Abstract
Background:
Careful consideration of the differential diagnosis of discordant thyroid function tests (TFTs) is fundamental to prevent mismanagement, waste of resources, uncover rare thyroid disorders, or other underlying critical conditions in patients referred for the evaluation of this issue. Here, we describe the frequency of underlying diagnoses and focus on cases in which the identification of analytical interferences led to the discovery of lymphoproliferative disorders.
Methods:
This is a retrospective cross-sectional study including all consecutive patients referred to the Department of Endocrine and Metabolic Diseases, Istituto Auxologico Italiano, for suspected central hyperthyroidism between January 2017 and February 2025. Inclusion criteria were based on laboratory findings of (1) elevated or inappropriately normal thyrotropin (TSH) with high FT4 and/or FT3, (2) elevated TSH with FT4 in the upper reference range, and (3) failure of TSH to normalize despite levothyroxine therapy, along with rising FT4 levels. The differential diagnosis was made by (1) reassessment of discrepant results in the hormone panel by different analytical methods or tandem mass spectrometry; (2) serum serial dilution or polyethylene glycol precipitation; (3) evaluation of rheumatoid factor, serum protein electrophoresis (SPEP), and cryoglobulins; and (4) deep clinical phenotyping and/or confirmatory genetic testing, as appropriate.
Results:
Among 144 patients referred for discordant TFTs mimicking central hyperthyroidism, 44% were genuine rare thyroid disorders—32% with resistance to thyroid hormone beta (RTHβ), 7% with TSH-secreting adenomas, and 5% other diagnosis—and 56% had assay interference. In this latter group, interfering clonal paraproteins were detected in 6/81 cases. One patient was ultimately diagnosed with multiple myeloma, another one with type 1 cryoglobulinemia due to indolent marginal zone B non-Hodgkin lymphoma, and the remaining four cases were found to have monoclonal gammopathy of uncertain significance. The clonal paraprotein was an IgM kappa/lambda in five cases and IgG kappa or IgA kappa in one case, respectively.
Conclusions:
In patients referred for discordant TFTs, RTHβ is the most prevalent thyroid disorder, but variable assay interferences represent the most frequent condition. In these cases, SPEP and hematological investigations are highly recommended.
Introduction
Immunoassays are not infrequently affected by analytical interferences, leading to diagnostic errors and inappropriate treatments. 1 Possible sources of interference include altered hormone-binding proteins (albumin, thyroxine-binding globulin, and transthyretin), biotin ingestion, rheumatoid factor (RF), 2 paraproteins, 3,4 antianalyte autoantibodies (e.g., macrothyrotropin [macro-TSH], antithyroxine [T4], and antitriiodothyronine [T3]), 5 heterophile antibodies, 6 human antianimal antibodies, 7 or antireagents antibodies (e.g., antistreptavidin and antiruthenium). 8,9
In contrast, nonimmunometric methods for assessing thyroid hormones (THs) 10 and steroids, 11,12 such as liquid chromatography/tandem mass spectrometry (LC-MS/MS), are not prone to these interferences but are not widely available for routine testing because of high costs, technical complexity, and limited accessibility in many laboratories. Automated immunoassays remain the most popular methods for endocrine assessments. The typical format of immunoassays for free TH, thyroid autoantibodies, and adrenal and gonadal steroids is competitive, whereas TSH, gonadotropins, prolactin (PRL), adrenocorticotropin hormone (ACTH), beta-human chorionic gonadotropin, and parathyroid hormone are quantified by noncompetitive “sandwich” immunoassays. 1,13
A combination of different methods is often necessary to uncover assay interferences. 1 The use of an alternative method is the most straightforward approach, followed by polyethylene glycol (PEG) precipitation to remove interfering antibodies. 14 However, PEG is not appropriate for free analytes, as the dilution changes the ratio between bound/unbound hormones. 7 Loss of linearity at serum serial dilution is suggestive of spurious results, although false negatives may occur in ∼40% of cases. 15 Heterophilic blocking tubes are another auxiliary method, but results are assay-dependent and false positives may occur. 16
Here, we report the distribution of the different diagnoses following the workup implemented in our department for evaluating discrepant thyroid function tests (TFTs), which led to the identification of six patients with multiple immunoassay interferences caused by clonal paraproteins associated with previously unknown lymphoproliferative disorders.
Materials and Methods
We performed a retrospective cross-sectional study including all consecutive patients referred to the Department of Endocrine and Metabolic Diseases of Istituto Auxologico Italiano for TFTs mimicking central hyperthyroidism (resistance to thyroid hormone beta [RTHβ] or TSH-secreting pituitary adenoma [TSH-oma]) between 2017 and February 2025. Inclusion criteria were based on the following laboratory findings: (1) elevated or inappropriately normal TSH with elevated FT4 and/or FT3, (2) high TSH with FT4 in the upper range of the reference interval, or (3) failure to normalize TSH despite levothyroxine therapy and rising FT4.
The Institutional Review Board of IRCCS Istituto Auxologico Italiano approved the study protocol (code: 05C821_2018), and the research was completed in accordance with the Declaration of Helsinki as revised in 2024. Participants gave written informed consent for using anonymized data and biological samples for research purposes.
Hormones were measured by electrochemiluminescence immunoassays (Roche Diagnostics, Germany) unless otherwise reported. PEG precipitation was systematically performed for detecting macro-TSH but also to screen for interference in the fT4 and fT3 immunoassay in a subset of patients, as previously described. 5,14,17 To control dilution and nonspecific adsorption to PEG, samples are always measured in parallel with euthyroid and overt hyperthyroid individuals (Supplementary Table S1). Serum serial dilutions were done manually using Elecsys universal diluent. 1 Total-T4 (TT4) was measured by time-resolved fluoroimmunoassay (Delfia, Perkin-Elemer). Androgens were measured by isotopic dilution-LC-MS/MS. 18
Results
A total of 144 cases were included. Figure 1 illustrates the diagnostic workflow for discrepant TFTs mimicking central hyperthyroidism that is currently implemented in our center.
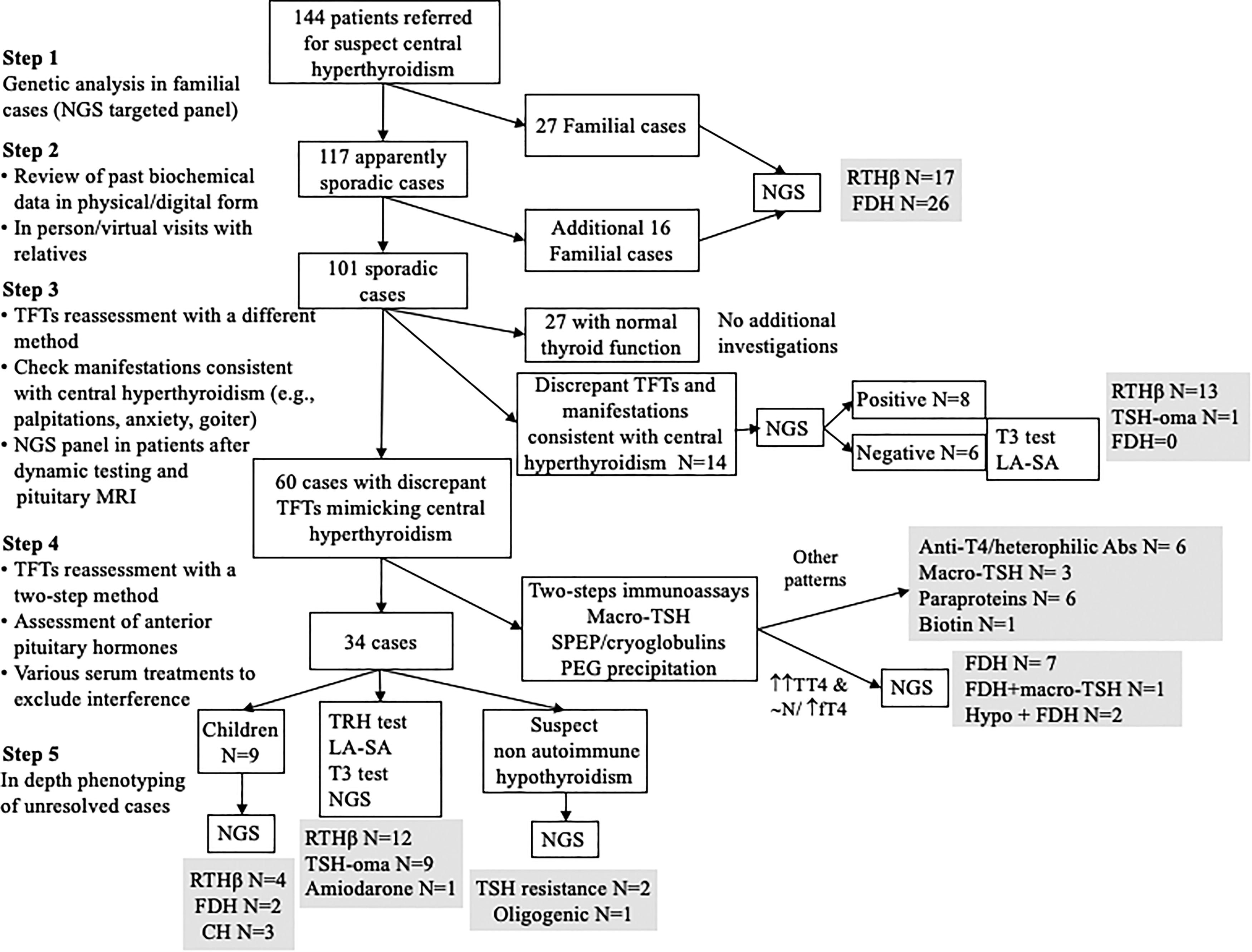
Flowchart applied at our center for the differential diagnosis of cases with thyroid function tests mimicking central hyperthyroidism. The diagnostic process is articulated in several sequential steps. The diagnostic yields are reported. In familial cases, the identification of relatives with similar hormone profiles guides genetic testing by NGS for mutations in relevant genes (THRA, THRB, SCL16A2, SECIPB2, DIOs, ALB, SERPINA7, TTR, AIP, and MEN1). Sporadic cases are initially approached by repeating TFTs with alternative assay methods. If discrepancies persist, additional investigations (e.g., serial dilutions, PEG precipitation, heterophilic antibody blocking agents, and additional biochemistry) are performed. Following these steps, for patients without interferences, dynamic testing, pituitary imaging, NGS molecular analysis, or direct sequencing are performed, as appropriate. 19 –21 CH, congenital hypothyroidism; FDH, familial dysalbuminemic hyperthyroxinemia; LA-SA, long acting-somatostatin analog; NGS, next-generation sequencing; PEG, polyethylene glycol; RTHβ, resistance to thyroid hormone type beta; SPEP, serum protein electrophoresis; TFT, thyroid function tests; TSH, thyrotropin; TSH-oma, TSH secreting pituitary adenoma.
The demographic and biochemical characteristics stratified by final diagnoses, as well as the origin of the cases, are described in Table 1. Overall, 42% of the patients were referred from health care providers without access to specialized laboratories. Blood tests were repeated in ≥2 different laboratories before referral, as recommended. 19 –21 However, no particular attention was given to the analytical method, as changing the laboratory did not imply a change in methodology. Additionally, laboratory reports often omitted the used method, hampering the method assessment. Among 117 patients referred as sporadic and having previously undergone resource-consuming second-level investigations (e.g., pituitary magnetic resonance imaging [MRI]; Fig. 1), we identified additional affected family members in 14%, who were finally diagnosed as RTHβ or familial dysalbuminemic hyperthyroxinemia (FDH; Fig. 1).
Origin, Demographic and Biochemical Characteristics of Patients Referred to Our Department for Discrepant Thyroid Function Tests Suggestive of Central Hyperthyroidism, Stratified by Most Frequent Final Diagnoses
This group does not include the three patients with macro-TSH.
Reference ranges: TSH 0.4–4.5 mU/L, fT4 11.5–24.5 pmol/L, fT3 2.8–7.1.
fT3 and fT4 both increased, N = 9; increased fT4 and normal fT3, N = 6; normal fT4 and increased fT3, N = 12 isolate increase.
Cardiologist, dentist, pediatricians, and andrologist.
Cases with congenital hypothyroidism, isolated TSH resistance, and hypothyroidism with oligogenic origin were excluded from this table but are discussed in detail in the main text.
FDH, familial dysalbuminemic hyperthyroxinemia; IAI, IRCCS Istituto Auxologico Italiano; IQR interquartile range; MRI, magnetic resonance imaging; RTHβ, resistance to thyroid hormone beta; TSH-oma, TSH-secreting pituitary adenoma.
The prevalence of assay interference was 56% (81/144), with variable numbers of patients diagnosed with biotin interference, macro-TSH, heterophilic antibody interference, and FDH (Fig. 2). All the 38 FDH subjects had increased or normal fT4 in one-step or two-step immunoassays, respectively; TT4 was always high, including six cases with heterozygous p.G6S TTR variant whose results in the binding affinity assays were conflicting. 22 Among these FDH subjects, 30 carried single heterozygous variants in ALB and 4 in TTR gene. Another female patient carried a mutation in the TTR gene, associated with macro-TSH and a genuine primary autoimmune hypothyroidism. The patient was referred with a suspicion of TSH-oma due to inability to normalize TSH levels (>50 mU/L) despite combination therapy with levothyroxine (75 µg/day) and T3 (42 µg/day). At referral, she was thyrotoxic with elevated fT3 (9.3 pmol/L) and normal fT4. Following the evidence of macro-TSH, a weight-adjusted dose of levothyroxine was prescribed. Three months later, despite a borderline elevated TSH (6.5 mU/L after PEG precipitation), TT4 was high that led to the discovery of the p.G6S TTR variant. One female patient carried mutations in both genes (p.R218H in ALB and p.G6S in TTR). Two FDH patients had primary nonautoimmune hypothyroidism with high TSH levels (6.5 and 5.7 mU/L): one carried a variant of unknown significance (VUS) in GLIS3 gene (p.G749R) and a mutation in ALB gene (p.R218H), and the other had a heterozygous mutation in the DUOX2 gene (p.W178C) and a variant of the TTR gene (p.T139M). One female with macro-TSH and primary autoimmune hypothyroidism on levothyroxine therapy was referred for RTHβ, based on TSH >100 mU/L with borderline high fT4 levels (22.8 pmol/L). Another female was referred due to persistently elevated TSH (48–50 mU/L), normal fT4, and a multinodular colloid goiter. Following PEG precipitation, TSH normalized in both patients, confirming analytical interference. We performed additional next-generation sequencing (NGS) investigation in the patient with goiter and identified two new gene variants, which may explain her phenotype: a premature stop codon (p.Y55X) in TUBB1 and a VUS in IYD (p.D186H). 23 The spurious results at referral were not confirmed in 27 patients (Figs. 1 and 2).
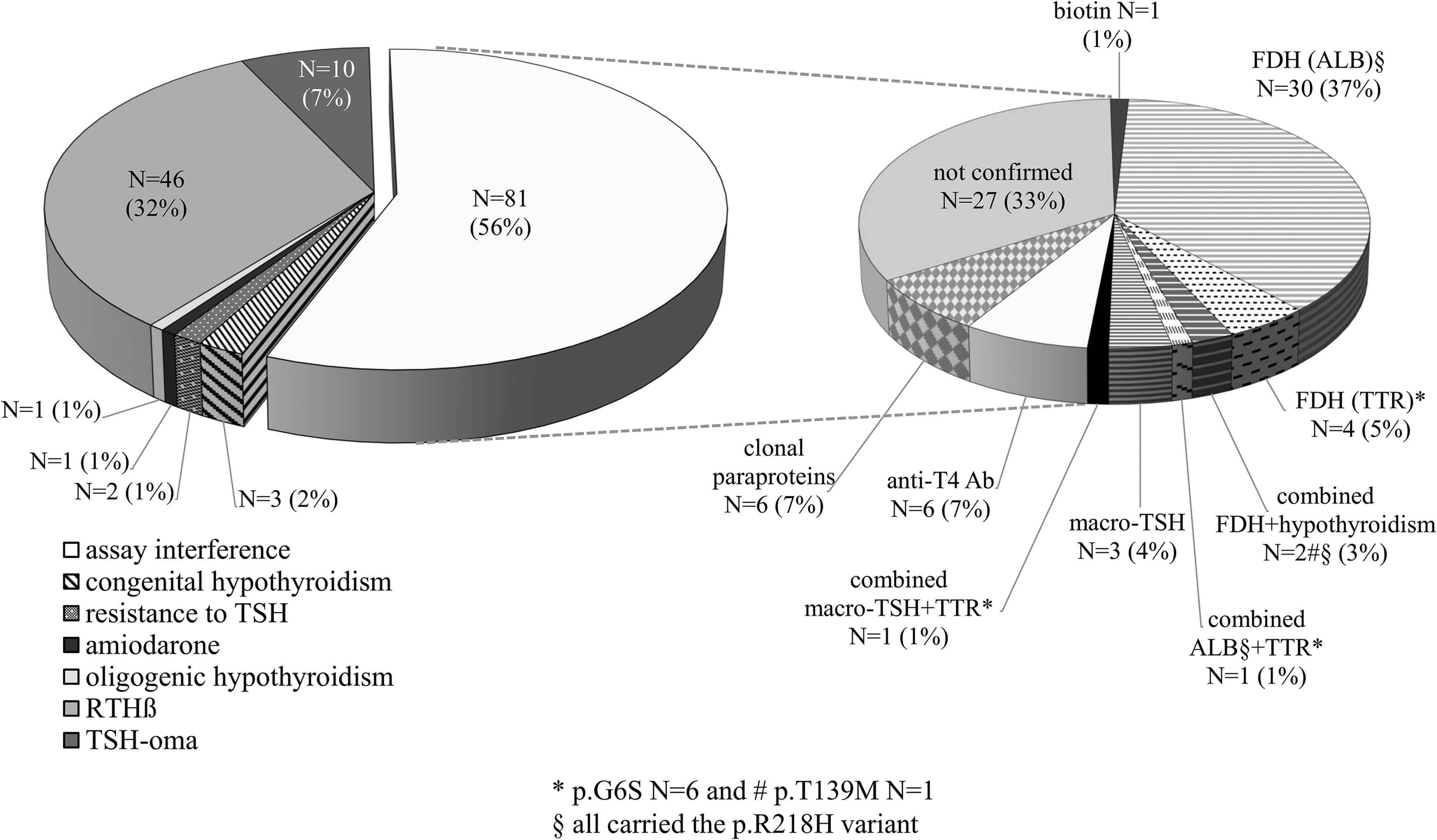
Prevalence of assay interference in this monocentric cohort of patients referred for discrepant thyroid function tests. The left pie chart displays the final diagnoses of the 144 patients. Interestingly, RTHβ was the most frequent genuine thyroid disease, but variable laboratory issues were the prevalent category. The right inset shows the numerical proportions of each type of immunoassay interference observed in the 76 patients with spurious thyroid function test results. *p.G6S variant in the TTR gene: N = 6. # p.T139M variant in the TTR gene: N = 1. § All carried the p.R218H mutation of the ALB gene. ALB, gene encoding human albumin; TTR, gene encoding transthyretin.
Finally, clonal paraproteins were found in six patients (6/144: 4.2%; Tables 2 and 3) after exclusion of biotin supplements and RF. Falsely positive antithyroid antibodies were found in 3/6 patients.
Results of Investigations Available at Referral in Patients with Interfering Paraproteins
Reference ranges.
Roche: TSH 0.4–4.5 mU/L, fT4 11.5–24.5 pmol/L, fT3 2.8–7.1, TPOAb < 34 U/L, TgAb <116 U/L, TRAb <1.8 U/L, calcitonin, F < 6 pg/mL, M < 10 pg/mL, TTST males 9.9–27.8 nmol/L, cortisol 5–25 µg/dL, DHEAS males 0.91–6.76 µmol/L, LH males 1.7–8.6 U/L; F postmenopause 7.7–58.5 U/L, FSH males 1.0–14 U/L; F postmenopause 23.8–134.8 U/L, PRL 4–15.2 M 5–20 F ng/mL, ACTH 8–50 pg/mL, estradiol 18.4–505 pmol/L, delta 4-A males 0.2–3.1 mcg/L, urinary free cortisol (UFC) <169 nmol/24 hours; SHBG males 10–70; females 20–100.
ADVIA-Centaur: TSH 0.55–4.80 mU/L, fT4 0.89–1.76 ng/dL, fT3 2.30–4.20, TTST 6.5–23.7 nmol/L, cortisol 5.27–22.45 µg/dL, DHEAS 0.94–15.4 µmol/L, ACTH 8–47 pg/mL, females 20–100 nmol; PRL males 2.1–17.7 ng/mL.
Beckman Coulter: TSH 0.35–4.5, fT4 7.8–17.3 pg/mL, fT3 3.4–6.45 pg/mL.
Delfia: fT4 9–20 pmol/L, TT4 68–141 nmol/L.
bpm, Beats per minute; delta 4-A, delta-4-androstenedione; DHEAS, De-Hydro-Epiandrosterone Sulfate; FDG, fluoro-deoxy-glucose; FNA, fine needle aspiration; GD, Graves’ diseases; HR, heart rate; MGUS, monoclonal gammopathy of undetermined significance; MRI, magnetic resonance imaging; NA, not available; SHBG, sex hormone binding globulin; TTST, total testosterone; US, ultrasonography.
Hormonal Results Measured After PEG Precipitation in the Six Cases with Interfering Paraproteins
Interfered tests are highlighted in bold.
Multiplied for the dilution factor when appropriate.
BNP, brain natriuretic peptide; NA, not assessed; TNT, high-sensitivity troponin T.
Case 1: A 52-year-old woman referred for a suspicion of TSH-oma associated with Graves’ disease (GD), with positive TPOAb, TgAb, and TRAb. Thyroid ultrasound showed a solid left nodule, but thyroid uptake was normal. Despite high fT4 levels >50 pmol/L, thyrotoxic manifestations were absent and sex hormonebinding globulin levels were normal (Table 2). PEG precipitation revealed spurious hyperthyroxinemia and several additional interferences (underestimation in sandwich and overestimation in competitive immunoassays). TFTs were normal when reassessed by DELFIA and Beckman–Coulter platforms. Serum protein electrophoresis (SPEP) revealed an IgM-kappa monoclonal component (MC) of 1.32 g/dL, leading to the diagnosis of monoclonal gammopathy of undetermined significance (MGUS).
Case 2: A 60-year-old male referred with a suspicion of TSH-oma associated with GD. Methimazole was initiated based on positive TSH-receptor autoantibodies despite the incongruity between very high fT4 (50 pmol/L), partially suppressed TSH (0.1 mU/L), a normal thyroid uptake, and the absence of thyrotoxicosis (Table 2). During methimazole treatment, discrepant TFTs emerged (Supplementary Table S3). A thyroid ultrasound showed a hypoechoic nodule with a Bethesda category III cytology, prompting a surgical referral. At our hospital, after methimazole withdrawal, we found low TSH and PRL levels unresponsive to thyrotrotropin releasing hormone (TRH) test, but we obtained normal results when reassessed after PEG precipitation (Supplementary Table S4). PEG precipitation uncovered additional assay interferences (Table 3). TSH was normal when measured by Siemens ADVIA-Centaur. SPEP revealed an MC of 0.3 g/dL. The patient was ultimately diagnosed with indolent non-Hodgkin lymphoma (NHL) and secondary type 1 cryoglobulinemia. Thyroid surgery was not indicated as the THY-3 nodule remained stable during a 4-year follow-up.
Case 3: A 53-year-old male referred for suspected RTHβ. The suspicion was raised during investigations for remitting-recurrent asthenia and positive borreliosis serology without a history of tick bite exposure. TgAb, TPOAb, and TRAb were positive (Table 2), but the thyroid ultrasound was normal. PEG precipitation uncovered variable and multiple assay interferences (Table 3). SPEP showed an IgM-lambda MC <0.30 g/dL, with a final diagnosis of MGUS. Reassessment raised the suspicion of false positive serology against Borrelia burgdorferi.
Case 4: A 63-year-old man referred for hyperthyroxinemia, biochemical androgen excess, and unilateral adrenal lesion discovered during investigation for erectile dysfunction. High total testosterone and De-Hydro-Epiandrosterone Sulfate, with unsuppressed gonadotropins, were found on different assay platforms. ACTH and cortisol assays gave normal results (Table 2). Upon referral, we found low ACTH with 17OH-P and cortisol within the normal range, and normally responsive to a 250 mcg Cosyntropin test. PEG precipitation identified multiple assay interferences with underestimation of ACTH and LH and overestimation of estradiol, fT4, fT3, and testosterone (Table 3). Steroids were normal when reassessed by LC-MS/MS (Supplementary Table S5). SPEP revealed an IgM-kappa MC of 0.3 g/dL with a final diagnosis of MGUS.
Case 5: A 86-year-old female referred with the suspicion of TSH-oma based on abnormal TFTs. She underwent surgery for lung adenocarcinoma 10 years earlier, with the incidental finding of multinodular goiter. Serum calcitonin, TSH, fT4, fT3, and TPOAb were normal at baseline. During follow-up, hyperthyroxinemia with raised TSH was detected by the Roche method, whereas fT3 levels were normal. Similar results were found with ADVIA-Centaur; conversely, testing with the Beckman–Coulter platform showed normal TSH and fT4 levels but elevated fT3 (Table 3). PEG precipitation detected spurious hyperthyroxinemia and underestimation of ACTH (Table 2). An IgG-kappa gammopathy (9.2 g/dL) was found, leading to a diagnosis of multiple myeloma (MM) and referral to a hematologist for treatment, but she passed away 18 months later.
Case 6: A 49-year-old patient previously diagnosed with RTHβ. A biochemical signature of central hyperthyroidism was detected during the evaluation for relapsing AF but mistakenly interpreted as GD, leading to the initiation of methimazole. He never received amiodarone. Two years later, he was referred to a different center, and methimazole was discontinued, following the evidence of normal thyroid ultrasound and negative TPOAb, TgAb, and TRAb. A TRH stimulation test was normal. Direct sequencing of THRB gene was negative. Pituitary MRI and thyroid uptake were normal. Lately, he sought a second opinion at our hospital. TFTs confirmed hyperthyroxinemia (Table 2), whereas fT4 was normal on a two-step method, while total T4 was raised (Table 1). SPEP revealed two IgA-kappa MC (0.5 g/dL and <0.3 g/dL, respectively) with a final diagnosis of MGUS.
The remaining 63 patients (44%) underwent deep clinical phenotyping. The most frequent diagnosis was RTHβ (N = 46/63, 73%), followed by TSH-oma (N = 10/63, 16%; Fig. 1). Among the patients with RTHβ, 41 carried pathogenic THRB mutations (Supplementary Fig. S1), while 5 (12%) were wild-type. They all had a normal TSH response to TRH test and no lesions at pituitary MRI. To exclude a TSHoma, three patients underwent a 3-month trial with long-acting somatostatin analogs without significant fT4/fT3 changes, and two had a conserved TSH suppression upon T3 suppression test. 21 Among patients with confirmed TSH-oma, three had macroadenomas and seven had microadenomas. Seven underwent pituitary surgery yielding a histological diagnosis of TSH-Pit-NET, and two declined surgery and are currently treated with somatostatin analogs, while one was lost to follow-up. Six patients who had congenital hypothyroidism or nonautoimmune hyperthyrotropinemia were referred to exclude RTHβ because of the findings of borderline high fT4 and elevated TSH levels (see Supplementary Table S2 for biochemical and genetic results). Finally, a 17-year-old male, with an inherited cardiomyopathy on amiodarone treatment, presented with persistently increased TSH and FT4, following the resolution of type 2 amiodarone-induced thyrotoxicosis. Molecular analysis of THRB, TTR, and ALB genes was negative, leading to a diagnosis of drug interference (Fig. 1). The discrepancy in TFTs disappeared after heart transplantation and discontinuation of amiodarone. Figure 3 shows a scatter plot illustrating the relationship between fT4 and fT3 and between TSH and fT4 across diagnostic groups. Although FDH generally presents with lower fT4/TSH and fT3/fT4 ratios, there is a wide overlap among groups, preventing an accurate differential diagnosis based on biochemical signature.
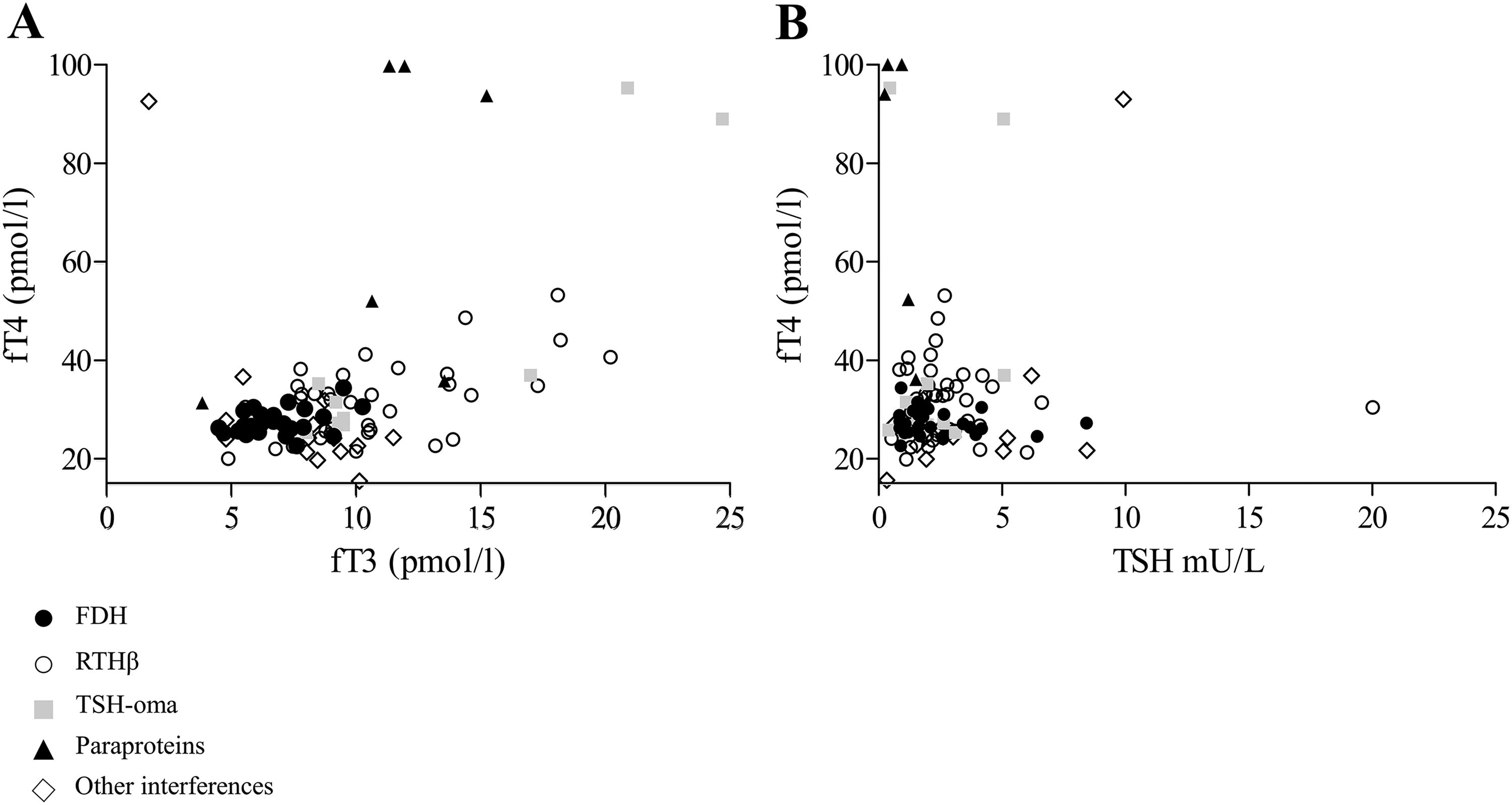
Scatter plots of thyroid function tests across diagnostic groups.
Discussion
In this real-life series of cases referred for discrepant TFTs mimicking central hyperthyroidism, we found variable assay interferences in 56% of cases. If appropriate biochemical tests had been anticipated, as indicated by expert clinical guidelines, 19 –21 several unnecessary clinical investigations could have been avoided with relevant cost savings. Despite existing recommendations, 19 –21 we found that family screening is still inconsistently applied. The identification of additional affected relatives in 14% of cases referred to as sporadic indicates that earlier recognition of familial patterns could prevent unnecessary second-level testing and directly prompt NGS analyses. Overall, 84 of 144 patients underwent genetic testing. This is important as NGS should become routine in the evaluation of patients with discrepant TFTs when similar biochemical anomalies are present in other relatives or after exclusion of assay artifacts (see Fig. 1).
Secure platforms for online consultations may improve caregiver–patient communication, possibly leading to the identification of additional affected relatives (Fig. 1). These systems could also enable the retrieval of previously normal TFTs, which would support either analytical interference or a TSH-oma. In this cohort, we found a high prevalence of assay interference, underscoring the complexity of the differential diagnosis of suspect central hyperthyroidism. As recommended, 19 –21 hyperthyroxinemia should be initially approached by repeating TFTs with alternative assay methods. If discrepancies persist, additional investigations such as serial dilutions and heterophile antibody blocking reagents should be implemented. While PEG precipitation is validated for the identification of macro-TSH and macro-PRL, its application to FT4 and FT3 assessment remains investigational 17 due to its effect on free/bound hormone equilibrium. In our study, PEG precipitation was selectively used in a few patients with complex analytical interference and results compared with well-characterized controls. Despite these limitations, PEG may be considered a practical and low-cost screening tool to detect antibody-bound hormone complexes, before proceeding to more advanced techniques. It is worth noting that even after excluding analytical interference, TFTs are insufficient for a definitive diagnosis in most patients. Nonetheless, the biochemical pattern may still offer useful diagnostic clues. For instance, a low fT3/fT4 ratio may raise suspicion for FDH in the appropriate clinical context (Fig. 3). These findings underscore the need for integrated evaluation, where biochemical data are interpreted alongside clinical features, dynamic testing, imaging, and genetic studies.
Several patients in our cohort presented with complex clinical and genetic profiles that challenged straightforward diagnosis and illustrated how genetic variants coexisting with biochemical interference can mimic pituitary feedback alterations. Among the FDH cases, two patients presented with primary nonautoimmune hypothyroidism and carried additional genetic variants in GLIS3 and DUOX2 genes. Another patient with macro-TSH carried variants in TUBB1 and IYD. Without the identification of such interference, these findings could have led to inappropriate treatments. Additionally, one female patient with FDH exhibited assay interference affecting both TSH and free T4. This dual interference not only complicated the differential diagnosis between FDH and TSH-oma but also raised significant challenges in the appropriate titration of levothyroxine therapy. Recognition of such analytical interference was crucial to guide a tailored treatment. Similarly, two patients with mild TSH resistance, due to heterozygous mutations in the TSHR gene, were initially misdiagnosed as having RTHβ because their FT4 levels were considered inappropriately high for borderline elevated TSH values. According to the phenotype of euthyroid hyperthyrotropinemia, 24 we did not prescribe levothyroxine.
Notably, among these cases, we describe six patients in whom the evidence of multiple immunoassay interferences led to the diagnosis of previously unknown lymphoproliferative disorders, including NHL and MM. Beyond the prescription of unnecessary investigations and useless therapies, the incorrect application of clinical protocols resulted in diagnostic delay for hematological malignancies.
Lymphoproliferative disorders encompass a heterogeneous group of malignancies and premalignant conditions such as monoclonal B-cell lymphocytosis (MBL) and MGUS, which can progress to chronic lymphocytic leukemia (CLL) and MM, respectively. 25 MBL 26 and MGUS are highly prevalent in the general population >60 years with a risk of progression of 1–2% per year. 25,27
Importantly, the identification of analytical interferences due to paraproteins can improve the patients’ clinical management. First, we diagnosed an early-stage NHL, and an MM promptly referred to a hematologist and three high-risk MGUS cases, since IgM or IgA MC is more likely to progress to MM, 28 lymphoma, CLL, amyloidosis, and Waldenström macroglobulinemia. 29 Early diagnosis is expected to improve survival because patients with MM who are under follow-up for MGUS have a better prognosis compared with those diagnosed at a later stage. 30,31 Second, the observed underestimation of ACTH levels in the fifth patient emphasizes the critical importance of recognizing such interferences in MM patients. One complication of MM is the paraneoplastic syndrome POEMS (polyradiculoneuropathy, organomegaly, endocrinopathy, monoclonal plasma cell disorder, skin changes), which may cause pituitary and adrenal insufficiencies. 32,33 Interestingly, paraproteins were found to lead to multiple interferences, with underestimation in sandwich immunoassay of pituitary hormones and overestimation in competitive immunoassays of cortisol, FT4, and testosterone. Thus, we recommend assessing MM patients by robust technologies, such as LC-MS/MS, to avoid underdiagnosis of endocrine insufficiencies, especially in the presence of neuropathy. Third, we identified a reliable method for the follow-up of patients with a genuine endocrine disease or specific risk factors. Finally, we discontinued a useless antithyroid treatment, thus avoiding the iatrogenic hypothyroidism in one of them.
Most of the patients included in this series underwent hormonal investigations in different laboratories, as suggested by expert opinions or guidelines 19 –21 ; however, the use of different methods was often overlooked, highlighting the importance of including the manufacturer’s name for the methods in the laboratory report, to avoid misinterpretations.
A potential limitation of our study is the referral bias inherent to its design as a single-center cross-sectional retrospective case series. As a tertiary endocrine referral center, we receive unresolved or complex cases from other centers, which may lead to an overrepresentation of rare/challenging conditions. Most patients were referred by endocrinologists from different clinical settings with a minority being referred from general practitioners or other specialists (about 10% of cases; Table 1). This referral pattern may have contributed to a skewed diagnostic spectrum. Despite these limitations, our study included all consecutive referred patients who underwent a standardized diagnostic workflow (Fig. 1), ensuring internal consistency. We believe the proposed diagnostic algorithm may serve as a practical reference for clinicians, particularly in first-level centers, to help identify patients who warrant further diagnostic investigation or referral to specialized services.
The lack of a causal link between the identified paraproteins and the assay interference represents another limitation. However, five patients had IgM or IgA MC, which are more frequently associated with analytical interference. 3,4 In fact, IgMs are pentameric/exameric complexes linked by disulfide bridges, 34 making them more susceptible to cross-reacting with multiple antigens. 35 Similarly, IgA-type paraproteins can be a mixture of monomeric and polymeric complexes. 36 Although specific quantitative data on interference at low concentrations are scarce, the multimeric nature of IgA paraproteins supports the possibility of analytical interference even at low concentrations.
In conclusion, identifying analytical interferences is critical for limiting needless investigations and avoiding potentially harmful therapies. Identifying these interferences entails watchfulness in recognizing discrepancies between serum levels of anterior pituitary tropins and target peripheral hormones, as well as between the hormonal levels, the patients’ clinical features, and imaging results. Although uncommon in the general population, multiple assay interferences due to paraproteins are frequent in patients with discrepant TFTs (∼4% in the whole referred cohort). As previously suggested, 21 we suggest performing at least SPEP and immunofixation in all patients with assay interferences, following the diagnostic algorithm explained here in detail.
Footnotes
Acknowledgments
The authors would like to thank the patients who consented the publication of their clinical and biochemical data.
Authors’ Contributions
A.M.: Data curation (equal), writing—original draft (lead), and formal analysis (supporting). G.F.A.G.: Data curation (equal) and writing—original draft (lead). S.R.: Data curation (supporting), formal analysis (supporting), and writing—review and editing (supporting). M.A.: Investigation (lead) and methodology (equal). I.B.: Investigation (equal) and methodology (equal). F.F.: Investigation (equal) and methodology (lead). V.M.: Investigation (equal), formal analysis (equal), visualization (equal), and writing—review and editing (equal). L.P.: Conceptualization (lead), supervision (lead), funding acquisition (lead), and writing—review and editing (lead). I.C.: Conceptualization (lead), methodology (lead), formal analysis (lead), and writing—review and editing (lead).
Data Availability
Datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request (doi:10.5281/zenodo.15738415).
Authors Disclosure Statement
The authors declare that there was no competing financial interest.
Funding Information
This research was funded by the European Union-Next Generation EU-NRRP M6C2-Investment 2.1 Enhancement and strengthening of biomedical research in the NHS (code: PNRR-MR1-2022-12375726 to Luca Persani).
Supplementary Material
Supplementary Data
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
