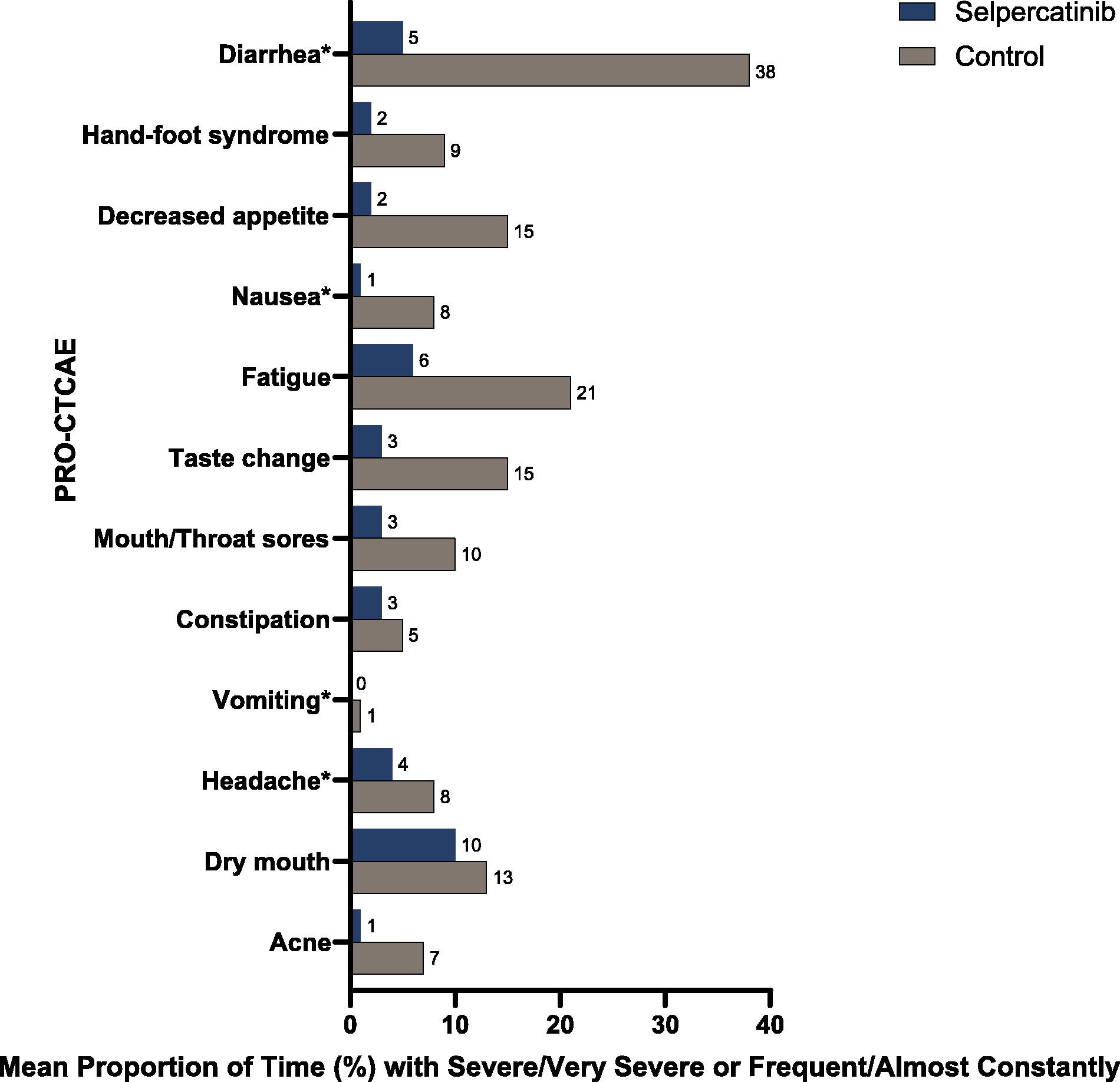
Mean proportion of time with severe to very severe categories on symptomatic adverse events, as measured from PRO-CTCAE. Pre-specified 13 symptomatic AEs based on reported frequent toxicities from selpercatinib, cabozantinib, and vandetanib in relevant clinical trials were analyzed. The mean proportion of time that patients experienced severe to very severe symptomatic adverse events was assessed using the PRO-CTCAE score 3 or 4 and compared between the two treatment groups. Analyzed in the tolerability evaluable population, which required patients to be on treatment for at least six months. The severity attribute is reported as: (1) frequency (for diarrhea, nausea, vomiting, headache, dry mouth) is scored as: never (score = 0), rarely (score = 1), occasionally (score = 2), frequently (score = 3), and almost constantly (score = 4); (2) severity (for hand-foot syndrome, appetite, nausea, fatigue, taste change, mouth/throat sores, constipation, vomiting, headache, and acne) is scored as: no side effects (score = 0), mild (score = 1), moderate side effects (score = 2), severe side effects (score = 3) and very severe side effects (score = 4); (3) interference (for appetite, fatigue, mouth/throat sores, headache) is scored as not at all (score = 0), a little bit (score = 1), somewhat (score = 2), quite a bit (score = 3), and very much (score = 4). All p < 0.001 except for constipation, vomiting, headache, and dry mouth. PRO-CTCAE, Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events. * Side effects with “frequently” to “almost constantly” scores (frequency score 4 or 5).
In the PRO evaluable population, post-baseline compliance rates of the Worst Pain Numeric Rating Scale were mostly greater than 80% at each week in both treatment groups. A higher proportion of patients reported worsening of weekly average of worst pain, as measured by the Worst Pain Numeric Rating Scale, in the control group compared with the selpercatinib group (Fig. 7).
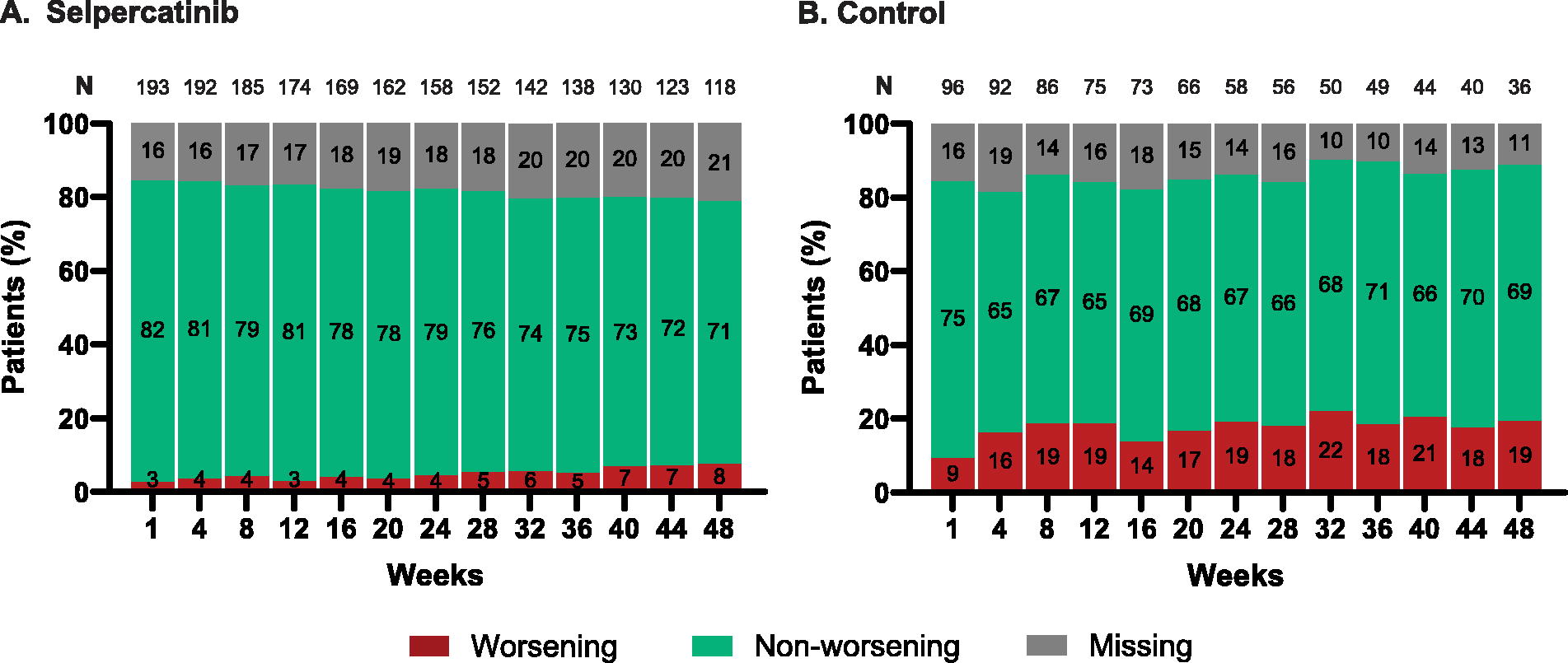
Proportion of patients with worsening and non-worsening pain from baseline. “Worsening” was defined as an increase of ≥2 points, and “non-worsening” as a change <2 points. N, number of expected patients.
Stool consistency and bowel movement frequency
In the PRO evaluable population, post-baseline compliance rates of the Bristol Stool Form Scale were mostly greater than 85% at each week in both treatment groups. At baseline, median scores indicated normal stools and were comparable between the two groups (4.3 [Q1–Q3: 3.6–5.0] in selpercatinib vs. 4.7 [Q1–Q3: 3.8–5.6] in control). After week 4, median weekly scores increased consistently in the control group. At week 18, the median weekly score in the control group reached 6 (Q1–Q3: 5.0–6.7), indicating diarrhea (versus 4 [Q1–Q3: 3.4–4.9] in the selpercatinib group) and remained close to 6 through week 48. Median weekly scores with selpercatinib were the same as baseline or lower, indicating normal stool. Related, at baseline, median bowel movement count was reported higher in the control group than the selpercatinib group (2.0 [Q1–Q3: 1.1–3.4] vs. 1.7 [Q1–Q3: 1.1–2.5]). In the PRO evaluable population, post-baseline, compliance rates of the Bowel Movement Count were greater than 85% at each week in both treatment groups. Post-baseline, median weekly bowel movement counts were consistently lower than baseline in the selpercatinib group (ranging from 1.3 to 1.6). In the control group, median weekly bowel movement counts post-baseline ranged between 1.7 and 2.7.
Discussion
To our knowledge, LIBRETTO-531 is the first to include the prespecified, alpha-controlled analysis of comparative PRT, a novel endpoint designed to quantify tolerability by capturing the individual patient-reported severity of burden by treatment side-effects while on treatment. This aligns with the current interest of regulatory agencies in evaluating the patient’s perspective on treatment tolerability. 16 Alpha-controlled PRO endpoints remain uncommon in oncology, leading to continued uncertainty as to how patients experience cancer treatments. In this analysis, first-line selpercatinib showed superior tolerability compared with the control treatment in patients with advanced, RET-mutant MTC. MTC is a chronic disease with a high predilection for distant metastases. Patients who have significant progression require long-term systemic therapy for adequate control; thus, there is great benefit in understanding the tolerability of treatment side effects.
A key aspect of the PRT endpoint was the choice of GP5 as a simple index of burden. Given the different types and severities of adverse events between selpercatinib and control treatments, it is complicated to determine which side effect profile imposes a bigger burden to patients. 17 GP5 helped adjudicate the variability in adverse effects to each treatment (selpercatinib and control) allowing for comparison with the same metric (proportion of time with high side-effect burden). Our PRT approach is consistent with the recent estimand framework for comparative tolerability. 18 The PRT analysis was based on the initial treatment assignment excluding the crossover period.
Complementary PRO results on symptomatic AEs and HRQoL were consistent with the comparative PRT findings. Patients on selpercatinib reported significantly less PTT with clinically meaningful HRQoL impairment compared with control despite a much longer duration of treatment. A higher proportion of selpercatinib patients also had clinically meaningful improvement or stable scores on the overall quality of life and functioning scores. Notably, patients treated with selpercatinib reported less PTT with severe or very severe symptomatic adverse events, including severe or very severe diarrhea (5% vs. 38% with control), a common paraneoplastic syndrome in patients with MTC. Thus, resolution or improvement in diarrhea scores indicates a good clinical response to drug therapy. Both treatment groups showed comparable PTT with severe or very severe constipation and dry mouth.
In terms of limitations, PRT cannot differentiate between more severe adverse events occurring later in treatment and intermittent adverse events for those with the same treatment duration. The open-label study design introduces the potential for bias in the collection of PRO data. However, we found that GP5 responses were well correlated with HRQoL (measured by the EORCT QLQ-C30 functioning scales), hospitalization events, and symptomatic adverse events (measured by PRO-CTCAE) in the prespecified psychometric analysis using blinded, pooled data from LIBRETTO-531. 13 This was consistent with published literature using real world data across multiple cancer types 4 and from industry-sponsored oncology clinical trial data. 6 In this analysis there were no PRO modules specific for thyroid cancer; however, as such this enhances the generalizability and broader applicability across randomized cancer studies beyond thyroid trials.
This analysis from the LIBRETTO-531 study showed superior comparative PRT for selpercatinib in patients with RET-mutant MTC compared with vandetanib/cabozantinib. These results complement the improved PFS and TFFS previously reported and further support selpercatinib use as the first-line standard of care for patients with advanced, RET-mutant MTC. This simplified, quantifiable PRT metric deserves further adoption as a complement to traditional clinical endpoints (e.g., PFS and overall survival) in future randomized-controlled clinical trials, especially in diseases with historically better outcomes where tolerability becomes a key factor in treatment success.
Footnotes
Acknowledgments
The authors thank the investigators and site personnel for their participation in the study, as well as the clinical study participants and their families and caregivers, without whom this work would not be possible. We thank Jennifer Wright, Lisa M. Hess, Boris Kin Lin, and Victoria Soldatenkova of Eli Lilly and Company, and Antoine Regnault, of Modus Outcomes, for their valuable insights in the conceptualization of comparative patient-reported tolerability.
Authors’ Contributions
R.E.: Formal analysis (equal), resources (equal), writing—reviewing and editing (equal). L.J.W.: Conceptualization (equal), methodology (equal), formal analysis (equal), resources (equal), writing—reviewing and editing (equal). J.C.: Formal analysis (equal), resources (equal), writing—reviewing and editing (equal). A.O.H.: Resources (equal), writing—reviewing and editing (equal). M.T.: Formal analysis (equal), writing—reviewing and editing (equal). E.J.S.: Formal analysis (equal), resources (equal), writing—reviewing and editing (equal). M.I.H.: Conceptualization (equal), formal analysis (equal), writing—reviewing and editing (equal). M.-H.G.: Formal analysis (equal), writing—reviewing and editing (equal). J.W.: Resources (equal), writing—reviewing and editing (equal). F.V.: Resources (equal), writing—reviewing and editing (equal). K.K.: Formal analysis (equal), resources (equal), writing—reviewing and editing (equal). J.K.: Formal analysis (equal), writing—reviewing and editing (equal). D.O.: Formal analysis (equal), writing—original draft (equal). C.C.: Conceptualization (equal), formal analysis (equal), writing—original draft (equal), writing—reviewing and editing (equal). P.M.: Formal analysis (equal), writing—original draft (equal), writing—reviewing and editing (equal). A.M.G.: Methodology (equal), formal analysis (equal), writing—original draft (equal), writing—reviewing and editing (equal). Y.L.: Formal analysis (equal), writing—reviewing and editing (equal). N.P.: Formal analysis (equal), writing—original draft (equal), writing—reviewing and editing (equal). B.R.: Resources (equal), writing—reviewing and editing. J.H.: Formal analysis (equal), resources (equal), writing—reviewing and editing (equal). M.S.B.: Formal analysis (equal), writing—reviewing and editing (equal).
Data Sharing Statement
Eli Lilly and Company provides access to all individual data collected during the trial, after anonymization, with the exception of pharmacokinetic, genomic, or genetic data. Data are available to request 6 months after the indication studied has been approved in the United States and European Union and after primary publication acceptance, whichever is later. No expiration date of data requests is currently set once data are made available. Access is provided after a proposal has been approved by an independent review committee identified for this purpose and after receipt of a signed data sharing agreement. Data and documents, including the study protocol, statistical analysis plan, clinical study report, and blank or annotated case report forms, will be provided in a secure data sharing environment. For details on submitting a request, see the instructions provided at ![]() .
.
Author Disclosure Statement
R.E. reports consultancy role for EISAI, Eli Lilly, BAYER, ROCHE, and IPSEN. L.J.W. reports fees from Bayer HealthCare Pharmaceuticals Inc., EISAI INC., Eli Lilly and Company, Ellipses, EMD Serono, Exelixis Inc., Illumina, Merck, Nested, and PDS Biotechnology Corporation. J.C. reports consultancy role in Novartis, Pfizer, Ipsen, Exelixis, Bayer, Eisai, Advanced Accelerator Applications, Amgen, Sanofi, Lilly, Hutchmed, ITM, Merck Serono, Roche, Esteve, Advanz; and research support from Novartis, Pfizer, Astrazeneca, Advanced Accelerator Applications, Eisai, Amgen, Gilead, Roche, Ipsen/Exelixis, ITM, and Bayer. A.O.H. reports research support from Eli Lilly, Novartis, Exelixis; advisory role for Knight and Eli Lilly and Company; and medical education provider for Bayer. M.T. reports advisory board role in AstraZeneca, Boehringer Ingelheim, Eisai, Eli Lilly, Merck KGaA, Merck Sharp, and Dohme. E.J.S. reports consultancy role in Eli Lilly, Eisai, Regeneron, AffyImmune; and research support from Eli Lilly, Regeneron. M.I.H. reports research support to institution from Eli Lilly and Company; and advisory role in Eli Lilly and Company. M.-H.G. declares no conflicts of interest. J.W. reports fees to institution for speaker/advisor fees from Lilly, Novartis, Eisai, AstraZeneca, Ipsen, Bayer, Roche and Incyte. F.V. reports advisory board role for Sanofi-Genzyme, Lilly, and Ipsen; speaker for Sanofi-Genzyme, Knight, Ipsen, Lilly, Merck, Bayer, and Onkos. Principal Investigator in clinical trials sponsored by AstraZeneca, Bayer, Lilly, Exelixis, and Novartis. K.K. reports consultancy role for Eli Lilly and Company. J.K. reports consultant role for Bayer Health Care, Ewopharma, Exelixis, Ipsen, Loxo, Lilly, Sanofi-Genzyme. Honoraria (clinical trials) and travel grants: AstraZeneca, Bayer Health Care, Eisai, Exelixis, Ipsen, Lilly, Novartis, Sanofi-Genzyme. D.O., C.C., P.M., A.M.G., Y.L., and N.P. report employment with Eli Lilly and Company with stock options. B.R. reports advisory role for Lilly, Exelixis, Bayer and Eisai. J.H. reports honoraria for speaker engagements, advisory roles or funding of continuous medical education from Eli Lilly, AAA, IPSEN, Roche, Pharma Mar, EISAI. Research grants from Novartis. Commercial medical education provider for AAA. Leadership roles in medical societies GTE, ATA, ITOG, and Co-Coordinator of the French network on refractory thyroid cancer, ENDOCAN-TUTHYREF. M.S.B. reports consultancy role for Aadi Bioscience, Bayer HealthCare Pharmaceuticals Inc., Blueprint Medicines Corporation, EISAI INC., Eli Lilly and Company, Exelixis Inc., Ipsen Biopharm Limited,
Funding Information
Supported by Loxo Oncology, a wholly owned subsidiary of Eli Lilly and Company.
Supplementary Material
Supplementary Appendix
Supplementary Tables
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
