Abstract
Introduction
Idiopathic pulmonary fibrosis (IPF) is a progressive fibrosing lung disease with high morbidity and mortality. Declining lung function and high symptom burden lead to poor quality of life and premature death. 1 Median survival is estimated to be 2-5 years from diagnosis. 2 Health-related quality of life is significantly impaired in patients with IPF, with marked deterioration as death approaches. 3 A joint policy statement by American Thoracic Society, American Academy of Hospice and Palliative Medicine, Hospice and Palliative Nurses Association, and Social Work Hospice and Palliative Care Network recommends incorporation of palliative care early in the care continuum for patients with serious respiratory illnesses including IPF. 4 Unfortunately, this is not frequently implemented in practice due to a variety of barriers, including lack of practice guidelines for providers as well as lack of prioritization of the palliative care needs of these patients. 5 There are limited descriptions of palliative care delivery and its impact in IPF, and even fewer studies on palliative care delivery models.
Oncology research has demonstrated numerous benefits of early integration of palliative care, including improved survival. Temel et al. showed that early referral to palliative care improved median survival in patients with metastatic non-small-cell lung cancer. 6 Several uncontrolled studies on palliative care in IPF have shown similar benefits, but its impact on survival has not been explored.7-10 In this paper, we describe 3 models of palliative care delivery and examine the impact of palliative care on survival in IPF. We hypothesize that delivery of palliative care will improve survival in IPF patients.
Methods
Study Design
This is a retrospective multicenter international study of 3 cohorts of patients with IPF. Adult patients (age >18 years) who were diagnosed with IPF based on American Thoracic Society (ATS) 2011 guidelines and seen between 2012-2018 in three interstitial lung disease clinics were included. Patients with missing data (patient moved or data not recorded) were censored at the time of leaving the region or loss to follow-up. We did not have any recorded data after patients moved away. Patients with a diagnosis of non-idiopathic pulmonary fibrosis were excluded.
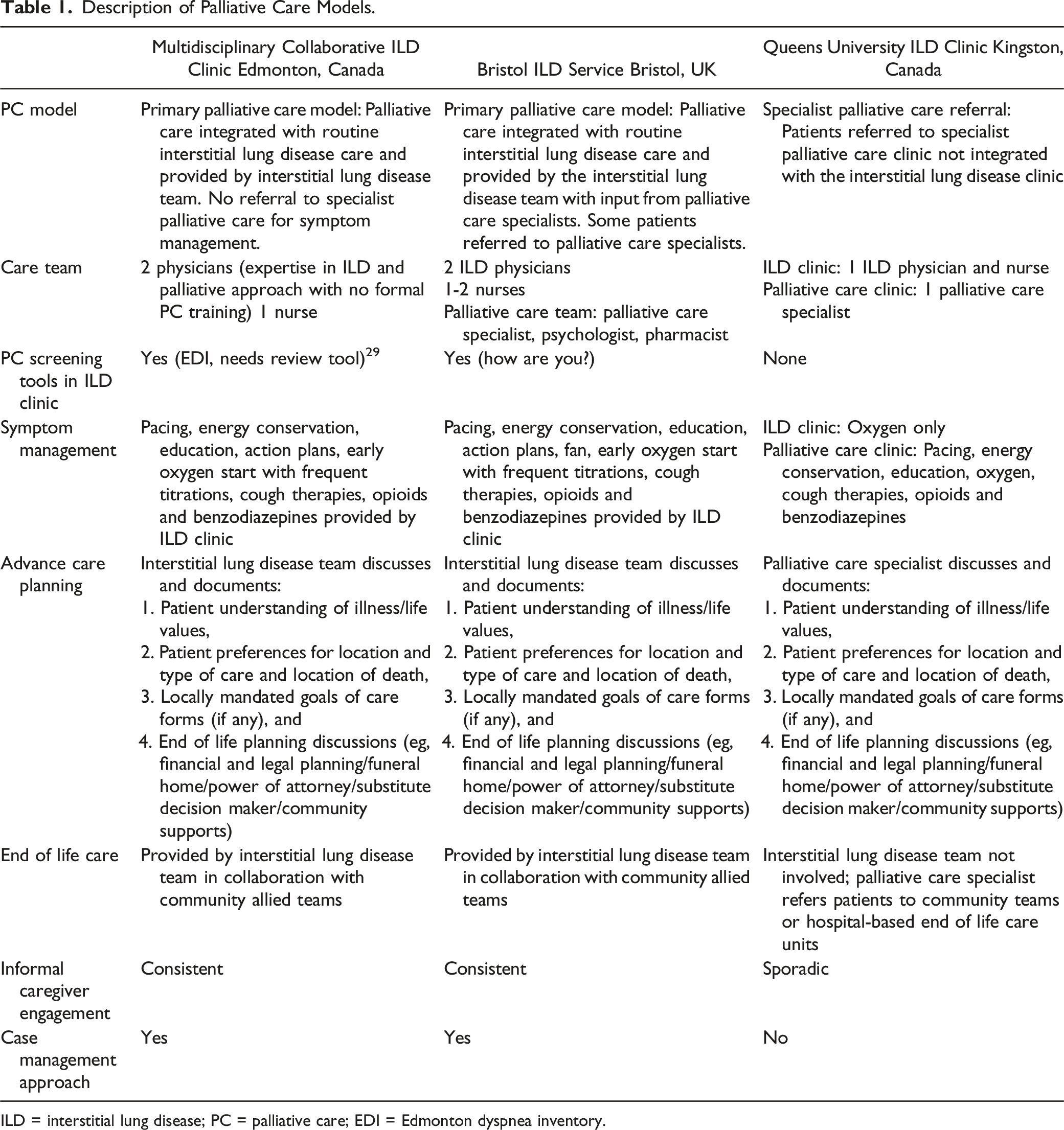
The models for palliative care delivery at the 3 sites are as follows (also described in Table 1): (1) (2) (3) Description of Palliative Care Models. ILD = interstitial lung disease; PC = palliative care; EDI = Edmonton dyspnea inventory.
Data Collection
The principal investigators developed a REDCap database for data collection; this was trialed by 2 sites to ensure adequacy prior to implementation. EC and BC data were entered into REDCap by respective site personnel. QC data were provided in an Excel spreadsheet and entered into REDCap by the principal investigator team. Data sources included electronic charts (eClinician at EC), paper charts (BC and QC), and the Queen’s University-Kingston Health Sciences interstitial lung disease clinic database and patient care system (electronic document management system).
Variables of interest included demographics, body mass index (BMI), smoking, pulmonary function tests, 6-minute walk distance, antifibrotics, opioids, supplementary oxygen needs, non-pharmacologic symptom therapies, advance care planning documentation, specialist palliative care referral, and location and cause of death if known for decedents. Deaths were confirmed based on report to the clinics at BC and QC, and via an online data repository at EC.
For this study, we used 2 criteria to classify patients as having received palliative care at EC and BC: any patient who had (i) a comprehensive assessment and management of symptoms and needs (both sites used their own needs assessment tools in the absence of a standard tool; management included any of non-pharmacologic strategies, oxygen, or opiates)
Data Analysis
Data were summarized by medians and interquartile ranges, counts and percentages as appropriate. Survival data were visualized using Kaplan-Meier curves and compared using a log rank test. Median survival and 95% confidence intervals were calculated for each center. Cox Proportional Hazards models were examined to determine the multivariable relationships between survival and palliative care intervention, care model, age, gender, forced vital capacity (FVC), diffusing capacity of the lung for carbon monoxide (DLCO), BMI, smoking history, antifibrotics, opioids, oxygen, and Gender-Age-Physiology (GAP) stage. GAP stage is used to determine the average risk of mortality in IPF and higher stage is associated with greater mortality. 12 We did unadjusted and adjusted analyses for all baseline predictors as above. Adjusted analyses are presented as 2 multivariable models. Model 1 was adjusted for age, gender, FVC, DLCO, BMI, oxygen, smoking history, antifibrotics, opioids, palliative care intervention and care model. Model 2 was adjusted for the same variables but age, gender, FVC, and DLCO were replaced by GAP stage which includes all 4 variables. Model residuals were examined for goodness of fit.
Given the nature of the palliative care intervention, which was initiated early when patients were first assessed in the EC and BC models but could be delayed after being referred to palliative care in the QC model, we addressed the potential for immortal time bias as follows: We divided the EC and BC cohorts into 3 groups, those who received palliative care at their initial visit, those who received palliative care at a later visit, and those who never received palliative care, and compared their clinical features. In order to control for potential immortal time bias, we performed a sensitivity analysis examining the subgroup of patients who had palliative care initiated at their first visit in the EC and BC cohorts and compared it to those who did not receive palliative care. For the QC cohort, we considered the referral date to palliative care as time zero for all survival analyses, instead of the date the patient was actually seen by palliative care.
All analyses were conducted using R v4.1.0 (Vienna, Austria).
Ethics Approval
The Health Research Ethics Boards in Alberta (REB Pro00082981); Ontario (HSREB 6025437); and Bristol (IRAS 254044) approved this study with waiver of informed consent due to minimal risk posed and difficulty obtaining informed consent from all participants for a variety of reasons, including loss to follow-up and death.
Results
Patient Characteristics (all Cohorts and by Site).
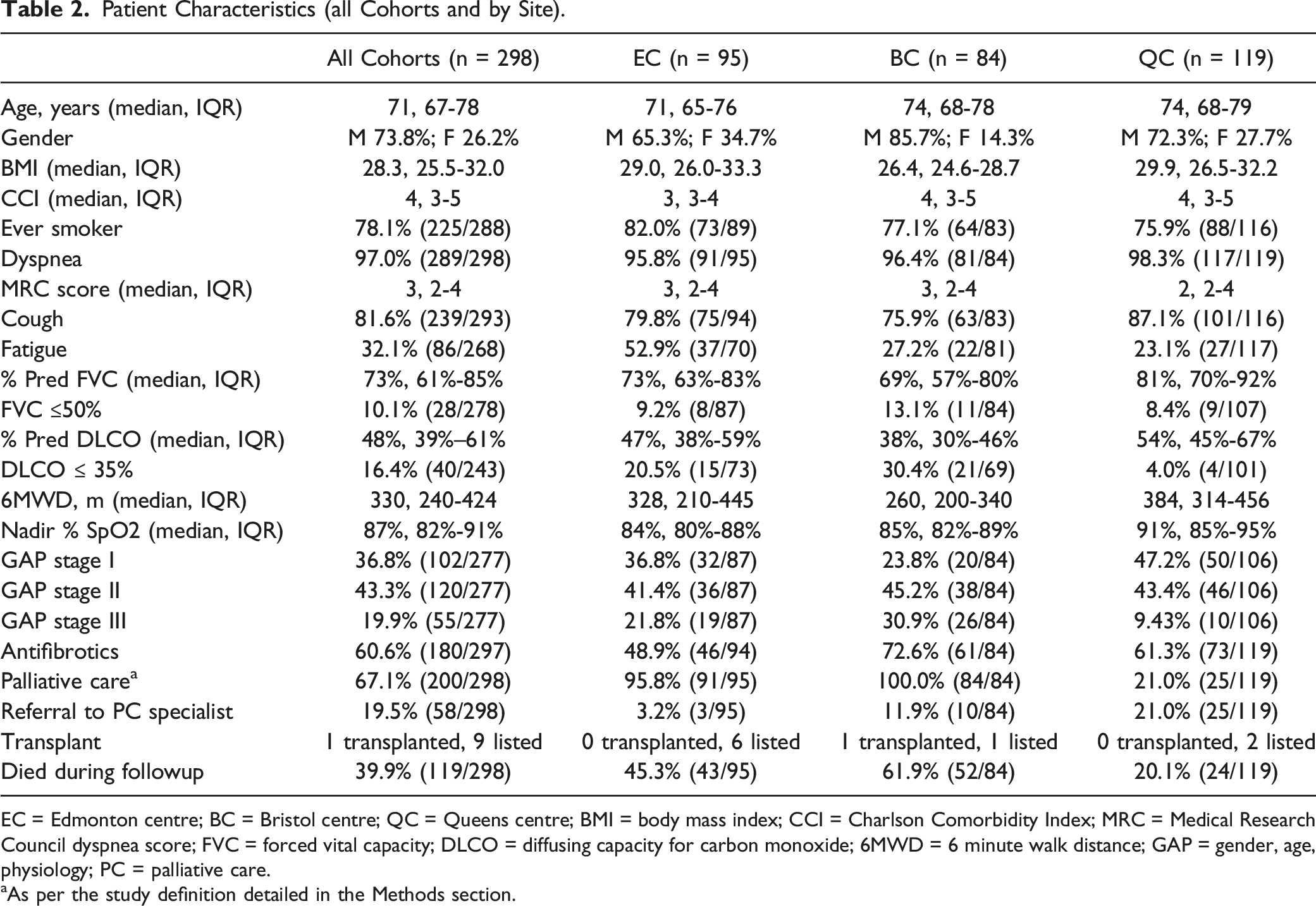
EC = Edmonton centre; BC = Bristol centre; QC = Queens centre; BMI = body mass index; CCI = Charlson Comorbidity Index; MRC = Medical Research Council dyspnea score; FVC = forced vital capacity; DLCO = diffusing capacity for carbon monoxide; 6MWD = 6 minute walk distance; GAP = gender, age, physiology; PC = palliative care.
aAs per the study definition detailed in the Methods section.
The panels in Figure 1 depict survival based on individual prognostic factors (adjusted according to Model (2) over the 4-year follow-up period. A univariate analysis demonstrated that a lower GAP stage, BMI >30 kg/m2, and antifibrotic use reduced the risk of death. Palliative care intervention did not show survival benefit in the unadjusted analysis, but in the adjusted analysis, it reduced risk of death even after accounting for antifibrotic use [model 2 aHR .28 (.12-.65)]. 66.0% of patients who received palliative care were also on antifibrotic therapy, compared to 49.0% of patients who did not receive palliative care (Table S3). Opioid use was associated with an increased risk of death in both models. Oxygen use was not a significant predictor of mortality in either model. BMI >30 kg/m2 had a protective effect on survival. The concordance index for the model was .76 (se .03). Adjusted Kaplan-Meier curves for various prognostic factors: (A) GAP stage, (B) Smoking, (C) BMI >30, (D) Oxygen therapy, (E) Antifibrotic therapy, (F) Opiate therapy, (G) Palliative care, and (H) SIte. Survival curves are adjusted according to model 2. There are 298 at risk at t = 0, 152 (51%) at risk at 1 year, 70 (23%) at 2 years, 36 (12%) at 3 years, and 16 (5%) at 4 years of follow-up. The EC and BC curves in panel H are overlapping because their corresponding HR was 1.01. N = no; Y = yes; GAP = gender, age, physiology; BMI = body mass index; PC = palliative care; EC = Edmonton centre; BC = Bristol centre; QC = Queens centre.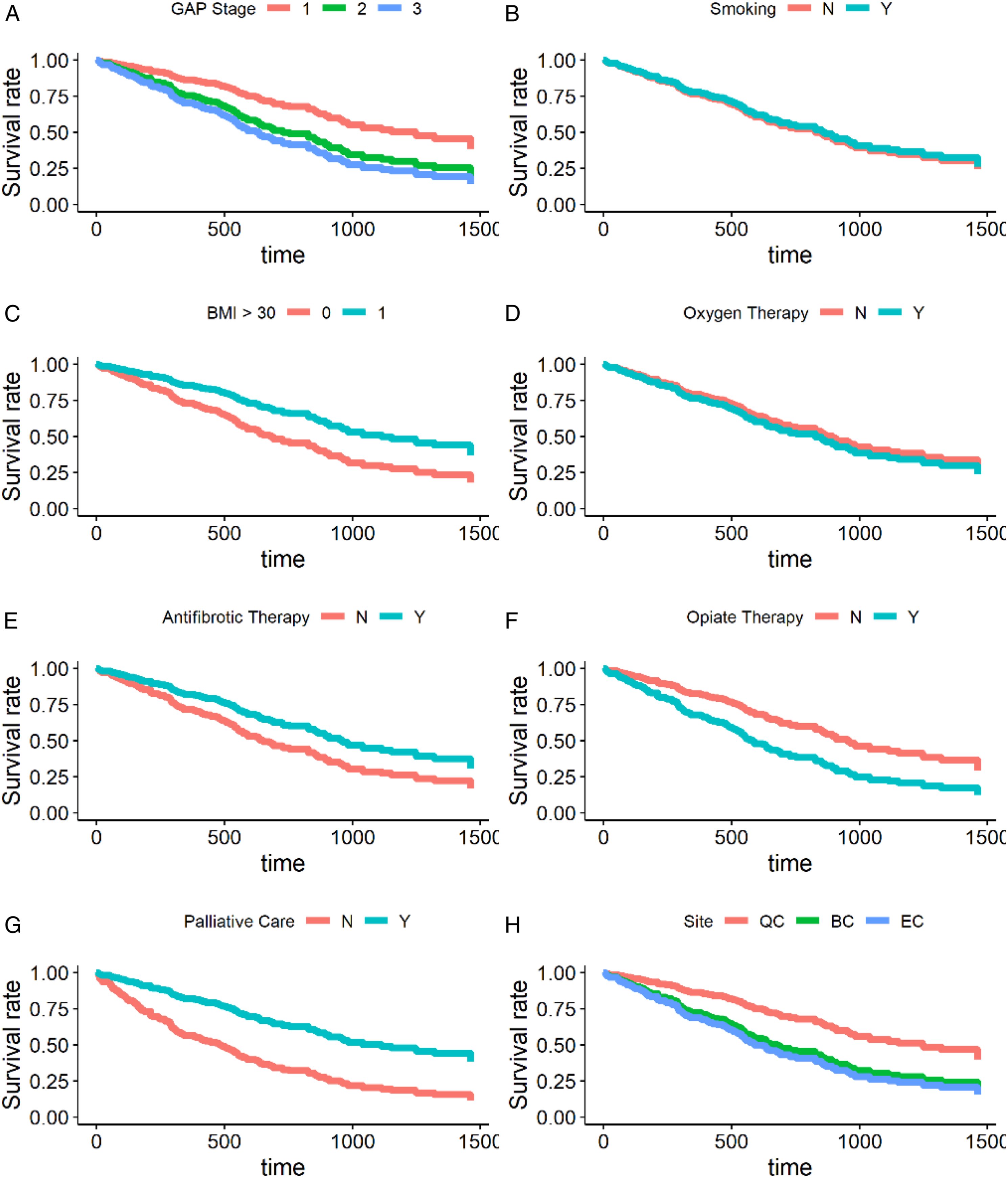
As variability in intervention duration may lead to favorable bias in survival analysis, we performed a sensitivity analysis based on time of onset of palliative care. Among the 200 patients who received palliative care as per the study definition, 76% (152/200) were initiated on it at the first visit. Compared to patients who received palliative care at the first visit, more patients who received palliative care later (visit 2 onwards) were in GAP stage I, corresponding to a lower disease severity (Table S3). In order to minimize bias further we repeated the univariate analysis with patients who were initiated on palliative therapies at first visit only, in other words excluding those who received palliative care later. The signal of benefit for palliative care intervention is preserved but not statistically significant [aHR .55 (.17-1.72)] (Table S1). If further stratified by GAP stage, the signal of benefit is only preserved in the GAP stage I patients (Table S2).
Discussion
This is the first study to describe various palliative care models in patients with IPF. In our international multicenter cohort, 67% received palliative care, with 76% being initiated at the first visit. This is in stark contrast to previous studies; Lindell and colleagues noted that only 14% of IPF decedents received palliative care, with a majority occurring in the last month of life. 13 The early onset of palliative care in our cohort is consistent with the recommendations in the 2022 ATS IPF guidelines. 1 In addition, we described 2 clinic models that integrated primary palliative approach with routine care in line with the 2022 ATS policy paper on palliative care for chronic respiratory diseases. 4 We are the first to report that a primary palliative approach can lead to more IPF patients receiving palliative care and earlier in the disease trajectory. Our hypothesis that palliative care can improve survival in IPF is also supported by our results, which showed that palliative care reduced the risk of death over the 4-year follow-up period. The pattern persisted after adjusting for known confounders including antifibrotic therapy and GAP stage. Our findings are further supported by a recent single center study of deceased fibrotic ILD patients demonstrating that patients who received palliative care had a significantly longer median survival at 23.9 months compared to 11.4 months in the usual care group. 14 Regardless, given the retrospective nature of our study, our results should be formally validated in a controlled prospective study.
The improved survival in QC compared to the EC and BC models (Figure 1(h)) could be attributed to more patients seen in earlier disease stages rather than the palliative delivery model itself. 91% of QC patients were in GAP stage I and II, resulting in an incomplete control of the confounding effect of GAP stage in the multivariable Cox models. This is a reflection of local referral patterns in the absence of community respirology clinics in that area. Interestingly, of all the variables analyzed in our model, palliative care was the strongest predictor of survival. This suggests that the survival benefit of palliative care stands, regardless of the care model used. Subgroup analysis of those who received palliative care at the first visit (with more advanced disease) also demonstrated a similar benefit.
Immortal time bias, which can occur when participants do not live long enough to receive an intervention, is important to consider in survival studies when a therapeutic intervention is evaluated. It is unlikely that immortal time bias played a significant role in our results because in the EC and BC models palliative care was initiated in most patients at the first visit, and in the QC model we defined the date of referral to palliative care as time zero rather than the date patients were actually seen by palliative care. We also attempted to mitigate the impact of immortal time bias with a subgroup analysis of patients who received palliative care at the first visit compared to those who did not receive palliative care at all. Although the benefit to survival was no longer statistically significant due to the reduced sample size, the magnitude of the palliative care benefit persisted.
The mechanisms by which palliative care may influence survival are unclear. Educating patients regarding illness trajectory and assessing health beliefs and preferences to promote shared decision-making can lead to more realistic treatment goals and improved symptom management. Intentional and timely management of dyspnea and cough, independent predictors of survival in IPF, may improve survival.15-18 Palliative care models in oncology highlight how integrated palliative care can achieve systematic symptom screening for timely care and consolidation of care teams for improved continuity and efficiency.
19
Patients with IPF have similarly complex care needs and desire comprehensive care in 1 location. A palliative referral-based model does not address this need and may lead to inequitable access, as demonstrated by our study, and fragmented care.
20
On the other hand, clinics may not have all the necessary resources like allied health services to meet patient needs, and clinicians may not have the training to deliver primary palliative approach, and thus it is imperative to integrate community resources and other expertise to help bridge personnel limitations. This not only improves access, but also promotes disease self-management in the community.21,22 Ideally, a majority of a patient’s care needs would be addressed within the framework of the interstitial lung disease clinic, but there is a role for specialist referral depending on the limitations of each individual program. Our proposed palliative care model is summarized in Figure 2. Model for integrated palliative care in IPF. ILD = interstitial lung disease, PC = palliative care, mMRC = Modified Medical Research Council Dyspnea Scale, EDI = Edmonton Dyspnea Inventory,
29
QOL = quality of life, 6MWT = 6-minute walk test, ACP = advance care planning, EOL = end of life. 1In select cases only, eg, refractory symptoms, hospice referral, challenging advance care planning discussions, etc.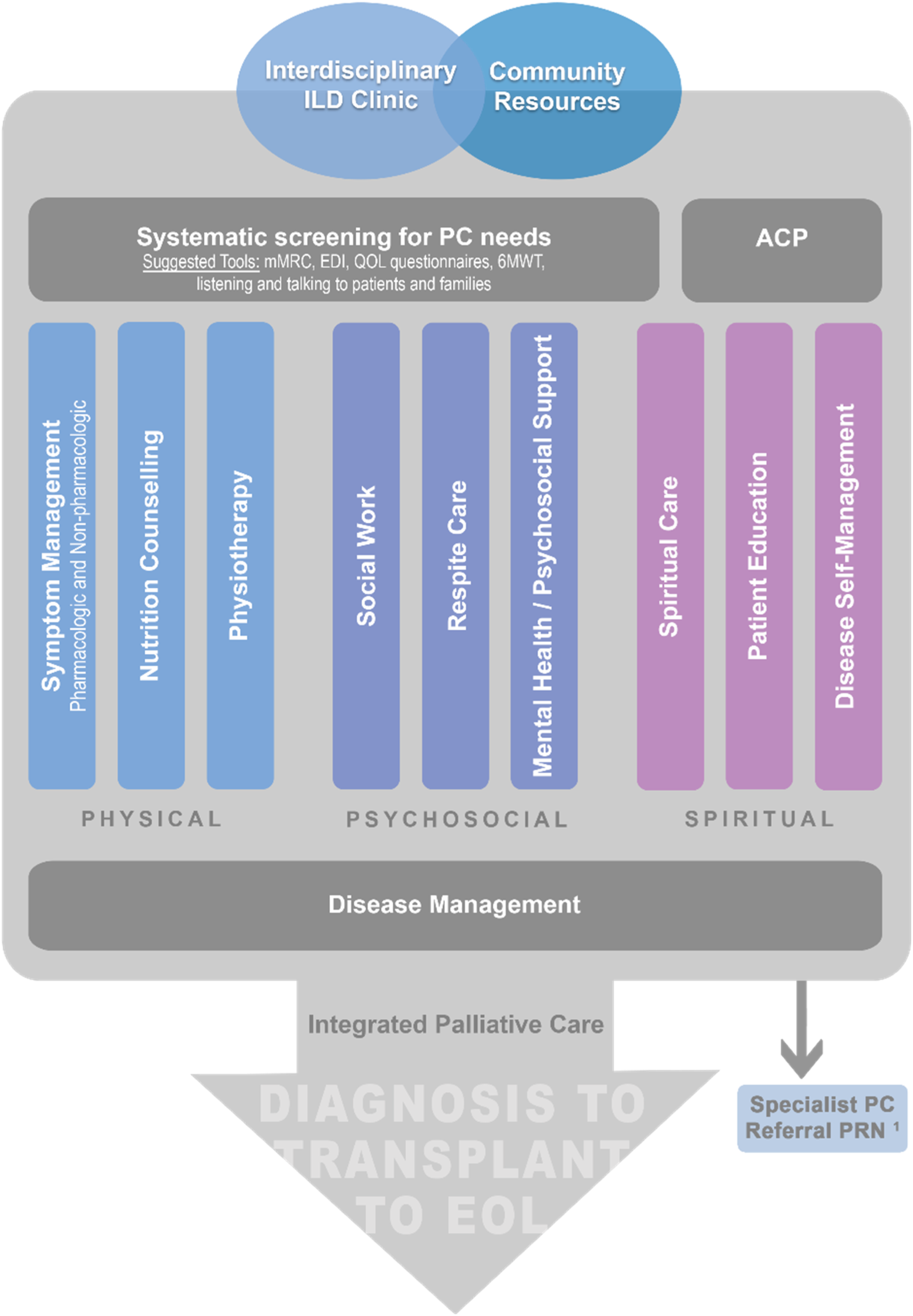
Patients who received opioids had worse survival after adjusting for covariates (Figure 1(i)). This is not in keeping with previous work by Bajwah and colleagues showing no increase in mortality or hospitalizations in patients on opioids. 23 This discrepancy may in part be because their study included non-idiopathic pulmonary fibrosis patients, and many may have been on opioids for reasons other than dyspnea. All patients in our study were prescribed opioids for dyspnea relief and likely represent a subset of patients with higher mortality risk by virtue of having more dyspnea. 15 While it is important to be cognizant of adverse effects, discomfort with opioid prescribing is a major barrier to adequate symptom management in IPF.
Multivariable Survival Analysis for all Study Subjects.
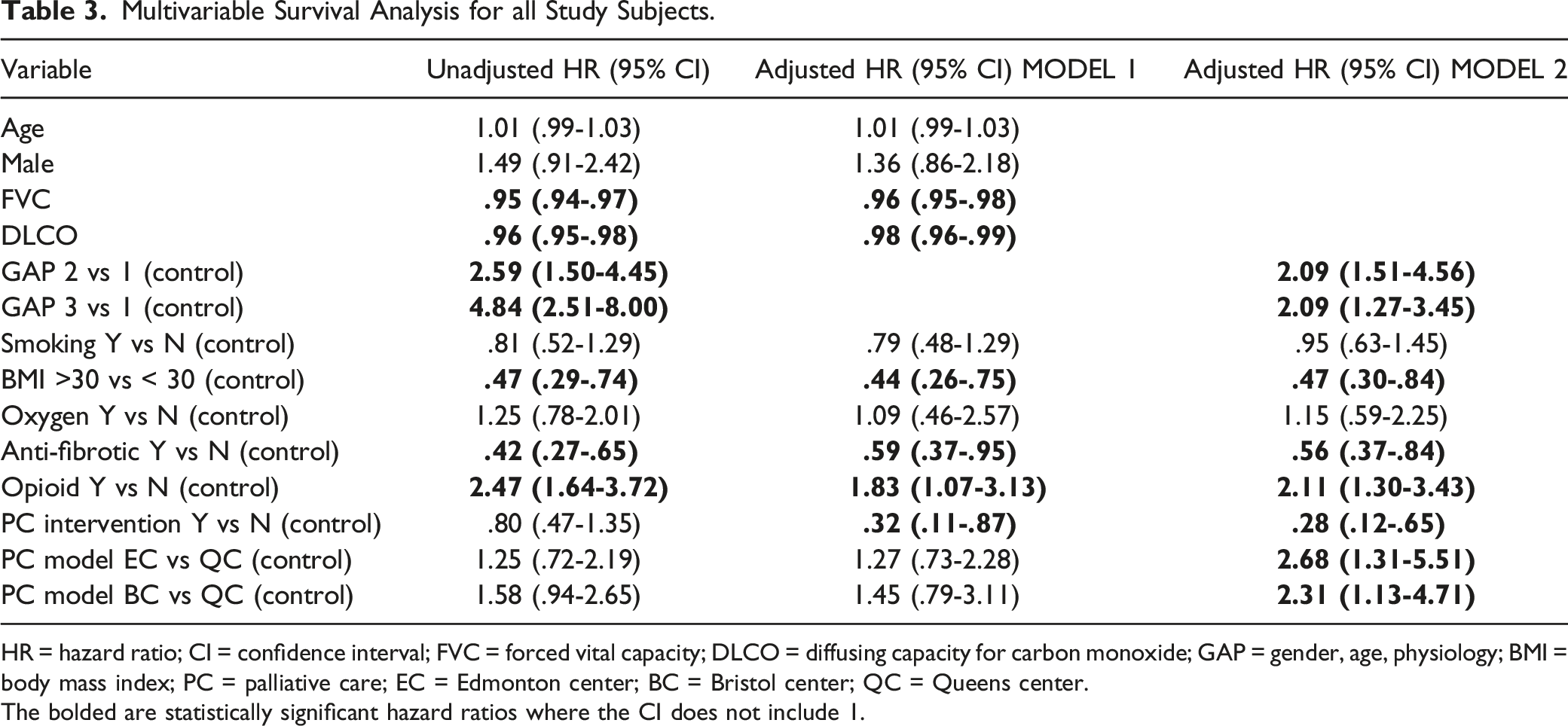
HR = hazard ratio; CI = confidence interval; FVC = forced vital capacity; DLCO = diffusing capacity for carbon monoxide; GAP = gender, age, physiology; BMI = body mass index; PC = palliative care; EC = Edmonton center; BC = Bristol center; QC = Queens center.
The bolded are statistically significant hazard ratios where the CI does not include 1.
Strengths and Limitations
The biggest strength of our study is utilization of data from a real-world setting with varying resources, clinic structure, and palliative care models. We acknowledge and adjust for known confounders including antifibrotics, GAP stage, and immortal time bias using statistical methods, and still demonstrate survival benefit of palliative care. In terms of the limitations of our study, a retrospective design has inherent biases from data not being collected specifically to address our question. The intervention was not uniform across study groups, which limits the assessment of the best method to apply palliative care, but this is also a strength as it demonstrates that real-world clinics are likely to have an impact on survival as we showed, despite different palliative care models. Future prospective studies utilizing a standardized model will hopefully shed more light on the best palliative care models for IPF.
We were not able to randomize our patients due to the retrospective nature of our study, but it is also important to note that randomization by disease severity alone may not ensure that groups are equivalent. The palliative care needs of individual patients are idiosyncratic and impacted by varying degrees of physical, psychosocial, and spiritual suffering and thus do not necessarily correlate with easily measured indices of disease severity. In addition, heterogeneity in time of onset and total exposure time to palliative interventions is inherent as shown in our study. Thus, our work highlights the potential challenges with conducting a controlled palliative care study in this population.
Implications for Future Research and Clinical Practice
Patients with IPF suffer from a high burden of symptoms and have complex care needs which escalate as they approach end-of-life. IPF registry studies have consistently observed GAP stage to correlate strongly with mortality.15,24-26 Many GAP stage II and III patients continue to experience large drops in FVC and DLCO compared to GAP stage I patients despite antifibrotic use. 27 Thus, the implication that a survival benefit from palliative care may complement that of antifibrotic therapy regardless of GAP stage is clinically relevant, and, if confirmed, may lead to a paradigm shift in the practice of pulmonologists. In view of the benefits that have been demonstrated with antifibrotics in progressive pulmonary fibrosis, it would be interesting to see if the benefits of palliative care can be replicated in this group of patients.1,28 Well-designed prospective trials are needed to further clarify the true impact of palliative care and best care models in pulmonary fibrosis. We have laid the foundation for such trials by demonstrating a signal of survival benefit and highlighting the challenges in trial design for palliative care intervention.
Supplemental Material
Supplemental Material - The Impact of Integrated Palliative Care on Survival in Idiopathic Pulmonary Fibrosis: A Retrospective Multicenter Comparison
Supplemental Material for The Impact of Integrated Palliative Care on Survival in Idiopathic Pulmonary Fibrosis: A Retrospective Multicenter Comparison by Jenny Lu-Song, MD, FRCPC, Jeffrey A. Bakal, PhD, PStat, Sarah Younus, BSc, MPH, Onofre Moran-Mendoza, MD, PhD, Ingrid Harle, MD, CCFP, Michelle Morales, RN, Naomi Rippon, RN, Shaney L. Barratt, MD, Huzaifa Adamali, MD, and Meena Kalluri, MD, FCCP in American Journal of Hospice and Palliative Medicine®
Footnotes
Acknowledgments
We acknowledge Drs. Sharina Aldhaheri and Ghadah Alrehaili for their contributions to data collection for this study. Study data were collected and managed using REDCap 1 electronic data capture tools hosted.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Alberta Respiratory Center, grant number [SC ZJ169] and Women and Children’s Health Research Institute at the University of Alberta.
Ethical Approval
This study was approved by the Health Research Ethics Boards in Alberta (REB Pro00082981); Ontario (HSREB 6025437); and Bristol, UK (IRAS 254044).
Contributorship
Data curation S.Y., N.R., H.A.; Formal analysis J.L., J.A.B., M.K.; Writing-original draft J.L.; Figures J.L., J.A.B.; Methodology I.H., O.M.-M., S.L.B., H.A., M.K.; Conceptualization M.K.; Resources M.K.; Supervision M.K. All authors contributed to drafting, revising, and finalizing the manuscript. All authors have read and agreed to the published version of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
Note
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
