Abstract
Background
Ideally, patients should self-report their symptoms. However, when this is not possible due to cognitive impairment, discomfort must be assessed through observation of the patient’s behavior. To the best of our knowledge, there are no tools for such assessment in palliative care.
Methods
A scale (Discomfort Scale for Palliative Care – DISPAC) was developed in 4 levels based on the frequency of signs of discomfort. The scale underwent face validity by 8 doctors and nurses experienced in palliative care. After that, DISPAC was compared with the DS-DAT and the VAS for construct validity using Spearman’s ρ. The comparison was made by 4 observers: 2 for DISPAC (DISPAC1 and DISPAC2), 1 for DS-DAT, and 2 for VAS. The intraclass correlation coefficient was used to measure the degree of consistency between raters, and Cronbach’s α was used to assess the internal consistency.
Results
The results of the face validity were as follows: the scale measures what it is intended to measure; the scale is useful for clinical practice; the scale can improve communication among healthcare professionals; and the scale is easy to use. The internal consistency was 0.95. The inter-rater reliability was also 0.95 (P < 0.001). The correlation coefficients between DISPAC1 and DISPAC2 and the VAS were 0.712 (P < 0.001) and 0.723 (P < 0.001), respectively. The correlation with the DS-DAT was 0.689 (P < 0.001) and 0.615 (P < 0.001), respectively.
Conclusion
DISPAC is a valid and reliable scale for assessing discomfort in patients who are unable to report it.
Keywords
Introduction
In advanced chronic diseases, especially during the last days of life, impaired consciousness frequently hinders the assessment of symptoms and patient comfort. Similar challenges can arise in sedated patients. In such situations, evaluating symptoms must be conducted indirectly due to the patients’ inability to communicate. Several tools have been developed for assessing specific symptoms, primarily pain1–3, as well as for evaluating discomfort in general.4,5 The majority were developed within the context of dementia.
As, by definition, symptoms are subjective experiences, the indirect methods of assessment can be flawed. When patients exhibit signs of discomfort, it can be challenging to determine the cause, whether it is pain, other symptoms, or a manifestation of delirium. The history and physical examination may clarify the cause of the signs of discomfort, and the result of the prescribed treatment may also be an essential element in that clarification.
The primary aim of palliative care is to provide relief from the suffering of patients with advanced diseases. To achieve that goal, it is essential to identify the sources and assess the intensity of discomfort. Several tools were developed for this purpose, relying on the patients’ reports, which is the best approach for subjective symptoms or problems. However, there are no tools specifically developed for palliative care to assess discomfort in patients with cognitive impairment who cannot report it. This lack of validated tools for assessing discomfort in palliative care drives researchers to use scales developed for other conditions, such as the Discomfort Scale – Dementia of Alzheimer’s Type (DS-DAT), for monitoring the level of discomfort in sedated patients. 6 A scale used in palliative care to assess discomfort had only face validity, which is insufficient for being deemed fully validated. 7 A recent review was unable to identify other validated tools for palliative care. 8
Due to the gap in available tools for palliative care, described above, there is a need for a validated scale to assess discomfort in this context. This study describes the development and validation of such a scale, designated as the Discomfort Scale for Palliative Care (DISPAC).
Methods
This study was carried out in a general hospital that includes a palliative care team. The hospital does not have a dedicated palliative inpatient unit. Instead, the team operates without assigned beds and provides outpatient consultation and home care.
The study was conducted on a convenience sample of inpatients with cognitive deterioration, unable to report their symptoms, followed by the palliative care team.
This scale was developed based on the frequency of signs of discomfort, stratified into 4 levels, ranging from no signs of discomfort to almost constant signs of discomfort (Appendix). The validation of the scale was carried out in 2 phases. The first phase involved evaluating the scale’s face validity. Eight doctors and nurses working in palliative care assessed the scale and responded to 4 questions using a five-point scale from “1 - strongly disagree” to “5 - strongly agree”: • Does the scale measure what it is intended to measure? • Can the scale be useful for clinical practice? • Can the scale improve communication among healthcare professionals regarding discomfort level? • Is the scale easy to use?
The second phase involved reliability and construct validation. Due to the lack of a gold standard for comparison, criterion validation could not be performed. For construct validation, the scale was compared with the DS-DAT, which has been used to assess comfort in palliative sedation studies, 6 and a visual analogue scale (VAS) measuring 100 mm anchored by the terms “no discomfort signs” and “discomfort at least almost constant”.
The DS-DAT, being a more complex scale that requires a more extended training period, was applied by the same 2 nurses, whereas several researchers used the DISPAC and the VAS. When a patient was identified as unable to report their symptoms due to cognitive disability, he/she was observed, without interacting with the patient, by 4 researchers for 5 minutes: one of the 2 nurses trained in the DS-DAT applied it, one doctor or nurse applied the VAS, and 2 researchers, doctors, or nurses applied the DISPAC simultaneously. Each of the 4 researchers independently scored the respective scales and recorded the results on separate forms developed for that purpose. The five-minute time frame was adopted because it is the standard for applying the DS-DAT. 5
The consciousness level was evaluated using the Consciousness Scale for Palliative Care. 9 This scale has 6 consciousness levels, which are: 1, awakes; when called by their name: 2, awakens and stays awake during the conversation; 3, awakens but falls asleep during the conversation, 4, reacts with movement or brief eye-opening, but without eye contact; trapezius muscle pinching: 5, reacts; 6, does not react.
For the statistical analysis, the association among the DISPAC, DS-DAT, and VAS was evaluated using Spearman’s ρ. The intraclass correlation coefficient was utilized to measure the degree of consistency between raters’ measurements. Cronbach’s α was employed to assess the internal consistency. 10
The patients included were inpatients followed by the palliative care team who were unable to report their symptoms. Therefore, they were unable to give informed consent; however, as the study did not involve any intervention, consent was requested from families when possible. Data recorded by the researcher were collected by the first author and inserted in an Excel datasheet. Only that researcher could access the database which was protected by a password. Patients were unidentifiable through the data inserted. The Ethics Committee of the hospital approved this study (022/CES/JAS).
Results
Demographic Data
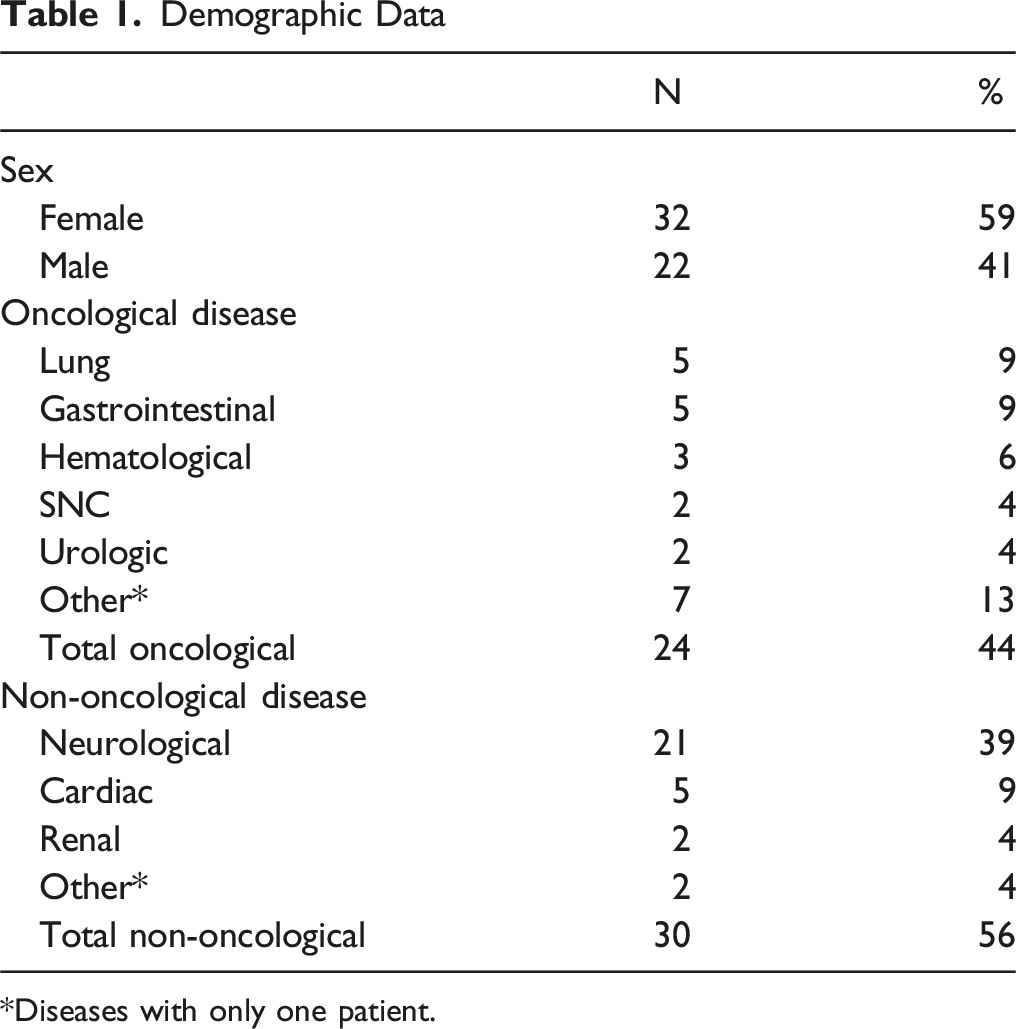
*Diseases with only one patient.
Correlation Between Scales–Spearman’s ρ
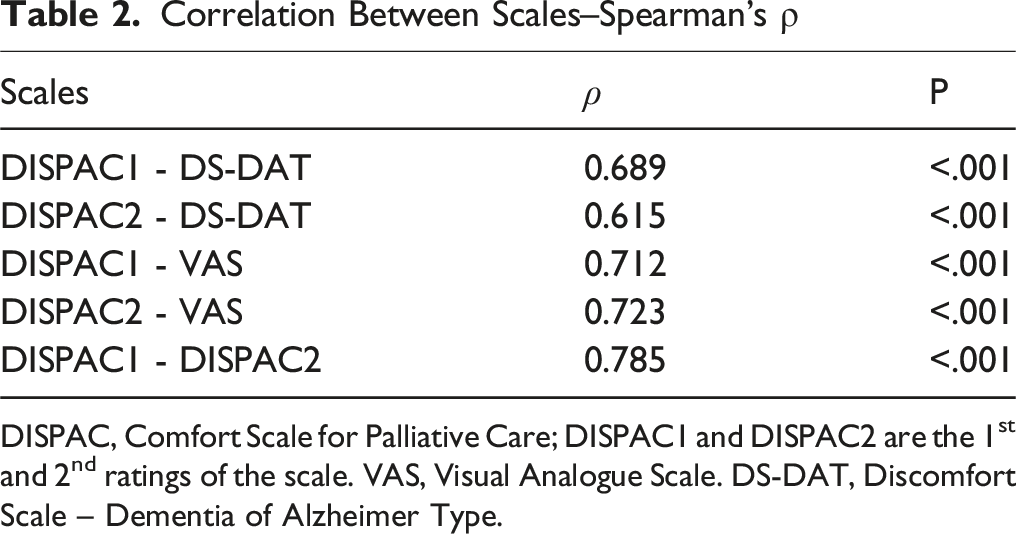
DISPAC, Comfort Scale for Palliative Care; DISPAC1 and DISPAC2 are the 1st and 2nd ratings of the scale. VAS, Visual Analogue Scale. DS-DAT, Discomfort Scale – Dementia of Alzheimer Type.
Face Validity
The results of face validity were: • Does the scale measure what it is intended to measure? Median 4 (3 to 5) • Can the scale be useful for clinical practice? Median 4 (3 to 5) • Can the scale improve communication among healthcare professionals? Median 5 (2 to 5) • Is the scale easy to use? Median 4 (4 to 5)
Internal Consistency
The internal consistency, measured with Cronbach’s α, was very high – 0.95.
Inter-rater Reliability
The degree of agreement between raters regarding the DISPAC was also very high, with an intraclass correlation coefficient of 0.95 (P < 0.001).
Construct Validity
The correlation coefficients between DISPAC1 and DISPAC2 and the VAS were good, 0.712 (P < 0.001) and 0.723 (P < 0.001), respectively. The correlation with the DS-DAT was slightly lower, 0.689 (P < 0.001) and 0.615 (P < 0.001), respectively (Table 2).
Discussion
This study addresses the assessment of suffering in patients with cognitive impairment in palliative care. The assessment of symptoms in these patients is a significant problem in medicine in general. The lack of tools developed for palliative care is surprising because the inability to communicate is frequent in this setting, mainly in the last days of life. On the contrary, numerous tools have been developed for dementia.5,11,12
Most tools have several items, such as vocalization, facial expression, body language, and breathing, that are observed and scored individually before being added to reach a global score. 5 One item that seems particularly difficult to interpret is the respiratory pattern. In several tools, it is scored negatively as a sign of suffering, 12 but, for example, a death rattle is usually not considered a sign of suffering, because patients are unconscious at that time. 13 For this reason, it is advisable to explain that to families to avoid unnecessary interventions, as they usually see it as suffering. In this study, the option was to classify globally the signs of discomfort and their frequency, without individually describing those signs, following the usual practice.
Face validity indicates that the scale measures what it is intended to measure, is helpful for clinical practice, can improve communication among healthcare professionals, and is easy to use. These encouraging results supported the next steps in the validation process: internal consistency, inter-rater reliability, and construct validity.
The internal consistency was very high, as was the inter-rater reliability, as assessed by Cronbach’s alpha and the intraclass correlation coefficient, both of which scored>0.90.10,14
In the absence of a gold standard to compare with the DISPAC, a criterion validity could not be done. Therefore, a construct validation was performed by comparing the scale with the DS-DAT and the VAS using Spearman’s ρ. The cutoff points to translate the correlations coefficient in descriptors like “weak”, “moderate”, or “strong” are somewhat inconsistent, but a correlation coefficient of >0.65 can be interpreted as “good”. 15 Accordingly, the correlation coefficient with VAS was good. With the DS-DAT, the correlation coefficient was slightly lower, particularly for DISPAC2, but still considered “good” for DISPAC1. This may be due to the DS-DAT being more complex with several variables to observe and score, including the noisy breathing, as discussed above.
This scale can be applied with minimum training, as it is easy to use. The scale may improve communication among healthcare professionals because the level of discomfort can be indicated numerically without needing to describe it. Therefore, the scale can be useful for clinical practice. The main limitation of this study is that it was carried out at only one institution, which raises questions about its generalizability. Another weakness of the scale is the time required for its application.
The 5-minute observation period results from the standard for the DS-DAT. This period is relatively long, but it is possible that it can be reduced in future research. Nevertheless, for now, the 5 minutes should be respected.
Conclusion
DISPAC is a valid and reliable scale for assessing discomfort in patients who are unable to report it. It is easy to use and may improve communication among health professionals by introducing a more precise and standardized terminology.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
