Abstract
Background
Advance care planning (ACP) is especially important in the context of cognitive impairment. Little is known about long-term effects of ACP on care partners.
Aim
To determine the sustained effect of facilitated ACP conversations on care partner-reported quality of communication (QoC) about end-of-life (EoL) care and readiness to engage in ACP at 24-months.
Design
A total of 273 participants were randomized 1:1 to a multicomponent ACP intervention (SHARE) or usual care. ACP was delivered between baseline and 6 months and dyads were followed through 24 months (N = 114). Outcomes included care partner responses to the 7-item QoC instrument and 6-item readiness to engage in ACP instrument. Group differences at 24 months were compared using ANCOVA, adjusting for baseline scores and cognitive impairment.
Setting/participants
Cognitively impaired older adults age 80+ and family care partners were recruited from 8 primary care clinics.
Results
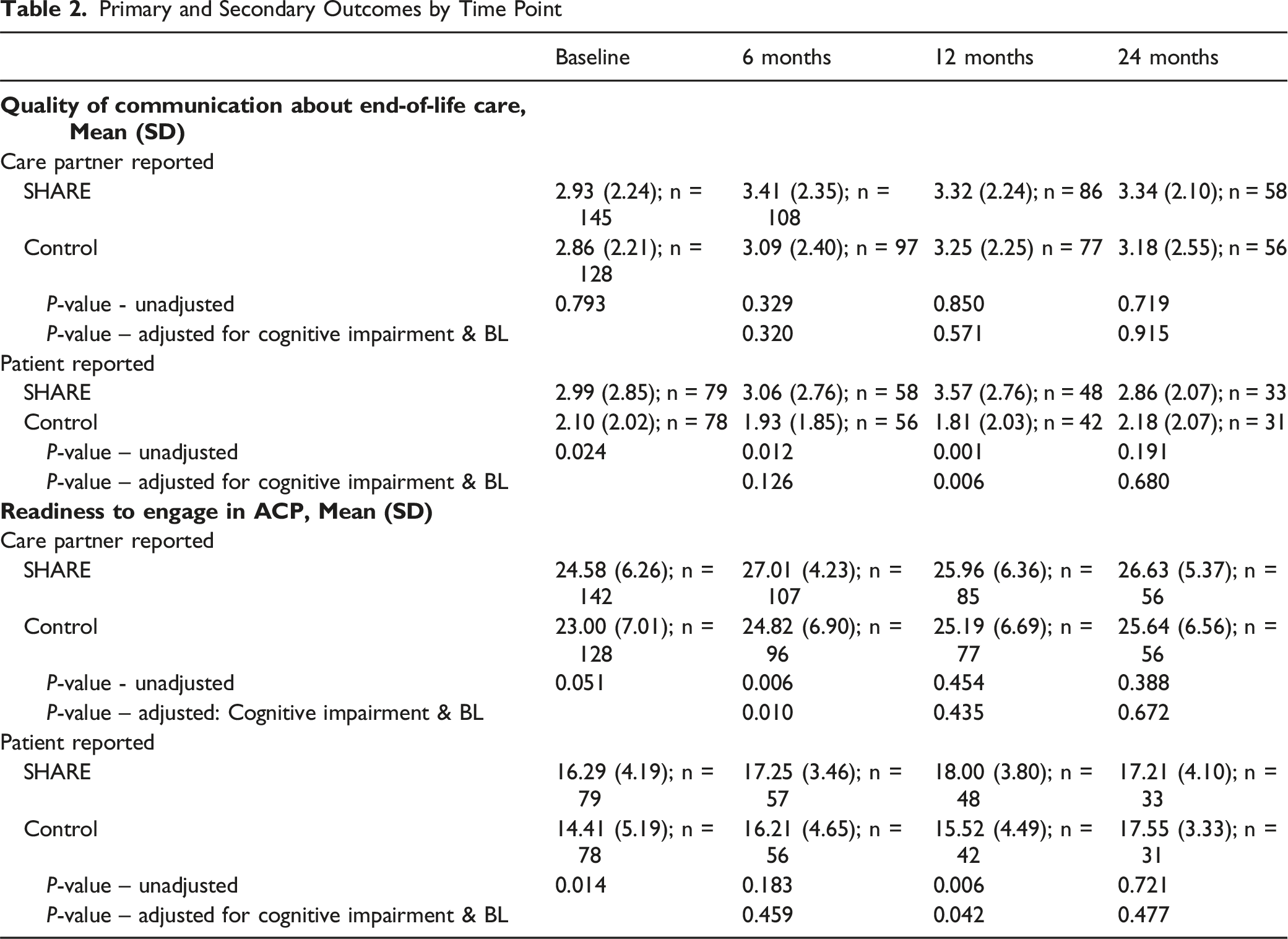
Care partners were mostly women (91.2%), average 65.5 years old, and adult children (66.7%) of the patient. 114 (42.8%) care partners completed 24-month assessments; One fifth of patients died within 24 months (n = 61; 22.3%). Caregiver QoC scores at 24 months were similar for intervention and control groups (3.34 vs 3.18; P = 0.92). Similarly, readiness to engage in ACP was comparable for intervention and control groups (26.63 vs 25.64; P = 0.67).
Conclusions
ACP conversations did not significantly affect QoC or readiness to engage in ACP at 24 months follow-up in this randomized trial. ACP may require an iterative approach for ongoing benefit in this population.
Keywords
Background
More than 7 million people in the U.S are living with Alzheimer’s Disease and Related Dementias (ADRD). 1 The progressive and debilitating effects of cognitive impairment on decision-making capacity amplifies the importance of including family in conversations about future care and treatment preferences early in the disease course2-5 through longitudinal advance care planning (ACP).2,6 ACP is associated with care partner preparedness for engaging in surrogate decision making and greater awareness and understanding of patient’s end-of-life wishes.7,8 However, barriers to initiating ACP conversations, such as discomfort and a tendency to avoid conversations about end-of life care, are notable.9,10 Also, family are not routinely engaged in ACP discussions between patients and providers. 11 ACP is especially challenging in the context of cognitive impairment2,12 and completion of advance directives are less common in persons living with ADRD. 13
Although prior research suggests ACP confers benefit, few studies have examined the longer-term effects, particularly for care partners who may benefit the most from ACP. 14 This study presents findings from a randomized controlled trial of a multicomponent dyadic communication intervention that included facilitated ACP conversations in a population of adults aged 80 and older with cognitive impairment. We previously described results at 6 and 12 months that found benefit for aspects of ACP processes and communication about end-of-life care. 15 This paper examines whether those beneficial effects were sustained for care partner-reported outcomes at 24 months.
Methods
Design, Participants, Recruitment and Setting
This analysis examines 24-month outcomes from a randomized trial, Sharing Healthcare Wishes in Primary Care (SHARE). The study was approved by the Johns Hopkins Medicine Single Institutional Review Board (00242431) and monitored by a Data Safety and Monitoring Board. The study tested a multicomponent communication intervention among older adults age 80+ years old with cognitive impairment and their family care partners. The study protocol has been registered (NCT04593472) and published. 16 Patients were recruited from 8 primary care practices across 2 health systems in the Baltimore-Washington region. Eligible patients were English speaking, screened positive for cognitive impairment with a cognitive screening tool and received unpaid care from a family member willing to participate. Care partners were aged 18+, English speaking, and attended doctor visits with the patient. Upon providing informed written or verbal consent and completing a baseline survey, patient-care partner dyads were randomized 1:1 to either the SHARE intervention or control group. The principal investigator, staff conducting the follow-up assessments and biostatisticians assessing the outcomes were blinded from randomization. Follow-up assessments were fielded at 6, 12 and 24 months. Recruitment, enrollment, and follow-ups were conducted from October 2020 to February 2025. The intervention processes 17 trial results, 15 and descriptive qualitative analyses have previously been reported.12,18
SHARE
Participants randomized to SHARE were mailed an introductory packet from the primary care practice, with an invitation to schedule a meeting with a facilitator trained to lead ACP conversations. Resources and educational materials included a person-family agenda setting checklist to align perspectives about the role of family and stimulate discussion about health care issues, a blank advance directive, a guide for communicating with clinicians, and instructions for completing proxy registration for the patient portal. ACP facilitators, who were not required to have a clinical background, received training in all elements of SHARE, which included the Respecting Choices ACP curriculum, focusing on ACP in the context of cognitive impairment and care partner involvement. 19 Participants were provided contact information for ACP facilitators and encouraged to request an appointment via video or teleconference. Facilitators led ACP conversations and assisted with completing advance directives. Conversations were audio-recorded and reviewed to monitor SHARE protocol, with feedback and support provided to facilitators as needed. Facilitators documented the ACP conversation in REDCap and the patient’s electronic medical record.
Outcomes
Older adult and care partner participants were administered assessments by phone or electronic REDCap surveys. The primary outcome was the care partner’s response to the 7-item end-of-life (EoL) subscale of the quality of communication (QoC) questionnaire - a validated instrument widely used in ACP research. Each item is rated on a scale from 0 (“worst”) to 10 (“best”) with a summary score representing the average of all items and higher scores indicating better QoC20,21 as previously described. We additionally examined care partner responses to a 6-item readiness to engage in ACP instrument with item responses rated on a 5-item Likert scale ranging from 1 (“I have never thought about”) to 5 (“I have already done it”)22,23 with summary score ranging from 0 to 30, and higher scores indicating greater readiness.
Covariates
Cognitive status in older adults was reported at baseline in 2 ways. First, patients were fielded the modified telephone interview for cognitive status (TICS-m),24,25 a widely used tool measuring one’s cognitive function where higher scores mean better cognitive function, which was categorized based on established cut-points: severe (0-27), moderate (28-31), and normal/mild cognitive impairment (32-50). Second, we examined care partner reports of older adults being previously diagnosed with dementia or cognitive impairment.
Statistical Analysis
Analyses of covariance (ANCOVAs) were used to evaluate treatment effects on continuous outcomes with randomly assigned treatment group as the primary independent variable and baseline score as a key covariate. We examined data for 114 care-partners who completed 24-month assessments. Quantitative analyses were conducted using SAS 9.4. Group differences in outcomes were compared using ANCOVA, adjusting for baseline scores and cognitive impairment level. We also descriptively examined differences in both outcomes for SHARE and control groups stratified by cognitive impairment: severe cognitive impairment (0-27) vs moderate/mild/no cognitive impairment (28+).
Results
Of the 273 patient-care partner dyads who enrolled in SHARE, 114 (41.8%) care partners completed 24-month data collection: 61 patients had died (22.3%) and 96 (35.2%) were lost to follow-up Figure 1). Table 1 describes the characteristics of patient-care partner dyads. SHARE CONSORT diagram Enrolled Study Participants Completed 24-Month Assessment Note. Percentages may not sum due to rounding. aBased on TICS-M cut point of <28 of 50; higher values indicate better cognitive function. bReflecting patient responses, when reported, and care partner, for those who did not self-report survey responses, with higher scores indicative of better quality of life using QOL-AD. Range: 13-52. cCaregiver burden measured with 13-item Zarit Burden Inventory +1 global item as reported by Bedard, 2001. Each item is scored 0-4 for a range of 0-52, higher scores indicating greater burden. dAnxiety measured using GAD-7; range 0-21, higher scores indicating greater anxiety.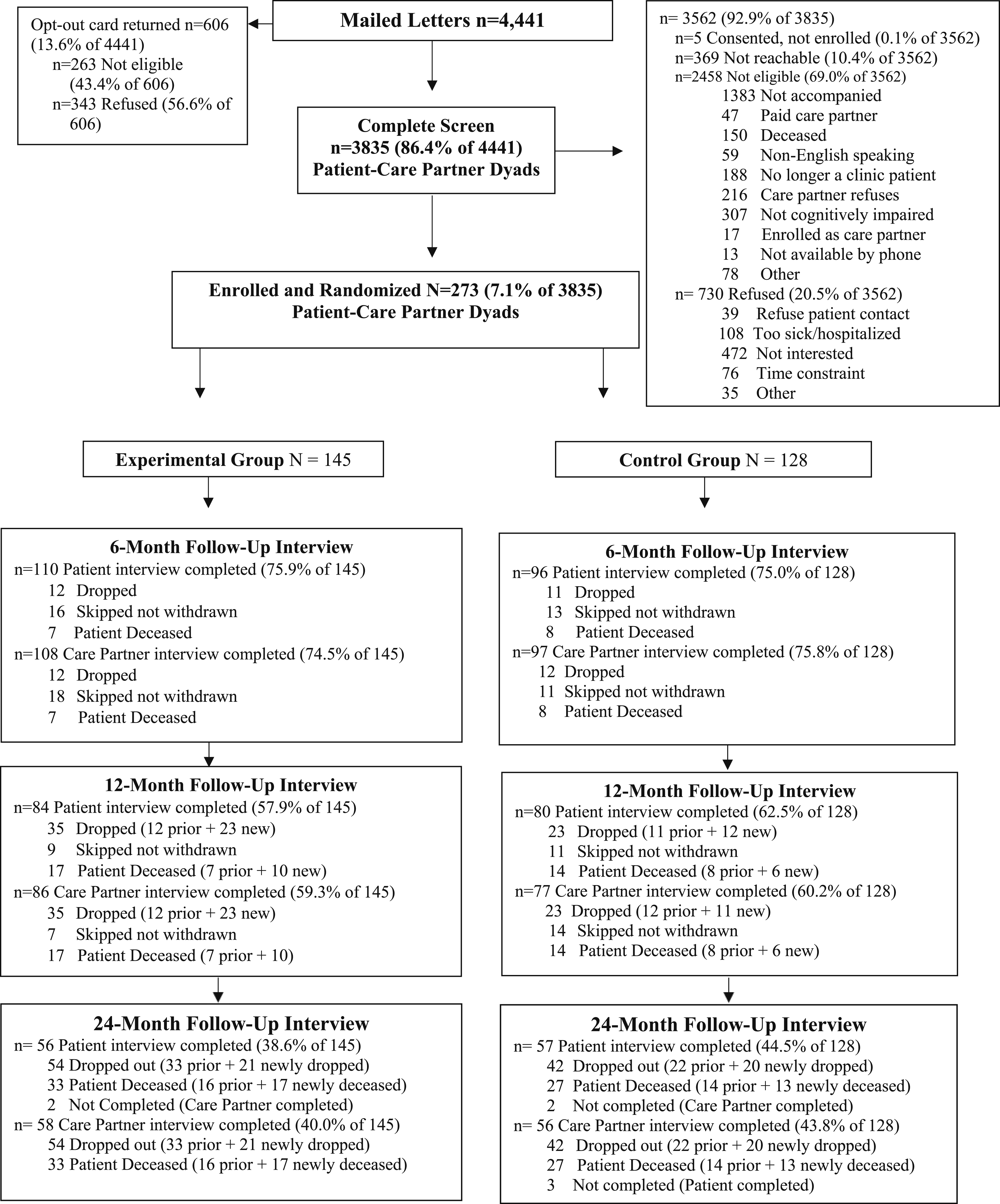
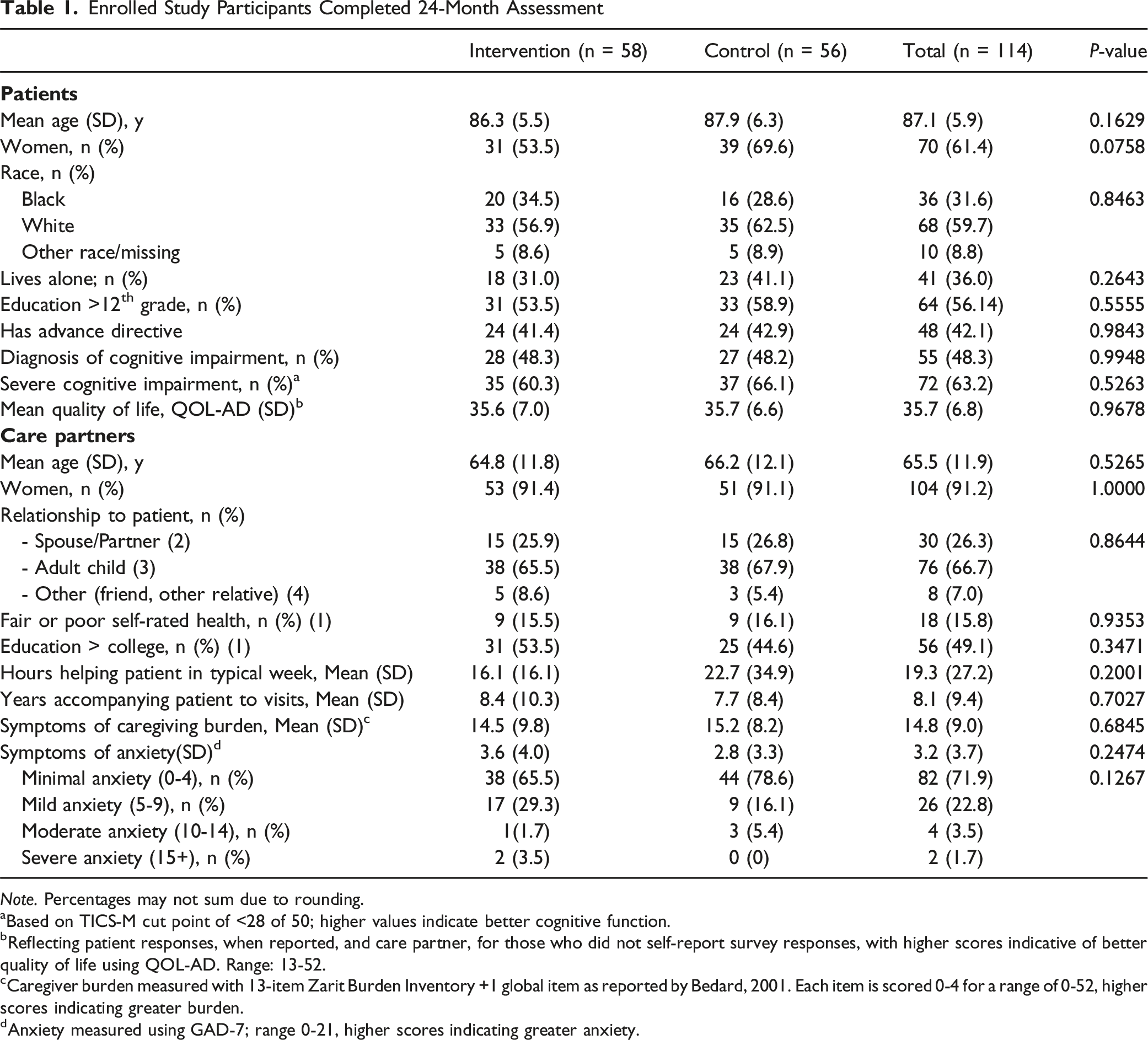
Care partners were on average 65.5 years at baseline and were mostly women (91.2%). Two-thirds (n = 76; 67.7%) were adult children with the remainder mostly being spouses of the patient (n = 30; 26.3%). Care partners provided 19.3 hours of care per week on average (ie, caregiving) and had accompanied patients to doctor visits for an average of 8 years. No between-group differences were observed in patient or care partner characteristics.
Quality of Communication
Primary and Secondary Outcomes by Time Point
Readiness to Engage in Advance Care Planning
Mean care partner-reported readiness to engage in ACP scores were somewhat higher in the SHARE (versus control) group at baseline (Ms = 24.58 vs 23.00; P = 0.05) and numerically increased in both groups at 24 months but were not significantly different across SHARE and control groups (Ms = 26.63 vs 25.64; P = 0.67) either before or after adjusting for baseline score and cognitive impairment. At 24 months, after stratifying by patient cognitive impairment level, no significant differences were observed between SHARE and control groups for care partners of patients with severe impairment (adj Ms = 25.83 vs 25.05, P = 0.55) or for care partners of patients with more mild or moderate cognitive impairment (adj Ms = 27.60 vs 26.90, P = 0.56).
Discussion
This study reports 24-month outcomes from a randomized controlled trial of an ACP intervention among older adults with cognitive impairment and their care partners in the primary care setting. We found no notable differences in care partner reported QoC about EoL or readiness to engage in ACP at 24 months, contrary to earlier effects showing improvements in these outcomes at 6 and 12 months after intervention onset.
Study findings contribute to a gap in understanding long-term outcomes of ACP. Previous research 26 examining long-term effects of ACP on patient-care partner congruence and decisional conflict on goals for end-of life care preferences reinforced the need to conceptualize and operationalize ACP as an iterative discussion to accommodate changing patient experiences. Revisiting care preferences is critical, not only to strengthen communication strategies between patients, family care partners, and providers, but also for ensuring that care preferences accurately reflect changes in health conditions, including cognitive decline.27-29 Our study findings align with the need to evolve from the idea of ACP as a single primary care event to ongoing discussions engaging care partners to better prepare them for patient end-of-life care and decision making.
Mixed outcomes of trialed ACP interventions and nuances in the assessment of outcomes have garnered attention30,31 and motivated some to question the value of ACP discussions in clinical care.6,32,33 Providers have reported challenges with delivering ACP in the context of cognitive impairment – recognizing that doing so may require additional time, questions about decisional capacity, the need for nuanced communication, and the involvement of family2,13 Efforts to develop more appropriate measures should be grounded in these key domains, since existing tools, such as the ACP engagement survey, may not be appropriate measures of ACP efficacy in this population.34,35
Our research drives home the importance of measurement in ACP interventional research. There is a compelling need for rigorous, responsive measures that reflect outcomes that matter: such measures are especially challenging in the context of dementia and care partner involvement.2,12,36 Prior dementia care-planning interventions that integrated an iterative approach by periodically revisiting care plans after the initial intervention have led to caregiver benefit as reflected by reduced burden associated with a surrogate role.37,38 However, important gaps exist with respect to assessment of other ACP-related outcomes. Sustaining improved ACP outcomes will require addressing not only current health priorities and preferences, but ongoing conversations to provide support and resources especially in circumstances where health conditions worsen.
Additionally, interventions that integrate a multidisciplinary care team (nurses, social workers, etc.) equipped to address end-of-life care needs and support longer-term ACP, including family involvement, could generate measurable impact.6,39 Caregiver education and training in navigating ACP discussions can be especially valuable and important in the context of dementia care, which often transpires over an extended period – and can be especially challenging in primary care given constrained time and resources.2,12,40 Equipping caregivers with practical tools to navigate ACP may potentially fill this gap. 41 While many initiatives42,43 have focused on clinician or nurse training in ACP communication tools, less attention has been directed at implementing a family-centered approach, which may directly benefit caregiver readiness to explore end-of-life care treatment options and attenuate emotional burden associated with surrogate decision making. 14
This study has several limitations. Just one-third of SHARE care partners provided 24-month outcome data for a range of reasons, including patient and care partner mortality, changes in primary care providers, and study attrition. The study design was notably altered from in-person to virtual modalities due to the covid-19 pandemic, which affected both the nature of ACP conversations as well as the strength of the connection of ACP facilitators to primary care practices. Still, this is the first study to report 24-month outcomes for care partners of patients 80 years and over with cognitive impairment in primary care.
Conclusion
Although significant treatment effects for the SHARE intervention were observed earlier in the trial, these effects were not sustained at 24 months post intervention. Additionally, we observed no significant group differences in treatment when analyses were confined to subgroups based on the level of patient cognitive impairment, This study adds to the ongoing research highlighting the importance of integrating regular ACP discussions in primary care visits, especially for older adults with cognitive decline. Our study suggests that a single ACP conversation is not sufficient to sustain improvement in quality of communication with providers. Future research should focus on scalable models for sustained engagement and improved outcome measurement.
Footnotes
Ethical Considerations
The study was approved by the Johns Hopkins Medicine Single Institutional Review Board (00242431) and the local Institutional Review Boards at the University of Maryland and MedStar Research Health Institute on April 13, 2020.
Consent to Participate
All human subjects provided verbal or written informed consent.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The SHARE Trial was funded by NIA (R01AG058671, PI: Wolff).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data can be obtained by directly contacting the corresponding author.
