Abstract
Background
Assessing and managing hospice patients’ pain is a common source of anxiety among hospice family caregivers (HFCGs), especially caregivers in rural communities who face special challenges including distance, limited access, and concerns about opioid misuse.
Objective
To pilot test Ready2Care, a pain management education intervention for rural HFCGs. We sought to determine whether there was a signal of benefit for clinically-relevant outcomes and to identify contextual factors pertinent to conducting a future randomized clinical trial of Ready2Care.
Methods
We conducted a multi-method, single-arm study, enabling completion of paired t-tests comparing pre- and post-intervention measures of caregiver anxiety, pain management self-efficacy, barriers to pain management, and reports of patient pain intensity and corresponding patient and caregiver distress. We concurrently conducted an embedded implementation evaluation via calculation of descriptive statistics (recruitment and retention data) and directed content analysis of brief caregiver interviews.
Results
Twenty-seven (n = 27) HFCGs participated; 15 completed the study. Among completers, significant improvement was observed in patient pain intensity (average 1.4 points decrease on 0-10 scale) and in overall pain experience. No statistically significant changes were detected in caregiver anxiety, barriers to pain management, or pain management self-efficacy. Facilitators to successful conduct of a future clinical trial included high acceptability of Ready2Care, driven by its perceived clarity and relevance to caregivers’ concerns. Barriers included lower-than-anticipated accrual and an attrition rate of nearly 44%.
Conclusion
A multisite clinical trial of Ready2Care is warranted; however, its success may require more effective recruitment and retention strategies for rural caregiver participants.
Introduction
Each year, millions of Americans with serious and life-limiting illnesses receive hospice services intended to promote comfort at the end of life. 1 In the United States, most hospice patients receive care in the community 1 with family members and friends providing the majority of their care, often with little training, preparation, or support. 2 Anxiety among these hospice family caregivers (HFCGs) is widespread,3,4 with clinically significant prevalence 6 times greater than that of the general population.5–7 A common source of anxiety among HFCGs is the challenge of assessing and managing hospice patients’ pain. 8
HFCGs frequently express discomfort administering pain medications, fearing under- or over-medicating patients.2,9,10 They struggle to assess pain 11 and worry about medication side effects, such as confusion, sedation, and constipation.2,9,10 Many report challenges communicating with healthcare providers about pain. 12 HFCGs who witness uncontrolled pain often feel highly distressed 13 and are at increased risk for poor long-term outcomes, such as social role impairment, decreased energy, negative perceptions of general health, and major depressive disorder. 14
HFCGs managing pain in rural communities face unique challenges. 15 Their homes are often geographically distant from friends, family members, and hospice agencies, limiting access to practical, informational, and emotional support related to pain management.15–17 At times, rural cultures emphasize self-reliance and toughness, potentially decreasing the likelihood that rural HFCGs will reach out to others for pain management assistance, further limiting their support. 18 Furthermore, HFCGs living in rural communities typically have limited access to specialty-trained palliative care professionals, community services (e.g., 24-hour pharmacies), and personal care providers. In addition, social concerns about pain medications (e.g., fear of addiction, stigma associated with use) are particularly pronounced in rural communities, which have been disproportionately affected by opioid misuse.19,20 Taken together, these realities underscore the pressing need for evidence-based interventions to support rural HFCGs’ pain management efforts.
Ready2Care is a remotely-delivered pain management education intervention for rural HFCGs that is adapted from Cagle and colleagues’ EMPOWER (Effective Management of Pain: Overcoming Worries to Enable Relief) intervention. 21 EMPOWER has been shown to increase HFCGs’ knowledge about pain management, reduce HFCGs’ concerns about pain and pain medications, and lower patient pain. 21 Ready2Care is delivered in 3 phases: (1) an initial 30 minute telephone screening and provision of tailored educational content based on participant concerns or misperceptions about pain management identified during the screening, (2) mail and/or email delivery of written educational materials (ie, the Ready2Care pain education brochure), which participants are asked to review, and (3) a 5-10 minute follow-up call 1 week later to confirm receipt of the written materials, address any new or remaining questions about Ready2Care content, and remind participants of ongoing pain management support from their hospice interdisciplinary team. For research purposes, a third 30-minute phone call 1 week later is used to explore participant response to the intervention and any remaining pain management concerns.
The Ready2Care educational brochure features common concerns about pain management in hospice care (addiction, tolerance, medication side effects, denying pain, perception of drug-seeking behavior, fear of overmedicating care recipient, not wanting to bother others, and concern treatments will not work) and evidence-based information to address each concern, including education about non-medication adjuvant treatments. The brochure also features information about how to manage medications, including how to stay informed, organized, and how to keep track of medication administration; how to use reminders; when and how to communicate with the hospice team; and hospice team contact information. To support health literacy accessibility, the brochure’s language reads at a 7.4 Flesch-Kinkaid grade level, indicating strong readability, and is presented in easy-to-read font. For the interactive component of Ready2Care, nurse interventionists verbally share scripted educational messages in response to identified pain management barriers (eg, addiction concerns, misperceptions about medication side effects), discuss HFCGs’ questions and concerns about these educational messages in the context of their care recipient’s personal needs, and coach HFCGs to discuss their concerns and questions with their interdisciplinary hospice team. In conversation, nurse interventionists show empathy and provide encouragement and affirmation.
Study Aim
Although randomized clinical trials (RCTs) are recognized as the gold standard for generating evidence on the effectiveness of interventions in hospice care, 22 they often fail to produce results that can be confidently applied in real-world clinical decision making. 23 To improve the eventual informativeness of clinical trials, Taylor and Kowalkowski 23 recommend conducting pre-RCT pilot studies guided by implementation science, enabling identification of facilitators and barriers to the conduct of future RCTs in addition to outcomes appropriate for more definitive investigation in future research. Consistent with this recommendation, our team of researchers and hospice clinicians conducted a pilot study of Ready2Care, a pain management education intervention for rural HFCGs. In doing so, we sought to determine whether there was a signal of benefit of Ready2Care regarding clinically relevant outcomes and to identify contextual factors pertinent to the conduct of a future randomized clinical trial of the intervention.
Methods
We conducted a multi-method, single-arm, pre-post pilot study with an embedded implementation evaluation informed by the Consolidated Framework for Implementation Research (CFIR), 24 Proctor’s conceptual model of implementation research, 25 and the checklist of goals for pilot studies of psychosocial interventions in palliative care created in collaboration with the Palliative Care Research Cooperative group. 26 CFIR is an evidence-based implementation framework designed to help researchers identify determinants (i.e., barriers or facilitators) of implementation outcomes, including both clinical outcomes that reflect the impact of an intervention (also referred to as “innovation outcomes”) and outcomes that measure the extent to which an intervention is successfully implemented in clinical settings. 24 Proctor’s model of implementation research links intervention and implementation strategies with 3 types of outcomes, including implementation outcomes (e.g., feasibility, acceptability), service outcomes (e.g., effectiveness, person-centeredness), and client outcomes (e.g., satisfaction, symptomatology). 25 Finally, the checklist of goals for pilot studies of psychosocial interventions in palliative care outlines specific objectives a pilot study within the palliative care domain should seek to achieve, such as testing recruitment methods and estimating participant retention. 26
Participants and Setting
HFCGs were eligible to participate in the study if they met the following criteria: were (1) age 18 or older, (2) an informal (i.e., unpaid) family caregiver receiving services from 1 of 2 rural hospice sites with which our team had an established research partnership, (3) able to speak and read English, and (4) caring for a hospice patient with a Palliative Performance Scale (PPS) score of 30% or higher (to avoid attempting to recruit family caregivers of patients who were actively dying and to allow sufficient time to deliver the Ready2Care intervention prior to hospice patient death). As is customary in hospice, we broadly defined “family caregiver” to include all individuals identified as such in the medical record; legal or biological relationships were not required. For this pilot study, HFCGs of patients with any diagnosis were included.
Data Collection
Eligible participants were recruited through partnership with a large hospice agency that included 2 sites serving predominantly rural areas of 1 Midwestern state. This agency authorized the research team to access, use, and disclose personal health information solely for the purposes of the study through signing a HIPAA authorization letter. Research staff generated a list of new hospice admissions and located the contact information for individuals listed as newly admitted patients’ family caregiver(s) of record. After confirming that each hospice patient had a PPS score of 30% or greater 27 and waiting a minimum of 4 days following the hospice admission to ensure regular hospice staff had an opportunity to complete required initial visits and assessments, research staff contacted HFCGs by telephone, briefly explained the study, gauged caregivers’ interest in learning more about the study, provided additional detail for interested parties, and invited potential participants to provide verbal consent to enroll in the study. Recruitment was paused for 1 month during the winter holidays.
Recruitment benchmarks for HFCGs in rural settings were not available due to an absence of published studies in the field. However, our team set recruitment goals based on data obtained from the rural site of a recently concluded HFCG study 21 that used a recruitment process similar to Ready2Care. In that study, research staff were unable to contact 9.5% of 262 eligible referrals from a rural hospice site. Of the remaining 237 HFCGs researchers were able to contact, 28.6% consented, with non-consenting caregivers reporting being “uninterested” and “too busy” as major reasons for declining participation. Based on these benchmark data and a prediction that we would identify an estimated 200 eligible HFCG referrals over the duration of the study, our goal was to consent 50 HFCGs into the Ready2Care pilot. Finding no published research on rural HFCG interventions of comparable length to Ready2Care, we opted not to establish a priori retention goals.
Design and Measures
In this pilot study, 2 trained researchers delivered the intervention. Both are registered nurses; 1 has a PhD in Nursing and the other is a candidate for a PhD in Nursing Science. During the first of 3 phone calls with study interventionists, HFCGs provided basic demographic data and completed a baseline survey that included a series of items and standardized measures assessing HFCGs’ concerns about pain and pain management (measured with the EMPOWER barriers to pain management scale) 28 ; self-efficacy in pain management (measured with selected items from the Modified Caregiver Self-Efficacy in Pain Management scale, CSEPMS) 29 ; reports of patient pain intensity and corresponding patient and caregiver distress (measured with the Family Pain Questionnaire 3-item experience subscale 30 and a single-item caregiver proxy pain rating using a scale from 0 [no pain] to 10 [worst possible pain]); and caregiver anxiety (measured with the Generalized Anxiety Disorder [GAD] 2-item scale). 31 After baseline data were provided, interventionists reviewed participants’ responses to the EMPOWER scale, verbally relayed scripted educational messages in response to any identified pain management barriers (e.g., concerns or misperceptions), discussed HFCGs’ questions and concerns about these educational messages, and coached participants to address concerns beyond the scope of the intervention with their hospice interdisciplinary team. Following this initial call, interventionists sent a copy of the Ready2Care pain education brochure to HFCGs via email and/or U.S. mail, according to HFCG preference.
During a second call approximately 1 week later, interventionists confirmed that HFCGs had received and reviewed the Ready2Care pain education brochure and repeated the 3-item Family Pain Questionnaire experience subscale, which assessed caregiver ratings of patient pain intensity and corresponding patient and caregiver distress.
During the third and final call, conducted about 1 week after the second call, interventionists again assessed HFCGs’ concerns about pain and pain management, pain management self-efficacy, reports of patient pain intensity and corresponding patient and caregiver distress, and caregiver anxiety. In addition, consistent with CFIR guidelines, 24 data on selected contextual factors as potential implementation determinants were collected. This included HFCGs’ access to technology and health literacy (measured with the BRIEF Health Literacy screening tool). 32 In addition, HFCGs’ perceptions of the intervention were explored via investigator-initiated items and brief qualitative interviews, which were audio-recorded with HFCGs’ consent and later transcribed by a third-party service. At the end of study participation, participants were thanked for their time with up to $50 (by check), depending on how many calls they completed.
The Washington University in St Louis Institutional Review Board approved the study (#202103035). All participants provided verbal consent prior to study enrollment. Participant data were securely stored on protected REDCap servers to maintain strict privacy standards.
Analysis
Descriptive statistics were used to measure recruitment (absolute number of referrals received, number of referrals received as a percentage of total agency admissions, percentage of referred caregivers who enrolled in the study), retention (percentage of enrolled participants retained in the study through days 7 and 14), and all study measures to provide statistical parameters helpful in planning future research. In addition, comparisons between pre- and post-Ready2Care caregiver-reported patient pain ratings, anxiety (2-item GAD) scores, EMPOWER barriers to pain management scores, CSEPMS individual item scores, and Family Pain Questionnaire subscale scores were made using paired t-tests.
A directed content analysis 33 using NVivo v. 12 was applied to participants’ responses to open-ended questions about perceptions of the Ready2Care intervention in the context of researcher observations and field notes. An initial list of predetermined codes, based on the study’s guiding conceptual frameworks and published research on HFCGs’ experiences with pain management,2,34 was applied and updated as needed to accommodate newly identified codes. Two researchers (LTS, KTW) independently coded the interviews, then met to compare coding and refine the code list via discussion and, as needed, resolution of conflicts regarding initial coding decisions. The first author then combined the codes to develop themes, which the research team then reviewed.
Results
Participant Characteristics
Summary of Participant Characteristics.
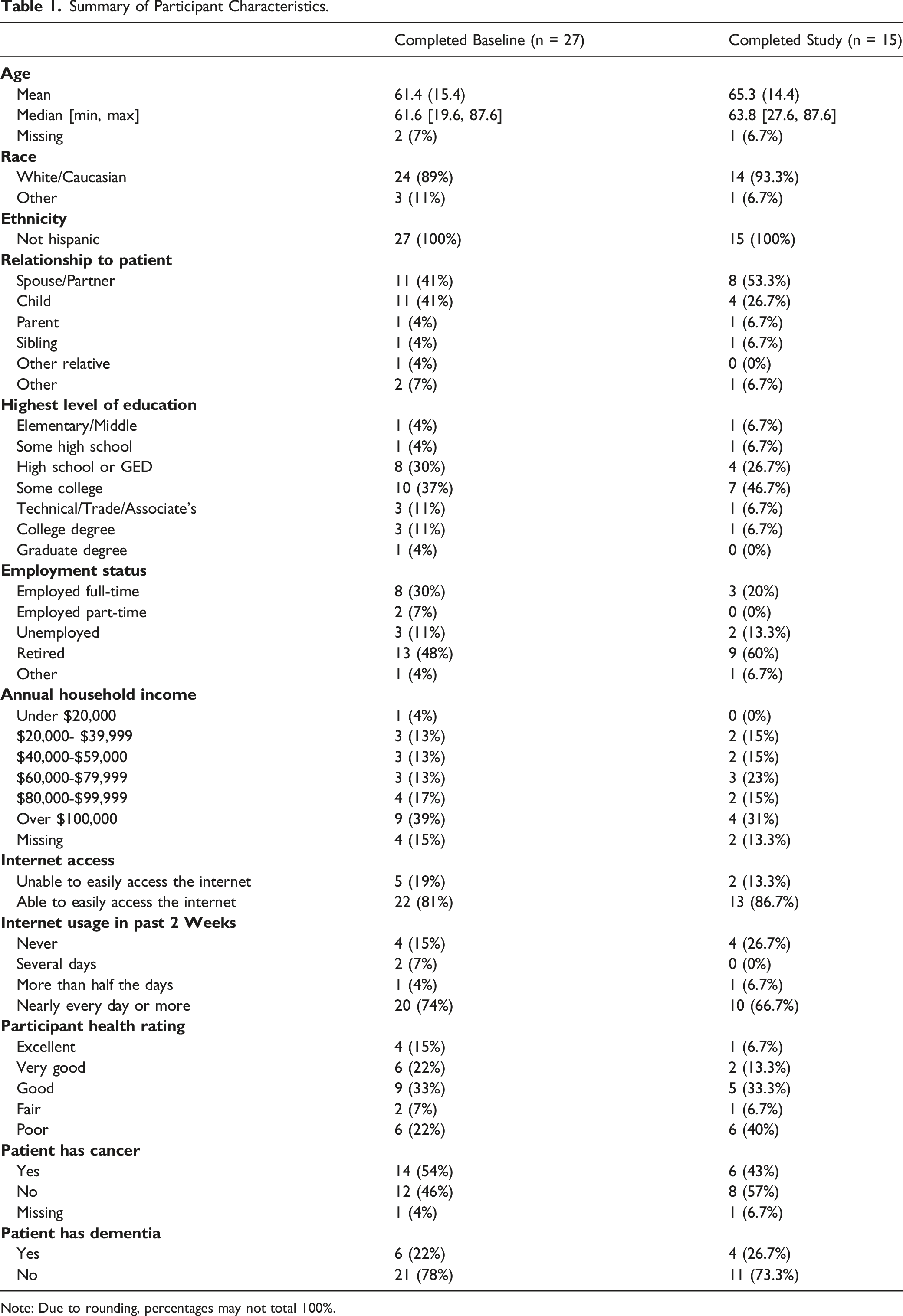
Note: Due to rounding, percentages may not total 100%.
Acceptability
Summary of Ready2Care Quality Ratings.

Insights About Implementation of the Ready2Care Intervention Among Rural Hospice Family Caregivers.

Table 3 also describes Ready2Care components caregivers recommended changing. A minority of caregivers recommended Ready2Care personalize content to a patient’s specific illness and symptoms (e.g., how to administer pain medication to a person with continuous hiccups). Caregivers of persons with dementia or cognitive impairment strongly valued the intervention, recommending researchers adapt Ready2Care to their specific informational needs, given limited understanding of non-verbal pain behaviors and assessing pain in this population. Similarly, a minority of caregivers reported wanting the intervention to include more detail (e.g., additional and/or more specific strategies to address constipation). Finally, a minority of caregivers recommended small changes to the brochure itself. For example, 1 caregiver recommended researchers revise the brochure language such that pain “concerns” are instead presented as recommended actions caregivers can take, and that the hospice telephone number be included on all pages for easy access.
Feasibility
Feasibility results were mixed. The feasibility of delivering Ready2Care to participants once enrolled in the study was supported, as we were able to conduct initial screenings and discuss tailored educational messages with 27 of the 30 HFCGs who consented. However, we identified numerous challenges associated with the feasibility of recruiting and retaining a sufficiently large sample of rural HFCGs to support future, large-scale testing of the intervention. Ultimately, we fell short of our recruitment goal of 50 HFCGs, and only half of the HFCGs who consented to study participation completed the full 14-day study protocol. Figure 1 presents the flow diagram of participant recruitment, enrollment, and study completion. Ready2Care family caregiver participant recruitment and retention.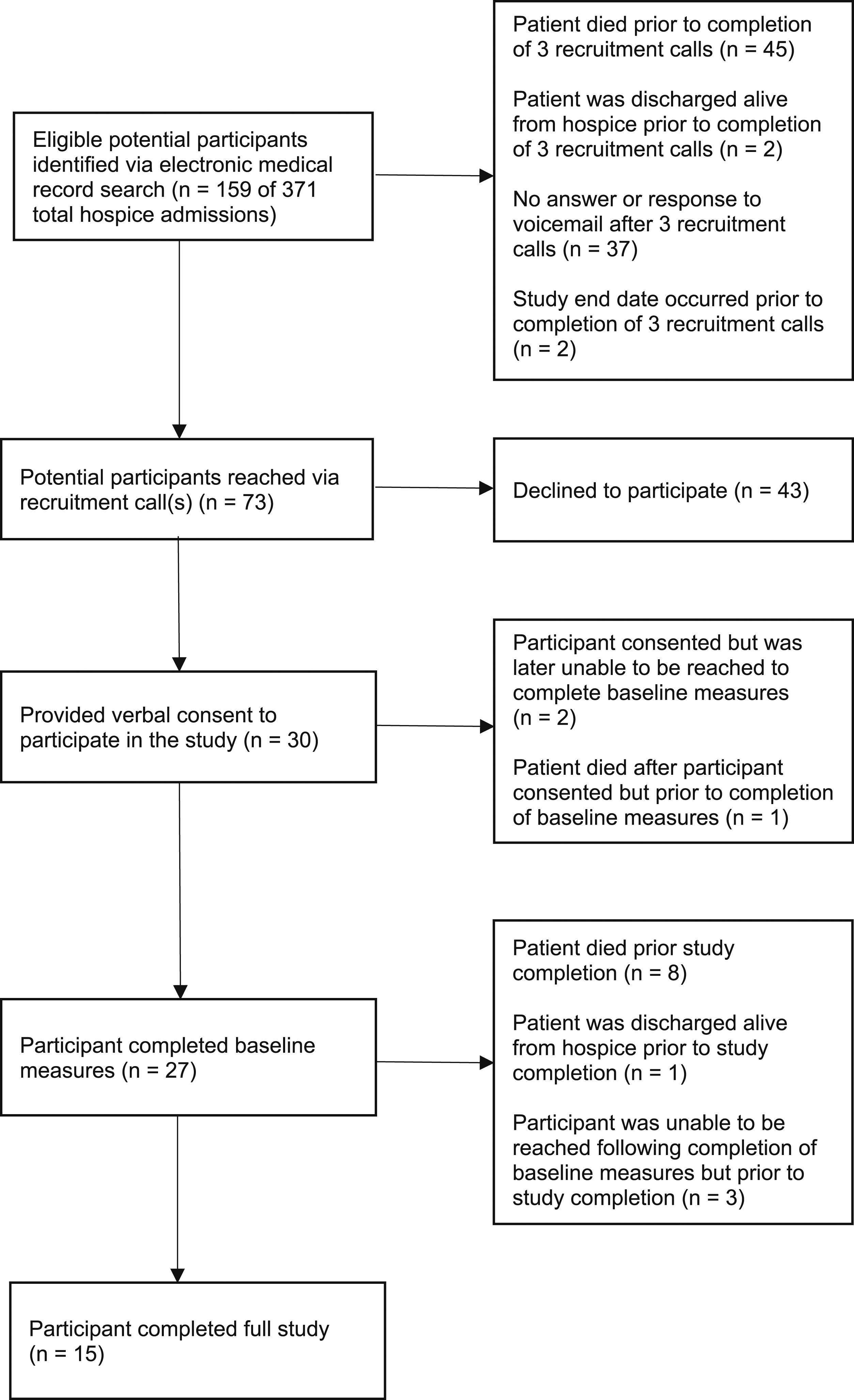
Recruitment
From September 7, 2021 to August 1, 2022, we identified 159 eligible referrals, which represented 42.9% of agency hospice admissions during this period. Of these referrals, 37 caregivers did not answer any of the interventionists’ 3 phone calls or respond to interventionist voicemails (23.3% of referrals), with 45 hospice patients dying prior to completion of the 3 recruitment calls (28.3%). Thirty participants provided verbal consent to participate in the study (18.9%), but only 27 caregivers completed the baseline assessment (17.0% of referrals).
Retention
Of 27 caregivers who completed baseline interviews, 19 were retained through day 7 (11.9% of referrals, 70.4% of participants who completed baseline interviews) and 15 caregivers completed the study (9.4% of referrals, 55.6% of participants who completed baseline interviews). Of the 19 caregivers who completed day 7, 14 (73.7%) had reviewed the Ready2Care brochure and 5 (26.3%) had not. Reasons caregivers gave for having not yet accessed the brochure by day 7 included lack of time or lack of access to mail.
Ready2 Care Outcomes
Full Study Participant Variables at Baseline and Post-Intervention for Ready2Care.
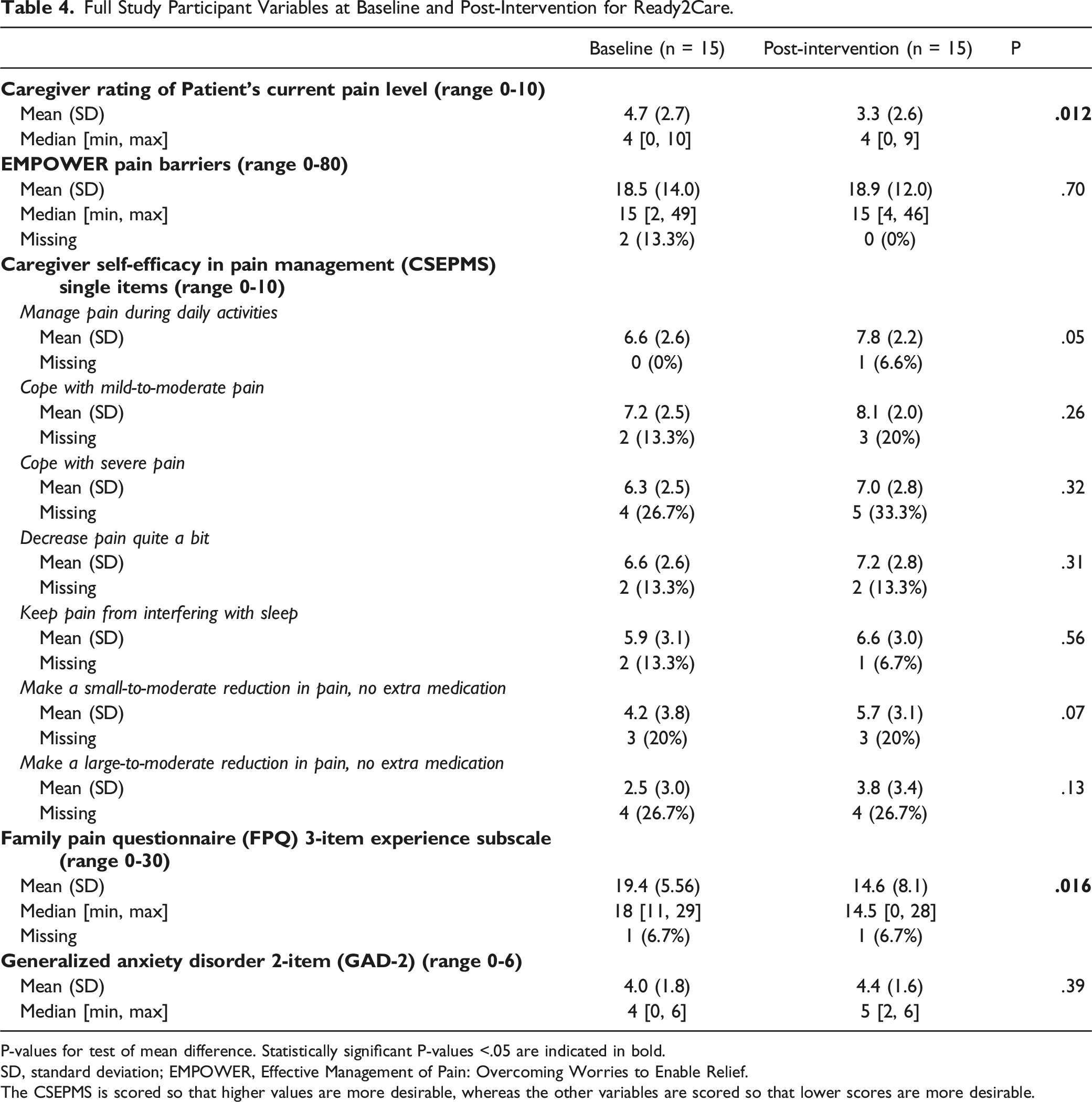
P-values for test of mean difference. Statistically significant P-values <.05 are indicated in bold.
SD, standard deviation; EMPOWER, Effective Management of Pain: Overcoming Worries to Enable Relief.
The CSEPMS is scored so that higher values are more desirable, whereas the other variables are scored so that lower scores are more desirable.
Caregiver-Reported Outcomes of the Ready2Care Pain Education Intervention.

Discussion
In this pilot study of an educational pain management intervention for rural hospice family caregivers, we found Ready2Care to be highly acceptable and feasible to implement clinically. Quantitative study results underscored the clinical promise of Ready2Care as a strategy to improve pain and related distress, while qualitative findings illustrated Ready2Care’s potential to improve pain management understanding and confidence among caregivers, facilitate action to relive pain among caregivers, improve pain-related communication across caregivers, and increase collaboration with the hospice team.
Research-oriented feasibility measures, however, highlighted pronounced difficulties recruiting and retaining HFCGs from rural hospice programs for research purposes. Our findings are consistent with other studies that indicate inherent challenges with recruitment and attrition in hospice research, typically due to the limited interest or availability of caregivers, short hospice enrollment periods, and patient death.35,36 More research is needed to understand optimal recruitment and retention methods for HFCG studies. Some HFCG researchers recruit participants after hospice agencies have pre-screened participants and presented intervention programs to participants during nurse or social worker home visits.35,37 This pre-screening referral process is significantly more resource and time-intensive than our team’s recruitment strategy yet may result in higher consent rates. For example, 1 recent study of an intervention for HFCGs that used this recruitment method yielded 47 eligible referrals from 2 non-rural hospice sites during a 9-month period; 43% consented to participate, 38% started the intervention, and 21% completed the intervention (n = 10). 37
Study findings also shed light on numerous contextual factors relevant to the conduct of a future randomized clinical trial of Ready2Care. Providing care for an individual nearing the end of life is often burdensome and can be both physically and emotionally exhausting. 38 This may be particularly true for caregivers who are themselves in fair or poor health, which was the case for nearly half of the participants who completed the Ready2Care intervention in this study. Overwhelmed family caregivers may be disinclined to participate in research or, if they consent to participate, may have insufficient time to engage in research-related activities (e.g., reviewing the Ready2Care pain education brochure) outside of scheduled encounters with the research team. Future Ready2Care studies should be designed with the busy lives of family caregivers in mind. Recruitment and retention challenges should be anticipated and accounted for in planning efforts (e.g., conducting power analyses, deciding upon the number of sites to engage), and required activities between intervention sessions should be minimized if not eliminated entirely. Difficulties with recruitment of hospice family caregivers in research is well documented.35,36 Hospice enrollment typically occurs late in the illness trajectory, often within the last 2 weeks of life, limiting the time frame researchers can conduct studies. Out of respect for families, we waited 4 days after hospice enrollment before contacting caregivers, further reducing our window of opportunity to engage HFCGs. HFCGs provide difficult hands-on care at an emotionally stressful and demanding time, reducing participant receptivity and challenging informed consent.35,36 Recruitment of hospice caregivers in rural settings is particularly challenging given lower adoption of hospice in rural areas 39 and the typically small size of rural hospice programs.
Early in the research process, we realized our recruitment goals were too high due to fewer than expected eligible referrals and a significantly higher non-response rate (23.3% non-response in our study compared to 9.5% in another recently completed clinical trial including similar participants). We attribute this difference to the difficulty of caregiving and, possibly, the phone numbers from which interventionists called participants, which did not match local area codes and may have been dismissed as suspected telemarketing. Future research will require more hospice agency partnerships and rural sites and use of phones with local numbers to decrease automatic refusal. In addition, the consent rate (18.9%) of Ready2Care was lower than the study we used to set recruitment goals (28.6%), which may be due to differences in the intervention requirements and perceived benefits. HFCG studies that use hospice staff to recruit participants demonstrate higher conversion rates 35 but also increase adoption barriers.
Difficulties retaining HFCG participants due to hospice patient death further compounded sample size issues.35,36 In Ready2Care, 90% of participants who verbally consented to participate completed the baseline interview, 63.3% were retained through day 7, and 50% completed the study. Based on other studies, hospice patient death and lack of time were likely contributing factors to attrition.35,36 Future research should account for high attrition and identify ways to increase retention among rural participants.
More specific findings regarding the acceptability of Ready2Care focused on its remote delivery. In both quantitative and qualitative reports, caregivers expressed strong positive sentiments and appreciation for the intervention as delivered via telephone. This finding is consistent with a recent systematic review that found telehealth interventions, specifically those conducted over the telephone like Ready2Care, to be effective at developing caregiving skills, improving psychological functioning among family caregivers of persons with chronic illnesses, and increasing collaboration. 40 As found in previous research, the experience of the interventionists, 2 registered nurses, and use of a structured tool to guide care practice positively contributed to discussing pain management concerns and best practices.41,42
Study participants also emphasized their appreciation for the socio-emotional support provided by Ready2Care interventionists, whom they described as caring and empathic, with some expressing sentiments similar to the 1 shared by a HFCG who explained that Ready2Care was “very good” because they had not previously “had anybody [they] could really talk with that seems to [understand].” Prior research has found loneliness or “a sense of profound isolation” to be an important contextual factor in the end-of-life caregiving experience, 43 suggesting that future iterations of Ready2Care will be best received and perhaps most likely to be effective if meaningful contact with interventionists is retained. That is not to suggest that additional technologies could not play a role in research with rural HFCGs. Web-based delivery of participant surveys, for example, appears to have a high likelihood of feasibility, as nearly 87% of study participants reported being able to easily access the internet. However, human contact in the delivery of the intervention itself appears to be an important feature to continue.
Participants’ expressed desires for personal interaction and feeling understood and their appreciation of Ready2Care’s support reflect a deep need for programs that address the social and emotional needs of HFCGs. Social support, which has multiple dimensions (emotional, appraisal/esteem, companionship, informational, and instructional 44 ) is a critical factor in improving caregiver mental and physical health and reducing caregiver burden.44–47 Unfortunately, as patients near the end-of-life, social support for caregivers from friends and family often decreases, 48 making support for HFCGs especially important. While it is unknown which social support dimensions Ready2Care addressed, expansion of social support features of Ready2Care may be warranted. In addition, clinicians should consider developing social support groups for HFCGs. Rural HFCGs unable to join in-person support groups due to distance or responsibilities may benefit from online support groups. One recent RCT found that hospice family caregivers who participated in a private online support group on Facebook experienced statistically significant decreases in depression compared with HFCGs in the usual care control group. 21
Study participants also indicated researchers should consider adapting Ready2Care to conditions commonly encountered in hospice, such as dementia and other illnesses that may result in cognitive impairment, as pain may be under-controlled in these populations due to caregiver difficulties assessing and managing pain among patients who are non-verbal or who experience other communication challenges, 49 highlighting the unique disease-specific contexts in which hospice caregiving takes place. Importantly, however, study findings suggest that any future changes to the Ready2Care pain education brochure should be made in such a way that its clarity and ease of understanding are retained, particularly given that the average of participants’ health literacy scores fell between cut-offs for “marginal” and “adequate” levels, and numerous participants commented positively about the lower literacy level at which the Ready2Care pain education brochure is currently written.
Although HFCGs reported numerous positive outcomes and actions they attributed to participation in Ready2Care, some variables (e.g., caregiver anxiety, caregiver pain management self-efficacy) did not improve in a statistically detectable manner. While this finding may be due to small sample size, interventionist field notes suggest that for some caregivers, notably those who did not initially think their family member experienced pain, concerns about pain and their management of pain may have increased as the patient began experiencing new pain or as HFCGs became more aware of pain through improved assessment promoted by Ready2Care. Similarly, evidence suggests that family caregivers experience greater anxiety as a patient approaches death, 50 which may explain why caregiver anxiety did not improve post-intervention. Additional research involving larger samples and more rigorous study design features (e.g., participant randomization, blinding of outcome assessors) are needed to more fully capture the impact of Ready2Care and to better understand how it affects dimensions of HFCGs’ emotional experiences.
Conclusion
Ready2Care, a remotely-delivered adaptation of Cagle and colleagues’ educational pain management intervention, EMPOWER, was shown to be highly acceptable among HFCGs in rural settings and innovative in its effectiveness at improving hospice pain management outcomes. Future research should explore better recruitment and retention methods to improve feasibility and should consider adaptations to common hospice conditions, such as dementia, in which pain is often poorly understood and under-treated. A multisite clinical trial of Ready2Care is recommended.
Footnotes
Acknowledgments
We acknowledge the signficant contributions of Sheryl Zimmerman, PhD, Lauren W. Cohen, MA, Laura S. Porter, PhD, Laura C. Hanson, MD, MPH, and David Reed, PhD, who were involved in the development and testing the of EMPOWER intervention, on which Ready2Care was largely based. We also acknowledge Tina Nolte, BA, who was instrumental in recruitment for the Ready2Care pilot study. In addition, we sincerely thank our clinical hospice partners and the hospice family caregivers who participated in this research and without whom it would not have been possible.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hospice Foundation of the Ozarks and the Foundation for Barnes-Jewish Hospital. The content is solely the responsibility of the authors and does not necessarily represent the official views of the study’s funders. In addition, Dr Starr was supported by the Ruth L. Kirschstein National Research Service Award training program in Individualized Care for At Risk Older Adults at the University of Pennsylvania, National Institute of Nursing Research (T32NR009356). JoAnn Jabbari was supported by the Jonas Nurse Leaders Scholars Program.
