Abstract
Background
People living with Parkinson’s disease (PD) and related parkinsonian disorders (PDRD) experience multidimensional needs that may benefit from palliative care (PC).
Objectives
To systematically review the impact of PC integration for people with PD/PDRD and their caregivers.
Methods
MEDLINE, Web of Science, Scopus, and Cochrane were searched for English-language articles to December 31, 2024. Eligible studies enrolled adults with PD/PDRD receiving any PC intervention vs usual neurological care. Studies enrolling mixed long-term neurological cohorts (including PD/PDRD) were included and treated as indirect evidence relevant to PD/PDRD. Outcomes included symptom management/burden (SMB), quality of life (QOL), satisfaction with care (patient/caregiver), and caregiver burden (CB). Risk of bias was assessed, and results were synthesized narratively. This review was registered in PROSPERO.
Results
Seven studies (n = 1621) were included (4 randomized trials; 4 from the United States of America). All studies assessing SMB reported improvements in intervention groups, though not across every symptom. Four of 6 studies reported significant QOL gains; 2 found no difference. Satisfaction findings were mixed (1 improvement, 1 no change). Three of 5 studies reported reductions in CB, particularly beyond 12 months. The qualitative study highlighted dissatisfaction with standard care and perceived benefits of PC in communication, interdisciplinary support, and linkage to community resources. Findings from mixed cohorts were consistent in direction but interpreted cautiously due to indirectness.
Conclusions
Integrating PC for PD/PDRD shows promise for improving symptoms, QOL, and caregiver outcomes. Clearer PD/PDRD-specific trials—using consistent terminology and reporting—are needed to strengthen inference.
Keywords
Introduction
Rationale
Parkinson’s disease (PD) is the second most common degenerative disorder, primarily affecting older adults. 1 Its hallmark motor symptoms—bradykinesia, rigidity, resting tremor, and postural instability,2,3—are often accompanied by a broad spectrum of non-motor manifestations, including pain, depression, dementia, and sleep disorders.3,4 PD also carries high-risk complications such as falls and severe dysphagia, contributing to increased mortality.3,5
Palliative care (PC) aims to enhance quality of life (QOL) through a holistic, multidisciplinary approach, 6 addressing symptom management/burden (SMB), advance care planning (ACP), caregiver support, and psychosocial and spiritual needs. 7 However, PC remains traditionally associated with hospice and cancer care. 8 In neurology, neuropalliative care—an emerging field with a bright future 9 —applies PC principles to chronic progressive neurological diseases. 10
Despite a symptom burden comparable to cancer, 11 individuals with PD often experience unmet needs—including underrecognized symptoms, reduced QOL, existential distress, and high caregiver burden (CB). 12 Yet, the role of PC in PD remains underexplored, with limited robust evidence supporting its integration.6,10 Referrals to PC are infrequent, and barriers to access are common. 13 Limited time, space, financing, and staffing have been identified as major obstacles to PC implementation in PD. 14
Emerging research suggests potential benefits of PC in PD, but effects on patient and caregiver outcomes remain inconsistent, 6 and evidence gaps hinder policy and service development for broader PC integration.1,6
Recent reviews have examined PC in atypical parkinsonian syndromes and other neurodegenerative diseases,15,16 yet a focused evaluation in PD and related parkinsonian disorders (PDRD) is still lacking.
Objectives
This systematic review aimed to answer the following research question: Does integrating PC improve patient and caregiver outcomes compared to standard care in individuals with PD or PDRD?
Methods
This systematic review followed the recommendations of the “Cochrane Handbook for Systematic Reviews of Interventions”, 17 and was reported in accordance with the “Preferred Reporting Items for Systematic Reviews and Meta-Analyses” guidelines. 18
Eligibility Criteria
Participants
Adults (≥18 years) with PD or PDRD (multiple system atrophy, progressive supranuclear palsy, corticobasal syndrome, and dementia with Lewy bodies) and/or their informal caregivers. Non-parkinsonian neurodegenerative diseases (eg, multiple sclerosis, amyotrophic lateral sclerosis, Huntington’s disease, primary cerebellar ataxias, essential tremor) were not considered PDRD. Studies enrolling multiple neurological diagnoses were eligible when PD/PDRD were explicitly included, even if the proportion of PD/PDRD was unknown.
Interventions
Any PC Intervention (Specialist or Generalist; Inpatient, Outpatient, Home-Based, or Tele-PC)
Comparators
Usual neurological care or another active control; single-arm studies were eligible if pre–post outcomes were reported.
Outcomes
Motor and non-motor SMB, QOL, CB, patient and/or caregiver satisfaction with care (SWC), using measures as reported by the original studies.
Study Design
Primary empirical studies of any design were eligible. We excluded reviews, protocols, conceptual papers, conference abstracts, book chapters, letters/editorials, theses, and calls for collaboration.
Articles not freely accessible or outside our Faculty’s subscriptions were excluded.
Information Sources
MEDLINE (via Pubmed), Web of Science, Scopus, and Cochrane Library databases were systematically searched for relevant articles published up to December 31, 2024. Each source was last consulted on January 4, 2025. No additional records were identified through manual search or other sources. No direct contact with authors was made to identify additional sources.
Search Strategy
The search combined free-text terms and MeSH terms using the following structure: (“palliative care” OR “hospice care” OR “end-of-life care”) AND (Parkinson*). Filters were applied to include only articles written in English, involving adult participants (≥18 years). The search strategy used in each database is summarized in Supplemental Table 1.
Selection Process
In the initial screening, all articles were reviewed by title and abstract independently by both authors. The full texts of potentially relevant articles were then assessed for eligibility, also independently by the authors. Any disagreements regarding study selection or data extraction were resolved through discussion and consensus. The reference manager software Mendeley® v2.122.0 for Windows was used to collect, organize, and remove duplicates.
Data Collection Process
A data-charting form was developed using Microsoft Excel (Microsoft Corp, 2016) and iteratively refined through testing. Initial data extraction was conducted primarily by the first author, with the second author independently cross-verifying and supplementing the data. Any disagreements regarding study selection or data extraction were resolved through discussion until consensus was reached.
Data Items
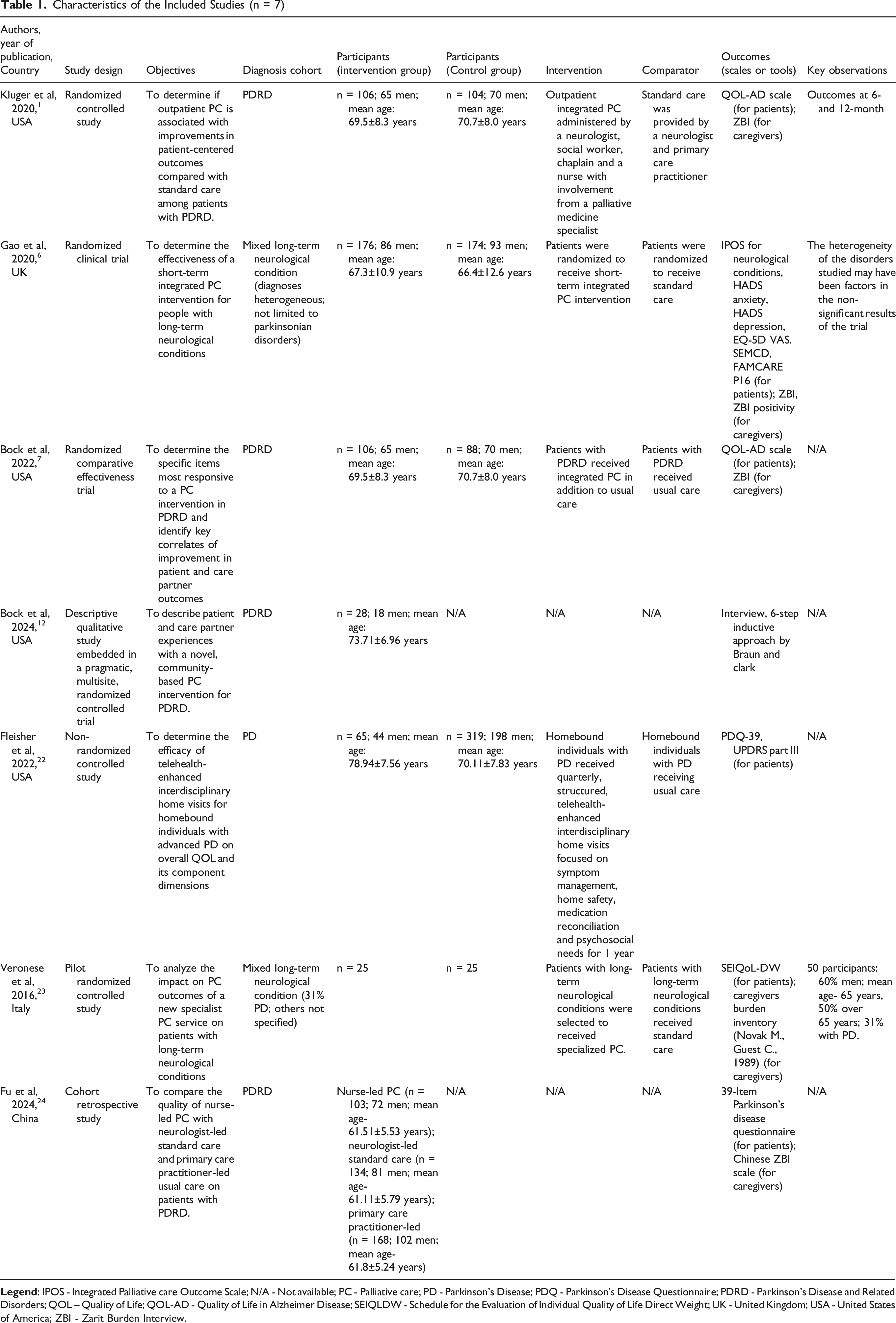
Characteristics of the Included Studies (n = 7)
Main Results of the Included Studies (n = 7)

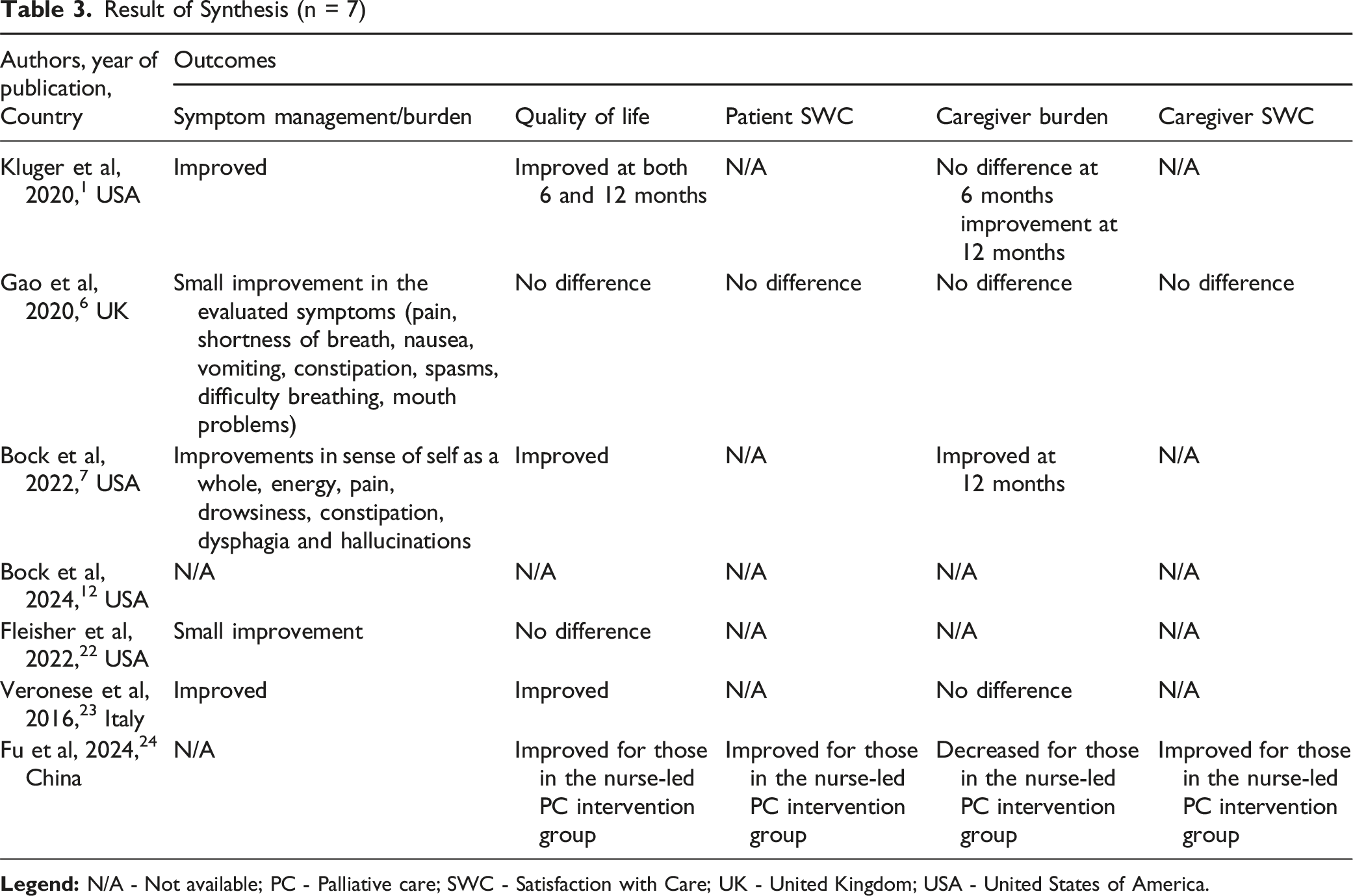
Result of Synthesis (n = 7)
Two reviewers independently extracted study characteristics and outcome data (SMB, QOL, SWC, CB), recording effect estimates or summary findings as reported by the original studies; disagreements were resolved by consensus. For mixed-diagnosis cohorts, we documented cohort composition where available (eg, % PD) and flagged non-separable PD/PDRD results to ensure transparent interpretation within the synthesis.
Study Risk of Bias Assessment
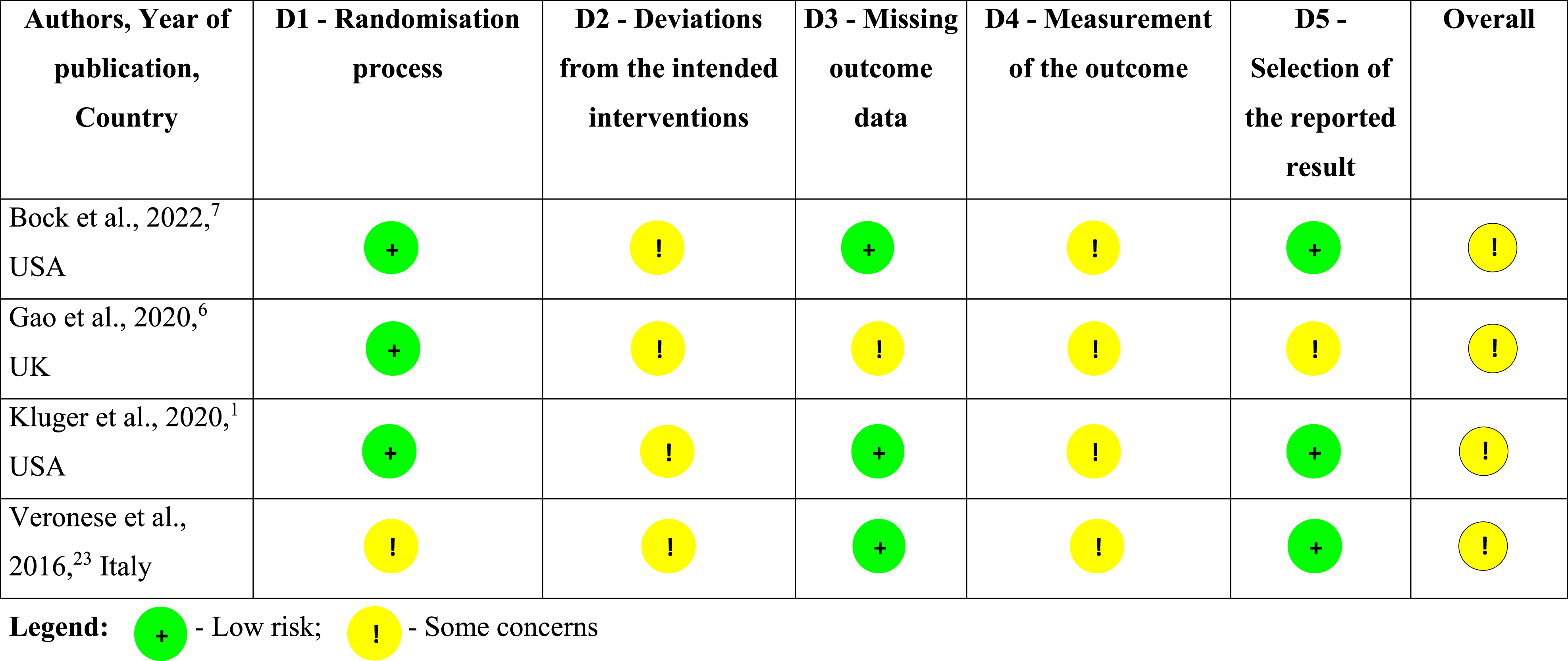
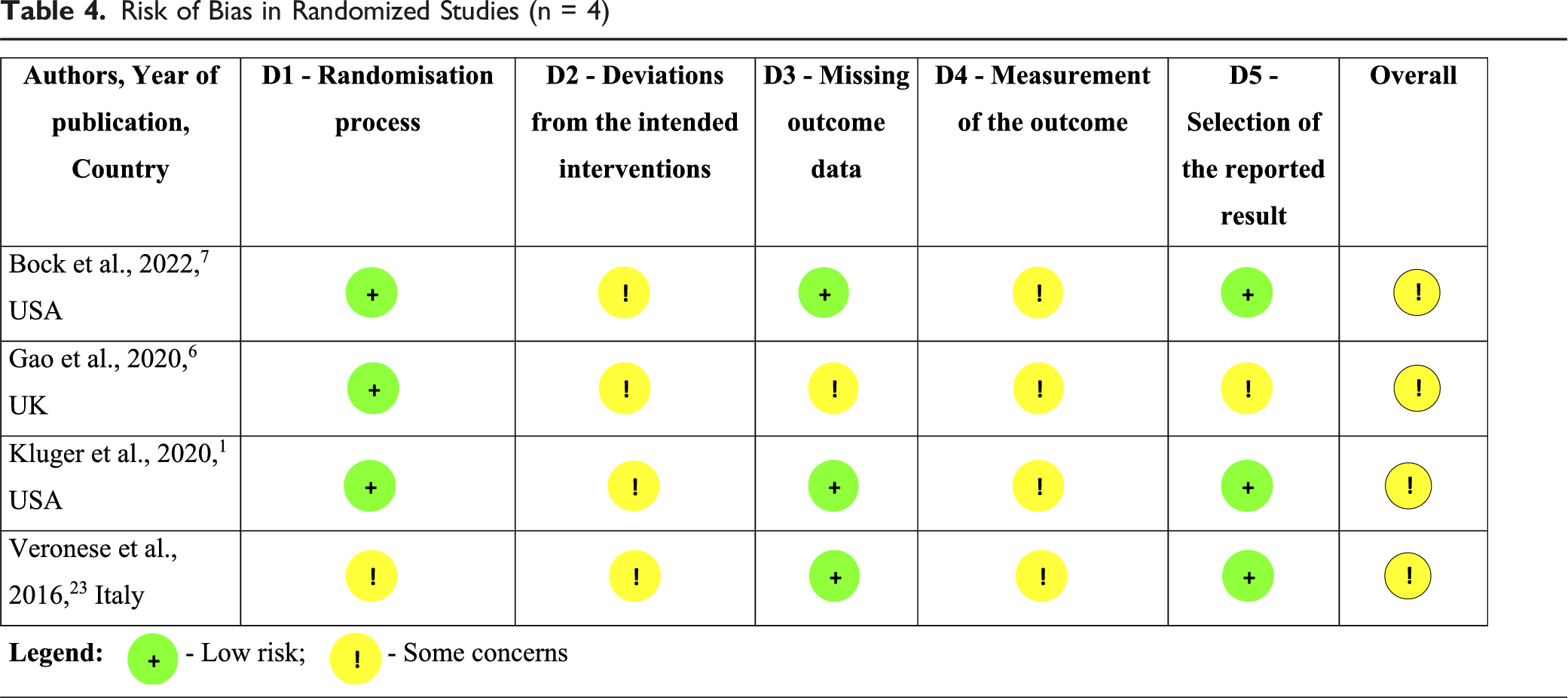
Risk of Bias in Randomized Studies (n = 4)
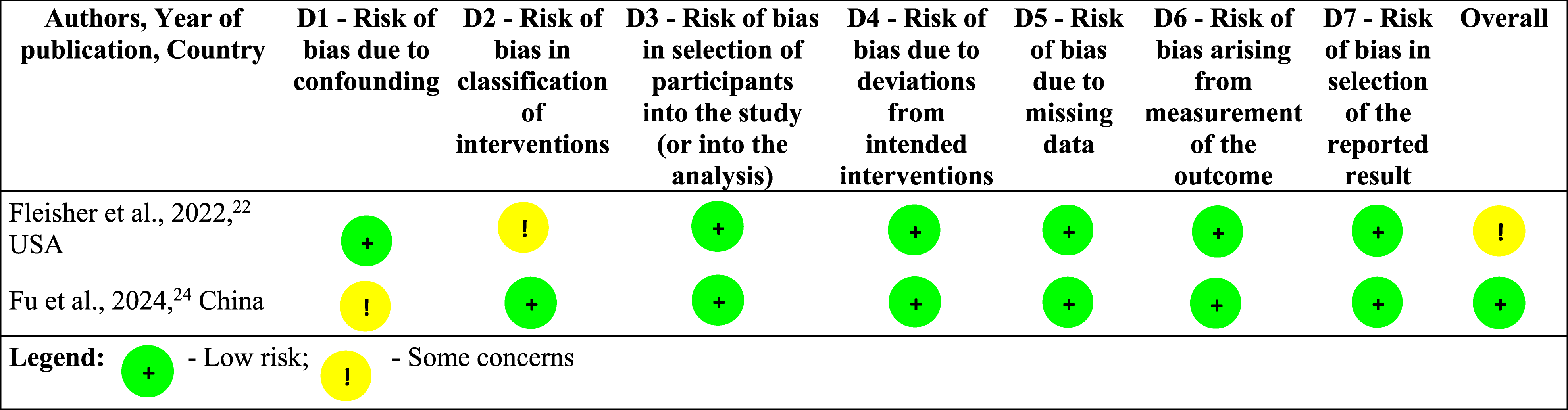
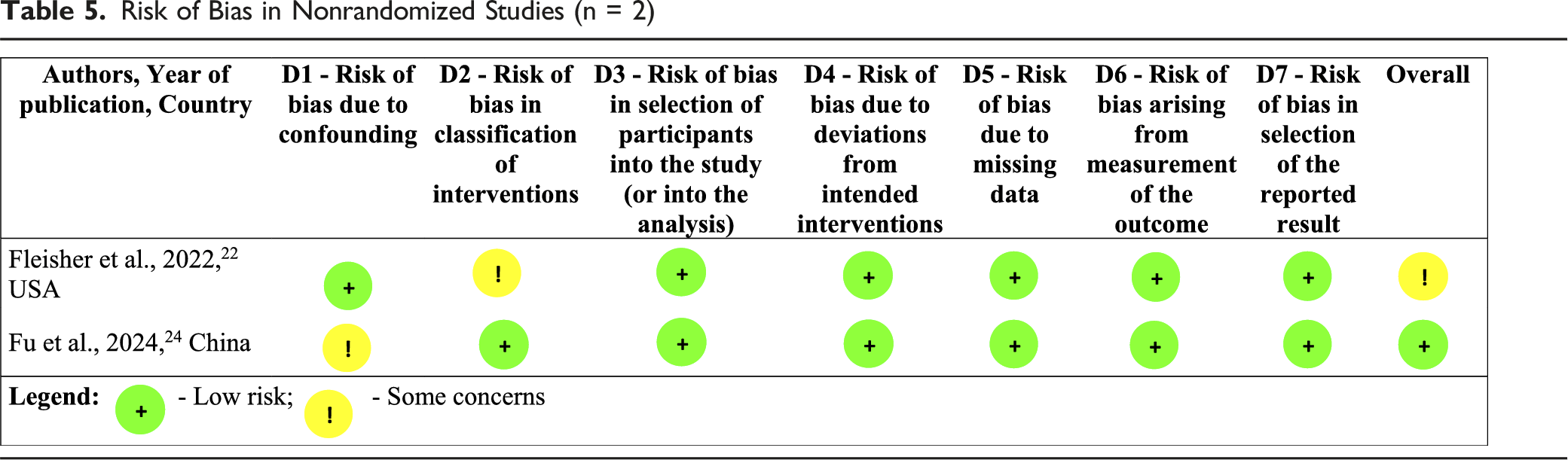
Risk of Bias in Nonrandomized Studies (n = 2)
Effect Measures
We accepted all the effect measures as declared by the study authors, for each outcome. These measures were utilized both in the synthesis and presentation of the results.
Synthesis Methods
A meta-analysis was not conducted due to the limited number of studies and the heterogeneity of interventions and outcomes, which precluded meaningful quantitative synthesis. Likewise, no formal evidence grading framework was applied; instead, study findings were synthesized narratively.
We synthesized all eligible studies (PD/PDRD-only and Mixed) together because the intervention is care-process oriented and outcomes are transdiagnostic. Mixed-cohort studies were flagged and their inclusion noted as indirect with potential to dilute PD/PDRD-specific effects. Where cohort composition was reported (eg, proportion PD), we describe it. We report summary statistics across studies and compare patterns qualitatively by cohort type.
The synthesis is organized according to the outcomes under study: SMB, QOL, patient SWC, caregiver SWC, and CB. In addition, 1 qualitative study exploring patient and caregiver experiences with PC was included to provide contextual and explanatory insights. Its findings were analyzed thematically and integrated narratively, with themes mapped onto the outcome domains.
To visually present the results of individual studies and the synthesis findings, data-charting forms were created and are presented in Tables 2 and 3, respectively.
Results
Study Selection
A total of 670 records were identified through database searching. After the removal of 139 duplicates, 531 records were screened by title and abstract, resulting in the exclusion of 512 records. The remaining 19 full-text articles were assessed for eligibility, of which 12 were excluded due to absence of relevant outcomes (n = 7), incompatible study design (n = 4), or lack of relevant intervention (n = 1). Seven articles met the inclusion criteria and were included in the review. The study selection process is illustrated in Figure 1. Flow Diagram of the Study Selection Process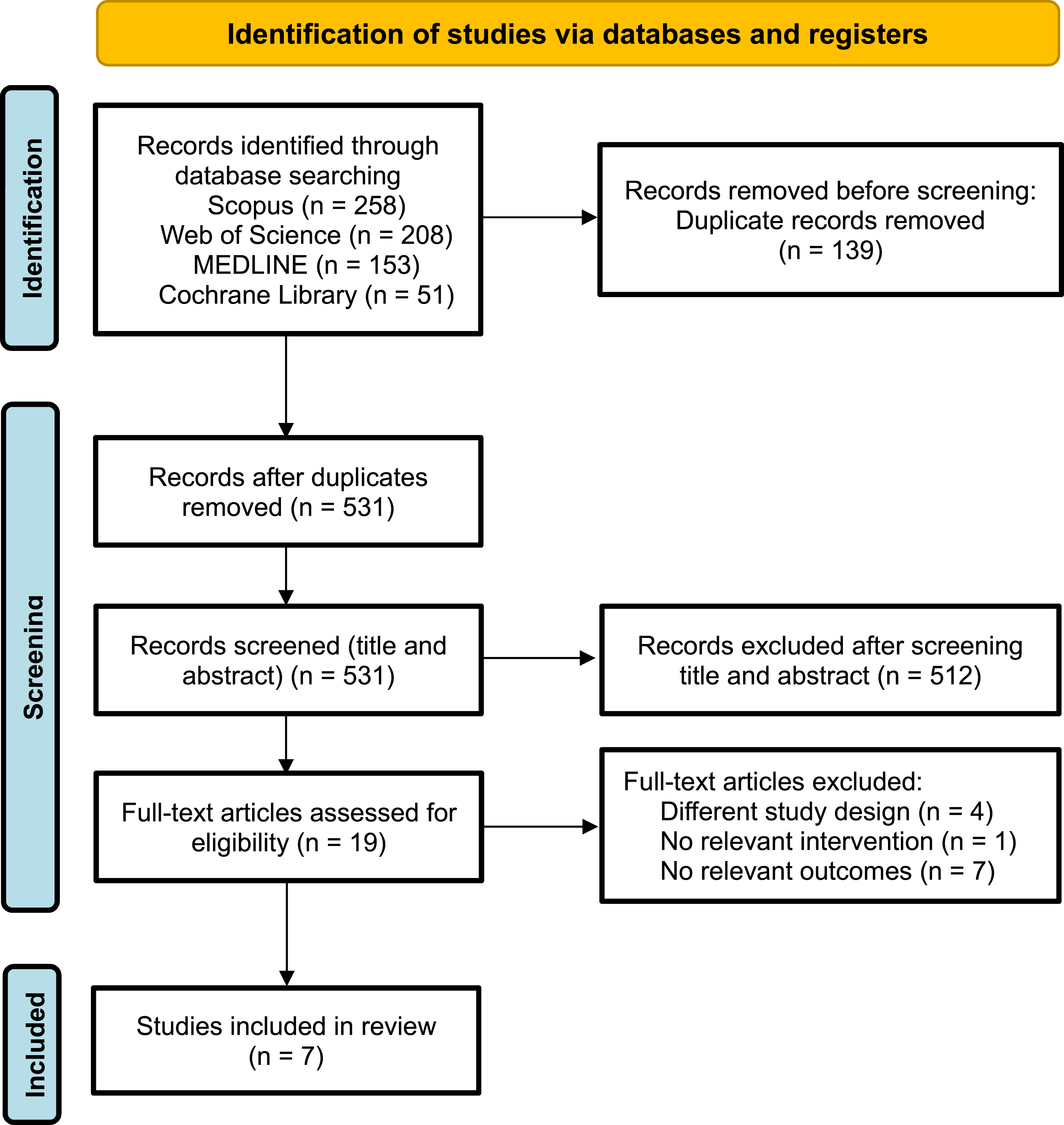
Study Characteristics
The seven studies originated from America (United States of America),1,7,12,22 Europe (United Kingdom, 6 and Italy 23 ), and Asia (China). 24 A variety of study designs were represented: two RCT,1,23 one non-RCT, 22 one randomized clinical trial, 6 one randomized comparative effectiveness trial, 7 one descriptive qualitive study, 12 and one retrospective cohort study. 24 All studies included patients with PDRD, although two also enrolled individuals with other long-term neurological conditions,6,23 in one, 31% of participants had PD, while the other did not report the PD/PDRD proportion. Sample sizes ranged from 28 participants, 12 to 405 patients. 24 The characteristics of the included studies are summarized in Table 1.
Risk of Bias in Studies
The overall risk of bias among the 7 studies was low to moderate (Tables 4 and 5).
In the included RCT, allocation sequence bias was generally low, using blocked or minimization randomization.1,6,7 Veronese et al 23 applied a parallel arm design with folders randomly selected by a secretary, which carried a moderate risk of bias.
Non-RCT by Fleisher et al 22 and Fu et al 24 lacked random allocation, but no clear evidence suggested high bias from this alone. Blinding of participants, caregivers, and providers was not implemented; however, Kluger et al 1 and Gao et al 6 blinded analysis teams until after data collection. Bock et al 7 and Veronese et al 23 did not report blinding.
Loss to follow-up occurred mainly due to death or clinical deterioration. Only Gao et al 6 had moderate bias risk from missing data, but no study showed meaningful impact on outcomes. Measurement methods were consistent across groups, though subjective scales for QOL and CB could introduce bias, acknowledged by Kluger et al. 1
Selective reporting was low risk in all studies,1,7,23,24 except Gao et al, 6 which reported only 12-week baseline data. Fu et al.’s 24 retrospective cohort and Fleisher et al.’s 22 non-RCT had low risk in most domains, with minor concerns.
Bock et al 12 was appraised using the CASP checklist, which indicated overall good methodological quality, although the rigor of data analysis was not fully described. The authors acknowledged that their sample might not represent PDRD patients with significant comorbidities, limiting transferability of findings.
Results of Individual Studies
Detailed results of individual studies are presented in Table 2.
Results of Synthesis
A summary of the synthesis results is presented in Table 3.
Symptom Management and/or Burden
All studies assessing motor and non-motor SMB reported improvements in the intervention group compared to the usual care group.1,6,7,22,23 However, the extent and statistical significance of these improvements varied by study and symptom domain. No study reported consistent improvement across all symptoms assessed. Overall, 60% of the studies described clear improvements, while 40% reported only modest gains. In the qualitative study, 12 participants perceived routine neurology care as overly focused on motor symptoms/medication, with unmet needs in non-motor symptom support and anticipatory guidance. Under the PC model, the multidisciplinary approach addressed a broader range of symptoms and needs.
Quality of Life
Six of the included studies assessed QOL.1,6,7,22-24 Of these, 4 reported statistically significant improvements in the intervention groups compared to standard care.1,7,23,24 The remaining 2 studies declared to have found no statistically significant changes.6,22 Caution is warranted in interpreting these findings, as all participants were aware of their treatment allocation, potentially introducing bias in self-reported QOL outcomes. In the qualitative study, 12 participants linked clear prognostic counseling, psychological support, and spiritual care (including chaplaincy) to improved coping and day-to-day functioning, suggesting pathways by which PC may positively influence QOL, even when quantitative QOL changes were modest. Chaplains, initially perceived by some as ominous, often became highly valued sources of spiritual support once engaged, highlighting psychosocial–spiritual gains not routinely met in standard neurology care. 12
Patient Satisfaction with Care
Only 2 studies evaluated patient SWC.6,24 One study reported no statistically significant differences between groups, 6 while the other described improvements in the intervention group compared to standard care. 24 In the qualitative study, 12 patients described greater engagement with the clinical team and more direct conversations about the future within PC, which they associated with higher satisfaction and a sense of being “seen” beyond motor symptoms. Across interviews, frustration stemmed from regional provider shortages, limited access to neurology/primary care, and fragmented communication. 12
Caregiver Satisfaction with Care
Similar to patient SWC, one study reported no statistically significant differences between groups, 6 while the other found improvements in caregiver SWC in the intervention group compared to standard care. 24 The limited number of studies and inconsistent findings make it difficult to draw firm conclusions on this outcome. In the qualitative study, 12 care partners reported that PC improved communication, validated caregiver roles, and facilitated referrals to community resources—features they viewed as enhancing satisfaction with the care experience. PC was perceived to streamline coordination and connect dyads to community-based resources. 12
Caregiver Burden
Of the 5 studies that assessed CB,1,6,7,23,24 3 reported improvements in the intervention group compared to standard care.1,7,24 Notably, 2 of these observed improvements only at the 12-month mark,1,7 while 1 reported improvement at 6 months. 24 These findings should be interpreted with caution, as caregivers were aware of the interventions received, which may have influenced their perception of burden. In the qualitative study, 12 caregivers frequently shouldered care-coordination tasks due to poor inter-provider communication in usual care. Under PC, team-based coordination and referrals partially mitigated this load, indicating potential burden relief mechanisms. 12
Discussion
Summary of Evidence
This systematic review (n = 7) assessed the impact of PC integration on patient and caregiver outcomes in PDRD. Six studies compared PC with standard care, while 1 examined unmet needs under usual care. All studies on SMB reported improvements with PC, though not consistently across symptoms. Four of 6 studies showed QOL benefits, while 2 found no difference. Evidence on SWC was mixed: of 2 patient studies, 1 found improvement and 1 no change; the same pattern was observed in caregiver SWC. Three of 5 studies reported reductions in CB, particularly at 12 months, though group awareness may have influenced results.
Patients voiced dissatisfaction with standard care, highlighting an overemphasis on motor symptoms, limited prognostic counselling, fragmented services, and inadequate interdisciplinary support. PC interventions addressed these gaps by broadening symptom focus, facilitating prognostic discussions, enhancing team-based care, and improving access to community resources.
Overall, findings suggest PC may benefit SMB, QOL, and CB in PDRD, with less consistent effects on SWC. However, the small number of studies, heterogeneity of designs, and variable outcome measures limit the strength of conclusions. High-quality, larger-scale research is needed to guide the effective integration of PC into routine PD management.
Symptom Management/Burden
Gao et al 6 reported a small but statistically significant reduction in overall symptom burden, despite not finding significant changes in non-key symptoms. The authors noted that the heterogeneity of their study population—which included individuals with multiple sclerosis—may have diluted the observed effects.
Among the studies that examined specific symptoms rather than global impact,7,22,23 the most notable improvements were reported in pain, tiredness, nausea, drowsiness, shortness of breath, bowel symptoms, apathy, and overall well-being. These findings align with Saleem et al. 25 ’s research showing that pain, fatigue, constipation, and drooling are among the symptoms most responsive to PC interventions. Alleviating these symptoms can significantly impact patients’ QOL. Importantly, several studies noted that the degree of symptom improvement increased over time, suggesting cumulative benefits of sustained PC.
Some non-motor symptoms may have identifiable and modifiable causes. For instance, joint mobilization and attentive skin care can help prevent pain in bed- or chair-bound individuals. 26 Despite the predominance of motor symptom management in standard care, non-motor symptoms often remain under-addressed. Bock et al 12 found that patients expressed a strong desire for psychological support and prognostic counseling, underscoring these as unmet needs. Notably, the same study reported that chaplains—initially perceived as ominous figures—often became the most valued source of spiritual support.
Regarding motor symptoms, these typically worsen over time due to diminishing pharmacologic responsiveness, with severe akinesia, postural instability, freezing of gait, dysarthria, and dysphagia being particularly burdensome. 27 While neurologists generally manage these symptoms, Kluger et al 1 demonstrated unexpected improvements in both motor and non-motor symptoms following PC integration. Fleisher et al 22 observed better preservation of mobility in the PC group compared to standard care. These findings highlight the value of combining pharmacologic with non-pharmacologic approaches to symptom management. For example, dietary modifications were considered the most effective non-pharmacologic intervention for non-motor symptoms, while resistance training and deep brain stimulation showed benefits for motor symptoms, particularly resting tremor, rigidity, and bradykinesia. 28 Furthermore, evidence suggests that PC may improve depressive symptoms in patients, even in the absence of changes in antidepressant use. 29
Quality of Life
Four of the included studies reported improvements in QOL following PC interventions.1,7,23,24 Consistent with this, a pragmatic trial enrolling 359 individuals with PDRD and 300 caregivers found that community-neurologist PC education plus team-based tele-PC was associated with better patient QOL at 6 months vs usual care. 30
Since QOL is strongly influenced by both motor and non-motor symptoms, Lum and Kluger 26 recommend regular assessment of domains such as pain, mood, psychotic and behavioral symptoms, sleep, energy, communication, autonomic function, swallowing, and nutrition.
Understanding how each individual patient and their family define and prioritize QOL is essential. This discussion should begin early in the disease trajectory,3,26 —ideally before cognitive decline limits participation—but should be approached sensitively to avoid overwhelming patients, particularly at the time of diagnosis.26,31
An additional dimension relates to the patient’s sense of “self as a whole.” Bock et al 7 reported that improvements in grief, mood, and spirituality under PC contributed significantly to patients’ self-perception and overall QOL. The erosion of self-worth and identity may be an underestimated source of distress in PDRD. 26
Assessment of QOL is inherently challenging, especially as disease progression can impair cognition and communication. Most available tools rely heavily on patient self-report. Therefore, there is a need for validated instruments that enable proxy or clinician-based evaluations. A flexible battery of tools is recommended to adapt to different settings and symptom domains, supporting a systematic and holistic approach to needs assessment – identification, evaluation, and response. 32
Notably, 2 studies employed tools originally developed for patients with Alzheimer’s disease, raising concerns about their appropriateness in the PDRD population.1,7 This underscores the urgent need to refine or develop specific QOL assessment tools to better capture the unmet needs of these patients. 32
Despite these challenges, the preferences of patients with mild-to-moderate dementia should not be disregarded. The “4 Cs” framework (understanding of Condition, Choices, Consequences, and Consistency of decisions) supports the notion that decision-making capacity should be judged based on specific choices rather than assumed globally, 26 offering a more nuanced approach to shared decision-making even as cognitive decline advances.
Patient Satisfaction with Care
There is limited evidence regarding the impact of PC on the SWC of individuals with PDRD, with only 2 studies including it as an outcome—and with contrasting findings.6,24 Veronese et al, 23 while not directly evaluating SWC, reported that PC improved social isolation and aspects of perceived care. However, it also noted a worsening in some psychological outcomes among the intervention group, possibly due to patients’ discomfort when interacting with professionals closely associated with end-of-life care, particularly when patients had not yet accepted themselves as terminally ill.
Patients and caregivers reported difficulties accessing primary physicians and neurologists, compounded by poor communication between providers, often leaving caregivers to coordinate care themselves. 12 To address these issues and improve SWC, care teams should be interdisciplinary and include PC physicians, nurses, social workers, spiritual care providers, bereavement support professionals, occupational therapists, speech and language therapists, and dietitians. 33
Responding to patients’ emotional needs is also vital to improving care satisfaction. The diagnostic moment is often experienced with great emotional weight. ACP discussions should be introduced early—ideally before disease progression limits communication capacity—to ensure they are proactive rather than reactive. 33 These conversations should include topics such as advance directives, decisions regarding resuscitation, ventilation, nutrition, hydration, symptom management, preferred place of death, and appointment of healthcare proxies. 31 Provision of team-based PC via telehealth and PC education for community neurologists may improve ACP. 30 To facilitate ACP discussions, communication strategies like SPIKES (Setting up the Interview, Assessing Patients’ Perception, Obtaining Patients’ Invitation, Giving Knowledge, Addressing Emotions, Strategy and Summary), 34 and NURSE (Name the emotion, Understand and legitimize the emotion, Respect the challenges and courage of the patient, Support the patient and family, Explore approaches to working with this emotion) 35 have been recommended. 26 These mnemonics help clinicians approach difficult conversations with empathy, structure, and clarity—preparing patients for what lies ahead, assessing their current understanding, addressing their emotions, and involving family members when appropriate. Given patients’ common reluctance to engage with future planning—due to fear, denial, hope, or a desire to focus on the present, 31 —these tools are essential in ensuring communication remains compassionate and patient-centered. Annual reassessment of patient readiness for these discussions is advised. 31
Caregiver Satisfaction with Care
As with patient SWC, evidence on caregiver SWC remains limited, with only 2 studies evaluating it and presenting divergent results.6,24 The subjective and multifaceted nature of this outcome complicates its assessment. Nonetheless, caregivers consistently report feelings of isolation, often perceiving themselves as the sole coordinators of what should be a multidisciplinary care team.12,33 Frustration stems from inadequate communication with healthcare professionals, regional shortages of providers, and a perceived lack of understanding of the patient’s complex needs. 12 Additionally, caregivers frequently express that they were not adequately informed about disease progression, prognosis, or available care options, including PC and ACP. 31 These gaps hinder timely decision-making and limit both patient and caregiver access to appropriate care.
Greater involvement of caregivers in planning and communication processes, along with improved professional education on PDRD and PC, is essential to enhance caregiver SWC and support. 33
Caregiver Burden
Care partners of individuals with long-term conditions such as PDRD often experience sleep deprivation, exhaustion, anxiety, depression, guilt, and impaired immune function, all of which negatively affect their emotional, social, and physical well-being. 26 The impact of PC on CB remains inconsistent across the 5 studies that addressed this outcome, making it difficult to draw firm conclusions.1,6,7,23,24 Consistently, a pragmatic stepped-wedge trial in PDRD found that community neurologist PC education plus telehealth did not reduce 6-month CB vs usual care. 30
Among the studies reporting improvements in CB, there is variation regarding the time required to observe benefits—two found statistically significant changes at 12 months,1,7 while 1 observed improvement by 6 months. 24 Conversely, 2 studies found no significant differences. Veronese et al 23 suggested that prolonged disease duration and delayed discussions about death may contribute to persistent caregiver distress, with higher levels of cognitive impairment in patients correlating with greater CB. 36 Bock et al 7 also found that patient engagement in ACP was associated with a small but significant reduction in CB, potentially due to both direct and indirect support offered through PC. However, not all patient-focused interventions lead to reductions in CB. For instance, despite improving patient condition, deep brain stimulation did not correspond to lower CB, possibly due to ongoing care demands, side effects, or unmet expectations. 36
To mitigate CB, the inclusion of social work and counseling services has been recommended.26,37 The National Consensus Development Conference for Caregiver Assessment highlights the need to identify primary caregivers, incorporate their needs into care planning, provide essential caregiving training, and conduct periodic reassessments.26,38 Timely referral to PC is crucial when home care becomes unsustainable, particularly when patients require additional assistance, transportation, or emergency services. 33 Barriers to PC access in PDRD include insufficient information, limited education, inadequate home-based care, inconsistent provider support, and socioeconomic or cultural disparities, while enhanced care coordination and caregiver education facilitate access. 39
Limitations
This review has several limitations. The included studies displayed considerable heterogeneity in design, sample size, interventions, and outcome measures, which precluded meta-analysis and limited the strength of conclusions. Many studies had small samples and short follow-up, reducing generalizability and robustness. Most lacked blinding of participants and caregivers, increasing the risk of bias, particularly in subjective outcomes such as QOL and SWC. Our synthesis included mixed-diagnosis studies in which PD/PDRD formed only a subset (or the proportion was unknown). This introduces indirectness and may attenuate PD/PDRD-specific effects. We therefore flagged these studies, interpreted findings cautiously, and highlight the need for future PD/PDRD-specific trials. Furthermore, the majority of studies were conducted in North America and Europe, limiting cultural applicability. Research in PC settings faces additional challenges, as patients often present with cognitive or physical limitations that may hinder participation and reliable data collection. The diversity of PC models across studies further complicates comparisons, as no standardized approach exists for PDRD. Caregiver outcomes were rarely assessed, and findings were frequently conflicting, highlighting an important gap in evaluating caregiver experiences.
With regard to the review process, language restrictions to English may have excluded relevant evidence, and the search was limited to databases available through our institution. Our search strategy, while comprehensive, did not include “neuropalliative care” as a specific term, which may have resulted in missed studies describing integration of PC principles within neurological care. No contact was made with study authors to obtain unpublished data or clarifications, which may have affected completeness. A formal evidence grading framework (eg, GRADE) was not applied, due to feasibility constraints, which reduces interpretability and clinical applicability. Future reviews should incorporate structured grading systems to strengthen the reliability of findings.
Finally, although the review was registered in PROSPERO, this occurred after the search and screening stages but before data extraction, which may affect transparency.
Despite these limitations, this review provides a valuable synthesis of current evidence and highlights key directions for future research and practice.
Implications of the Results for Practice, Policy, and Future Research
The findings of this review highlight the potential of PC to improve the management of PDRD, particularly in addressing multidimensional SMB, enhancing QOL, and reducing CB. However, inconsistent implementation and heterogeneity of outcomes across studies point to key implications for clinical practice, health policy, and research.
Clinicians should consider integrating PC principles early and systematically into PDRD care. Multidisciplinary approaches that go beyond motor symptom control—incorporating psychological support, prognostic communication, and caregiver needs—may offer added value. Training neurologists and other professionals in core PC competencies is a practical next step to expand access and improve coordination.
Health systems and service planners should recognize the value of embedding PC within neurology and movement disorder services. This may require investment in team-based structures, development of referral pathways, and clearer guidance on when and how to integrate PC in neurodegenerative conditions. Reimbursement models should reflect the complexity and longitudinal nature of care required by PDRD populations.
There is a need for high-quality, adequately powered studies using standardized outcome measures to assess PC effectiveness across settings and populations with PDRD. Research should also explore optimal timing, delivery models, and long-term impacts of PC, as well as patient and caregiver experiences. Comparative effectiveness and implementation studies may further guide scalable, evidence-based integration of PC into neurology care.
Conclusions
This systematic review assessed the effectiveness of PC vs standard care in PDRD, focusing on SMB, QOL, SWC, and CB. The findings suggest that PC may offer meaningful benefits, particularly in improving symptom control, QOL, and reducing CB over time. Evidence on SWC was limited and mixed.
Qualitative findings revealed persistent gaps in standard care, including fragmented delivery, limited interdisciplinary involvement, and poor communication. PC may address these through comprehensive, team-based care models.
However, variation in study design, outcomes, and interventions limits generalizability. More rigorous, larger-scale studies are needed. Standardized outcomes, longer follow-up, and studies across disease stages will be essential to guide practice and inform policy on PC integration in PDRD care.
Supplemental Material
Supplemental Material - Integrating Palliative Care in Parkinson’s Disease and Related Disorders: A Systematic Review of Patient and Caregiver Outcomes
Supplemental Material for Integrating Palliative Care in Parkinson’s Disease and Related Disorders: A Systematic Review of Patient and Caregiver Outcomes by Maria-Inês Barros-Ferreira, Paulo Reis-Pina in American Journal of Hospice and Palliative Medicine®.
Footnotes
Author Contributions
This study was conceptualized by MIBF and PRP. MIBF and PRP conducted searches and screening of articles, analyzed data, designed the review protocol, wrote the manuscript, reviewed it, and approved the definitive version of the paper.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data relevant to this study are included within the article. No additional datasets, code, or materials were generated or used.
Registration and Protocol
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.