Abstract
Background
In palliative care, drug delivery methods must be simple, rapid and acceptable especially in homecare settings to relatives as non-professional caregivers. This study compared intranasal (IN) and subcutaneous (SC) administration performed by medical laypersons under standardized conditions.
Methods
31 volunteers without medical training participated in a non-randomised crossover study. After receiving instructions, participants performed both SC and IN administration in a simulated environment using placebo medication. Primary endpoints were preparation and administration time. Secondary endpoints included perceived ease, comfort, safety and handling difficulties. Data were collected using structured questionnaires. Statistical analyses used paired t-tests and binomial testing.
Results
All participants (27 female, median age range 50-60 years) completed both procedures. Mean administration time: 4:49 minutes (SD 1:20) for SC, 1:16 minutes (SD 0:20) for IN (p < .001, d = 2.97). >90% preferred IN and rated it as easier (30/31, 97%), more comfortable (30/31, 97%), faster (30/31, 97%) and safer (21/31, 68%). SC was consistently rated more complex (30/31, 97%) and cumbersome (30/31, 97%). Handling errors were more frequent with SC application. Nearly half of participants (15/31) had prior SC experience, while all had personal experience with nasal sprays, though not in administering them to another adult. Participants emphasized the importance of training for safe and accurate administration.
Conclusion
Medical laypersons strongly preferred IN over SC administration. IN was significantly faster, more acceptable and associated with fewer handling problems. These findings support IN administration as a practical and caregiver-friendly alternative in palliative care.
Clinical Trial Number
Not applicable.
Trial Identification
173-02, protocol Version 02, 03.11.2022.
Introduction
In palliative care, rapid and simple drug administration is often more decisive for patient comfort than the availability of new substances. There have been limited new drug approvals and several medications are used off-label. To enhance the care of palliative care patients, it is essential to optimize the route of drug administration, prioritizing those methods that minimize patient burden while also being straightforward for caregivers to administer.
A commonly used method is subcutaneous (SC) injection. The injection is administered into the SC adipose tissue, typically in the abdominal or thigh region. This method is minimally invasive and allows for a slow, steady release of the drug into the bloodstream. It is commonly used for the administration of insulin in diabetic patients or heparin for thromboprophylaxis. Furthermore, sedatives like midazolam are often administered off-label through this route. A key advantage of SC administration is the feasibility of self-injection by the patient. However, this method requires expertise and may cause hesitation or discomfort for both patients and caregivers due to the risks associated with needle-based injections. 1
An alternative, potentially gentler and more accessible administration method is intranasal (IN) delivery. The IN medication administration has become a well-established method in emergency medicine and for seizure management due to its rapid onset of action. 2 Medication can be absorbed into the systemic circulation through the highly vascularized nasal mucosa, bypassing the first-pass effect.3,4 However, only small volumes, ideally 0.3 to 0.5 mL per nostril, with a maximum of 1 mL, can be administered. Larger volumes may risk leakage into the nasopharyngeal area, potentially delaying or reducing efficacy.5,6 IN drug administration has also emerged as an effective and convenient treatment method particularly for palliative care patients. 7 In the palliative care setting, several medications have shown efficacy when administered IN such as fentanyl (indication: breakthrough pain management, with rapid onset and long-term safety), 8 ketamine (promising for treating depression, also in palliative care patients)9,10 and midazolam which is currently under investigation for terminal agitation treatment.11,12 Compared with SC injections, nasal sprays are familiar, non-invasive and require fewer handling steps, which may lower barriers for untrained caregivers.
While both administration methods, IN and SC, have their advantages, the choice often depends on individual patient factors, the medication being used and the clinical context. In palliative care, the timely relief of acute symptoms is often facilitated by delegating the administration of as-needed medications to caregiving relatives. To ensure both safety and effectiveness, these medications must be designed for ease and practical use, enabling individuals without formal medical training to administer them correctly. Simple and user-friendly application not only supports reliable symptom control but also helps to reduce the emotional and practical burden on family caregivers, who are frequently untrained in medical procedures.13,14 Overall, the limited body of research suggests that IN administration may offer advantages for medical laypersons in certain situations, particularly in terms of ease of use. However, there are neither direct comparative studies on IN vs SC administration by laypersons nor further studies to evaluate its effectiveness, safety and patient preferences across various contexts such as palliative care. To address this gap, we conducted a simulation study in which medical laypersons performed both IN and SC administration under standardized conditions. The aim was not only to measure objective parameters such as preparation and administration time but also to gather subjective feedback from participants, since these insights may inform how relatives can be better supported in administering medication at home. Direct, controlled head-to-head comparisons of IN vs SC performed by laypersons remain limited. Our study adds data to an area where intuitively held assumptions are rarely tested under controlled conditions.
Methods
Study Design and Setting
The research project was designed as a monocentric, crossover, open label, non-randomised simulation study compared IN and SC administration under standardized conditions. Each participant performed both routes once on a dummy model in a prepared treatment room designed to approximate a real-world environment. The sequence of IN and SC administration was not prespecified, as it was not considered a variable that would affect outcomes. Blinding was not possible due to the nature of the study procedures.
Participants and Recruitment
Medical laypersons were defined as individuals who may be employed in the healthcare sector but do not have direct patient contact and do not provide clinical care. They had no formal professional training as physicians, nurses, medical assistants or other healthcare professionals involved in patient care. Participants were recruited on a voluntary basis via email or in person. Rather than issuing individual invitations, several hospital departments meeting the inclusion criteria were contacted and asked to circulate study information through internal distribution lists. This recruitment strategy aimed to include participants from different non-medical professional backgrounds, with varying levels of medical knowledge (none to moderate). Apart from the requirement of legal adulthood, no selection based on age or gender was applied. Eligible participants were voluntary, healthy adults aged ≥18 years who were able to understand the nature and scope of the study. Exclusion criteria included non-consent and physical limitations that could impair handling of ampoules, syringes or nasal spray bottles.
Training and Procedures
Participants were pre-trained on the procedure for both IN and SC-bolus administration via video presentation and written instructions at the study site. Following the training by qualified research group members, the participants were required to perform each administration method on a dummy. No specific order of the two administration methods was specified. No time limits were set. The duration of each procedure was measured and participants were subsequently asked to identify which method they found easier and which one they would prefer for themselves and others. Problems and questions that arose during the implementation were also documented. The quality of the training materials was assessed by the participants. Study results were recorded on paper data collection forms. Personal data including age and gender for the demographic description of the participants and previous experience with medication administration and exclusion of participants with medical professional training were collected. No directly identifiable personal data were collected and individual participants cannot be identified from the dataset.
Time measurement was conducted by a member of the research group and documented together with any deviations or issues encountered by the participants during the drug application. Timing covered the intervall from the start of preparation to completion of the drug administration process. Minor deviations related to time measurement inaccuracies (for example small systematic or random delays in starting or stopping the timer) were considered negligible, as the analysis focused on time ranges rather than exact second-by-second accuracy and on substantial differences between ready-to-administer and manually prepared dosage forms.
After performing both administration methods, a final evaluation and comparison was made by the participants.
The assessment of preference and acceptance for each administration route was conducted using a survey questionnaire, with participants answering the questions in the data collection form (see Appendix 1 and 2). The study participants had the opportunity to provide additional assessments in the form of free-text responses. In addition, concluding questions were used to assess the quality and suitability of the training materials. This ensured that participants met the requirements for the correct preparation and administration and were able to put this knowledge into practice.
Materials
The dummy drugs were prepared at the local hospital pharmacy (31 ampoules of 1 mL water for injection (solvent of the final medicinal product Solu-Decortin H 25 mg, MERCK, 9238.01.00, medicines in glass ampoules were selected to simulate everyday clinical practice at the site), along with the necessary injection materials). Other study materials included disposable gloves, disinfectant (alcoholic skin antiseptic), 5 mL syringes, 27 G needles, 19 G needles and bandages (if necessary). Additionally, six nasal spray bottles were filled with approximately 7.5 mL of water for injection purposes and labelled as placebo. These nasal spray bottles could be reused by different participants during the study.
Ethics and Safety
As approved by the ethics committee, informed consent was provided verbally, based on an information letter, as no study interventions were performed on humans. The study was performed in accordance with the ethical standards of the Declaration of Helsinki.
Sample Size Rationale
No formal a priori sample size calculation was performed. The research project was planned as a pilot study and a minimum of 30 participants were included, corresponding to 25 participants based on the central limit theorem, plus an additional 20% dropout rate.
31 were ultimately enrolled. The paired (within-participant) design was chosen to increase statistical efficiency.
Statistics
All data from participants who completed the study correctly (per-protocol) were evaluated. Data was not personalized. Quantitative data for approximately normally distributed parameters are presented as means and standard deviations. For non-normally distributed data, median and range are reported. Qualitative data are described by their absolute and relative frequencies. To examine differences in application duration between IN and SC administration, a t-test for dependent samples was performed using time [minutes] as the dependent variable. Additionally, Cohen’s effect size (d) was calculated. To determine whether participants’ preferences for administration methods were statistically significant rather than due to chance, binomial tests were performed. These tests assessed whether the observed proportion of participants preferring 1 method over the other significantly differed from an expected 50:50 distribution (p = 0.5) that would occur if there was no inherent preference. One-sided binomial tests were used as we hypothesized, based on preliminary research and clinical experience, that intranasal administration would be preferred for positive attributes (easier, more comfortable, safer, faster), while subcutaneous administration would be associated with negative attributes (more burdensome, more complex). For each attribute, the statistical “success” category was assigned to the method hypothesized to be associated with that attribute. A p-value <.05 was considered statistically significant. Statistical analyses were performed using R Studio (R Core Team, 2024).
Results
N = 31 participated the study (27 female, median age range 50-60 years), as shown in Figure 1. No protocol deviations occurred. Flow Chart Participant flow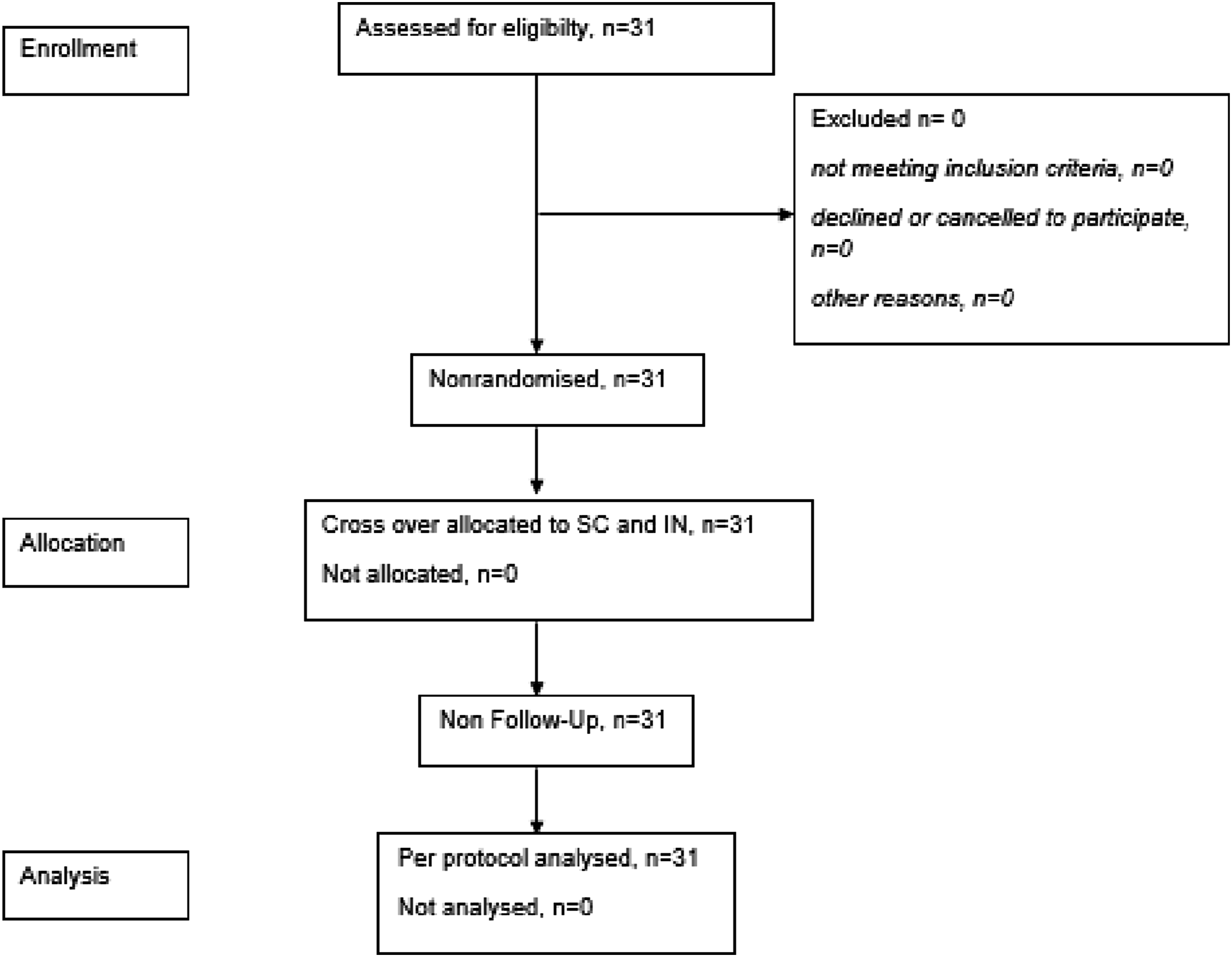
For the SC administration, participants required an average of 4:49 minutes (SD = 01:20, Range [3:00; 7:37]), while for the IN administration, the average time was 1:16 minutes (SD = 0:20, Range [0:42; 1:59]) as depicted in Figure 2. These differences between application methods were found to be highly significant (t(30) = −16.6, p < .001) with a large effect size (d = 2.97). Comparison of time [minutes] required for drug administration between subcutaneous and intranasal application methods by medical laypersons. Box plots show the distribution of time (in minutes) needed to complete the administration process for each method. Boxes represent interquartile range with median line; whiskers extend to minimum and maximum values within 1.5 times the interquartile range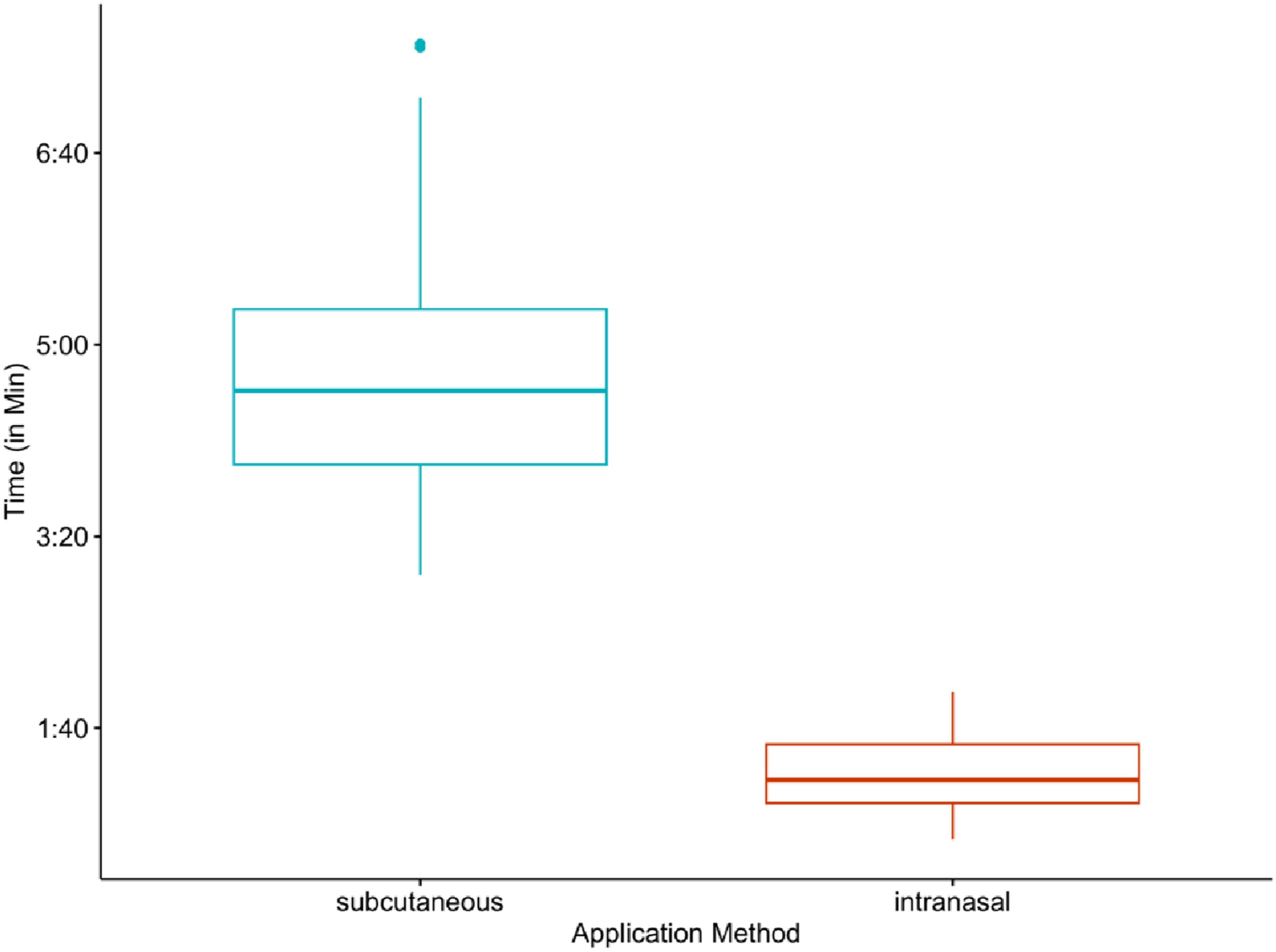
Participant Preferences and Perceptions of Intranasal Versus Subcutaneous Administration Methods
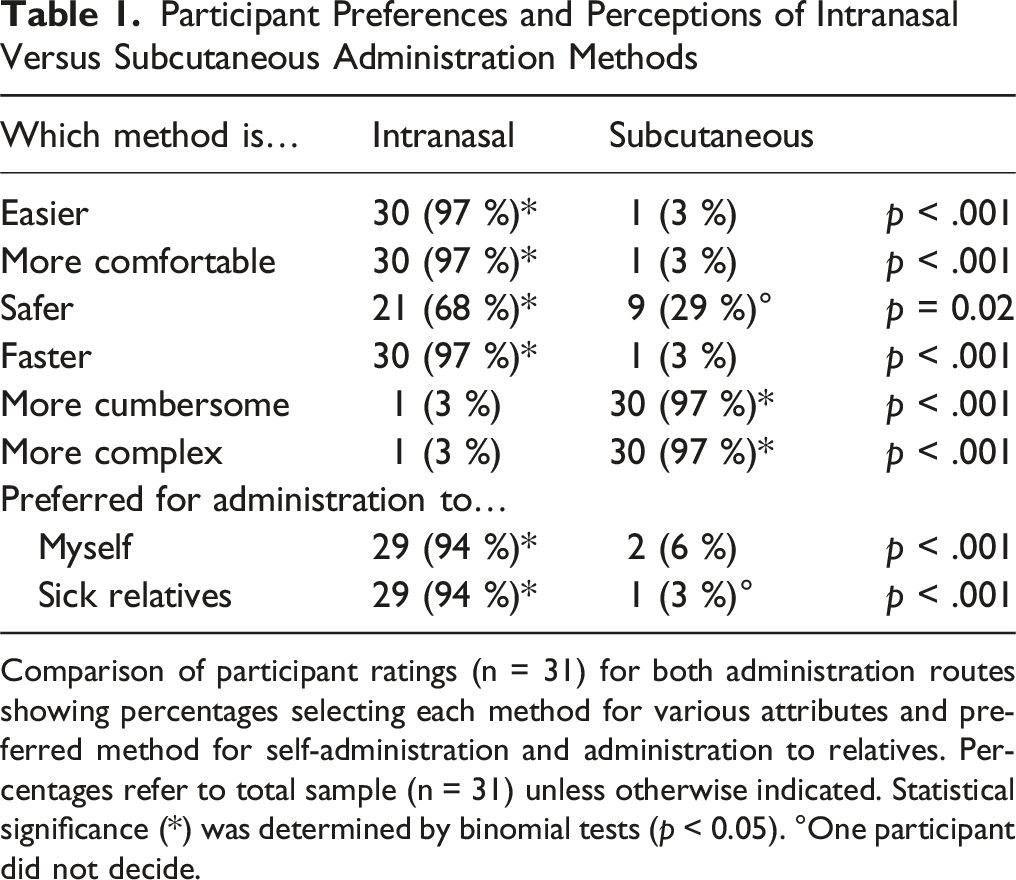
Comparison of participant ratings (n = 31) for both administration routes showing percentages selecting each method for various attributes and preferred method for self-administration and administration to relatives. Percentages refer to total sample (n = 31) unless otherwise indicated. Statistical significance (*) was determined by binomial tests (p < 0.05). °One participant did not decide.
Evaluation of Administration Including Feasibility and User Experience
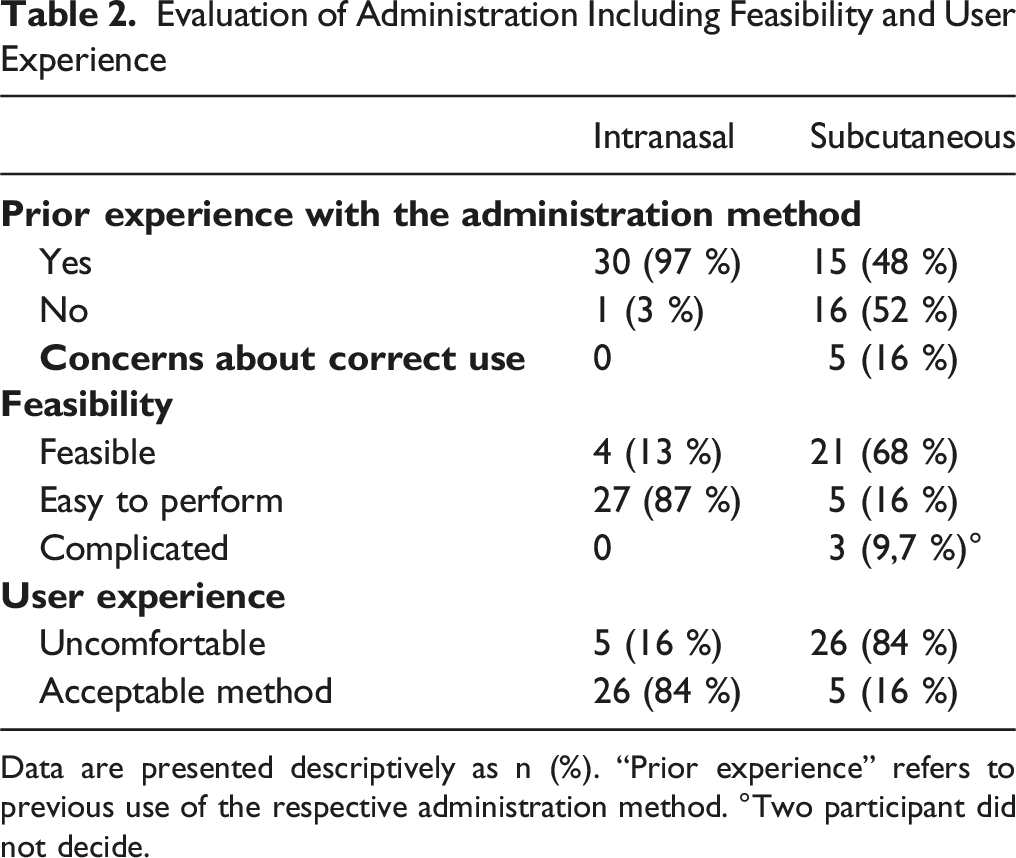
Data are presented descriptively as n (%). “Prior experience” refers to previous use of the respective administration method. °Two participant did not decide.
Types and Frequency of Issues

The training materials for SC administration were rated as good by 22 participants (71%) and as only sufficient by nine participants (29%). For IN administration, 26 participants (84%) rated the training materials as good, while five participants (16%) rated them as only sufficient.
Discussion
This study demonstrated that medical laypeople strongly preferred IN over SC administration under standardized conditions. Participants completed IN administration significantly faster than SC administration and rated it as easier, more comfortable and less cumbersome, with over 90% preferring IN administration for both themselves and when providing care to others. While previous studies and case reports indicated that IN administration leads to rapid drug absorption and is generally easier to perform than SC administration, this study provides to the best of our knowledge the first direct comparison of these two routes under controlled study conditions. This creates a stronger rationale for choosing one of the two dosage forms as administration method, considering the minimal burden on both the patient and their caregiving environment. The study was specifically designed within the context of palliative care 12 where ease of use is of particular importance.
Regarding the gender distribution (27/31 female participants), it can be noted that this imbalance roughly corresponds to the distribution of female caregivers in the literature, estimated at approximately 75%. 15 Participants with no or only minimal medical knowledge were deliberately selected to better reflect the majority of caregivers who would likely benefit the most from simple and uncomplicated medication administration methods. This approach ensured that the results reflected real-world caregiving conditions as the majority of informal caregivers lack professional medical or nursing backgrounds and therefore have limited in-depth knowledge in the administration of medications.
The study results were remarkably clear and consistent. The number of participants (n = 31) was already sufficient to achieve robust results with sufficient statistical power. Essentially, nearly all participants rated the IN administration as easier to handle. Notably, several participants identified difficulties with handling needles or the act of injecting another person. For family caregivers who may be hesitant or unable to inject the patient, IN administration offers a viable alternative and provides relief, particularly in stressful situations such as emergency interventions or end-of-life care.
The more tolerable and easily implementable IN administration could significantly enhance caregivers’ perceived self-efficacy, particularly in emotionally demanding circumstances such as pain exacerbations or terminal agitation. Caregiving self-efficacy, defined as caregivers’ confidence in their ability to manage care-related tasks, is strongly associated with reduced caregiver burden. 16 When caregivers possess higher self-efficacy, they experience lower stress levels and greater overall well-being. Caregivers who perceive themselves as capable of managing caregiving tasks are better equipped to handle anxiety and emotional distress, potentially leading to improved mental health outcomes for both themselves and their patients. 17 By reducing psychological strain, enhanced self-efficacy contributes to a more compassionate and effective caregiving experience.
Another key point of this study was the significantly shorter duration of application for IN administration. Using ready-to-administer nasal sprays was clearly superior in this regard and the direct comparison clearly substantiated the advantage of faster execution for IN applications. These ready-to-administer nasal sprays are also easier to store compared to syringes for SC administration, offering additional practical benefits in home care settings. While prefilled syringes could likely withstand this comparison, they were not evaluated within the scope of this study. At the study site, preserved, industrially manufactured prefilled syringes are not available. Therefore, syringes must be prepared in advance by nursing or caregiving personnel. This can be considered a potential hygienic risk, as unpreserved injection solutions should not be prepared more than one hour in advance. Since it is often unpredictable when they will be needed, medications and materials are left in place so that relatives can prepare them at short notice, as simulated in our study, as a single dose SC-bolus injection. In addition, indwelling needles were not included in the comparison, as agitated patients may occasionally remove them, and prolonged dwell time can increase the risk of skin infections.
A potential learning effect from prior experience must be acknowledged. About half of the participants reported earlier experience with SC injections, while all were familiar with nasal sprays in daily life, though none had administered them to another adult. It appears that there are no relevant differences in SC administration times between experienced and inexperienced participants, and the strong preference for IN administration across the entire cohort suggests that prior exposure did not materially bias the results. This was not part of the study and is therefore only an assumption and interpretation when considering the results. Prior experience may have involved prefilled syringes rather than the full process depicted here, including glass ampoule handling and syringe preparation. Nonetheless, the absence of a systematic assessment of prior IN experience represents a limitation of this study.
The study focused on feasibility and handling rather than precise dosing accuracy. Therefore, small differences in the exact administered volume between application forms cannot be entirely ruled out.
Further limitations include the lack of comparison with other SC administration options that have only been evaluated theoretically, such as SC injection via indwelling needles and the use of pre-filled syringes or ready-to-use syringes for SC administration. As a result, the preserved nasal spray may appear inherently superior as a ready-to-use dosage form in this comparison. At the same time, this reflects a practical advantage, as nasal sprays can be prepared by any public pharmacy and are immediately available when needed.
Some participants were non-native German speakers, which may have affected the comprehension of training materials and instructions. This underscores the importance of clear, accessible and linguistically appropriate educational resources when implementing such interventions in routine care.
In summary, this study, conducted within a controlled setting, demonstrated significant advantages of IN over SC administration in medical laypersons. The results of this study add to the evidence base suggesting that alternative routes such as IN are perceived as more user-friendly. The IN application was favoured for its faster and easier administration. Our findings provide systematic confirmation and help explain the underlying reasons, such as fewer handling steps and greater familiarity with nasal sprays. In this way, the study contributes to a better understanding of why IN administration may be more acceptable for caregivers and how this knowledge can be translated into practical guidance and training for everyday palliative care.
Thus, IN administration represents a meaningful viable alternative to SC administration, particularly in fields such as emergency medicine and palliative care. However, it should be noted that not all medications are suitable for IN application. Within the spectrum of medications used in palliative care, most opioids and benzodiazepines are particularly well-suited for IN administration, offering rapid absorption and a quick onset of action.6,18 The results of this study highlighted the greater ease of IN application, which can be an important relief for caregiving relatives, especially in difficult and stressful situations, such as terminal agitation in highly palliative care terminal patients.
Supplemental Material
Supplemental material - Feasibility of Intranasal Versus Subcutaneous Drug Administration: A Non-Randomised Crossover Study
Supplemental material for Feasibility of Intranasal vs Subcutaneous Drug Administration: A Non-Randomised Crossover Study by Hanna Hirschinger, Constanze Rémi, Eric Thanbichler, Wolf-K. Hofmann, Deniz Gencer and Tobias Boch in American Journal of Hospice and Palliative Medicine®.
Footnotes
Acknowledgments
Svetlana Hetjens, Department of Medical Statistics, Biomathematics and Information Processing, University Medical Center Mannheim for statistical advice. Palliative care ward of the 3rd Department of Medicine, Medical Faculty Mannheim for providing the dummy model. Anja Spiegel, Pharmacy, Mannheim University Hospital for assistance with the practical implementation. The 31 participants.
Ethical Considerations
Conducted in accordance with the Declaration of Helsinki, reported to Ethics Committee II of the University of Heidelberg, Medical Faculty Mannheim (TEMP830842-AF 5). No further ethic approval necessary.
Consent to Participate
Informed consent was provided verbally, based on an information letter, as no study-related procedure involved humans.
Author Contributions
[HH] planned and realized the trial with [DG and CR]. [ET] and [TB] did the statistical analysis. [WKH] is head of the working group. [HH] drafted the manuscript, which was critically revised by [CR, ET, WKH, DG and TB]. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Fund by 3rd Department of Medicine, Medical Faculty Mannheim, University Medical Center Mannheim, Heidelberg, Theodor-Kutzer-Ufer 1-3, 68167 Mannheim, Germany. For the publication fee we acknowledge financial support by Deutsche Forschungsgemeinschaft within the funding programme “Open Access Publikationskosten” as well as by Heidelberg University. Open Access funding enabled and organized by Projekt DEAL.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The minimal dataset necessary to interpret, replicate, and build upon the findings reported in the article is included within the article. The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
