Abstract
Background
Life Review Interventions (LRI) are gaining attention in palliative care as a therapeutic approach to support psychological and spiritual needs. However, their effectiveness remains uncertain.
Objectives
This systematic review aimed to analyze the effects of LRI in adults with advanced chronic or terminal illness receiving palliative or end-of-life care.
Methods
MEDLINE, Scopus, Cochrane Library, and Web of Science were searched for English-language articles (2018-2024). Included studies involved adults with advanced chronic illness receiving any form of LRI; control groups, where applicable, received usual care. Primary outcomes were symptom burden, spiritual well-being (SWB), and quality of life (QOL). Studies were critically appraised. Due to heterogeneity, a narrative synthesis was conducted. The review was registered in PROSPERO.
Results
Eight studies were included, most of moderate to high methodological quality. Half were conducted in North America, with the remainder from Europe and Asia. The total sample comprised 598 participants, 89% of whom had cancer. The review identified eight distinct LRI, each with varying formats and delivery methods. For symptom-related outcomes (n = 8), four studies reported significant improvements in anxiety, depression, or general well-being. For SWB (n = 6), three studies demonstrated positive effects. Regarding QOL (n = 4), only one study showed meaningful improvement; the others found no significant change.
Conclusions
LRI may provide psychological and spiritual benefits in palliative and end-of-life care. However, current evidence for their effectiveness in improving QOL is limited, highlighting the need for further high-quality research.
Keywords
Introduction
Rationale
A diagnosis of advanced illness, such as cancer, often profoundly disrupts quality of life (QOL), bringing psychological, spiritual, and existential distress to the forefront.1,2 This distress can exceed physical symptom concerns, 1 and highlight our mortality. 3 Depression, anxiety, hopelessness, desire for hastened death, and reduced QOL are common. 4 Addressing these issues is essential, as spiritual distress can exacerbate physical symptoms. 5
Palliative care (PC), as defined by the World Health Organization, aims to improve QOL by preventing or relieving suffering through early identification and management of physical, psychosocial, and spiritual issues. 6 Psychosocial and spiritual interventions, such as reminiscence therapy, are increasingly recognized as supportive in PC. 4
Life Review Interventions (LRI) are narrative-based tools that help individuals find meaning, resolve emotional conflicts, and reframe life experiences. 6 Through structured recollection across life stages, LRI foster expression, validation, and perspective shifts.1,6 This process can reduce anxiety and depression, enhance spiritual well-being (SWB), and improve coping and QOL. 6 SWB ranges from distress to wellness and is often challenged in those receiving PC. 6 Spiritual distress—marked by fear, hopelessness, or loss of meaning—can worsen psychological symptoms, while spiritual wellness promotes peace, purpose, and fewer symptoms. 6 Symptom burden (SB) reflects the perceived frequency and severity of symptoms, contributing to physical and emotional strain. 7 By cultivating purpose and peace, LRI may reduce SB, enhance SWB, and support QOL at end of life.2,6
Two prior systematic reviews assessed LRI in PC but had limitations. The 2017 review included only randomized trials in cancer populations. 8 The 2020 review addressed QOL outcomes but only included studies up to July 2018. 9 Given the growing interest in psychosocial and spiritual care, an updated synthesis is needed.
Objectives
This systematic review aimed to respond to the following research question: What is the effectiveness of LRI among adults with advanced chronic diseases or terminal illness receiving palliative or end-of-life care?
Methods
This systematic review followed the recommendations of the “Cochrane Handbook for Systematic Reviews of Interventions,” 10 and was reported in accordance with the “Preferred Reporting Items for Systematic Reviews and Meta-Analyses” guidelines. 11
A preliminary protocol was developed and refined during the review process, and its final form is reflected in the structure of this article.
Eligibility Criteria
Participants
Adults (≥18 years old) of any gender diagnosed with advanced chronic illness, either oncological or non-oncological, receiving PC or end-of-life care.
Interventions
Any LRI, as defined by the original authors.
Comparators
The control group, if present, received usual care.
Outcomes
Included studies had to report on at least one of the following outcomes: SB, SWB, or QOL. Any definition and any scale of assessment were accepted.
Study Design
Any, excluding literature reviews, study protocols, descriptions of protocols, conceptual models, conference abstracts, posters at conferences, book chapters, letters, editorials, commentaries, academic theses, proposals for collaboration or calls for collaboration. Articles were excluded if they were not subscribed to by our faculty.
Information Sources
MEDLINE (via PubMed), Scopus, the Cochrane Library, and Web of Science were systematically searched for studies published between January 1, 2018, and December 31, 2024. This period updates and extends findings from two prior systematic reviews on LRI,8,9 the most recent of which included studies only up to July 2018. 9 Starting the search in 2018 ensured inclusion of newer evidence while avoiding duplication. A backward citation search of all included studies was also performed. All sources were last accessed on January 11, 2025. No authors were contacted for additional data.
Search Strategy
The search employed both free-text terms and MeSH terms, structured as follows: “life review” AND (“palliative care” or “terminal care” or “end-of-life care” or “hospice care”). The search strategy is detailed in Supplemental Table 1.
Selection Process
Titles and abstracts were independently screened by both authors. Full texts of potentially eligible articles were then reviewed by two independent reviewers. Disagreements were resolved by discussion and consensus. No automation tools were used.
Data Collection Process
A data-charting form was developed in Microsoft Excel (Microsoft Corp, 2016) and refined through pilot testing. The first author led data extraction, with independent cross-checking by the second author. Discrepancies were resolved through discussion until consensus. No automation tools were used, and original authors were not contacted.
Data Items
Characteristics of the Included Studies (n = 8)
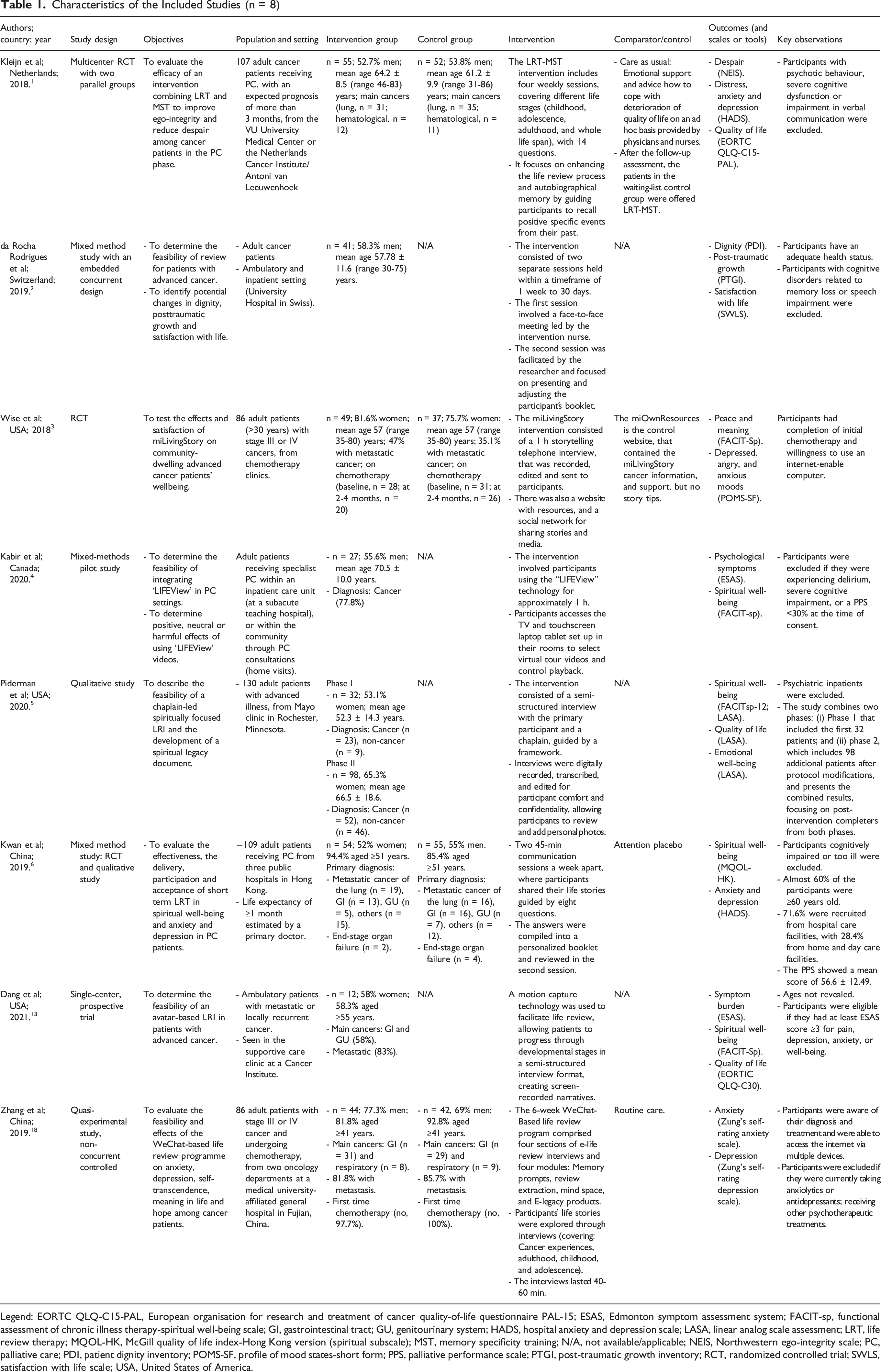
Legend: EORTC QLQ-C15-PAL, European organisation for research and treatment of cancer quality-of-life questionnaire PAL-15; ESAS, Edmonton symptom assessment system; FACIT-sp, functional assessment of chronic illness therapy-spiritual well-being scale; GI, gastrointestinal tract; GU, genitourinary system; HADS, hospital anxiety and depression scale; LASA, linear analog scale assessment; LRT, life review therapy; MQOL-HK, McGill quality of life index-Hong Kong version (spiritual subscale); MST, memory specificity training; N/A, not available/applicable; NEIS, Northwestern ego-integrity scale; PC, palliative care; PDI, patient dignity inventory; POMS-SF, profile of mood states-short form; PPS, palliative performance scale; PTGI, post-traumatic growth inventory; RCT, randomized controlled trial; SWLS, satisfaction with life scale; USA, United States of America.
Results of the Included Studies (n = 8)

Legend: ESAS, Edmonton symptom assessment system; FACIT-sp, functional assessment of chronic illness therapy-spiritual well-being scale; HADS, hospital anxiety and depression scale; MQOL-HK, McGill quality of life index-Hong Kong version (spiritual subscale); N/A, not available/applicable; PDI, patient dignity inventory; PTGI, post-traumatic growth inventory; SWLS, satisfaction with life scale; USA, United States of America.
Study Risk of Bias Assessment
Two reviewers independently assessed the risk of bias for all included studies. Discrepancies were resolved through discussion and consensus, with reference to study reports and appraisal criteria as needed. No third reviewer was involved, and no inter-rater statistic was calculated.
Risk of bias for randomized controlled trials1,3 was assessed using the revised Cochrane risk-of-bias tool (RoB 2). 12 For the single-center prospective trial, 13 the ROBINS-I assessment tool was applied. 14 For mixed-methods studies,2,4,6 the Mixed Methods Appraisal Tool was utilized. 15 The qualitative study 5 was evaluated using the Checklist for Qualitative Research, 16 while the JBI Checklist for Quasi-experimental Studies 17 was used for the quasi-experimental study. 18 Study quality was classified by the proportion of “Yes” responses: low (<0.5), moderate (0.5-0.7), or high (>0.7), based on author-defined thresholds.
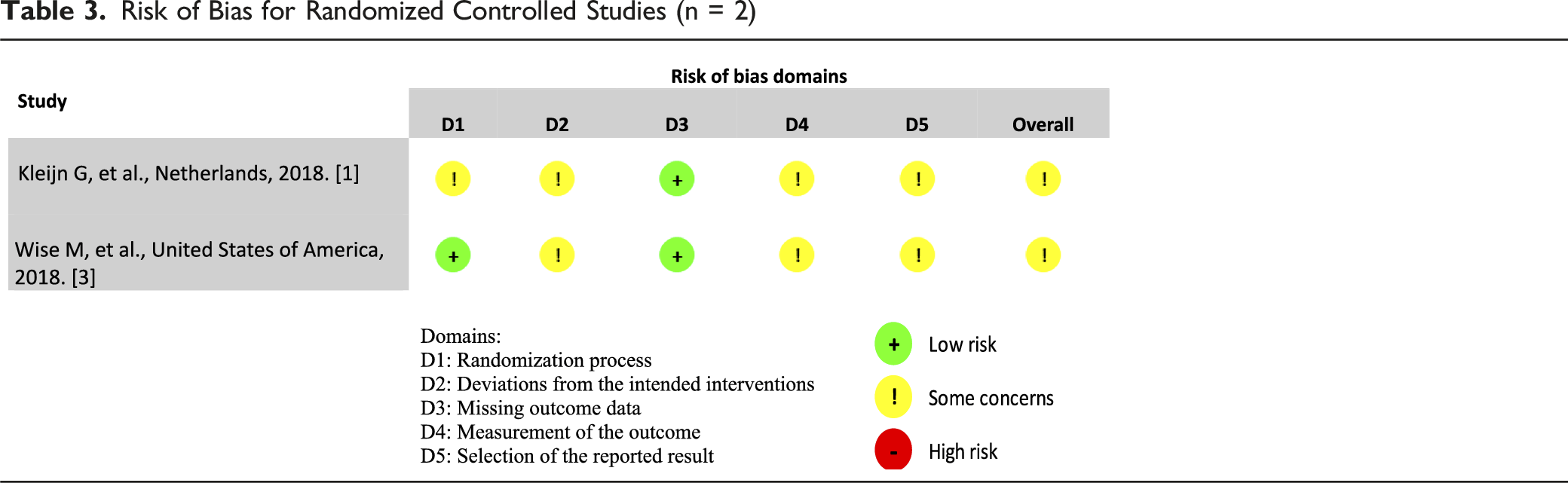
Risk of Bias for Randomized Controlled Studies (n = 2)
Risk of Bias for Non-Randomized Controlled Studies (n = 1)

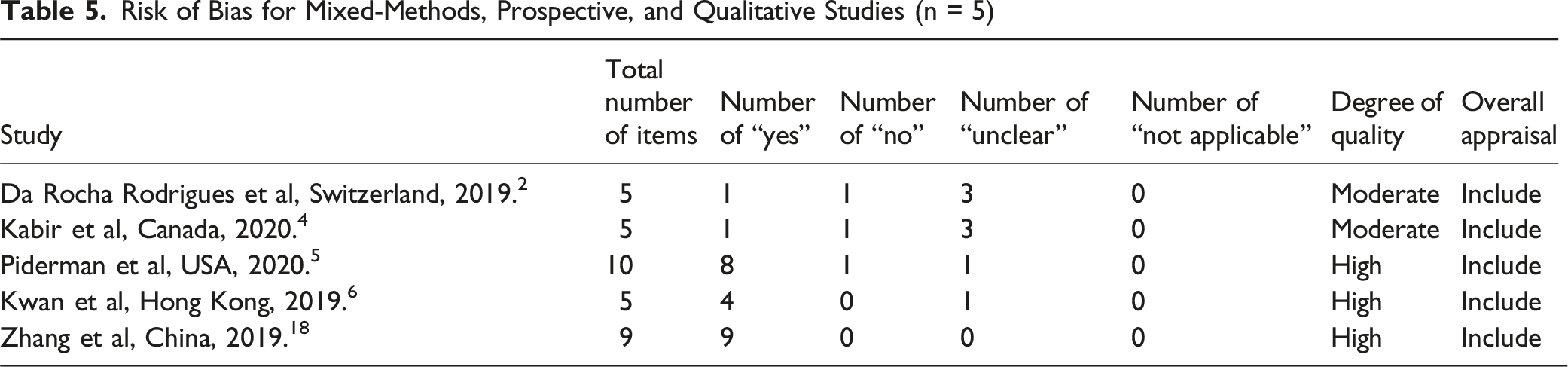
Risk of Bias for Mixed-Methods, Prospective, and Qualitative Studies (n = 5)
Effect Measures
We accepted all the effect measures as declared by the study authors, for each outcome. These measures were utilized both in the synthesis and presentation of the results.
Synthesis Methods
Meta-analysis was not conducted due to the small number of studies and marked heterogeneity in participants, interventions, comparators, outcomes, and follow-up. Meta-regression was likewise not feasible. Evidence was narratively synthesized, retaining original measurement scales and reporting effect direction and magnitude as described by the authors. No formal evidence grading framework was applied.
Findings are organized by outcome: SB, SWB, and QOL. Summary data are presented in Table 1 (Study Characteristics), Table 2 (Main Findings), and Tables 3–5 (Risk of Bias).
Results
Study Selection
The database search yielded 74 articles. After removing 19 duplicates, 55 records were screened. Title screening excluded 17 articles, and 11 more were excluded after abstract review. A total of 27 full texts were assessed, with 19 excluded: 16 for lacking relevant interventions and three for ineligible outcomes. Eight studies met inclusion criteria. The selection process is detailed in Figure 1. Flow diagram of the studies selection process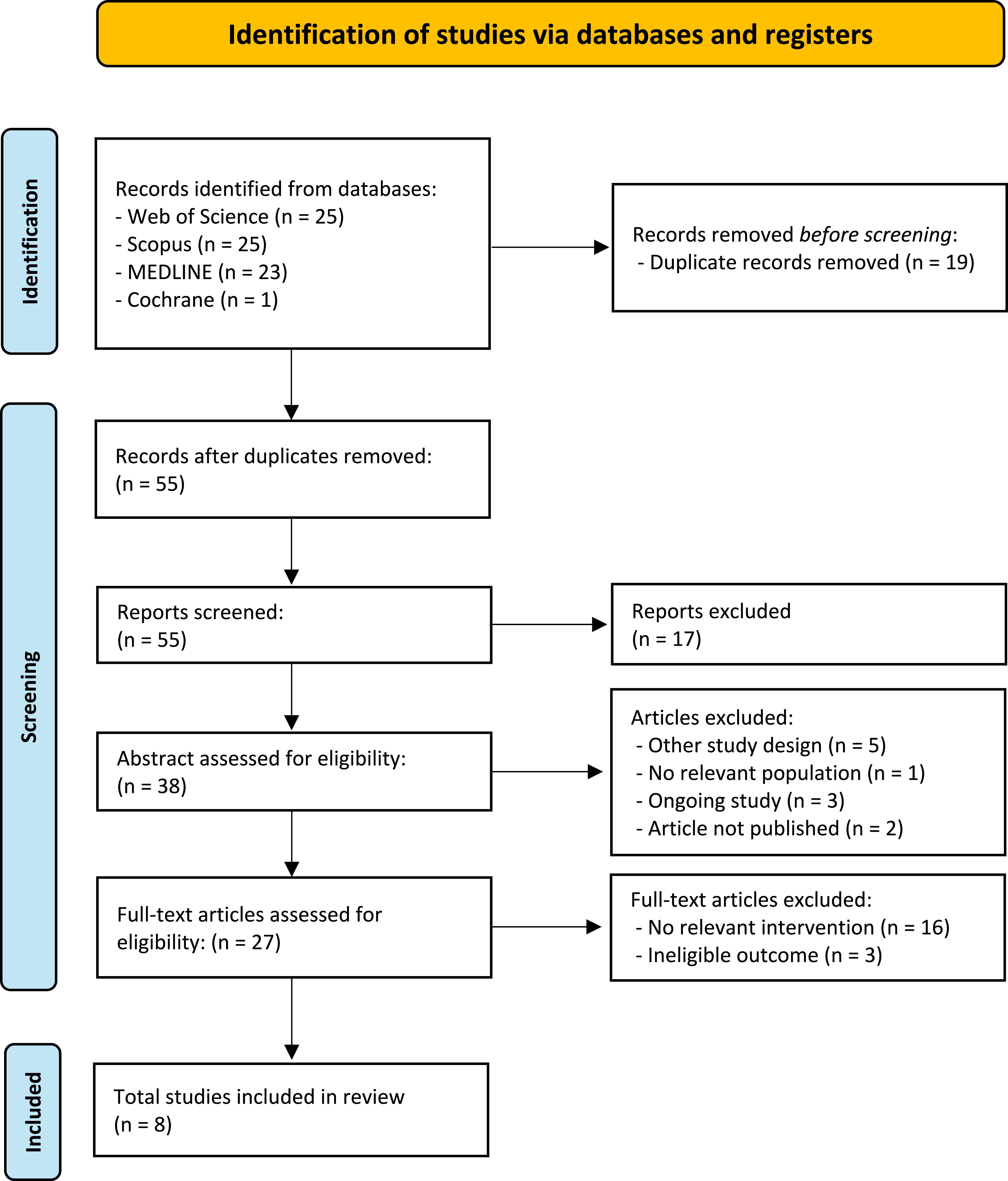
Study Characteristics
Our review included: three mixed-methods studies,2,4,6 two randomized controlled trials,1,3 one single-center prospective trial, 13 one qualitative study, 5 and one quasi-experimental study. 18 The studies originated from America (the United States,3,5,13 and Canada 4 ), Europe (the Netherlands, 1 and Switzerland 2 ) and Asia (China6,18). The total number of participants across all studies was 598, with 531 of them being cancer patients. Detailed characteristics of the studies are presented in Table 1.
Life Review Interventions Included in This Review
Eight distinct LRI were identified, each varying in format and delivery: Life Review Therapy + Memory Specificity Training: A 4-week program exploring life stages via 14 guiding questions, enhancing recall of specific positive memories.
1
Revie⊕: Two-session format (1-30 days apart); involved a nurse-led meeting followed by researcher-guided review of a personalized booklet.
2
miLivingStory: A 1-h recorded storytelling phone interview, followed by editing and distribution to participants, with optional website and social sharing.
3
LIFEView: A digital life review using a television and touchscreen system to access self-guided virtual tours (∼1 h).
4
Chaplain-Led Spiritually Focused Life Review: A semi-structured, chaplain-led interview, recorded and transcribed for participants, with the option to add photographs.
5
Short-Term Life Review: Two 45-min sessions one week apart, guided by eight questions and culminating in a personalized story booklet.
6
Avatar-Facilitated Life Review: Used motion capture and screen recording to guide participants through semi-structured life review across life stages.
13
WeChat-Based Life Review: A 6-week, four-module e-intervention using interviews and digital content (eg, Memory Prompts, Mind Space, E-legacy).
18
Risk of Bias in Studies
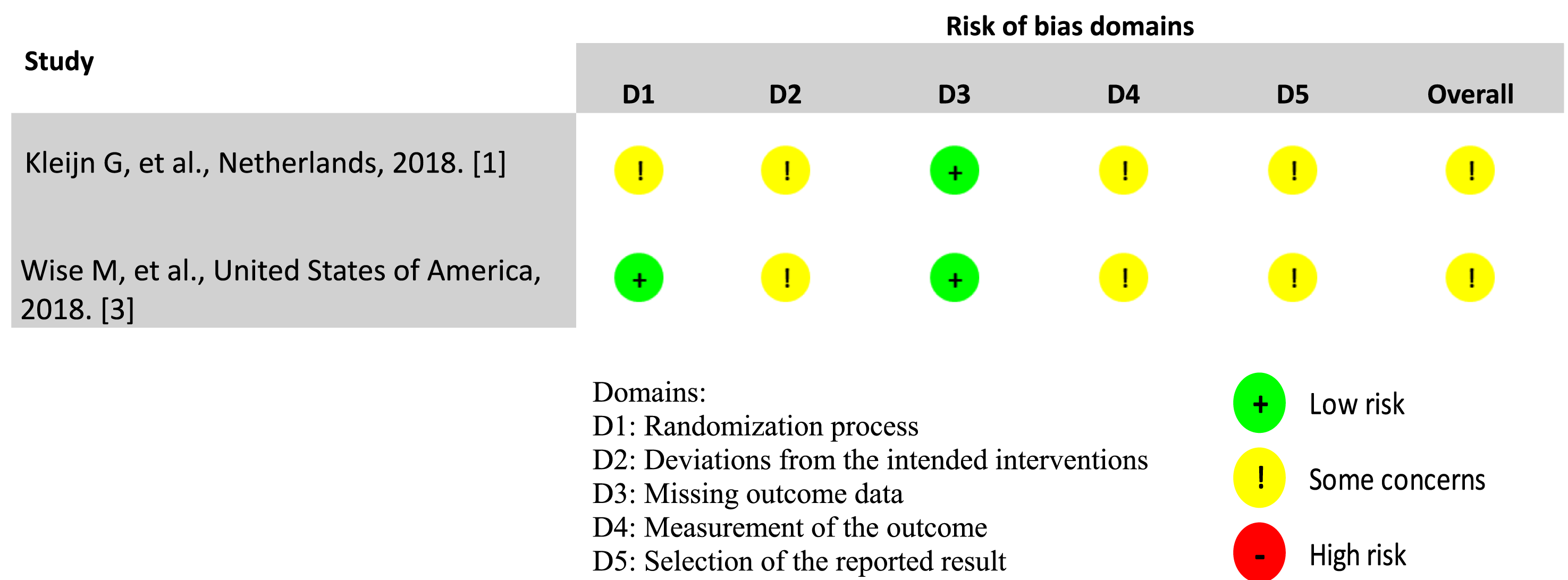
The overall risk of bias within the two randomized controlled trials was moderate (Table 3).
The allocation sequence was considered low risk, as both studies used block randomization.1,3 Wise et al 3 employed a random number generator with blocks of ten, ensuring a controlled allocation process. 3 This randomized controlled trial was conducted without blinding participants to their assigned intervention, however participants were blinded to whether their intervention was the control or the treatment condition. Kleijn et al 1 employed centrally conducted randomization by an independent researcher, with block size of twenty. 1 However, patients and psychologists were aware of treatment allocation. 1 Potential deviations from intended interventions may have occurred due to contextual factors in the trials.1,3 Wise et al 3 reported a 22% attrition rate, with no significant baseline differences between completers and dropouts. Kleijn et al 1 reported a 28% dropout rate, mainly due to disease progression and death.
There was no clear evidence in any of the randomized controlled trials indicating differences in outcome measurement methods between groups.1,3,6 However, potential bias remained a concern. Wise et al 3 noted that while participants were blinded to whether their intervention was control or treatment, they were aware of their assigned group. Similarly, Kleijn et al 1 did not implement blinding for participants or psychologists.
The single-centre prospective study 13 had low risk across all domains, except for confounding (critical risk) and outcome measurement (moderate risk). Due to the critical risk of bias in confounding, the overall risk of bias was deemed critical (Table 4).
Among the mixed-methods studies, two were deemed to have a moderate risk of bias,2,4 while one was classified as low risk. 6 (Table 5).
According to the Checklist for qualitative research, Piderman et al, 5 has seven criteria with a “yes,” supporting its inclusion (Table 5).
According to the JBI Checklist for Quasi-experimental Studies, the study 18 by received “yes” responses for all criteria, confirming its inclusion (Table 5).
The risk of selective reporting was a concern in all studies due to the lack of confirmation of a pre-specified analysis plan before data became available.
Results of Individual Studies
Detailed results of individual studies are presented in Table 2.
Results of Syntheses
Symptom Burden
Eight studies address this outcome.1-6,13,18
One study found that 80% of patients had unchanged or improved well-being scores after 1 month. 13 Another study observed improvements in 46.6% of participants over time. 5 In contrast, Kabir et al found no significant differences in Edmonton Symptom Assessment System-revised scores between pre- and post-“LIFEView” assessments, though minor improvements were noted in well-being, tiredness, and anxiety. 4
Regarding anxiety, three studies found no statistically significant differences between the intervention and control groups over time.1,3,6 However, one study reported significant improvements in anxiety in the experimental group compared to the control group, which showed no significant changes. 18 Additionally, two studies showed significant changes over time in the intervention group.6,18
For depression, three studies noted no significant differences in the intervention group compared to the control group.1,3,6 Conversely, one study found significant improvements when comparing the two groups. 18 Additionally, in two studies the intervention group showed a decrease in depression with statistical significance over time.6,18
For angry mood, one study found no statistically significant differences at either the 2-months and 4-months intervals after the intervention, when compared the two groups. 3
In assessing despair, when comparing the intervention and control groups, one study noted no significant changes in dispair. 1
Regarding distress, one study found no significant differences over time in symptom distress and existential distress. 2 When comparing the intervention and control groups, one study noted no significant changes in distress. 1
Spiritual Well-Being
Six studies address this outcome.2-6,13
Based on total Functional Assessment of Chronic Illness Therapy - Spiritual Well-Being (FACIT-Sp) scores, one study noted significant improvements, 13 while two studies showed no significant improvements.4,5
In terms of SWB scores, one study found significant improvement in the intervention group compared to the control group 6 and another study found significant changes over time, 5 while one study found no significant improvement over time. 13
Regarding peace and meaning, one study reported no significant difference in the peace and meaning subscale between the intervention and control groups at 2 months. 3 At 4 months, the intervention group showed a significant improvement in peace, but no significant differences were found in the meaning subscale. 3 Another study, showed no significant changes over time for the Patient Dignity Inventory subdimension of peace of mind, or in the Post-Traumatic Growth Inventory subdimensions of spiritual changes and appreciation of life. 2
Quality of Life
Four studies address this outcome.1,2,5,13
One qualitative study reported statistically significant improvements in the intervention group in QOL from baseline to follow-up, 5 while another study did not find significant differences. 13 Additionally, one study did not find significant differences in QOL between the intervention group and the care-as-usual group. 1
One study noted no significant differences over time in appreciation of life subdimensions and noted a strong sense of life satisfaction, although it was not significant. 2
Discussion
Summary of Findings
This systematic review assessed the impact of LRI on SB (n = 8), SWB (n = 6), and QOL (n = 4) in adults with advanced or terminal illness. Eight distinct LRI were identified, each with varying formats and delivery methods. For SB, four studies reported significant improvements in anxiety, depression, or general well-being, while the remaining four found no effect. For SWB, two of three studies showed significant improvements, including higher total scores and positive changes in FACIT-sp subdomains. In contrast, only one of four studies on QOL reported a significant improvement over time; the others found no differences. Overall, LRI demonstrated potential benefits in selected outcomes, particularly in SB and SWB, though findings were heterogeneous and effects on QOL were inconsistent.
Our findings both align with and extend those of earlier systematic reviews conducted prior to 2018. Wang et al (2017) included eight RCTs in advanced cancer populations and reported pooled benefits of LRI on SWB, general distress, and QOL, although sustained effects were observed only for QOL at short-term follow-up. 8 Despite these signals, the authors noted substantial methodological limitations and heterogeneity. 8 Huang et al (2020) focused on QOL and included six studies with mixed designs and populations, reporting a significant improvement in QOL following LRI. 9 However, that review lacked a clear operational definition of LRI, used diverse outcome instruments, and included only studies published up to July 2018. 9 In contrast, this review incorporates studies published through 2024, includes broader populations and delivery modes (including digital and technology-assisted LRI), and systematically examines outcomes beyond QOL, namely SB and SWB. While our findings corroborate prior evidence suggesting benefits for psychological and spiritual domains, they also highlight limited and inconsistent effects on QOL. These findings emphasize the need for standardized interventions, consistent outcome measures, and high-quality trials to clarify the role of LRI in PC.
Life Review Interventions and Symptom Burden
Our findings suggest that LRI may alleviate psychological symptoms such as anxiety and depression in some patients, although effects were inconsistent across studies. This variability may reflect differences in intervention format, delivery setting, duration, and participant characteristics, including baseline symptom severity. Our results are consistent with prior literature. Lan et al, 19 in a review of 15 studies, concluded that LRI is a worthwhile approach for older adults, with significant improvements in depression and hopelessness. Similarly, Lui et al, 20 in a meta-analysis, demonstrated positive effects on depression, anxiety, and mood among older adults with life-threatening illness, though without significant change in general well-being. Ando et al 21 reported decreased anxiety and depression among terminally ill cancer patients receiving a short-term LRI in hospital. Korte et al 22 found that LRI were more effective than usual care in reducing depressive symptoms at post-treatment and follow-ups and improved anxiety and positive mental health. Lamers et al 23 observed that an online-guided LRI improved depressive symptoms compared to waitlist, but was not superior to expressive writing, raising questions about intervention specificity. Al-Ghafri et al 24 found moderate short-term effects on depression that diminished at follow-up. Westerhof et al 25 also found significant improvements in depressive symptoms, but no consistent advantage over other psychosocial interventions, and no effects on despair, loneliness, or well-being. Pot et al 26 reported reduced depression with LRI, though anxiety also decreased in the control group.
Taken together, both our review and the broader literature highlight LRI as a potentially useful tool for addressing mood symptoms in serious illness but also point to variability in outcomes. This may reflect heterogeneity in delivery, underlying psychological distress, or other moderating factors such as illness stage, readiness for reflection, or therapeutic alliance.
Life Review Interventions and Spiritual Well-Being
Our review found mixed effects of LRI on SWB, with only a few studies reporting statistically significant improvements. These inconsistencies may reflect variations in how spirituality is conceptualized and measured across studies. Some tools assess existential or meaning-based constructs, while others focus on religious or faith-oriented domains. Additionally, cultural background, personal beliefs, and prior spiritual engagement influence outcomes. Previous literature suggests potential benefits of LRI in promoting SWB, particularly among patients facing end-of-life situations. In 2017, Wang et al 8 reported a desirable effect of LRI on the meaning of life domain of SWB. In 2007, Ando et al 27 found significant improvements in spirituality and orientation following life review interviews. In 2008, the same group 28 observed increased FACIT-Sp scores after short-term LRI in terminally ill cancer patients, and again in 2010, Ando et al 21 confirmed significantly greater improvements in FACIT-Sp in the intervention group. Similarly, Xiao et al 29 demonstrated improved perceived support, reduced alienation, and higher valuation of life following a life review program.
Taken together, these findings suggest that LRI may enhance aspects of SWB in some patients, but effects are not consistent across studies. Differences in measurement tools, delivery formats, and participant readiness to engage in spiritual reflection may all influence outcomes.
Life Review Interventions and Quality of Life
Findings on QOL were inconclusive, with only one of four studies reporting significant improvements following LRI. The remaining studies found no meaningful differences either over time or between groups. This variability underscores the complex, multidimensional nature of QOL, shaped by physical health, emotional state, social support, and subjective life experiences. These overlapping influences make it difficult to isolate the direct effects of LRI on overall QOL. In the broader literature, Wang et al 8 reported a statistically significant effect of LRI on overall QOL in a meta-analysis, particularly at short-term follow-up. Lui et al 20 noted moderate improvements in QOL and life satisfaction, whereas Lan et al 19 found no significant impact. Similarly, Ando et al 27 observed increased QOL after life review interviews, and Xiao et al 29 found higher QOL scores in the intervention group both post-intervention and at 3-week follow-up.
These findings suggest that while LRI may positively influence certain dimensions of QOL, evidence remains inconsistent. Outcomes may depend on baseline distress levels, intervention timing, delivery method, or individual receptiveness.
Strengths and Limitations
This systematic review has several strengths. It addresses an underexplored area by evaluating the impact of LRI on SB, SWB, and QOL in palliative and end-of-life care. By including diverse study designs, the review captures both quantifiable outcomes and patient-reported experiences. A rigorous critical appraisal using validated tools enhances the trustworthiness of the findings. The review was registered with PROSPERO prior to data extraction and followed a systematic methodology with predefined eligibility criteria. The search strategy covered four major databases over a 7-year period (2018-2024), reflecting the most recent evidence. Both physical and psychosocial outcomes were examined, aligning with the multidimensional nature of PC. The focus on adults with advanced chronic or terminal illness provides a clinically relevant and updated synthesis of the psychological, spiritual, and QOL effects of LRI.
Despite these strengths, several limitations must be acknowledged. First, the absence of funding restricted access to subscription-based databases such as Embase, CINAHL, and PsycINFO. Consequently, only four databases were searched, which may have reduced comprehensiveness. Nonetheless, overlap between databases likely mitigated the risk of missing key studies. Additionally, grey literature was not searched.
The search strategy was developed by the authors without direct input from an information specialist or medical librarian, which may have affected the comprehensiveness of the search.
Language restrictions were applied, as only English-language studies were included, potentially introducing selection bias. Starting the search from 2018 may have excluded earlier yet relevant evidence, although this time frame was selected to build upon prior reviews. No author contact was made to obtain missing data.
The included studies exhibited substantial heterogeneity in methodologies, participant characteristics, intervention format and delivery (eg, duration, structure), outcome measures, follow-up periods, and statistical analyses. Control conditions, typically labeled as “usual care,” also varied by setting. These differences limit comparability across studies and hinder the ability to draw robust, generalizable conclusions about the specific benefits of LRI.
Risk of bias was moderate in most RCT and high in non-randomized studies, which compromises the overall reliability of the evidence. While validated instruments were used to assess SWB and psychological outcomes, these measures remain inherently subjective and difficult to standardize.
Due to the heterogeneity of study designs and outcomes, a narrative synthesis was performed. This precluded meta-analysis and the generation of pooled effect estimates but allowed for a more nuanced interpretation of the complex and evolving evidence. The small number of eligible studies, most of which involved cancer populations with limited sample sizes, also restricts the generalizability of findings to broader PC populations.
Lastly, no formal evidence grading framework (eg, GRADE) was applied, reducing the interpretability and clinical applicability of the findings. Although the protocol was registered with PROSPERO prior to data extraction, its registration occurred later in the review process, which may not fully align with best-practice standards for transparency.
Implications of the Results for Practice, Policy, and Future Research
Practice
This review suggests that LRI may offer meaningful psychological and spiritual support to adults with advanced or terminal illness, particularly in reducing symptoms such as anxiety and depression and in enhancing SWB. However, the variability in outcomes indicates that these interventions are not universally effective. Healthcare professionals should consider individual preferences, cultural context, and baseline psychological or spiritual needs when integrating Life Review into holistic, person-centered care plans.
Policy
Although the findings are encouraging, the evidence supporting LRI remains mixed, especially regarding improvements in QOL. Policymakers should approach the integration of Life Review into PC frameworks cautiously, treating it as a potentially beneficial but not yet fully established practice. Investments in implementation research, professional training, and pilot programs can help determine its feasibility and acceptability in diverse care settings.
Future Research
There is a clear need for robust, well-designed studies to better understand the effectiveness of LRI across patient populations. Future research should apply standardized protocols, validated and multidimensional outcome measures (including culturally sensitive and theoretically grounded tools for SWB), and extended follow-up periods to assess sustainability. Studies should clarify which QOL domains are most responsive to LRI and explore which patient subgroups are most likely to benefit, considering variables such as diagnosis, disease stage, baseline psychological distress, and readiness to engage in reflection. Comparative trials examining different delivery formats (eg, digital vs in-person), and evaluations of cost-effectiveness and integration into routine PC services, are also needed to inform future implementation.
Conclusions
This review suggests that LRI may help alleviate psychological distress in patients with advanced or terminal illness, with more limited and inconsistent effects on SWB and QOL. The evidence remains mixed, reflecting heterogeneity in study design, delivery formats, and outcome measurement.
To advance the field, future research should prioritize methodological rigor, including standardized protocols, validated instruments, and longer-term follow-up. Identifying patient profiles most likely to benefit and testing innovative, scalable delivery formats will be essential to establish LRI as a meaningful component of end-of-life care.
Supplemental Material
Supplemental Material - Life Review Interventions in Palliative Care: A Systematic Review of Patient-Centered Outcomes
Supplemental Material for Life Review Interventions in Palliative Care: A Systematic Review of Patient-Centered Outcomes by Margarida Pascoal-Carvalho and Paulo Reis-Pina in American Journal of Hospice and Palliative Medicine®.
Footnotes
Author Contributions
This study was conceptualized by MPC and PRP. MPC and PRP conducted searches and screening of articles, analyzed data, designed the review protocol, wrote the manuscript, reviewed it, and approved the definitive version of the paper.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data relevant to this study are included within the article. No additional datasets, code, or materials were generated or used.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.