Abstract
Background
As cancer remains a leading cause of death for older adults, ensuring high-quality end-of-life (EOL) care is a critical component of cancer care. Prior studies have predominantly assessed EOL care for racial and ethnic disparities or among one cancer type. Therefore, using a population-based sample, our objective was to describe EOL care among decedents with cancer across various social and clinical characteristics.
Methods
This retrospective cohort study used 2003-2019 data from The University of North Carolina at Chapel Hill Cancer Information and Population Health Resource, which links cancer registry to administrative claims data in North Carolina. We examined five EOL practices within the last 30 days of life including: >1 emergency department (ED) visit, intensive care unit (ICU) admission, hospital admission, intravenous (IV) chemotherapy, and no hospice use. We assessed in-hospital death and late hospice initiation in the last 3 days of life. We descriptively assessed differences in EOL by various demographic characteristics.
Results
Among 150 412 decedents, 48% were female, 23% were non-Hispanic Black, and 61% were Medicare beneficiaries. The most frequent EOL care practice was hospital admission (54%). Over time, no hospice use, ICU admission, hospital admissions, and in-hospital deaths decreased. However, in 2019, hospital admissions and no hospice use were still high (52% & 38%, respectively). Compared to patients with other insurance, Medicaid decedents had the best quality EOL care: ED visit (9%), hospital admission (45%), and hospice initiation in the last 3 days (6%).
Conclusions
Additional research is needed to understand what could be driving these observed differences. Interventions, such as EOL patient navigation, could improve upon EOL care in patients with cancer.
Background
Cancer is the second most frequent cause of death in the US, and for adults under the age of 85, cancer is the leading cause of death. 1 Therefore, it is important to understand how we care for individuals with cancer at the end of their lives. 2 According to the National Cancer Institute, end-of-life (EOL) care is meant to provide comfort & control symptoms (ie, social, physical, emotional, spiritual) to patients who are near the end of their lives and, specifically within the cancer context, who have stopped cancer treatments, whether curative or not. 3 For example, certain EOL care practices, such as chemotherapy in the last month,4-6 is not considered appropriate as this practice continues care and/or does not provide comfort care at the end of life. Alternatively, initiating hospice use within the last few months of life (as opposed to in the last few days of life) is considered an appropriate way to care for patients at the end of their lives as hospice aims to increase comfort at the end of the patient’s life. 7
However, there are subsets of patients who are more likely to receive inappropriate EOL care, such as patients who are of racial and ethnic marginalized groups or individuals with low incomes.8,9 This can be attributed to access to care issues including underinsurance and limited patient and caregiver knowledge about various services available toward the end of life, such as hospice and palliative care. Furthermore, in a commentary by Perry and colleagues, other reasons for EOL disparities include patient values and preferences, medical mistrust, and the number of primary care visits. 10 As many studies have focused on racial and ethnic disparities or only a specific cancer type when assessing EOL cancer care,11-14 there is a need for studies that systematically and comprehensively examine EOL care and subsequent disparities across various sociodemographic factors, clinical characteristics, and over time. Therefore, using a population-based sample, our objective was to describe the quality of EOL care among patients with cancer over time and to identify age-, race-, place-, and/or insurance-based disparities in EOL care. We have focused on these characteristics as prior research has found that disparities exist for these factors across various cancer-related outcomes including survival.15-17
Methods
Study Design and Participants
This retrospective cohort study used data from the University of North Carolina at Chapel Hill (UNC) Cancer Information and Population Health Resource (CIPHR). CIPHR links North Carolina Central Cancer Registry (CCR) data to Medicare, Medicaid, and private health insurance administrative claims. 18 Overall, 100% of patients with cancer who are diagnosed within North Carolina are included in the CCR and, of those, 85% are linked to insurance claims. Inclusion criteria for our study included the following: patients diagnosed with first or only cancers (as we were interested in understanding how the primary cancer diagnosis affected EOL care. Similar logic was utilized in a similar study conducted by our group 11 ); who were aged ≥18 years old; who had a record of death between 02/2003 and 12/2019; insurance coverage in the month of and preceding their death; and one of the following cancers of interest: breast, prostate, lung, colorectal, melanoma, endometrial, stomach, liver, cervix, myeloma, larynx, or pancreas. Cancers of interest were chosen as these were either the most common cancer types diagnosed in our catchment area or individuals in our catchment area had the highest mortality among these cancer types. Decedents diagnosed with cancer from autopsy or death certificate were excluded since their diagnosis occurred after death. This study was approved by UNC’s Institutional Review Board (IRB 22-2598).
Outcomes: End-of-Life (EOL) Care Practices
We examined seven end-of-life care quality measures; five of these measures were within the last 30 days of life and all of the following EOL care practices are considered inappropriate.6,19-28 Each outcome was categorized as yes or no. 1. >1 emergency department (ED) visit in the last 30 days of life 2. Intensive care unit (ICU) admission in the last 30 days of life 3. Hospital admission in the last 30 days of life 4. Intravenous (IV) chemotherapy in the last 30 days of life 5. No hospice use in the last 30 days of life 6. Late hospice initiation (in the last 3 days of life) 7. In-hospital death
Variables of Interest
The following variables were included in our assessment: year of death, age at death (18-39, 40-64, 65+), sex (female, male), race and ethnicity (non-Hispanic Black, non-Hispanic American Indian or Alaskan Native, Hispanic/Latino(a) origin or descent, non-Hispanic Asian, non-Hispanic White), county of residence, insurance coverage at death (Medicaid, Medicare, Private, dual-eligible), and cancer types (breast, prostate, lung, colorectal, melanoma, endometrial, stomach, liver, cervix, myeloma, larynx, pancreas). Using decedents’ county of residence at diagnosis, we were able to classify counties as either rural or urban using the Rural-Urban Continuum Codes (RUCC). Specifically, RUCC codes 1, 2, and 3 for urban residency, and codes 4-9 for rural residency. Year of birth, sex, and race and ethnicity were all self-reported by decedents.
Statistical Analysis
Descriptive statistics were calculated for our study cohort using frequencies and percentages for categorical variables. Across our seven EOL care outcomes, we assessed for differences by year of death, age at death, sex, race and ethnicity, geography (rurality and county-level), insurance status, cancer type, and the intersection between race & ethnicity by rurality. We assessed the intersection between these two variables because individuals of marginalized racial groups who live in the eastern and western counties of North Carolina have a larger cancer burden when compared to White patients.29,30 To compare utilization of end-of-life care practices between groups of interest, we utilized Chi-Square tests and reported the corresponding P-values. For any groups with cell suppression (frequencies less than 11), Fisher’s Exact test was utilized instead. The statistical significance level was set to 0.05. All data management and analyses were performed using SAS© software, version 9.4 (SAS Institute, Cary, NC). Figures were made using Tableau software, version 2025.1 (SalesForce, Seattle, WA).
Results
Sample Characteristics
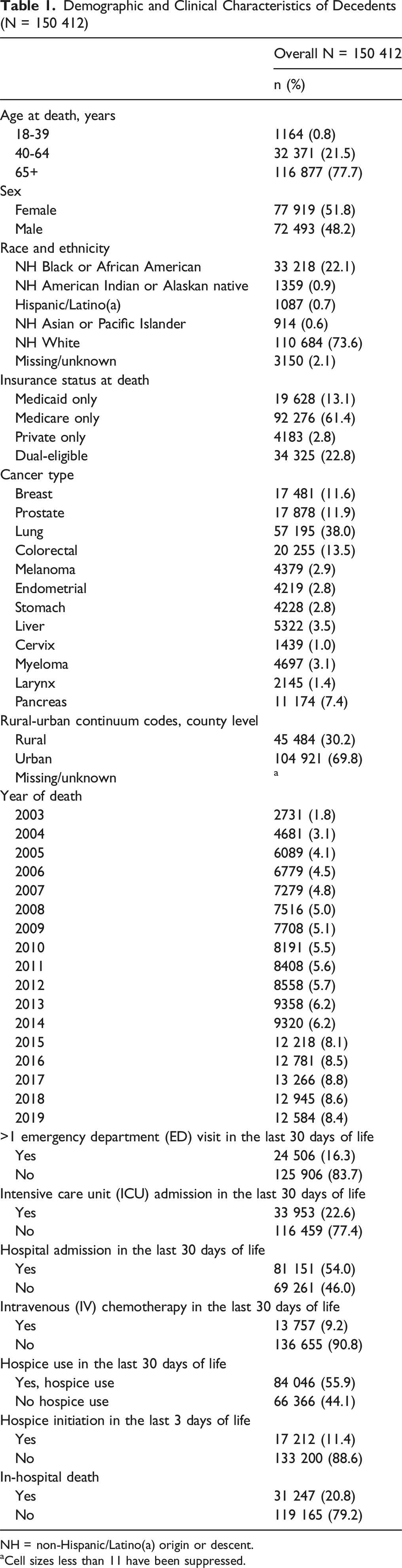
Our analytic cohort included 150 412 eligible decedents (Figure 1). Overall, 48% of decedents in our study were female, 30% lived in rural counties, and 23% were non-Hispanic Black or African American (Table 1). Most often, decedents were Medicare-only beneficiaries (61%). The three most frequent cancer types included lung (38%), followed by colorectal (14%) and prostate (12%). Cohort selection diagram Demographic and Clinical Characteristics of Decedents (N = 150 412) NH = non-Hispanic/Latino(a) origin or descent. aCell sizes less than 11 have been suppressed.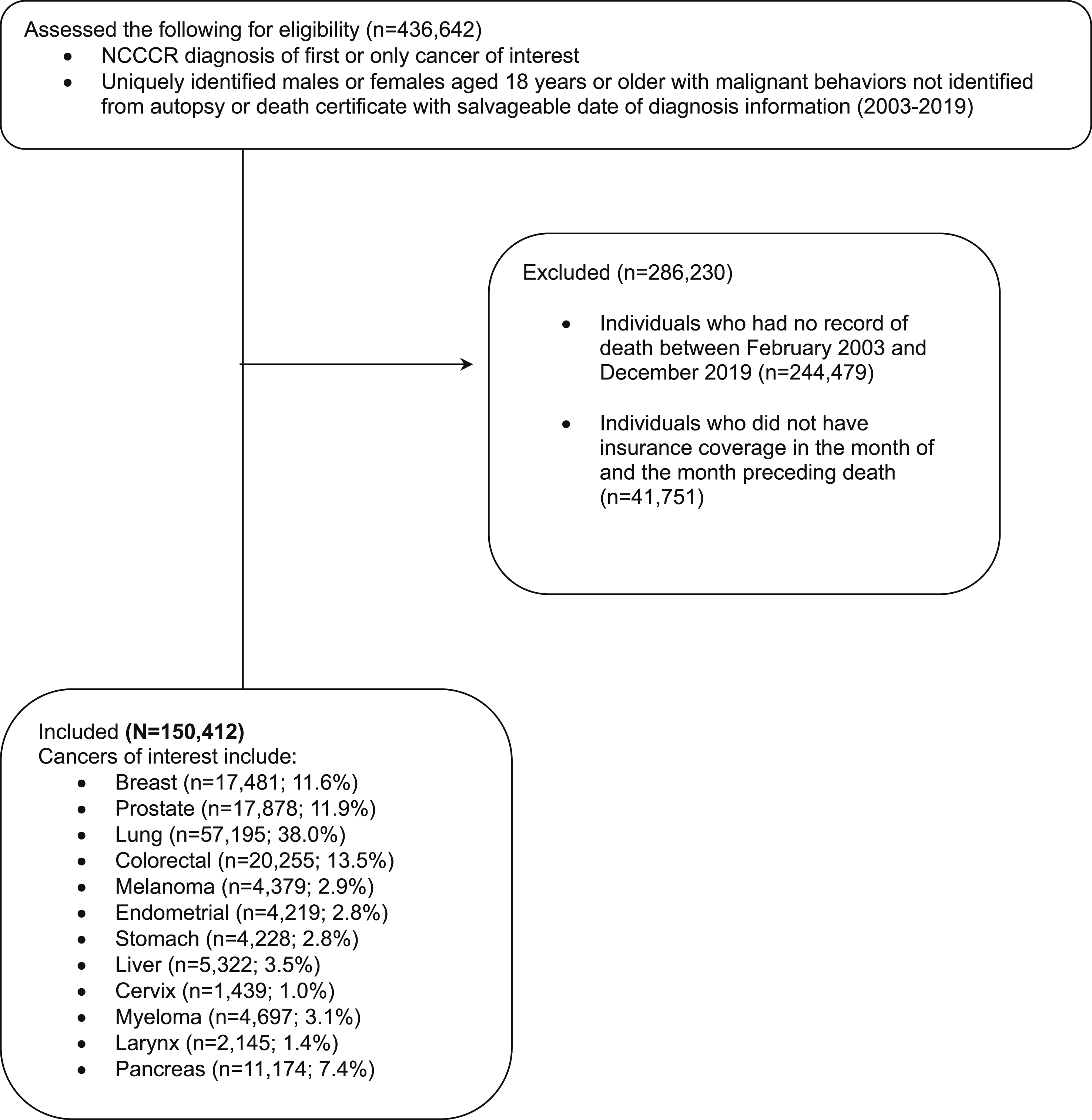
Trends and Disparities in End-of-Life Care
Overall
Among cancer decedents, the most frequently observed poor EOL care practice was hospital admission in the last 30 days of life (54%), followed by no hospice use in last 30 days of life (44%), and ICU admission in the last 30 days of life (23%; Table 1).
Year of Death
From 2003 to 2019, the proportion of decedents with >1 ED visit increased from 13% to 20% and hospice initiation in the last 3 days of life increased from 7% to 14%. Four measures decreased over time: no hospice use in the last 30 days of life decreased from 62% to 38%; hospital admissions in the last 30 days of life decreased from 66% to 52%; in-hospital deaths decreased from 30% to 18%; and ICU admissions decreased slightly (27% to 24%). Only IV chemotherapy use in the last 30 days of life was consistent across years, at about 9% (Figure 2). Differences in end-of-life indicators by year of death from 2003-2019 (N = 150 412)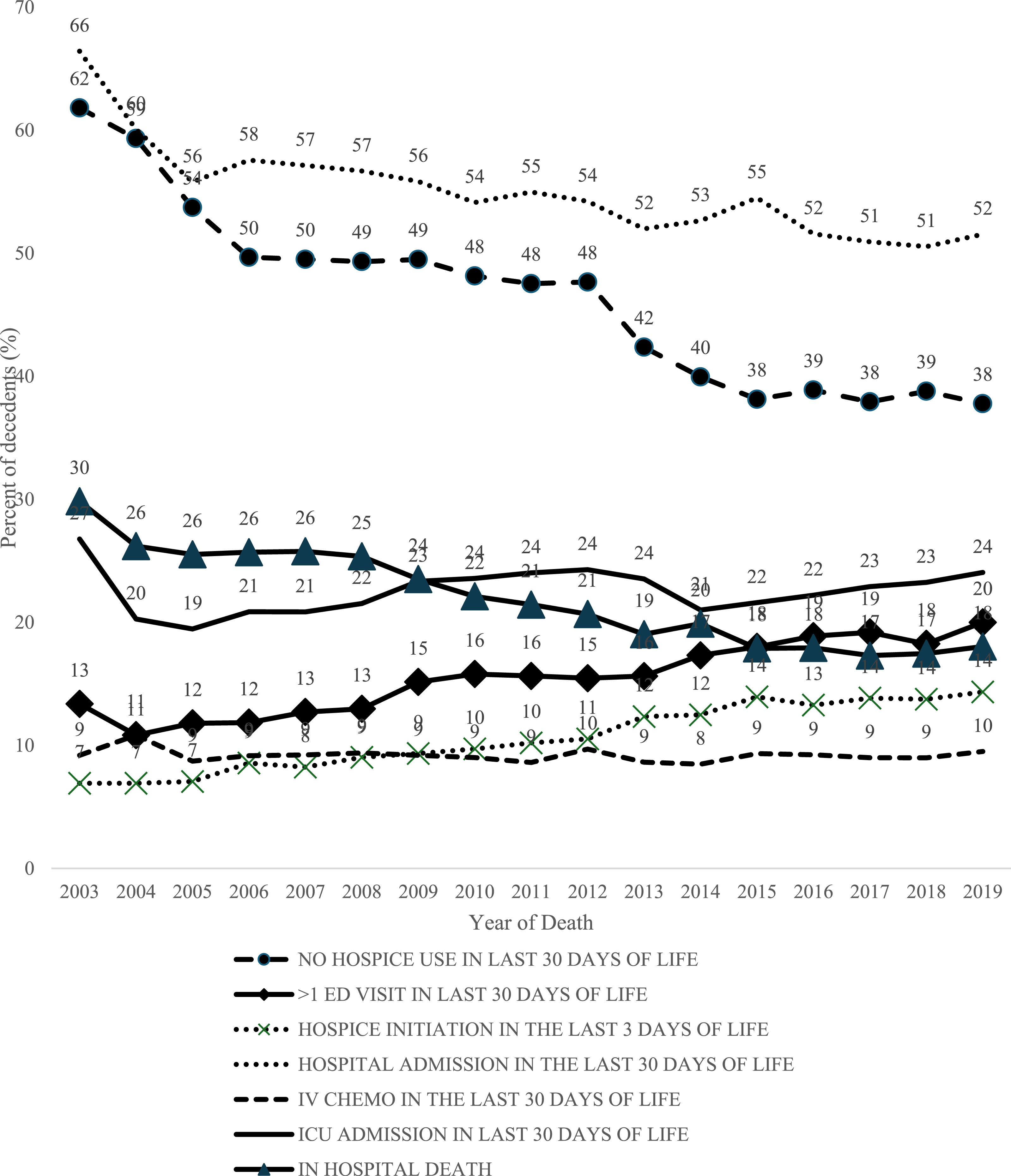
Age at Death
Decedents younger than 65 more often had an ICU admission than decedents 65 or older (18-39: 32%; 40-64: 30%; 65+: 20%; P < .0001; Supplemental Figure 1). When compared to patients who died as younger adults, older decedents less often received IV chemo in the last 30 days of life (18-39: 15%; 40-64: 12%; 65+: 8%; P < .0001). Patients who died between the ages of 18 and 39 more often did not use hospice in the last 30 days of life compared to older decedents (68%; 46-64: 60%; 65+: 39%; P < .0001).
Sex
Though not a large difference, male decedents more often had no hospice use within the last 30 days of life compared to females (47% vs 41%; P < .0001; Supplemental Figure 2). We did not find differences in the six other EOL measures by sex.
Race and Ethnicity
When compared to non-Hispanic White decedents, we observed that non-Hispanic American Indian or Alaskan Native decedents more often had ICU admissions in last 30 days (29% vs 21%; P < .0001; Supplemental Figure 3) and non-Hispanic Black decedents more often did not use hospice in last 30 days (54% vs 41%; P < .0001). Additionally, non-Hispanic Black decedents more often had a hospital admission in last 30 days compared to Hispanic or Latino(a) decedents (59% vs 50%; P < .0001).
Rurality and County of Residence
While we did not observe differences between rural and urban counties (Supplemental Figure 4), there was variation at the county-level. Most often, eastern counties had higher percentages of inappropriate EOL care practices including hospital admission in the last 30 days, in-hospital death, and IV chemotherapy in the last 30 days of life (Figure 3). Western counties had the highest use of hospice initiation in the last 3 days of life. Differences in end-of-life indicators among decedents county of residence in North Carolina: (A) > 1 emergency department visit in last 30 days of life; (B) intensive care unit admission in last 30 days of life; (C) hospital admission in last 30 days of life; (D) intravenous chemotherapy in last 30 days of life; (E) no hospice use in last 30 days of life; (F) hospice initiation in last 3 days of life; (G) in-hospital death (N = 150 412)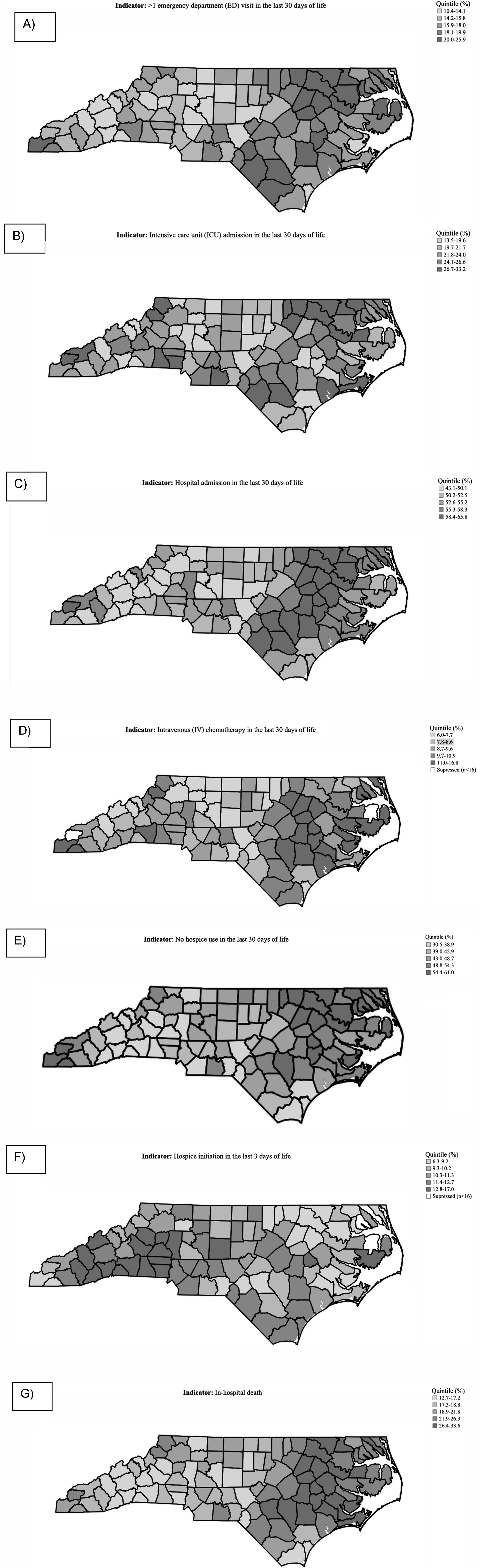
Race and Ethnicity by Rurality
Frequencies and Percentages of Individuals Meeting End-of-Life Care Practices Stratified by Race & Ethnicity and Rurality (N = 150 412)
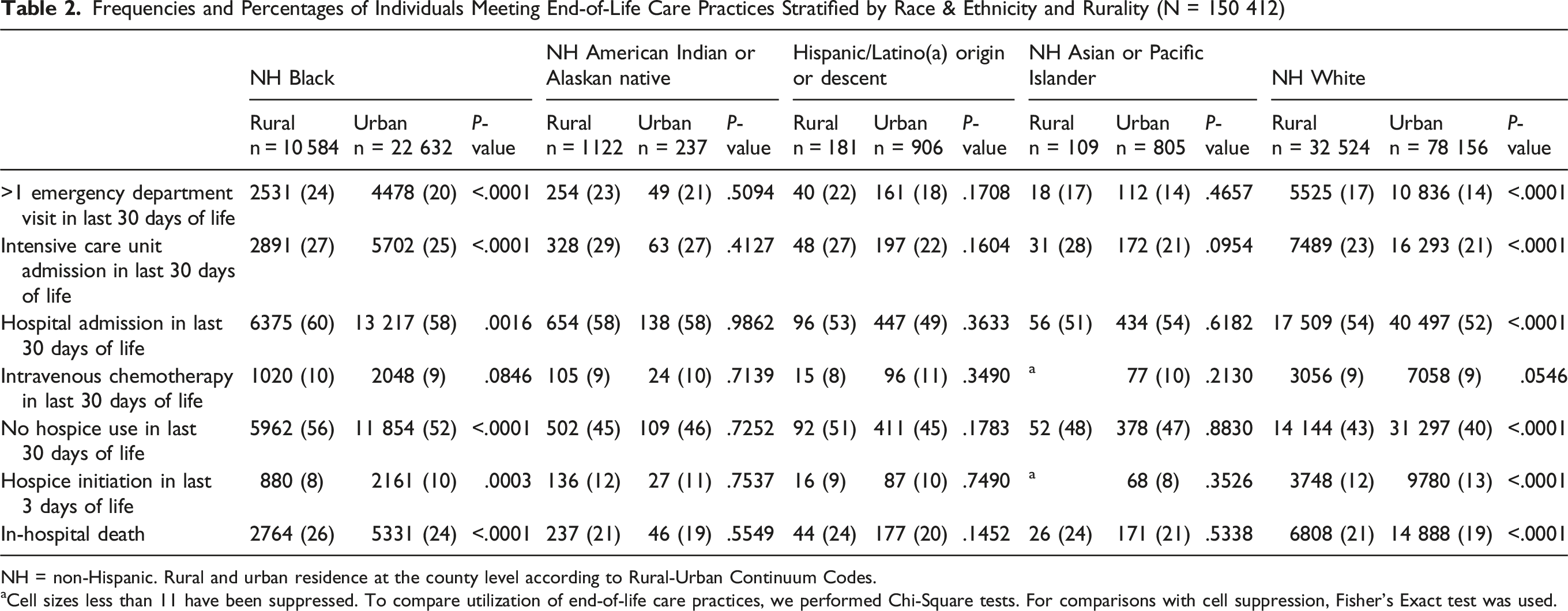
NH = non-Hispanic. Rural and urban residence at the county level according to Rural-Urban Continuum Codes.
aCell sizes less than 11 have been suppressed. To compare utilization of end-of-life care practices, we performed Chi-Square tests. For comparisons with cell suppression, Fisher’s Exact test was used.
Insurance Status
Compared to decedents with all other insurance types, decedents covered by Medicaid at the time of death had the lowest use (ie, the best quality EOL care) across the following EOL measures: ED visit (9%), hospital admission (45%), and hospice initiation in the last 3 days (6%; Figure 4). However, Medicaid decedents (71%) more often did not use hospice in the last 30 days of life compared to Medicare (38%), private (53%), or dual-eligible decedents (44%). Private or Medicare beneficiaries had the highest use of inappropriate EOL care quality, including hospital admission (Private: 63%), chemotherapy (Private: 17%), hospice initiation in the last 3 days (Medicare: 13%), and in-hospital death (Private: 29%). Additionally, about one in five (19%) patients with Medicaid died in hospital, which was similar to patients who were dual-eligible or enrolled in Medicare (both 21%). Differences in each EOL care practice between insurance groups were statistically significant at P < .0001. Differences in end-of-life indicators according to insurance status: (A) > 1 emergency department visit in last 30 days of life; (B) intensive care unit admission in last 30 days of life; (C) hospital admission in last 30 days of life; (D) intravenous chemotherapy in last 30 days of life; (E) no hospice use in last 30 days of life; (F) hospice initiation in last 3 days of life; (G) in-hospital death (N = 150 412)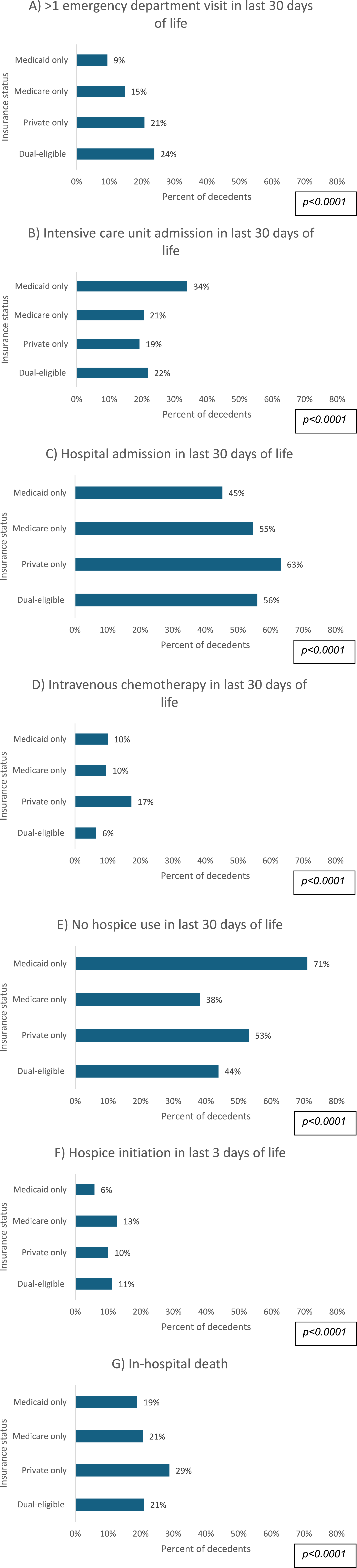
Cancer Type
There were notable variations across cancer sites for IV chemotherapy in the last 30 days of life, with the highest percentages observed for myeloma (16%), pancreas (12%), and lung (11%; Supplemental Figure 5). Furthermore, decedents with myeloma had the highest percentages for four other inappropriate EOL care practices: >1 ED visit in the last 30 days of life (20%), hospice initiation in the last 3 days of life (13%), hospital admission in the last 30 days of life (64%), and in-hospital death (26%). Differences in each EOL care practice between cancer types were statistically significant at P < .0001.
Discussion
Using a population-based sample linking cancer registry data to multiple payers’ administrative claims, we descriptively assessed EOL care quality for decedents with cancer across a largely diverse state. North Carolina has substantial racial, ethnic, and geographic diversity; 38% of our populations are Black, American Indian, or people of color and 33% are rural residents.31,32 We observed substantial differences in EOL care quality across several measures by insurance type, region, and over time. Among our sample, we observed that Medicaid beneficiaries had the best quality EOL care on all indicators; except for no hospice use in the last 30 days of life. Additionally, over time the utilization of most inappropriate EOL care practices decreased.
Interestingly, Medicaid cancer decedents were often less likely to receive inappropriate EOL care. This finding differs from previous literature; a study among Medicaid patients with cancer in New Jersey found that the majority received inappropriate EOL care. 33 Additionally, among Medicaid decedents in Georgia and Illinois, patients with cancer had significantly higher costs at EOL than individuals without cancer. 34 Other studies, however, have found that individuals with low incomes are often unfamiliar with or have no knowledge of hospice care, resulting in underutilization. 35 As patients with private insurance often have higher incomes, they may be more willing to pursue intensive EOL care, given their greater ability to pay for their healthcare costs. 36 Given the range of findings, more research (including more nuanced, multivariable analyses) is needed to understand the specific drivers of and mechanisms underlying this insurance-based difference.
Despite declining over time, the lack of hospice use within 30 days of death remains at an unacceptably high level. Prompting hospice use at the end-of-life should be a key priority because hospice use is associated with improved quality-of-life and fewer hospital admissions37-39 (which we also found remained high at the EOL). A systematic review by Parajuli and colleagues found several barriers to hospice care for patients with cancer including lack of communication with providers and health insurance policies that may limit access to hospice care providers. 40 Interventions such as culturally tailored EOL patient navigation could decrease these barriers and increase appropriately timed hospice care referrals.41-43 EOL navigators help patients plan for the end of life, including how they want to spend their final days, how they want to die, and how they want to be remembered while honoring their personal and cultural values. 43 A randomized controlled trial by Fischer and colleagues found their culturally tailored patient navigator intervention—included home health visits by a bilingual navigator— improved palliative care outcomes for Hispanic patients. 44 While this study was among individuals without cancer, more research is needed to understand if this model could be expanded to individuals with cancer and across other cultural backgrounds.
We also observed that the eastern and western counties of North Carolina had higher utilization of inappropriate EOL care. While we did not observe differences in EOL care practices between the intersection of race and ethnicity and rurality (which could be due to the descriptive nature of our analysis), eastern counties are more often rural and have a higher percentage of individuals who are medically underserved (including individuals of marginalized racial and ethnic groups).45,46 Furthermore, marginalized individuals in this area have a disproportionately larger cancer burden; a study using cancer registry data found that Black or African American patients in eastern counties in North Carolina had the highest cancer incidence and mortality compared to other racial and ethnic groups. 29 Similarly, in the western counties of North Carolina, the highest rates of cancer incidence are found among the American Indian population. 30 The American Indian population is also more likely to experience increased hospital admission, intensive care admission, and ED use in the last 30 days of life compared to White decedents with cancer. Interventions are needed in these particular regions that focus on racial and ethnic groups who face poorer EOL outcomes. 47
Furthermore, we did not observe any differences in EOL care practices between rural and urban residents, which contrasts with the literature.48-55 For example, Crouch and colleagues found that Medicare expenditures within the last six months of life were lower for rural residents than urban residents with cancer. 48 Additionally, a study by Watanabe-Galloway and colleagues found that Medicare beneficiaries living in rural counties had fewer days in the ICU and were less likely to use hospice within the 90 days before the end of life compared to urban counterparts. 49 Reasons for this are likely structural and systematic. In a review article by Anwar and colleagues, various barriers to EOL care for rural patients were identified, including lack of access, ability to retain palliative care providers, and restricted economic support. 50 While we did not find rural-urban differences, more research is needed to isolate rurality as there were likely confounding effects that masked our findings.
Our study should be considered in light of several limitations. While our study is strengthened by the robust data representing multiple payer types, it does not include uninsured patients. Because our sample is limited to a single state, our results may not be generalizable to the broader US population. Additionally, our time range was capped at 2019; differences in EOL care may have changed since the COVID-19 pandemic, which is an important topic for future analyses. Finally, our study was descriptive in nature as our goal was to understand observed differences in our catchment area. These results provided a broad overview of EOL care quality and differences; future research is needed that takes a deeper dive into understanding and fully characterizing potential disparities in EOL care quality.
Conclusion
In our study of decedents with cancer, despite observed decreases in several inappropriate EOL care practices over time, inappropriate EOL care practices remained high. In particular, we observed disproportionate utilization of end-of-life care practices among patients with private and public health care plans. More research is needed to understand the mechanisms driving these observed differences. Supportive services, including EOL patient navigation and other care coordination approaches that encourage earlier consideration of hospice services, may have the potential to improve EOL care.
Supplemental Material
Supplemental Material - Trends in End-of-Life Care Quality Among Patients With Cancer, 2003 to 2019
Supplemental Material Trends in End-of-Life Care Quality Among Patients With Cancer, 2003 to 2019 by Nicole E. Caston, Bradford E. Jackson, Chris D. Baggett, Stephanie B. Wheeler, Mya L. Roberson, Tess Thompson, Hayley Morris, Katie Johnson, Lisa P. Spees in American Journal of Hospice and Palliative Medicine®
Footnotes
Author Contributions
Study concept/design: LS, SW, CB.
Provision of study material or patients: NA.
Data collection/assembly: BJ, CB.
Data analysis and interpretation: NC, BJ, LS.
Manuscript writing: All authors.
Final approval of manuscript: All authors.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Work on this study was supported by the Cancer Information and Population Health Resource, UNC Lineberger Comprehensive Cancer Center, with funding provided by the University Cancer Research Fund via the state of North Carolina. The findings and conclusions in this publication are those of the authors and do not necessarily represent the views of the North Carolina Department of Health and Human Services, Division of Public Health. Support was also provided by the Cancer Center Core Support Grant [grant number 5-P30-CA016086]. Dr. Caston is supported by the National Cancer Institute’s National Research Service Award sponsored by the Lineberger Comprehensive Cancer Center at the University of North Carolina at Chapel Hill (T32 CA116339). Dr. Spees was supported by the National Institutes on Minority Health and Health Disparities of the NIH (K01 CA250989-01A1; PI: Spees).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Due to contractual obligations with data partners like the North Carolina CCR, the data are not accessible to the public. Nonetheless, access may be granted upon obtaining appropriate approval and adhering to usage agreements. Requests can be made to utilize the data within the UNC remote-access computing environment following project evaluation and approval by the Institutional Review Board, contingent upon having fully executed, project-specific data sharing agreements.
Disclosures
Dr. Wheeler has received salary support paid to her institution for unrelated work from Pfizer Foundation/National Comprehensive Cancer Network and AstraZeneca. Dr. Spees has received salary support paid to her institution for unrelated work from AstraZeneca. Dr. Roberson has received consulting fees from the National Committee of Quality Assurance outside of the scope of the submitted work; she has received research funding paid to her institution for unrelated work from Gilead Sciences.
Prior Presentations
This was presented as a poster at the 2025 Catchment Area Data Excellence (CADEx) Conference.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
