Abstract
Background:
Thyroglobulin (Tg), stimulated or unstimulated by recombinant human thyrotropin (TSH), is a static marker of recurrent or persistent disease, and the Tg-doubling rate (Tg-DR) is a dynamic prognostic factor. This study evaluated the prognostic value of an unstimulated Tg (uTg) and Tg-DR papillary thyroid carcinoma (PTC).
Methods:
This retrospective study included 1818 Tg antibody (Tg-Ab)-negative patients who underwent curative intent total thyroidectomy for PTC without distant metastasis. The uTg was measured 1–3 months post-surgery under TSH suppression (<0.1 mIU/mL). We calculated the Tg-DR for patients, of whom postoperative Tg levels could be measured three or more times under TSH suppression.
Results:
Eighty-eight (4.8%) and 32 (1.8%) patients had respective local and distant recurrences (median follow-up period, 7.2 years; 25th percentile 4.7 years, 75th percentile 9.8 years). Of 1818 patients, 131 had a uTg ≥3 ng/mL and were more likely to display local and distant recurrences in univariate and multivariable analyses (p < 0.001). We divided 1212 patients with no adjuvant radioactive iodine treatment, of whom uTg and Tg-DR data were available, into four categories A, uTg ≥3 ng/mL and Tg-DR ≥0.33/year; B, uTg <3 ng/mL and Tg-DR ≥0.33/year; C, uTg ≥3 ng/mL and Tg-DR <0.33/year; and D, uTg <3 ng/mL and Tg-DR <0.33/year. The lymph node recurrence-free survival rate was significantly worse from category A to D (A vs. B, p < 0.001, hazard ratio or HR [CI]: 5.083 [1.994–12.955]; B vs. C, p = 0.001, HR [CI]: 2.654 [1.462–4.824]; C vs. D, p < 0.001, HR [CI]: 27.420 [15.100–4.980]). The distant recurrence-free survival rate (DR-FS) of category B did not differ from that of category C (p = 0.419), but DR-FS of category D was better (p < 0.001) than those of B and C, and that of category A tended to be worse (p = 0.087) compared with those of B and C. Patients in category A, categories B and C, and category D could thus be classified as high-risk, intermediate-risk, and low-risk for distant recurrence, respectively.
Conclusions:
This study demonstrates the prognostic value of postoperative uTg and Tg-DR in Tg-Ab-negative patients with PTC under TSH suppression after total thyroidectomy. Prospective studies are needed to confirm these findings.
Introduction
Although papillary thyroid carcinoma (PTC) is generally indolent, several static markers for prognosis have been identified. 1 –3 Serum thyroglobulin (Tg) level suggests disease persistence in patients who undergo total thyroidectomy and are negative for the anti-Tg antibody (Tg-Ab). The guidelines of the American Thyroid Association recommend evaluating Tg levels under stimulation by recombinant human thyrotropin (rhTSH) for postoperative follow-up. 4
In Europe and the United States, the indication of adjuvant radioactive iodine (RAI) treatment is based on the initial risk of recurrence, integrating stimulated or unstimulated Tg (uTg) levels. 4,5 In Japan, adjuvant RAI administration is utilized less often than in Western countries because hospitalization to a specially prepared room is mandatory for patients who undergo administration of RAI >30 mCi, and the capacity for the treatment room is limited. Furthermore, postoperative follow-up using stimulated Tg is uncommon, and disease progression after total thyroidectomy is evaluated by uTg. Miyauchi et al. first focused on the serial change in uTg levels as a prognostic factor and demonstrated that short Tg-doubling time (Tg-DT; <1 and <3 years) was significantly associated with the prognosis of Tg-Ab-negative patients following total thyroidectomy. 6 DT is a well-recognized method for analyzing and expressing changes in serum tumor marker levels or tumor volumes over time. However, it has two major limitations: (1) if some patients show a decrease in these values over time, their DTs are given negative values, resulting in discontinuities compared with DTs with positive values, and (2) the DT values are inverse to the growth rate values. To resolve these limitations, Miyauchi et al. proposed the inverse of DT (1/DT) and nominated this index as the doubling rate (DR). DR is more suitable for evaluating patient prognosis because it resolves the two limitations of DT. 7
In Japan, PTC has an excellent prognosis similar to that in other countries, and only a small proportion of tumors show locoregional or distant recurrence. Here, we investigated whether and how the combination of postoperative uTg and Tg-DR is associated with local and distant recurrences in patients with PTC.
Materials and Methods
Ethics statement
This retrospective cohort study was approved by the Ethics Committee of Kuma Hospital (Ethical Committee Certification number: 20200709-1). All patients provided signed written consent for study participation.
Patients
Between 2012 and 2022, 3385 patients with PTC underwent initial surgery at Kuma Hospital. All patients were diagnosed with PTC on the basis of pathological examination. We excluded patients who tested positive for Tg-Ab, with distant metastasis at surgery (M1), with other coexisting thyroid malignancies, who underwent less than a total thyroidectomy, who underwent locally noncurative surgery, and those who were followed up for <1-year post-surgery. The details of participant flow are shown in Figure 1. Notably, we excluded 1567 patients with TSH ≥0.1 µIU/mL and 573 patients whose Tg under TSH suppression was measured fewer than three times because of missing data.
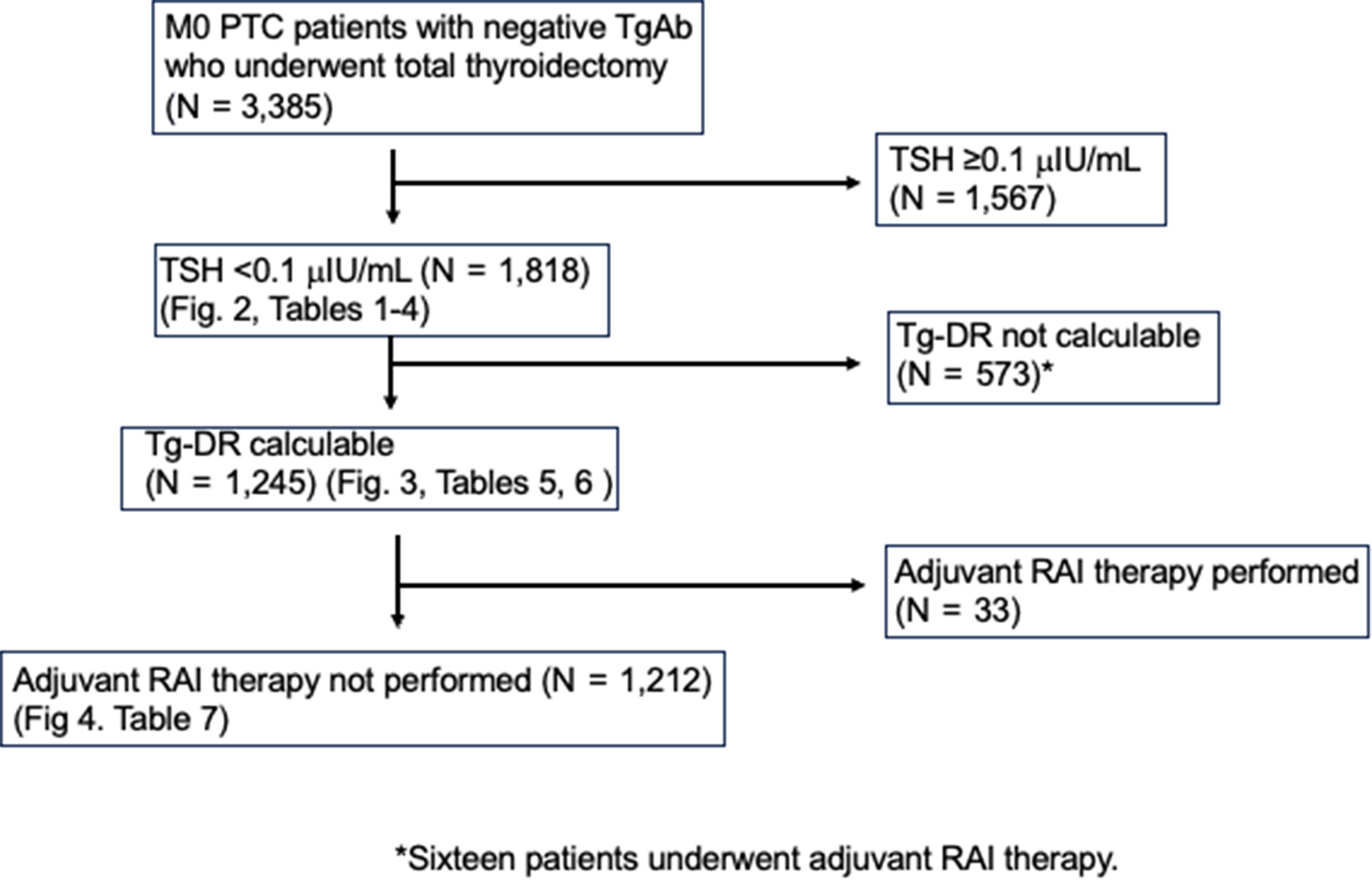
Flowchart of study patients. PTC, papillary thyroid carcinoma; RAI, radioactive iodine; Tg-Ab, antithyroglobulin antibody; TSH, thyrotropin; Tg-DR, thyroglobulin doubling rate.
Patients were asked to visit our outpatient clinic every 6 months or at least once a year if their condition was stable for blood examination for TSH, free triiodothyronine, Tg, and, sometimes, Tg-Ab. Ultrasonography was performed at least once a year to screen for local recurrence, and systemic examinations, such as computed tomography (CT) scan and positron emission tomography–CT, were also performed according to clinicians’ discretion. The Tg values collected for this study were all unstimulated (i.e., uTg).
Calculations of Tg-DT and Tg-DR
The formula to calculate DT has been previously described. 6 We provided it on our website for researchers worldwide (https://www.kuma-h.or.jp/kumapedia/kuma-medical/detail/?id=290) as the “Doubling time, Doubling Rate & Progression Calculator.” We calculated Tg-DT for patients who had their Tg measured three times or more under a TSH value of <0.1 mIU/mL. 6 Undetectable Tg levels were excluded from the calculations. DR is calculated as 1/DT to combat its aforementioned limitations. Therefore, for further analysis, we used Tg-DR. For analysis of Tg-DR, we adopted Tg data only when the TSH level was <0.1 mIU/mL, 6 and patients whose Tg were measured less than three times were excluded.
RAI administration
The use of RAI for remnant ablation (defined by an administered dose of approximately 30 mCi) or adjuvant treatment (defined by an administered dose of ≥100 mCi) was based on individual clinicians’ discretion. RAI adjuvant treatment was generally reserved for patients with PTC of advanced clinicopathological stage. In such cases, we calculated the Tg-DR at least 3 months after RAI administration.
Measurements of Tg, Tg-Ab, and TSH
For Tg measurement, we adopted Elecsys Tg and Tg II (Electrochemiluminescence Immunoassay; Roche Diagnostics, Rotkreuz, Switzerland) from 2012 to March 2015 and April 2015, respectively (the lowest functional sensitivity, 0.04 ng/mL). For Tg-Ab measurements, Elecsys anti-Tg electrochemiluminescence immunoassay (Roche Diagnostics) was used. For TSH measurement, Architect TSH (Chemiluminescence Immunoassay; Abbott, Abbott Park, IL), Elecsys TSH, and Elecsys TSH version 2 (Electrochemiluminescence Immunoassay; Roche Diagnostics) were used from 2012 to 2018, 2019, October 2020, and November 2020, respectively.
Statistical analyses
The primary and secondary outcomes were distant and local recurrence-free survival. We adopted clinicopathological factors regarded as high-risk according to the guidelines of the Japan Association of Endocrine Surgery (such as node metastasis [cN] >3 cm, extrathyroidal extension corresponding to T4a, extranodal tumor extension [LNEx], and tumor size >4 cm). 8 The primary and secondary outcomes were distant and local recurrence-free survival. We used the chi-square test to compare variables and developed a multivariable logistic regression model for multivariable analysis. For time series, the Kaplan–Meier method with log-rank test was used as univariate analysis in Figures 2 and 3. Since the proportional hazard hypothesis could not be established by the Schoenfeld residuals test according to the Grambsch and Therneau test (Fig. 4), we adopted p values and hazard ratios (HRs) from the Cox proportional hazard model instead of the log-rank test. The multivariable analysis for time series was performed using the Cox regression hazard model, and factors in univariate analysis (<0.20) were used in multivariable analysis. Missing data were excluded from the analyses. Statistical significance was set at p < 0.05, and we regarded 0.05 ≤ p < 0.1 as having a strong tendency. All analyses were performed and confirmed by a statistician (M.Y.) using R software.
Results
Prognostic value of uTg ≥3 ng/mL after total thyroidectomy (on TSH suppressive therapy)
Table 1 shows the background and clinical features of the 1818 patients included in the study. Our study included 327 Stage III, 263 Stage II, and 1228 Stage I cases. Overall, 131 patients (7.2%) had uTg ≥3 ng/mL after total thyroidectomy. Table 2 reports the relationship between uTg levels and clinicopathological features. High uTg levels were associated with tumor size >4 cm (p = 0.002), tumor extension corresponding to T4a (p < 0.001), clinical node metastasis (cN) >3 cm (p < 0.001), positive for LNEx (p < 0.001), and age <55 years (p = 0.032)—a well-known favorable prognostic factor in contrast to the former four.
Background and Clinical Features of 1818 Patients
Relationship Between Clinicopathological Features and Postoperative Unstimulated Thyroglobulin Values
LNEx, extranodal extension of metastatic lymph nodes; uTg, unstimulated thyroglobulin.
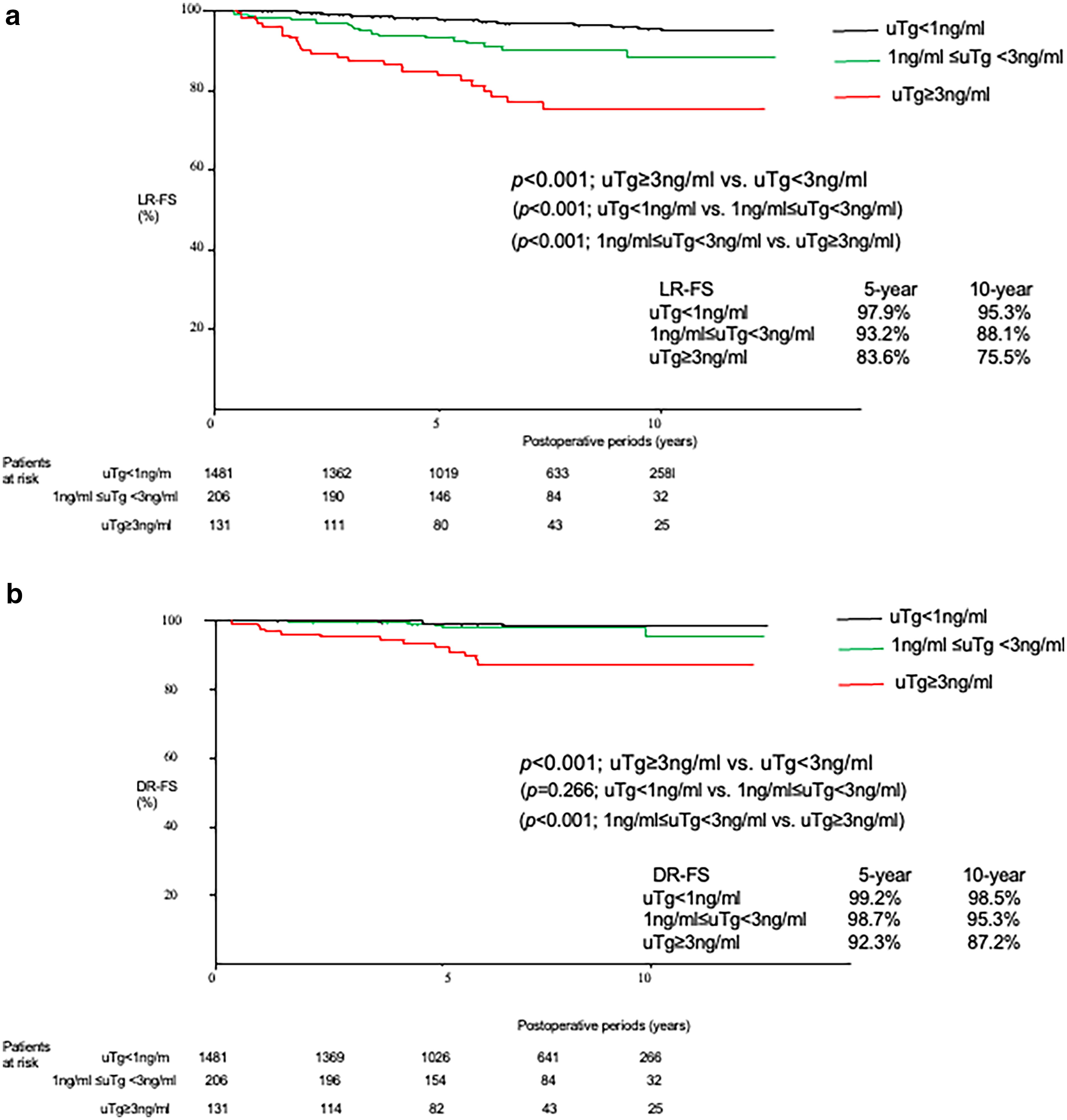
The follow-up period ranged from 1 to 12.7 years (median follow-up period, 7.2 years; 25th percentile 4.7 years, 75th percentile 9.8 years). To date, 88 and 32 patients have experienced disease recurrence in local and distant organs, respectively. Figure 2 shows the difference in prognosis according to the uTg values. The local recurrence-free survival rate (LR-FS) was observed to be significantly worse according to uTg in the following order: uTg <1 ng/mL, 1 ng/mL ≤ Tg < 3 ng/mL, and uTg ≥3 ng/mL (10-year LR-FS [confidence interval or CI]: 95.3% [93.2–96.7], 88.1% [82.3–94.0], and 75.5% [66.8–84.1], respectively, p < 0.001; Fig. 2a), and the distant recurrence-free survival rate (DR-FS) of patients with uTg ≥3 ng/mL was significantly poorer (10-year LR-FS [CI]: 87.2% [80.6–87.9]) than that of those with uTg <1 ng/mL and with 1 ng/mL ≤ uTg < 3 ng/mL (10-year LR-FS [CI]: 98.5% [97.6–99.3] and 95.3% [89.7–100], respectively, p < 0.001; Fig. 2b).
Tables 3 and 4 summarize the results of univariate and multivariable analyses for static factors associated with local and distant recurrences, respectively. We found that uTg ≥3 ng/mL was independently associated with local (p < 0.001, HR and [CI]: 5.00 [3.28–7.63]) and distant recurrences (p < 0.001, HR [CI]: 8.37 [4.61–15.20]), respectively (after adjustment with some conventional prognostic factors).
Univariate and Multivariable Analyses Results of Static Factors of the Local Recurrence of Papillary Thyroid Carcinoma
Factors with p < 0.20 in the univariate analysis were adopted for the multivariable analysis. The p value of male sex could not be calculated using a forward–backward stepwise selection method.
cN, clinical lymph node metastasis; CI, confidence interval; PTC, papillary thyroid carcinoma.
Univariate and Multivariable Analyses Results of Static Factors of Distant Recurrence of Papillary Thyroid Carcinoma
Factors with p < 0.20 in the univariate analysis were adopted for the multivariable analysis. However, the p values of uTg >3 ng/mL and cN >3 cm could not be calculated using a forward–backward stepwise selection method.
Prognostic value of Tg-DR ≥0.33/year after total thyroidectomy
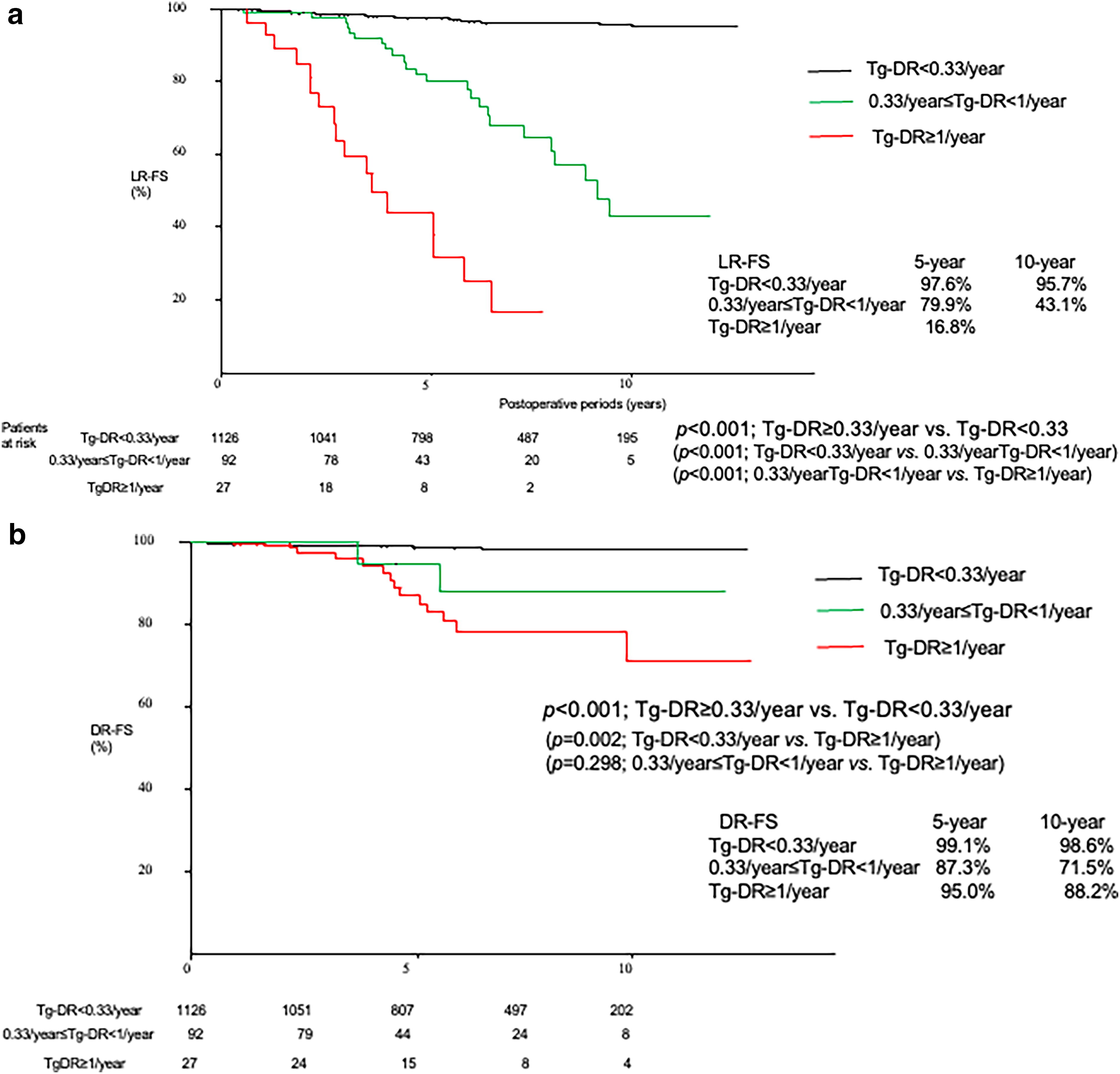
In our study, 119 of the 1245 patients (9.6%) in whom Tg-DR values were calculated had Tg-DR ≥0.33/year. Tg-DR ≥0.33/year was significantly associated with age ≥55 years (in contrast to uTg ≥3 ng/mL), tumor >4 cm, and T4a tumors according to the chi-square test (Table 5). However, Tg-DR ≥0.33/year was not associated with uTg ≥3 ng/mL (p = 0.448), suggesting that these two values reflected different oncological aspects—the tumor growth rate (a dynamic factor) and the residual tumor volume (a static factor), respectively. In multivariable logistic analysis, Tg-DR ≥0.33/year was independently associated with age >55 years (p < 0.001, HR [CI]: 2.265 [1.507–3.406]) and tumor exceeding 4 cm in size (p < 0.001, HR [CI]: 2.733 [1.605–4.651]) (Table 6). LR-FS was observed to be significantly worse (p < 0.001) in the order of patients with Tg-DR <0.33/year, 0.33/year ≤ Tg-DR <1/year, and Tg-DR ≥1/year (10-year LR-FS [CI]: 95.7% [94.2–97.2], 43.1% [24.9–61.3], and 0%, respectively, p < 0.001; Fig. 3a). Figure 3b shows the DR-FS for these three subsets. The 10-year DR-FSs [CI] of patients with Tg-DR <0.33/year, 0.33/year ≤ Tg-DR < 1/year, and Tg-DR ≥1/year were 98.6% [97.8–99.4], 71.5% [54.7–88.2], and 88.2% [72.6–100], respectively. Patients with Tg-DR <0.33/year had significantly better DR-FS than those with Tg-DR ≥0.33/year (p < 0.001).
Relationship Between Postoperative Thyroglobulin DR and Clinicopathological Features a
Patients for whom Tg-DR could not be calculated were excluded (N = 573).
Tg-DR, thyroglobulin doubling rate.
Multivariable Logistic Analysis Results for Factors Relating to Thyroglobulin Doubling Rate ≥0.33/Year
Factors with p < 0.20 in the univariate analysis were adopted for the multivariable analysis.
Subject to tumor-node-metastasis (TNM) staging.
Patients who underwent adjuvant RAI administration
Only 49 of 1818 patients (2.7%) underwent adjuvant RAI administration (≥100 mCi), and 291 (16.0%) underwent remnant thyroid ablation (30–50 mCi). Patients with large tumors (>4 cm) (p = 0.001), T4a tumors (p < 0.001), large cN1 (p = 0.001), LNEx (p < 0.001), and with uTg ≥3 ng/mL (p < 0.001) more often received adjuvant RAI administration. The LR-FS and DR-FS were poorer (p < 0.001) in patients who underwent adjuvant RAI administration than in other patients. The LR-FS of patients who underwent RAI ablation was worse than that of those who did not undergo RAI ablation or undergo only scintigraphy (≤13 mCi) (p < 0.001, HR [CI]: 4.56 [2.87–7.26]) and that of those who underwent adjuvant RAI administration was worse than that of those with RAI ablation (p = 0.031, HR [CI]: 2.06 [1.07–3.98]). The DR-FS of patients with RAI ablation was worse than that of those without RAI administration or with scintigraphy (p < 0.001, HR [CI]: 37.93 [11.05–130.20]) and that of those with adjuvant RAI administration was worse than that of those with RAI ablation (p < 0.001, HR [CI]: 3.81 [1.83–7.93]).
Prognosis of patients classified into four categories based on uTg and Tg-DR levels
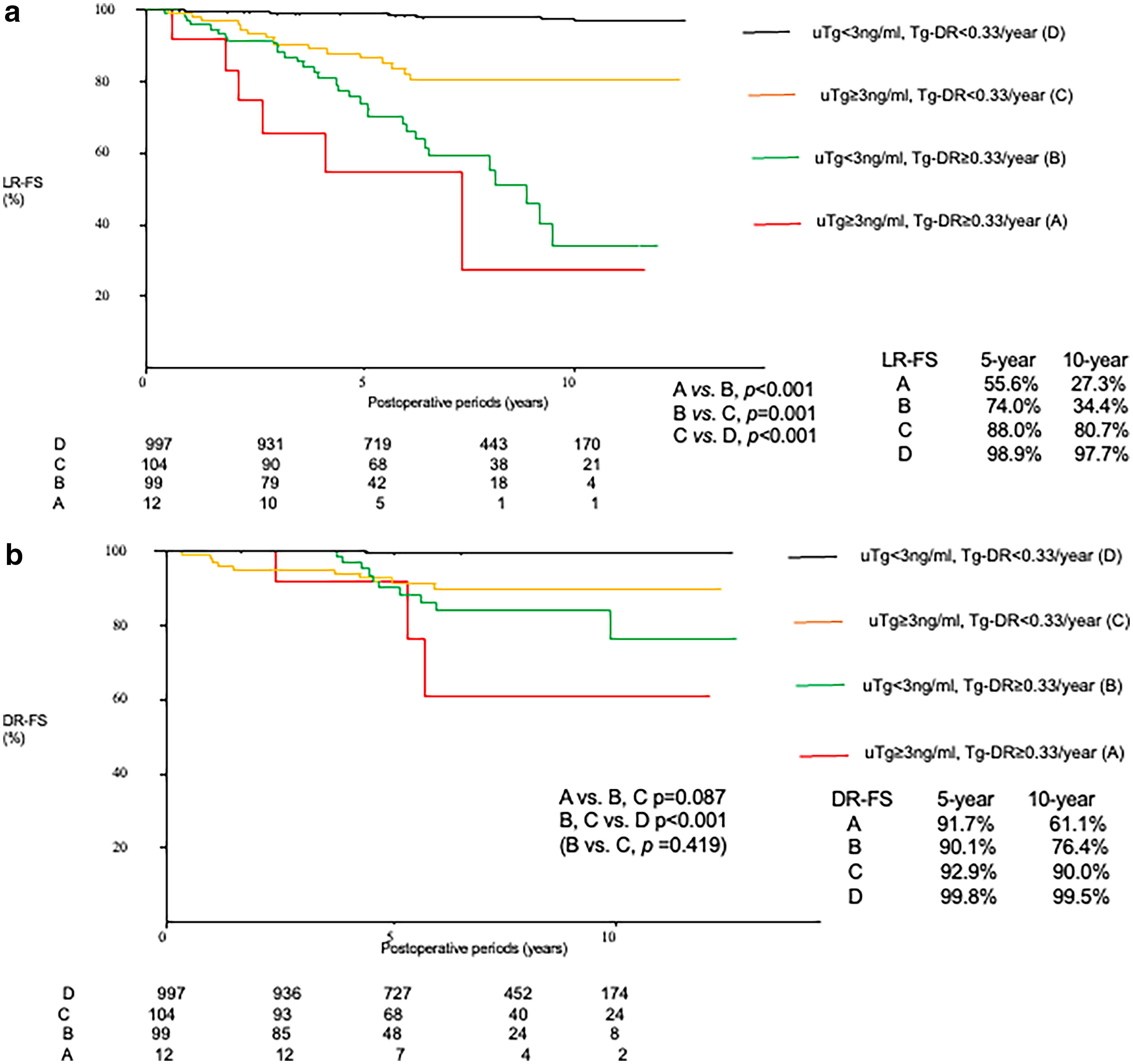
For further analysis, we included 1212 patients (including 195 PTC ≤10 mm) who did not receive adjuvant RAI and for whom Tg-DR could be calculated. This subgroup analysis included 291 patients who underwent small amounts of RAI (30 mCi) for the purpose of remnant thyroid ablation. Patients who underwent adjuvant RAI administration (≥100 mCi) were removed from the analysis (N = 33). We classified our patients into four categories: patients with uTg ≥3 ng/mL and Tg-DR ≥0.33/year (category A, N = 12), patients with uTg <3 ng/mL and Tg-DR ≥0.33/year (category B, N = 99), patients with uTg ≥3 ng/mL and Tg-DR <0.33/year (category C, N = 104), and patients with uTg <3 ng/mL and Tg-DR <0.33/year (category D, N = 997). Figure 4a shows the LR-FSs of patients in these four groups. The LR-FS was poorer in patients with high Tg-DR (categories A and B) than in those with low Tg-DR (categories C and D), and in both groups, patients with high uTg showed poorer LR-FS than those with low uTg (category A vs. B and category C vs. D), indicating that the LR-FS became poor in order of categories D, C, B, and A. Ten-year LR-FS of patients of categories A, B, C, and D were 72.7% (CI: 31.9–99.9), 65.4% (46.1–84.3), 19.3% (10.8–27.8), and 2.3% (1.2–4.6), respectively. The LR-FS was observed to be significantly better from category A to D (A vs. B, p < 0.001, HR [CI]: 5.083 [1.994–12.955]; B vs. C, p = 0.001, HR [CI]: 2.654 [1.462–4.824]; C vs. D, p < 0.001, HR [CI]: 27.420 [15.100–4.980]).
DR-FSs of those groups are shown in Figure 4b. Ten-year distant recurrence rates (DRRs) of patients in categories A, B, C, and D were 38.9% (CI: 2.8–75.0), 23.6% (6.8–40.1), 10.0% (3.7–16.3), and 0.5% (0.0–1.1), respectively. The DR-FS in category A patients tended to be worse (p = 0.063) compared with that in category B patients, and the DR-FS in category D patients was significantly better (p < 0.001) than that in category C patients; however, the DR-FS of patients in categories B and C did not differ from each other (p = 0.419). Subsequently, we reanalyzed the DR-FS after combining categories B and C into a single group. The DR-FS of category A patients tended to be poor compared with that of category B or C patients (p = 0.087, HR [CI]: 2.899 [0.856–9.811]), and the DR-FS of category B and C patients was significantly worse than that of category D patients (p < 0.001, HR [CI]: 27.166 [9.237–79.872]). Taken together, the risk of distant recurrence could be divided into three categories based on uTg and Tg-DR: high-risk (category A), intermediate-risk (categories B and C, 10-year DRR [CI]: 14.9% [76.1–91.0]), and low-risk (category D).
Discussion
Previous studies have shown the prognostic value of uTg and Tg-DR/Tg-DT. Sun et al. reported that uTg >10.1 ng/mL was associated with a poorer prognosis in patients who underwent reoperation for structural locoregional recurrences. 9 However, uTg as a prognostic factor requires further elucidation. Here, we demonstrated that uTg ≥3 ng/mL following total thyroidectomy had an independent prognostic value for LR-FS and DR-FS in multivariable analysis. Following Miyauchi et al.’s presentation of the prognostic significance of Tg-DR/Tg-DT, 6 other studies began to depict Tg-DR/Tg-DT as a prognostic factor for patients with differentiated thyroid carcinoma (DTC). Rossing et al. showed that the mortality risk of patients with DTC with a Tg-DT <5 months, compared with a Tg-DT of >14 months, was more than twofold. 10 Zhang et al. illustrated that short Tg-DT of the recent four data had a high correlation with patients with progressive metastatic DTC and those who underwent RAI therapy. 11 Miyauchi et al. reported that short Tg-DT (large Tg-DR) was associated with high Ki-67 labeling index and low disease-free survival in patients with PTC. 12 Ito et al. also demonstrated that high Tg-DR, along with high Ki-67 labeling index, significantly predicted excision-site recurrence of PTC following airway resection. 13
A value of uTg ≥3 ng/mL is more often detected in patients with some conventional factors predicting poor prognosis: large tumor, T4a tumor, large node metastasis, and LNEx (Table 2). 1 –3 Regarding patient age, uTg ≥3 ng/mL tended to occur more frequently in patients <55 years old, which is a sign of favorable prognosis. However, univariate and multivariable analyses (Tables 5 and 6) more frequently presented the detection of high levels of Tg-DR in patients ≥55 years than in those <55 years. This suggests that younger patients with recurrence often have well-differentiated PTC, which remains treatable (RAI or surgery), leading to a better prognosis. Older patients may have less differentiated PTC that is more difficult to treat, particularly if RAI resistance develops.
Measurements of uTg ≥3 ng/mL and Tg-DR ≥0.33/year were associated with worse LR-FS and DR-FS (Figs. 2 and 3). Additionally, in combination analysis, study patients could be clearly divided into four groups according to the LR-FS; patients with high Tg-DR showed a poorer LR-FS than those with low Tg-DR, and in these Tg-DR-high and -low groups, high uTg groups showed a poorer LR-FS than patients with low uTg (Fig. 4a). These observations suggest that Tg-DR may be more important for predicting patients’ LR-FS, whereas uTg may play a subsidiary role. For the DR-FS, patients with low uTg and Tg-DR showed the best prognosis, whereas those with high uTg and Tg-DR had the poorest prognosis. However, patients with high uTg and low Tg-DR and those with low uTg and high Tg-DR showed similar DR-FSs (Fig. 4b), which suggests that dividing patients into three groups is appropriate for evaluating the DR-FS: high-risk (high uTg and Tg-DR), intermediate-risk (high uTg and low Tg-DR or low uTg and high Tg-DR), and low-risk (low uTg and Tg-DR). Indeed, the DR-FS of these three risk groups significantly differ, as shown in Figure 4b.
This study has some limitations. First, the study is retrospective, and there may be some bias in the treatments (particularly relating to RAI use, as this was not standardized). Second, we needed to exclude many patients due to missing data. Furthermore, we excluded 1569 patients from the study because their TSH level was ≥0.1 µIU/mL, and hence, the findings of our study cannot be generalized to patients who do not undergo TSH suppressive therapy. We previously reported that free triiodothyronine becomes normal under TSH suppression, and it often becomes low under normal TSH levels, 14 that is why we performed TSH suppression therapy for patients who underwent total thyroidectomy. We recognize that TSH suppression to TSH values <0.1 µIU/mL) could be considered overtreatment, especially for patients with low-risk PTC. Indeed, this strategy differs from the recommendation in the guidelines of the American Thyroid Association 4 and the Japan Association of Endocrine Surgery. 8 We excluded patients who underwent RAI adjuvant treatment and included a small number of patients with RAI ablation, so the findings may not be generalizable to these groups. The findings are also not generalizable to patients with Tg antibodies.
Conclusions
We believe that postoperative uTg and Tg-DR are very valuable prognostic markers for Tg-antibody-negative patients with PTC who underwent total thyroidectomy and are taking TSH suppressive therapy. We believe that dynamic risk assessment using uTg levels and Tg-DR may inform optimal postoperative management of patients with PTC who have undergone total thyroidectomy and are receiving TSH suppression therapy. Further prospective studies are necessary to obtain a more reliable conclusion about this issue.
Footnotes
Acknowledgment
The authors thank Makoto Kawakami for his assistance with data collection using our electronic medical records system.
Authors’ Contributions
Y.I.: Primary data collection, formal analysis, investigation, project administration, and writing. A.M.: Conceptualization, supervision, and writing. M.Y.: Statistical analysis. M.K.: Data analysis, interpretation, editing, and article review. N.O.: Conception, article review, and editing. A.M.: Data analysis, interpretation, and editing.
Author Disclosure Statement
The authors declare that they have nothing to disclose.
Funding Information
No funding was received for this study.
