Abstract
Background
Continuous palliative sedation (CPS) and continuous opioid infusion (COI) are two widely used interventions to relief suffering and manage symptoms in the terminal phase. National guidelines exist for both. This study investigated guideline deviations in a hospital setting.
Aim
The main objective was to assess the prevalence of deviations from the Dutch guidelines for CPS and COI, potentially leading to inappropriate care. The secondary objective was to investigate whether involvement of the palliative consulting team and pain team resulted in less guideline deviations.
Design
Patient records from hospitalized patients who deceased between December 2022 and December 2023 who received CPS and/or COI as end-of-life care, were retrospectively analysed.
Results
101 patients who received CPS and 129 patients who received COI as end-of-life care were included. In patients receiving CPS, clinically relevant guideline deviations were found in 52.5% of patients, mostly related to change in opioid dose of COI during CPS and other opioid-related use during CPS. In 76.7% of the patients with COI, one or more guideline deviations were found. These were mostly related to opioid (starting) dose and use of morphine in patients with impaired renal function. Involvement of the palliative consulting team and pain team resulted in slightly less guideline deviations.
Conclusion
This evaluation on CPS and COI guideline deviations found one or more deviations in the majority of patients. Further research and training is required to identify methods to improve knowledge and improve the use of the guidelines on CPS and COI as end-of-life care.
Introduction
The main goals of care in the terminal phase are adequate symptom management and relief of suffering. Two widely used interventions in this context are continuous palliative sedation (CPS) and continuous opioid infusion (COI). In CPS, the consciousness of a patient is deliberately lowered to relieve otherwise refractory symptoms in the terminal phase. For a patient to be eligible for CPS, the Dutch guideline states that life expectancy should be shorter than two weeks, at least one refractory symptom should be present and approval should have been given by either the patient or their representative. 1 Since this guideline was introduced in 2005, the use of CPS in the Netherlands has increased significantly, rising from 8.2% in 2005 to 26% in 2022.2,3 National and international evaluations on adherence to (local) guidelines have reported a wide variety in adherence ranging from 7% in Canada to 100% in Spain. 4 In the Netherlands, a study from 2008 reported palliative sedation with only opioids in 22% of the patients. However, this is not in concordance with the guidelines and the investigators emphasized the need for the increased involvement of palliative consulting teams.5,6 Similar research conducted with a survey under physicians in the Netherlands in 2025 concluded the same. 3 Currently, palliative consulting teams are involved more regularly, however whether this involvement leads to less guideline deviations is yet to be investigated. 7
Likewise, opioids play a central role in palliative care, especially for the treatment of pain and dyspnoea. If oral administration is not possible, COI administered via intravenous or subcutaneous infusion is an important alternative. 8 Despite existing guidelines, COI is not always used in accordance with these recommendations. 9 A recent study showed that up to 44% of the COIs may be potentially inappropriate, due to high opioid infusions in opioid-naïve patients or due to more than three increases in opioid dose within 24 hours. 10 The investigators also reported that the involvement of palliative care teams decreased the prevalence of potentially inappropriate infusions. Whether this also applies to patients who received COI or CPS in a Dutch hospital is unknown. Additionally, the studies discussed above mainly focussed on the process leading up to the start of CPS and COI, while not including the follow-up steps. Therefore, we have retrospectively investigated patients who received CPS and/or COI as terminal care at Maastricht UMC+ (MUMC+). The aim was to assess and identify the prevalence of deviations from the Dutch guidelines for CPS and COI.1,9,11 The secondary objective was to investigate whether the involvement of the palliative consultation team and/or pain team was associated with the prevalence of guideline deviations.
Methods
This retrospective study was given a non-WMO declaration (Dutch act on medical research involving human subjects) by the medical-ethical committee of AzM/UM (METC 2024-0071 and METC 2024-0036). This study was conducted according to the STROBE guidelines. 12
Study Population and Sample Size
Electronic health records from patients, 18 years or older, who deceased between December 1st 2022 and December 31st 2023 and received CPS and/or COI at a general ward at the moment of death were included. CPS was defined as at least one record of continuously administered midazolam or levomepromazine subcutaneous or intravenously. COI was defined as at least one record of continuous opioid infusion subcutaneous or intravenously administered. Patients who deceased at the emergency department, operation room or intensive care unit were excluded.
A convenience sample of 100 patients with CPS and 100 patients with COI who deceased at a general ward at MUMC+ were investigated. If a patient received both CPS and COI, they were assigned to both the CPS and COI groups.
Data Collection
Patient files, nurse reports and medication lists were screened by one of the research team members which consisted of two palliative care specialized nurses and a researcher, who were all familiar with the guidelines.
For CPS, guideline deviations were divided in clinically relevant deviations and administrative deviations. Clinically relevant deviations were defined as deviations potentially compromising patient safety, including deviations related to type and dose of medication used for CPS (midazolam or levomepromazine), type and dose of COI during CPS, or start of COI with of during CPS, and the use of opioid boluses. Missing reports on life expectancy, presence of refractory symptoms, precautionary measures at the start of CPS (including: stop implantable cardioverter-defibrillator [ICD], stop nutrition, continuation of essential medication, and presence of urine catheter) were categorized as administrative deviation. For COI, guidelines deviations were based on opioid use before start of COI, type and dose of COI at start, type and dose of COI at time of death and indication for start COI.
The data extraction form can be found in Appendix III. Potential deviations from the guidelines and inappropriate COI and CPS were discussed within the research team and if necessary with one of the physicians in the team.
Involvement of the palliative consulting team was defined as at least one note of the team in the patient file discussing CPS or COI before or during CPS or COI. The involvement of the pain team was defined as at least one note of the team in the patient file discussing COI.
Statistical Analysis
All data was captured and analysed using SPSS version 28 (IBM Corp., Armonk, NY, USA). Results are depicted descriptively. Continuous variables are displayed as mean and range and categorical variables as number and percentage. The primary and secondary objectives are depicted as number and percentage. Considering the descriptive nature of the study, no additional statistical analyses were performed.
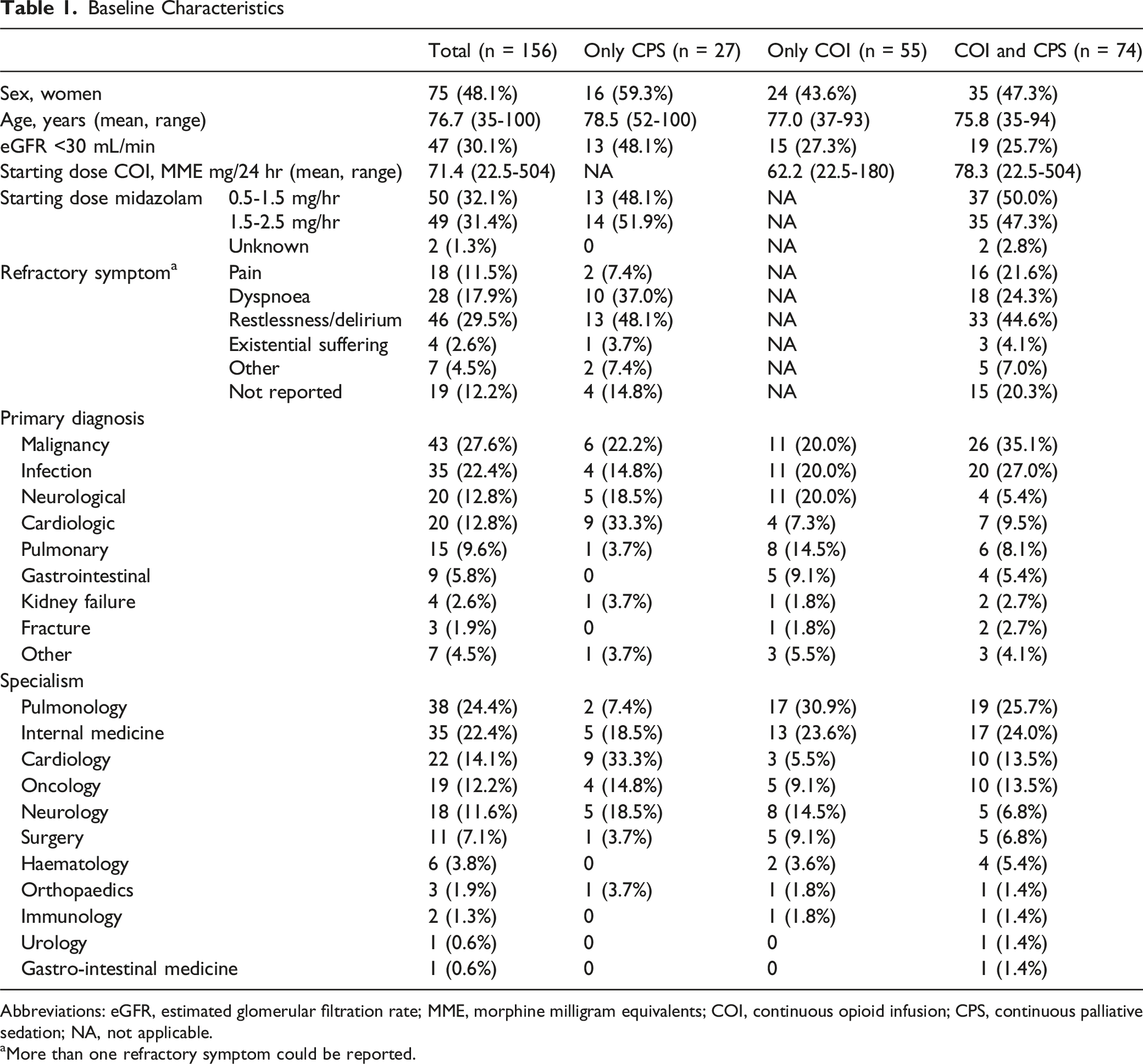
Results
To reach the aimed sample size of 100 patients with COI and 100 patients with CPS, 362 patient files were screened of which, 156 patients received COI and/or CPS (Figure 1). Approximately half of the patients were female (48.1%) and mean age was 76.7 years (range 35 - 100). The most common primary diagnoses were malignancy (27.6%) and infection (18.6%) and patients most often were admitted for pulmonology (24.4%) or internal medicine (21.8%). Since a large proportion of patients received both COI and CPS, no relevant differences were reported between the two groups except for the type of refractory symptoms (Table 1). A larger proportion of patients receiving only CPS had dyspnoea or restlessness/delirium as refractory symptom. Of the patients who received only CPS, 7.4% had pain as refractory symptom compared to 21.6% of the patient who received both CPS and COI. Flow-chart Baseline Characteristics Abbreviations: eGFR, estimated glomerular filtration rate; MME, morphine milligram equivalents; COI, continuous opioid infusion; CPS, continuous palliative sedation; NA, not applicable. aMore than one refractory symptom could be reported.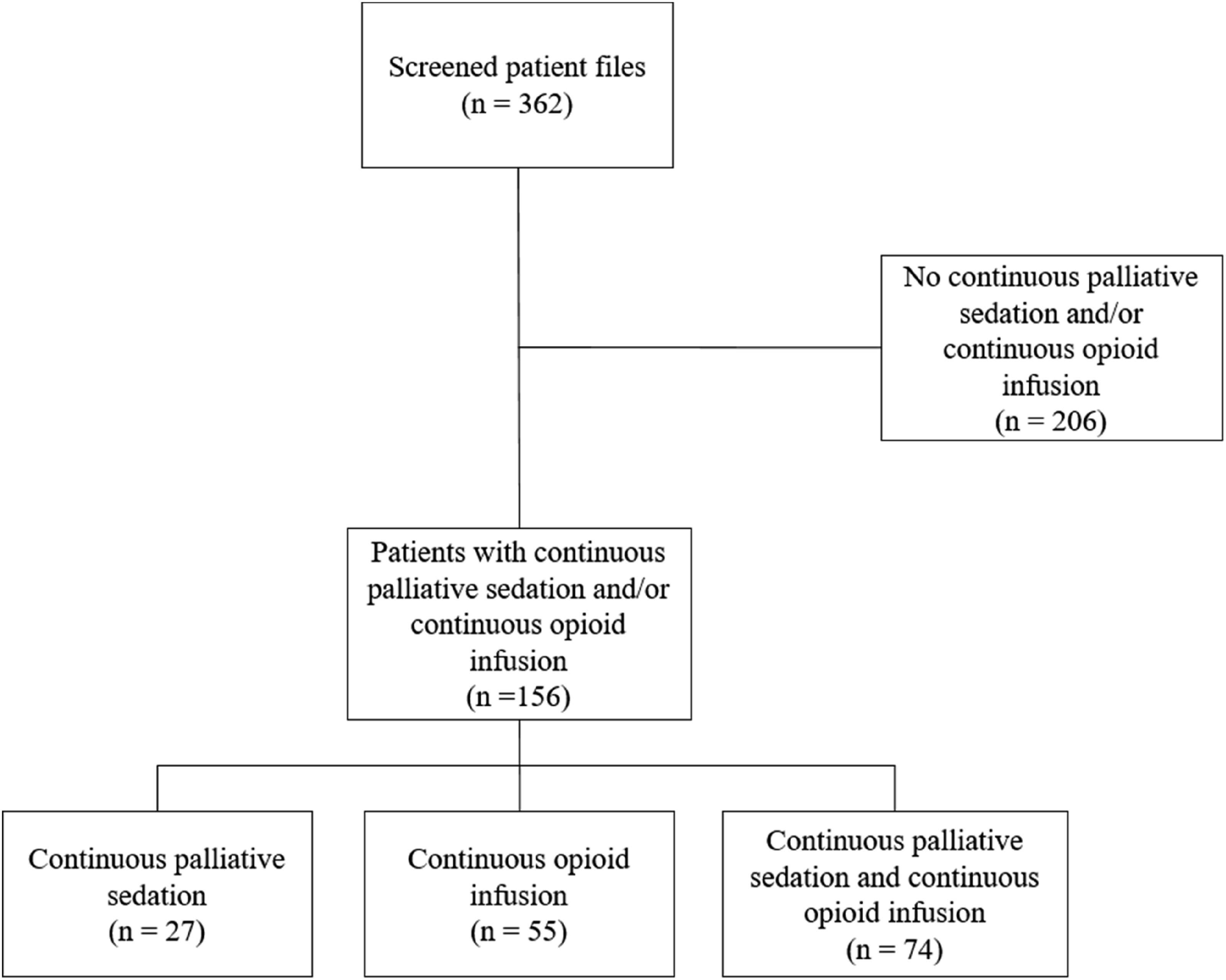
Continuous Palliative Sedation
The mean CPS duration was 29.7 hours ranging from <1 hour to 168 hours. In total, clinically relevant guideline deviations were found in 54 of the 101 patients (52.5%) who received CPS. In these patients, start of COI with or during CPS (37.7%) and increase of COI during CPS (35.8%) were most common (Figure 2). Overview of guideline deviations in CPS. Left panel; proportion of patients with a clinically relevant deviation of CPS guidelines. Right panel; specific reason of guideline deviation. *Except for the increase of 0.5 mg/hr to 1.0 mg/hr of midazolam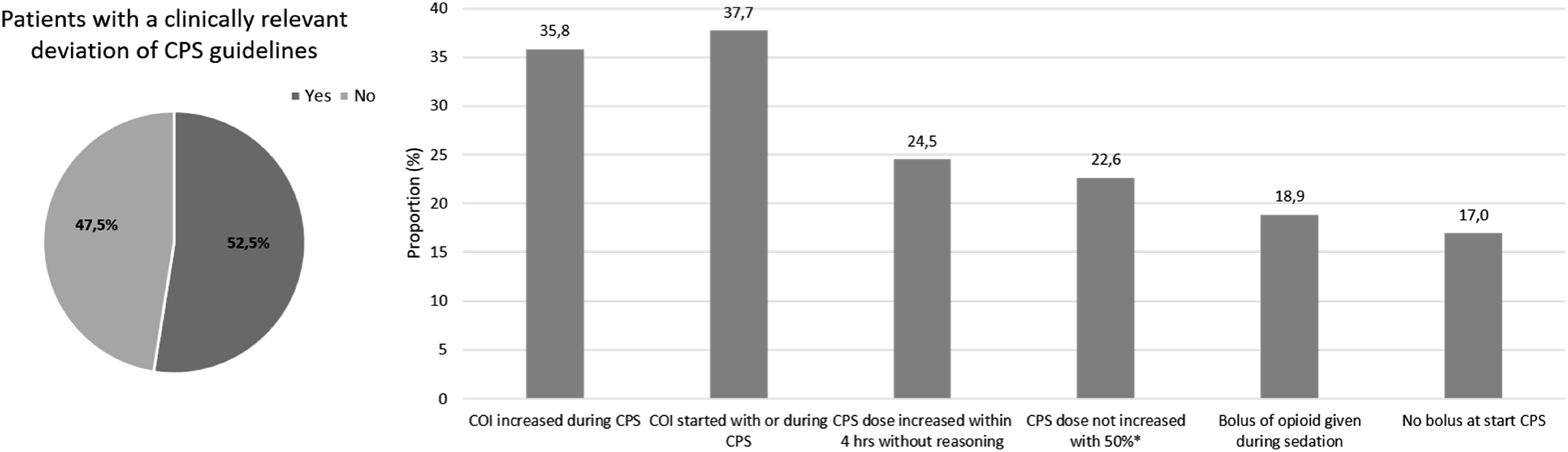
Administrative guideline deviations occurred in 86 patients (85.1%), all missing a report of life expectancy (100%). Other administrative guideline deviations were no report of a refractory symptom (22.1%), no report of start CPS (5.8%) and continuation of NaCl infusion during CPS (2.3%).
Continuous Opioid Infusion
COI duration was 44.4 hours on average and ranged from <1 hour to 377 hours. In 99 patients (76.7%) one or more COI guideline deviations were found. Most common COI guideline deviations were a high starting dose of the systemic opioid compared to their previous oral opioid dose (57.6%), high COI dose for indication of dyspnoea (>15 mg/24 hr intravenously or subcutaneous morphine in an opioid naive patient) (22.2%), COI started or increased during CPS (respectively 18.2% and 19.2%) and morphine as COI in a patient with impaired renal function (eGFR <30 mL/min) (19.2%) (Figure 3). Regarding the latter, 34 patients who received COI had impaired renal function prior to the start of COI. Of these patients, 19 (55.9%) received morphine as COI. Overview of guideline deviations in COI. Left panel; proportion of patients with a guideline deviation in COI. Right panel; specific reason of guideline deviation. +An increase of more than 50% was considered too high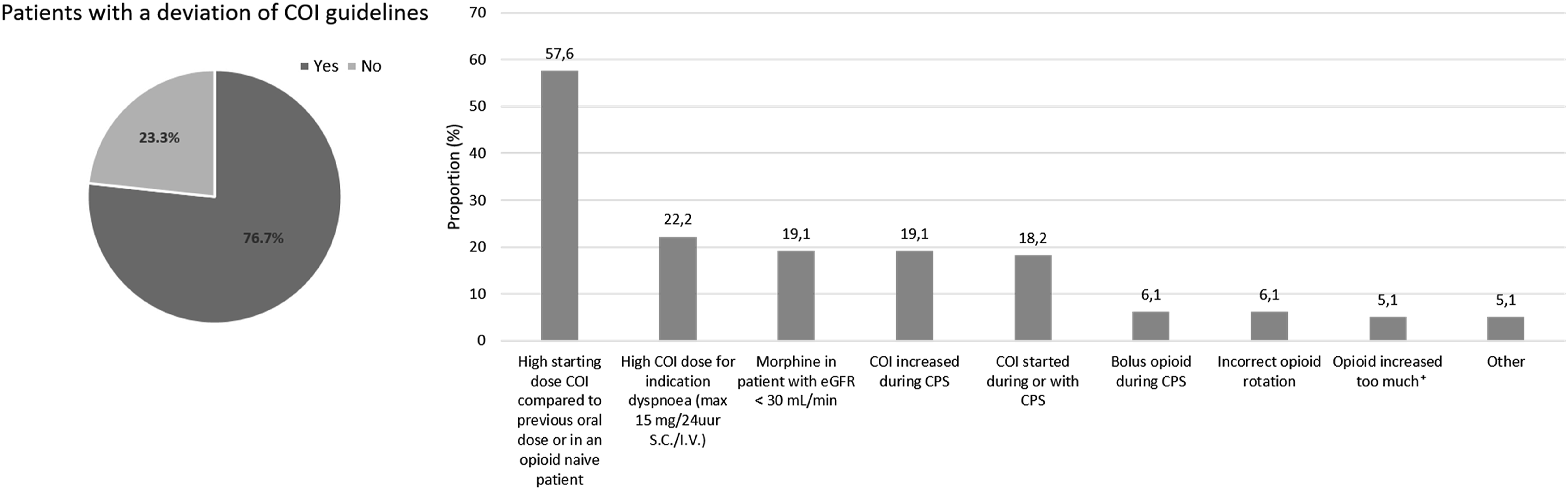
Involvement Palliative Consulting Team and Pain Team
The palliative consulting team was involved in 52 of the 101 patients (50.5%) receiving CPS and in 63 of the 129 patients (48.8%) receiving COI. Involvement of the palliative consulting team resulted in less clinically relevant CPS guideline deviations (48.1 vs 57.1%) and less COI guideline deviations (73.0% vs 80.3%). This is further specified in Appendix I.
The pain team was involved in 14 patients receiving COI (10.9%). For COI, 10 out of 14 patients (71.4%) had a guideline deviation if the pain team was involved. Notably, where the pain team was involved none of the patients received morphine as COI, compared to 19 of the 115 patients in which they were not involved. This is further specified in Appendix II.
Discussion
In the vast majority of patients receiving COI and patients receiving CPS, one or more (clinically relevant) guideline deviations was found. For patients receiving CPS, this was mainly attributable to the use of opioids during CPS.
Previous research mainly focused on self-reported knowledge of medical professionals on the existence of guidelines for CPS or on the start and implication of CPS, while the procedure after the start of palliative sedation remained largely unknown.6,13,14,15 To the best of our knowledge, we are the first to also report on deviations from the guidelines occurring after the start of palliative sedation and on COI guideline deviations in end-of-life care.
Continuous Palliative Sedation
To be eligible for CPS, one of the criteria is the presence of one or more refractory symptoms, meaning that no other (medical) intervention can relieve these symptoms quickly enough or without unacceptable side effects. 1 The addition of COI after the start CPS or the application of an opioid bolus can therefore be considered inappropriate care, potentially negatively impacting the effectivity of the sedation and introducing adverse effects. As already stated by both Reuzel et al. and Maréchal et al., sedation by an opioid is a side effect of an attempt to alleviate pain and thus will therefore not further improve the sedation when already started.5,13 Reuzel et al. reported that 46% of the medical specialists did not use benzodiazepines (including midazolam and levomepromazine) but opioids or antipsychotics as sedative. In the current study, we did not see the use of opioids with the intent to use as palliative sedation, only to alleviate pain or dyspnoea symptoms.
The report of a refractory symptom and life expectancy were often missing, but we expect that this is more likely due to a lack of reporting, rather than ineligibility of patients for CPS. Especially since the duration of CPS in all patients was shorter than two weeks and since the mean duration was comparable to the CPS duration reported previously.16,17
Continuous Opioid Infusion
Regarding COI guideline deviations, most deviations occurred in both COI and CPS, since COI was often increased or started during CPS. The type of COI was often matched with the previously used opioid, as is in concordance with the guideline for COI. 9 In opioid naïve patients starting with COI, morphine was the opioid of choice most frequently, consistent with previous literature. 18 Although an impaired renal function is a contraindication for the use of morphine, 55.9% of the patients with impaired renal function still received this as COI.9,19 Continuous infusion of morphine in patients with a GFR <30 mL/min can result in accumulation of active metabolites, potentially leading to adverse events. Therefore, COI in these patients is best initiated with a different opioid, such as fentanyl or oxycodone.
The guidelines for dyspnoea in the palliative phase recommend treatment with opioids of 15 mg/24 hr intravenously or subcutaneous MME in opioid-naïve patients.11,20 Therefore, if opioid-naïve patients received COI above this dose for this indication in the current study, this was reported as a guideline deviation. However the evidence for the application of opioids for dyspnoea is low-graded and a higher doses up to 30 mg/24 hr were investigated from which no clear optimal dose could be found.21,22 Further research is needed to elaborate on the preferred opioid dose for refractory dyspnoea.
Involvement Palliative Consulting Team and Pain Team
Even though not specifically investigated, the high number of guideline deviations of CPS and COI might be due to a lack of understanding of and education regarding the guidelines. Frequent and timely consultation of the palliative consulting team and, for COI specifically, the pain team might help to reduce the number of guideline deviations. Although the involvement of the palliative consulting team and/or pain team did not result in substantially lower numbers of guideline deviations, it is important to note that both teams were often involved after the start of CPS or COI. Since both teams only have a consulting function and although we did not report on this, noticed that the teams were often only consulted after difficulties in CPS or COI occurred. Furthermore, guideline deviations that were later corrected were still considered as guideline deviations in this study. Education by the palliative team on the guidelines for CPS in a Spanish study did result in a 100%-score when investigating alignment with the guidelines in the years following this. 6 This Spanish study mainly focused on the starting phase of CPS, however the follow-up procedures were not investigated. This approach and timely involvement of the palliative consulting team and/or pain team might also help to reduce the number of guideline deviations after the start of CPS or COI.
Study Limitations
This study has some limitations. Considering the retrospective nature of the study and the transition to another patient file system between the study period and the time of evaluation, some information regarding CPS or COI might have been missing. This mainly lead to missing information related to timing and dosing of boluses. Therefore, lacking information on boluses in between the increase of the CPS dose, was not considered as a guideline deviation. Reports of refractory symptoms and life expectancy should have been present in the available files and missing this information was therefore not considered a consequence of the transition to another electronic patient file system. Additionally, to generalize the results of this single-centre evaluation, additional research is needed.
Conclusion
In conclusion, this evaluation on deviations of COI and CPS guidelines found one or more deviations in COI and/or CPS in the majority of patients. Further research is required to identify methods to improve knowledge and improve the use of the guidelines on COI and CPS as end-of-life care.
Supplemental Material
Supplemental Material - Retrospective Evaluation of the Use of Continuous Palliative Sedation and Continuous Opioid Infusion in Hospitalized Patients in End-Of-Life Care
Supplemental Material for Retrospective Evaluation of the Use of Continuous Palliative Sedation and Continuous Opioid Infusion in Hospitalized Patients in End-Of-Life Care by J.L. Gulikers, J. Hamers, M. van der Zande, M. Theunissen, J.J.A.O. Schoenmaekers, M.H.J. van den Beuken-van Everdingen in American Journal of Hospice and Palliative Medicine®
Footnotes
Ethical Considerations
A non-WMO declaration by the medical-ethical committee of AzM/UM. Consent to participate and consent for publication were not applicable and waived by the ethics committee.
Author Contributions
JG: Investigation, Data curation, Formal analysis, Visualisation, Writing – original draft. JH: Investigation, Writing – review & editing. MZ: Investigation, Formal analysis, Visualisation, Writing – review & editing. MT: Conceptualising, Data curation, Methodology, Writing – review & editing. JS: Conceptualising, Writing – review & editing. MB: Conceptualising, Supervision, Writing – review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
