Abstract
Background: Opioids are known to induce delirium, but few studies have closely investigated differences in incidence of delirium among different opioids. Objectives: To determine whether there is a clinically significant difference in the incidence of delirium between oral opioids in previously opioid-naive patients. Methods: Subjects were 259 opioid-naive in-patients with cancer who were started on morphine sulfate, oxycodone hydrochloride, or tapentadol hydrochloride extended-release tablets at our hospital between August 1, 2014, and September 30, 2018. The incidence of delirium during the first week of treatment was compared between the drugs. Results: The incidence of delirium was 4.8% (n = 83) for morphine sulfate, 6.9% (n = 131) for oxycodone hydrochloride, and 6.7% (n = 45) for tapentadol hydrochloride. The incidence did not significantly differ between oxycodone hydrochloride (OR = .69, 95% CI = .20–2.30,
Background
Delirium is a major issue in palliative care and occurs in 10%–25% of hospitalized patients and 14%–40% of cancer patients. 1 Management and prevention of delirium is critical because of delirium negatively impacts the QOL of patients and their family members. Factors that contribute to delirium include age, smoking, alcohol abuse, comorbidities such as dementia, and medications. 2 Drug-induced delirium can be prevented by selecting drugs with the lowest risk of delirium. Drugs that induce delirium include opioids, benzodiazepines, steroids, H2 antagonists, anticonvulsants, antihistamines, and anticholinergics. Of these, opioids are the most likely to cause delirium. 1
When treating a patient with opioid-induced delirium, it is recommended to try opioid rotation3,4 and administration of an antipsychotic.5,6 In addition, the research indicates that non-pharmacologic approaches addressing factors such as hospital room lighting, undernutrition, and poor sleep may be beneficial in management of delirium. 7 However, few studies have compared the incidence of delirium in previously opioid-naive patients receiving opioids for cancer pain. In 2017, we investigated the incidence of delirium in patients receiving intravenous opioids for cancer pain and found that the incidence was significantly lower with intravenous fentanyl citrate compared with intravenous morphine hydrochloride. 8 In addition, Morita et al 3 found that opioid rotation to fentanyl significantly reduced the severity of delirium in cancer patients with morphine-induced delirium.
However, no study has yet investigated the incidence of delirium with oral opioids in opioid-naive patients. Fentanyl has low bioavailability of 31% (95% CI = .24–.33) when administered orally, 9 and thus an extended-release oral formulation has not been developed. However, the approach of using an oral formulation as the first opioid for opioid-naive patients still merits consideration as it may prevent delirium. Three first-line oral opioids are approved for use in Japan: morphine sulfate extended-release tablets, oxycodone hydrochloride extended-release tablets, and the relatively new option marketed from 2014, tapentadol extended-release tablets. Tapentadol acts on μ-opioid receptors and also inhibits reuptake of norepinephrine.10,11 These dual synergistic effects deliver equally potent analgesic effects compared with other strong opioids, while also having less severe gastrointestinal effects (e.g., constipation and nausea). 12 However, the degree to which tapentadol induces delirium is not known.
Accordingly, in this study, we aimed to compare the incidence of delirium between morphine sulfate extended-release tablets, oxycodone hydrochloride extended-release tablets, and tapentadol hydrochloride extended-release tablets in previously opioid-naive patients with cancer pain.
Methods
Patients
Subjects were 259 in-patients with cancer who started morphine sulfate extended-release tablets (M group), oxycodone hydrochloride extended-release tablets (O group), or tapentadol hydrochloride extended-release tablets (T group) at our hospital between August 1, 2014, and September 30, 2018. Only patients receiving opioids as medication for the first time were included; those with a history of intravenous or oral opioid use were excluded. Patients who had gastrointestinal obstruction that would make them unable to take oral medications, or who were discharged from the hospital within 1 week of starting opioid therapy were also excluded from the study. Patients who also received opioids for breakthrough pain were eligible for inclusion, but only if they received rapid-release oral morphine (if in the M group) or rapid-release oxycodone (if in the O group), and not any other drug. Rapid-release tapentadol has not been approved in Japan, and thus patients in the T group were eligible for inclusion if they received rapid-release oral morphine or rapid-release oxycodone for breakthrough pain, but not any other drug.
The attending physician selected which opioid to use in consideration of factors such as the nature and cause of the pain, liver and kidney function, and performance status (PS), with reference to clinical guidelines for cancer pain management published by the Japanese Society of Palliative Medicine, 13 European Society for Medical Oncology, 14 and National Comprehensive Cancer Network. 15
Data Analyzed
Researchers retrospectively reviewed electronic medical records written by physicians, nurses, or pharmacists during the first week of opioid therapy in each group, and classified patients who met at least 4 of 8 Intensive Care Delirium Screening Checklist (ICDSC) criteria as having developed delirium. 16
Data on dose, age, sex, PS, brain tumor/metastasis, disease of the central nervous system, dementia, past alcohol use, use of ropivacaine hydrochloride for epidural anesthesia, renal failure, and hepatic failure were analyzed as patient characteristics that could contribute to delirium. Renal impairment was defined as increased serum CRE of Grade 1 or higher and hepatic impairment as increased blood AST/ALT of Grade 1 or higher per the Common Terminology Criteria for Adverse Events (CTCAE) Ver. 5.0. Equivalent doses were calculated using the ratio morphine/oxycodone/tapentadol = 3:2:10.13,17,18
Use of concomitant drugs that can cause delirium, namely, benzodiazepines, anticholinergics, antidopaminergics, antihistamines, H2 antagonists, and steroids, within 1 week before and after starting opioid therapy was also determined, and rates of concomitant use were calculated for each group.
Statistical Analysis
The incidence of delirium was compared between groups using Fisher’s exact test. For patient characteristics, dose and age were compared using Student’s t-test, and all other factors were compared using Fisher’s exact test. Rates of concomitant use of other drugs that can cause delirium were compared using Fisher’s exact test. A significance level of 5% was used for all tests.
Ethical Considerations
This study was conducted in compliance with the Ethical Guidelines for Medical and Health Research Involving Human Subjects and with the approval of our hospital’s institutional review board (Approval No. 30-J153). Consent was obtained via an opt-out method using the hospital’s bulletin.
Results
Subjects
All 259 patients consented to participate in the study. There were 83 patients in the M group, 131 in the O group, and 45 in the T group.
Differences in Incidence of Delirium
As shown in Figure 1, the incidence of delirium was 4.8% in the M group, 6.9% in the O group, and 6.7% in the T group. The incidence of delirium did not differ significantly between the O group (OR = .69, 95% CI = .20–2.30, Incidence of Delirium with Each Oral Opioid, n (%).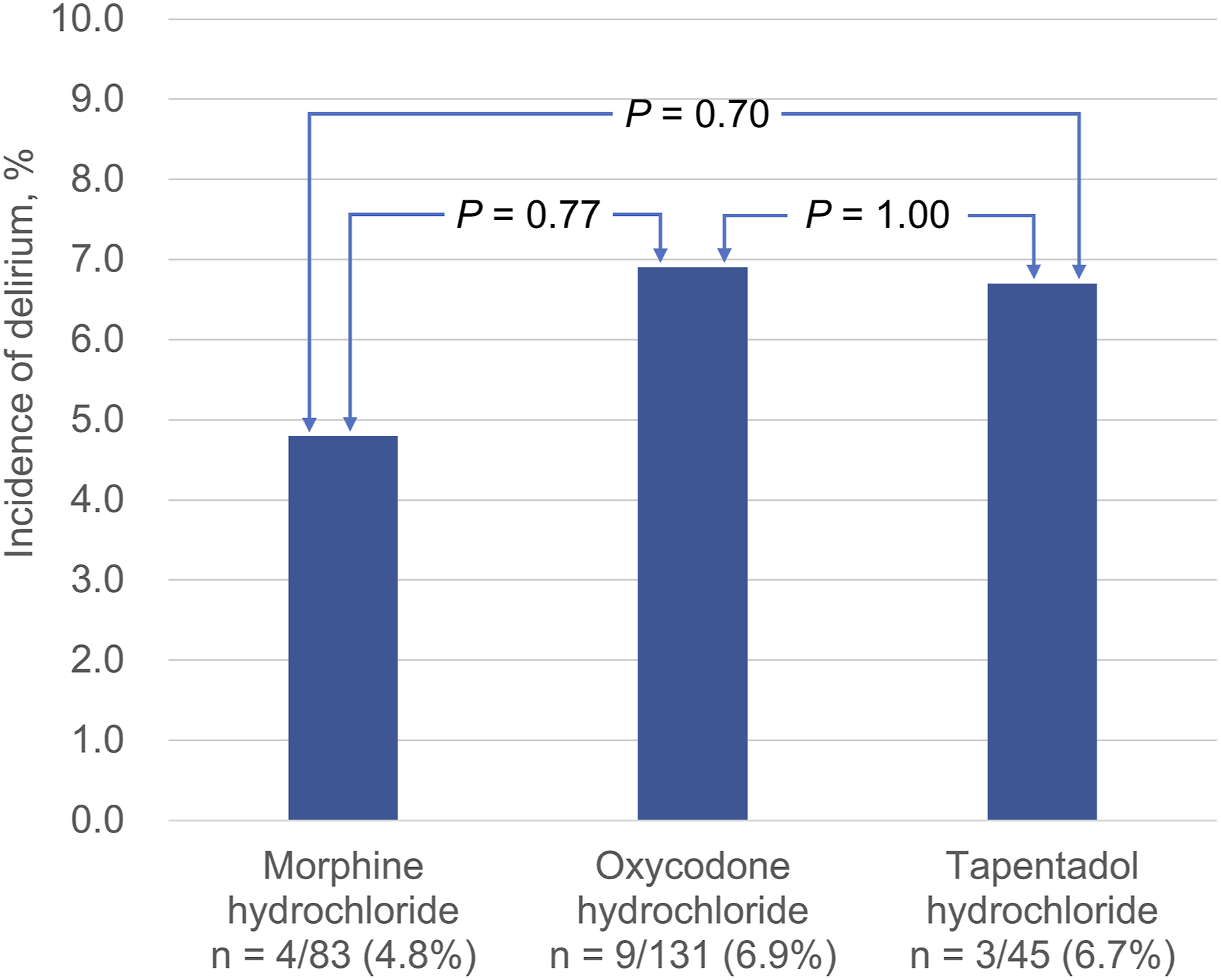
Differences in Patient Characteristics
Patient Characteristics.
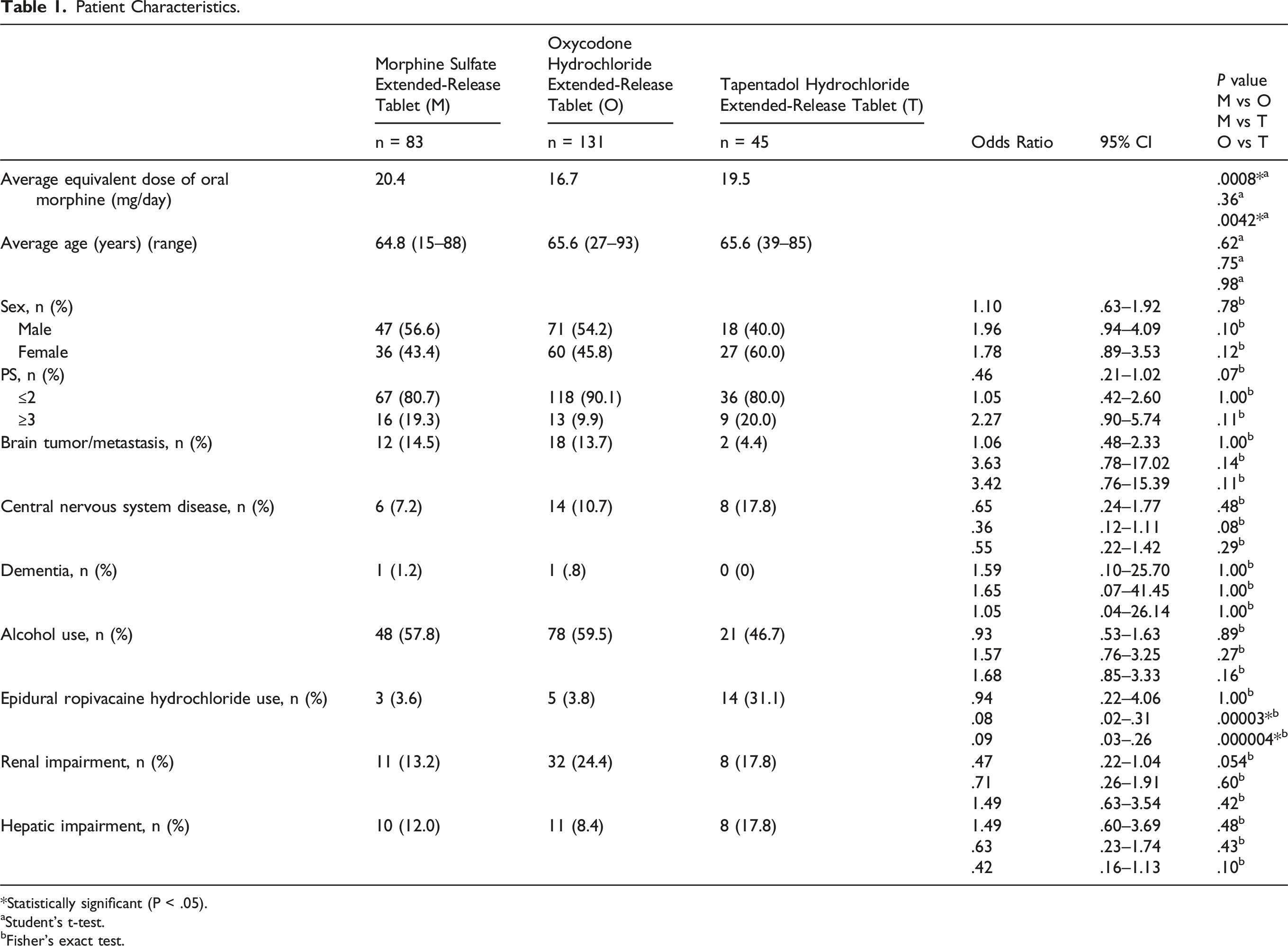
*Statistically significant (P < .05).
aStudent’s t-test.
bFisher’s exact test.
Differences in Concomitant Drug Use
Concomitant Drugs.
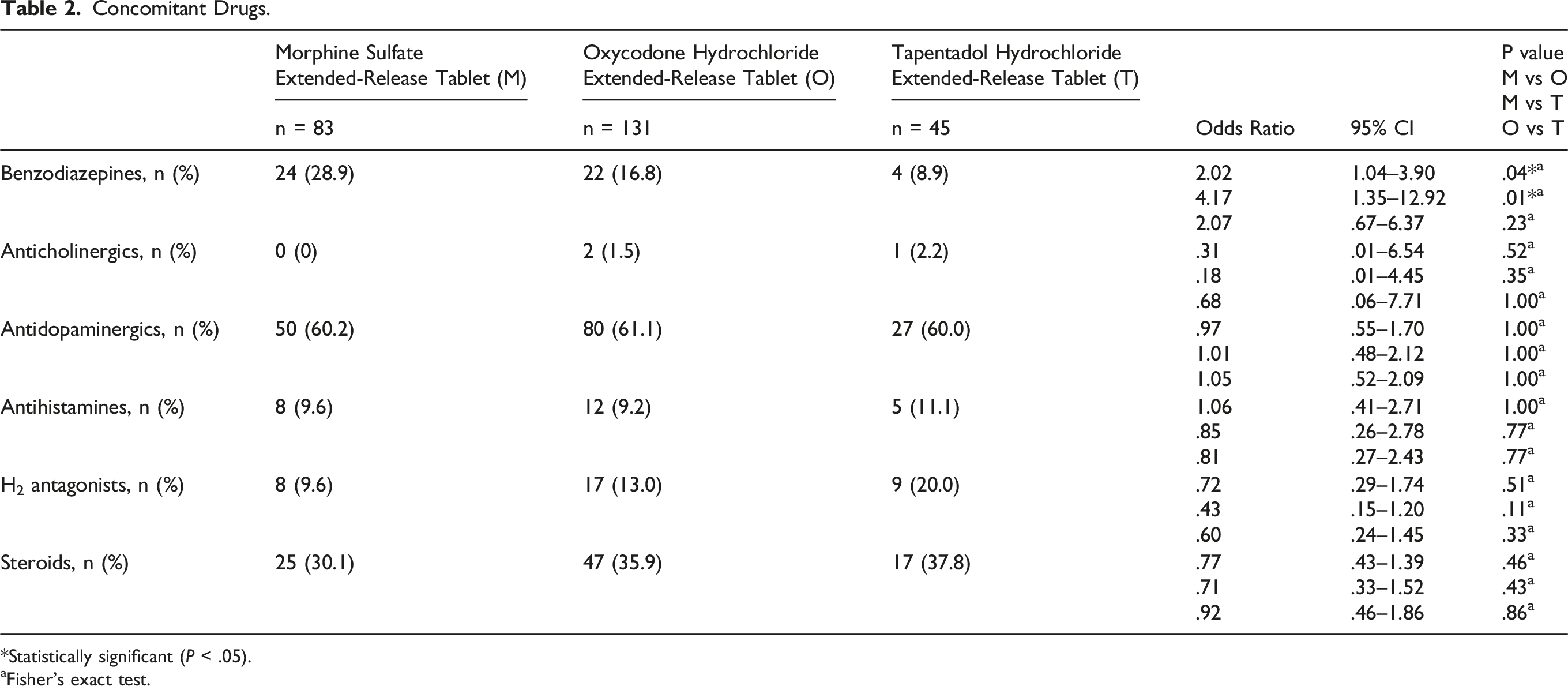
*Statistically significant (
aFisher’s exact test.
Discussion
In this study, we found no significant difference in the incidence of delirium among morphine sulfate extended-release tablets, oxycodone hydrochloride extended-release tablets, and tapentadol hydrochloride extended-release tablets in previously opioid-naive patients. This suggests that any of these oral opioids are equally good options from the perspective of preventing delirium.
This is consistent with our previous finding 8 that intravenous morphine and oxycodone cause delirium at comparable rates when used as the first opioid. However, we did find that incidence of delirium was lower with the oral formulation than with the intravenous formulation for both morphine and oxycodone (morphine PO in this study vs IV in previous study = 4.8% vs 28.9%, oxycodone PO in this study vs IV in previous study = 6.9% vs 19.5%). In many cases when IV opioid is selected as the first-line opioid for cancer pain, it is because the patient cannot take oral medication due to issues such as gastrointestinal obstruction or poor PS, and these patients are more prone to pain, undernutrition, and electrolyte imbalance, which are risk factors for delirium. 19 The findings from this study are more practical and useful because overall oral opioids are more frequently used as the first opioid than intravenous opioids in clinical practice. However, one limitation that arises from the retrospective nature of this study is that opioid selection was left to the discretion of the attending physician, which may have produced bias. Prospective studies with clear criteria for opioid selection may be necessary to obtain higher-quality evidence in the future.
Although tapentadol has a similar structure to tramadol, which has a high incidence of delirium when used to manage postoperative pain, 20 we found that the incidence of delirium with tapentadol was comparable to that of other opioids in this study. Tapentadol has been shown to have higher selectivity for μ-opioid receptors than tramadol, and less of an effect on serotonin reuptake. 21 These differences in mechanism of action may help explain our finding. Like fentanyl, tapentadol has little effect on δ- and κ-receptors, and thus would presumably be less likely to cause delirium, but we did not observe such a trend in our study.
One limitation of our study is that we could not rule out possible effects of rapid-release oral morphine or rapid-release oxycodone for breakthrough pain in patients who received tapentadol hydrochloride extended-release tablets. We would like to conduct further research once a rapid-release formulation of tapentadol is developed. In this study, the average equivalent dose of oral morphine was significantly lower in the O group than in the M group or T group. In our hospital, the smallest dose of 12-h extended-release tablets is 10 mg (equivalent dose of oral morphine, 10 mg) for morphine hydrochloride, 5 mg (equivalent dose of oral morphine, 7.5 mg) for oxycodone hydrochloride, and 25 mg (equivalent dose of oral morphine, 7.5 mg) for tapentadol hydrochloride. This means that if the smallest-dose tablet were administered to previously opioid-naive patients, the equivalent dose of oral morphine must have been smaller in the O group and T group than in the M group, and this may be one of the reasons for the above differences. The incidence of delirium is reported to be increased when the daily equivalent dose of oral morphine exceeds 90 mg, 22 but the daily dose was lower than that in all of our groups. Thus, we think that the significantly lower average equivalent dose of oral morphine in the O group than in other groups was not a substantial enough difference to impact on the clinical effect of these 3 oral opioids.
Another factor known to contribute to delirium is use of epidural anesthesia.23,24 Analysis of patient characteristics showed that a larger percentage of patients were using epidural ropivacaine hydrochloride in the T group than in the M or O groups, and this may have increased the incidence of delirium in the T group. In addition, analysis of concomitant drugs showed that a larger percentage of patients were using benzodiazepines in the M group than in the O or T groups, and this may have increased the incidence of delirium in the M group. A final limitation is that we were unable to compare blood electrolyte concentrations (Na+, K+, and Ca2+) at the start of opioid therapy because these values were unavailable for some patients, and thus we cannot rule out their effect on our results. Another factor that may have influenced the results is differences in the skill level of hospital staff caring for each patient in implementing non-pharmacological approaches to address issues such as hospital room lighting, undernutrition, and poor sleep. 7
To summarize, although we cannot completely rule out the effects of confounding factors due to the retrospective nature of this study, it appears that oral morphine, oxycodone, and tapentadol have comparable incidences of delirium when used as the first opioid and thus are equally good choices for oral opioid therapy.
