Abstract
Pilot and feasibility randomized controlled trials (RCTs) help determine whether to proceed to a full-scale trial and identify necessary modifications. Qualitative research embedded in these trials can offer insight into feasibility, acceptability, and implementation of interventions, which is not always evident through quantitative data alone. It remains unclear how qualitative findings have influenced decisions to progress to full-scale RCTs. A scoping review was conducted, guided by Joanna Briggs Institute methodology. MEDLINE was searched for pilot and feasibility RCTs of health intervention published between 2017 and 2022 in four target journals, focusing on those using qualitative research to inform progression decision-making. Eligible studies were published in English with freely available full-text results. Titles and abstracts were screened independently by two reviewers, with full texts screened by one reviewer and a random 20% sample double-screened by a second reviewer. Data were extracted on trial characteristics, progression criteria, qualitative methods, and decision-making processes. Of the 932 records screened, 69 trials (across 94 publications) met inclusion criteria. Two approaches to using qualitative research in progression decision-making were identified: the application of prespecified progression criteria and the integration of qualitative and quantitative data. Of 18 trials which reported prespecified progression criteria, five included qualitative progression criteria. Eight trials employed mixed-methods integration techniques. Three used both integration techniques and qualitative progression criteria. The findings highlight limited but promising examples of meaningful integration. Clearer guidance is needed to support the consistent and transparent use of qualitative research in informing progression decisions in pilot and feasibility RCTs.
Keywords
Background
To facilitate smoother implementation of a full-scale randomized controlled trial (RCT), researchers may undertake a pilot or feasibility RCT in which components such as acceptability, intervention delivery, and recruitment and retention are first evaluated on a smaller scale (Bond et al., 2023; Eldridge et al., 2016; NIHR; Skivington et al., 2021). These preliminary studies help researchers and funders determine whether progression to a full-scale RCT is justified and what modifications, if any, are necessary prior to doing so (Leon et al., 2011). Prespecified progression criteria are recommended in pilot and feasibility RCTs to guide these decisions and assess feasibility (Skivington et al., 2021). However, such criteria are often underreported, and where they are presented, the basis for their development, justification, and use in evaluating study processes and interventions is frequently unclear (Mbuagbaw et al., 2019; Mellor et al., 2021).
Moreover, progression criteria tend to be based primarily on quantitative data, with less attention given to the contribution of qualitative findings (Baldeh et al., 2020). Qualitative research can illuminate issues that may not be captured through quantitative methods alone, such as whether an intervention is acceptable to prospective participants or whether trial processes are feasible in practice. For example, qualitative research has shown how potential participants view hypothetical trial scenarios and how these views can indicate that a planned RCT may not be feasible (Lavender & Kingdon, 2009). The Qualitative Research in Trials (QUART) study similarly demonstrated that qualitative methods can clarify whether aspects of an intervention or trial design require modification before progressing to a full-scale RCT (O’Cathain et al., 2014). Although pilot and feasibility RCTs have a different purpose from full-scale RCTs, the potential contributions of qualitative research to understanding feasibility and acceptability are equally important in these early-phase studies.
Current guidance encourages the integration of qualitative research within pilot and feasibility RCTs to help explain how and why improvements to the study may be required, which would not be possible with quantitative data alone (O’Cathain et al., 2014, 2015; Pearson et al., 2020; Thabane et al., 2010). More recently, Aschbrenner et al. (2022) proposed five planning steps for optimizing mixed methods in pilot and feasibility studies. These include (1) identifying the feasibility domains to be examined with mixed methods; (2) aligning quantitative and qualitative data sources; (3) determining the timing of data collection; (4) planning integrative analyses using joint displays; and (5) drawing meta-inferences about feasibility. This framework offers practical guidance for integrating data and highlights where and when qualitative methods can help interpret quantitative progression criteria. However, other guidance on progression criteria remains focused on quantitative metrics. While understanding how qualitative research enhances the interpretation of progression criteria is valuable, specific guidance on using qualitative findings in pilot and feasibility study progression decision-making is lacking.
Currently, the underreporting of qualitative findings in pilot and feasibility RCTs limits understanding of their impact on trial progression decision-making. For example, a review found that while 71% of external pilot trials reported plans for qualitative research, only 51% reported the findings of that research (Mellor et al., 2021). Additionally, in only one-third of the studies, the qualitative findings, like intervention acceptability, informed progression decision-making. Following from these findings, recommendations for progression criteria in external pilot trials were developed (Mellor et al., 2023). These recommendations also supported the use of qualitative findings in pilot and feasibility study progression decision-making, but specific guidance on how to apply qualitative findings into these decisions remains lacking.
As a first step in addressing this gap, the aim of this scoping review was to explore how qualitative research has informed decisions about progression from pilot and feasibility RCTs to full-scale RCTs. The objectives were to: (1) Identify pilot and feasibility RCTs that have utilized qualitative research findings to inform decisions about progression to a full-scale RCT. (2) Explore which study components were assessed using qualitative methods. (3) Describe how decisions about progression from pilot and feasibility RCTs to full-scale RCTs have been made, including through the use of qualitative findings.
Methods
A protocol for this scoping review was registered on the Open Science Framework: osf.io/vyfgx (Björklund et al., 2023). The review was informed by Joanna Briggs Institute guidance (Peters et al., 2015, 2020) and is reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for scoping reviews (PRISMA-ScR) (Tricco et al., 2018).
Inclusion Criteria
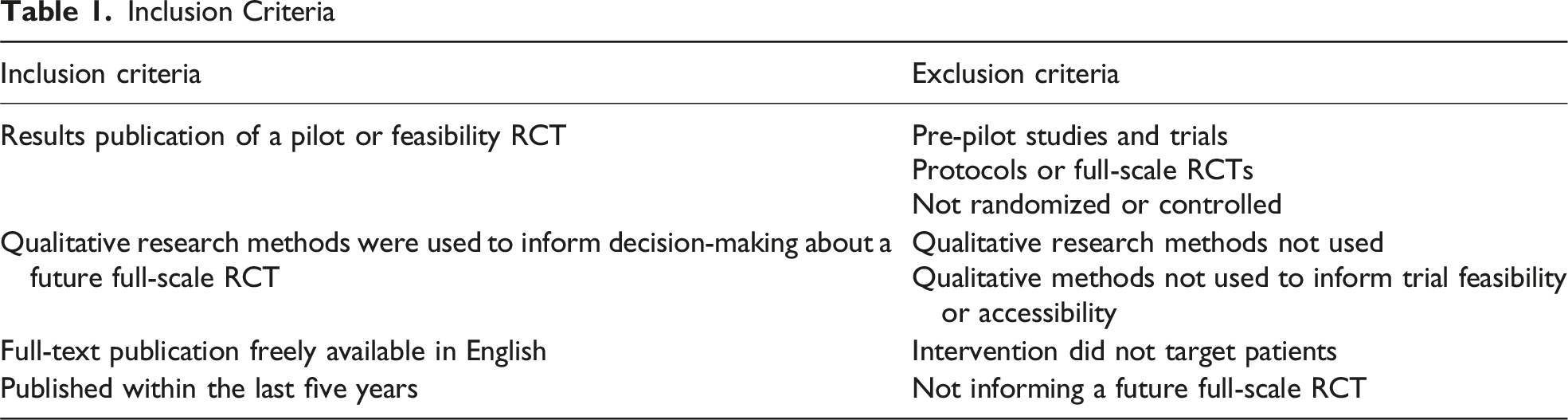
In line with previous methodologically focused reviews (Mbuagbaw et al., 2019; Mellor et al., 2021), four journals were searched via MEDLINE: BMJ Open, Pilot and Feasibility Studies, Trials, and PLOS One. These journals were selected as they had the highest number of citations with the terms ‘pilot’, ‘trials’, and ‘qualitative’ in the title, based on a preliminary MEDLINE search. Reference lists of included pilot and feasibility RCTs were screened for additional publications relating to the same study, for example, where qualitative findings were reported separately from primary trial results. Search terms included ‘pilot’, ‘feasibility’, ‘trial’, ‘study’, and ‘qualitative’ (see Supplemental File 1 for full search strategy).
Duplicates and publications with ‘protocol’ in the title were removed in EndNote (The EndNote Team, 2013). The remaining records were uploaded to Rayyan (Ouzzani et al., 2016) for screening. All titles and abstracts were screened independently by two reviewers (MB and NC). Uncertainties or disagreements were resolved through discussion with a third reviewer (CJ). Full-text screening followed, using an eligibility form developed by the review team (MB, NC, MH, and CJ). One reviewer (MB) screened all full texts, with a random 20% of texts being double-screened by another reviewer (NC, MH, or CJ). Any disagreements were discussed until consensus was reached. Publications relating to the same pilot and feasibility RCT were combined and reported as a single study.
Data extraction was conducted by one reviewer (MB), with data from a random 20% of studies also extracted by another reviewer (NC, MH, or CJ) to ensure accuracy. Extracted data included general study characteristics (e.g., country and intervention type), qualitative research methods, progression criteria, trial progression recommendations (e.g., how decisions were made), and the decision-making process. Findings are presented through a descriptive summary and synthesis. Feasibility domains were determined inductively during data analysis and grouped according to common areas of focus in pilot and feasibility RCTs. Domains such as fidelity and implementation were distinguished by whether the extracted data related to the delivery of the intervention during the pilot or feasibility RCT (fidelity) or to broader issues related to the context of the trial (implementation).
Results
In total, 932 articles were identified. There were no duplicates. This reduced to 202 after title and abstract screening. A further 129 articles were excluded when full-text articles were assessed for eligibility. The most common reason for exclusion was that the studies were not pilot or feasibility RCTs (n = 93) or not informing a future RCT (n = 30). The rationale for this exclusion was that the decision-making process would not be relevant to report for these studies. Twenty-one additional articles were identified through bibliography searching of included studies. In total, 69 pilot and feasibility RCTs were included in the review, reported in 94 publications. Twenty-two of the included studies reported results in more than one article, including 11 that reported the qualitative findings separately from the main results publication (see Figure 1). PRISMA-ScR flowchart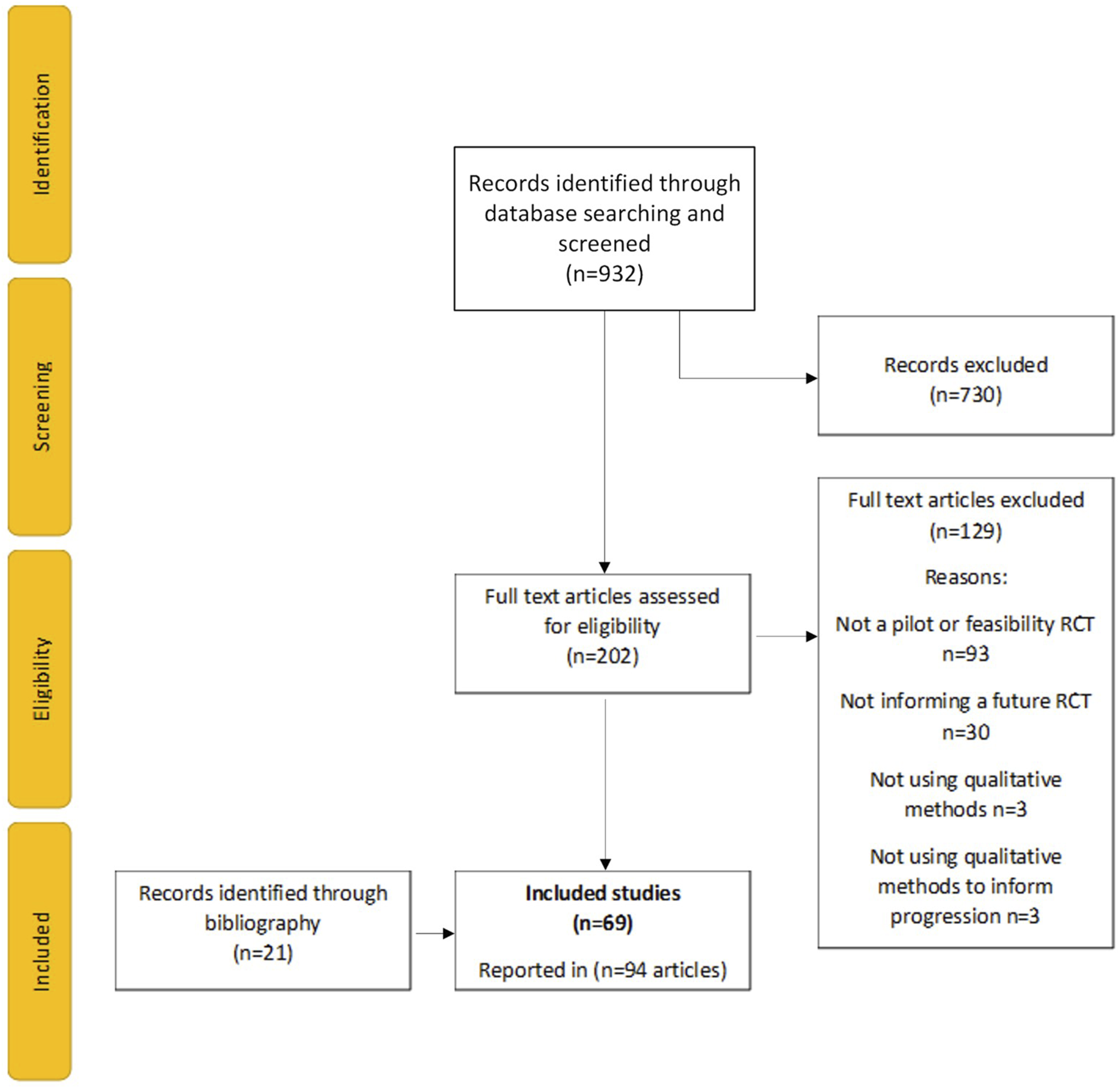
Characteristics of Included Pilot and Feasibility RCTs
Study Characteristics
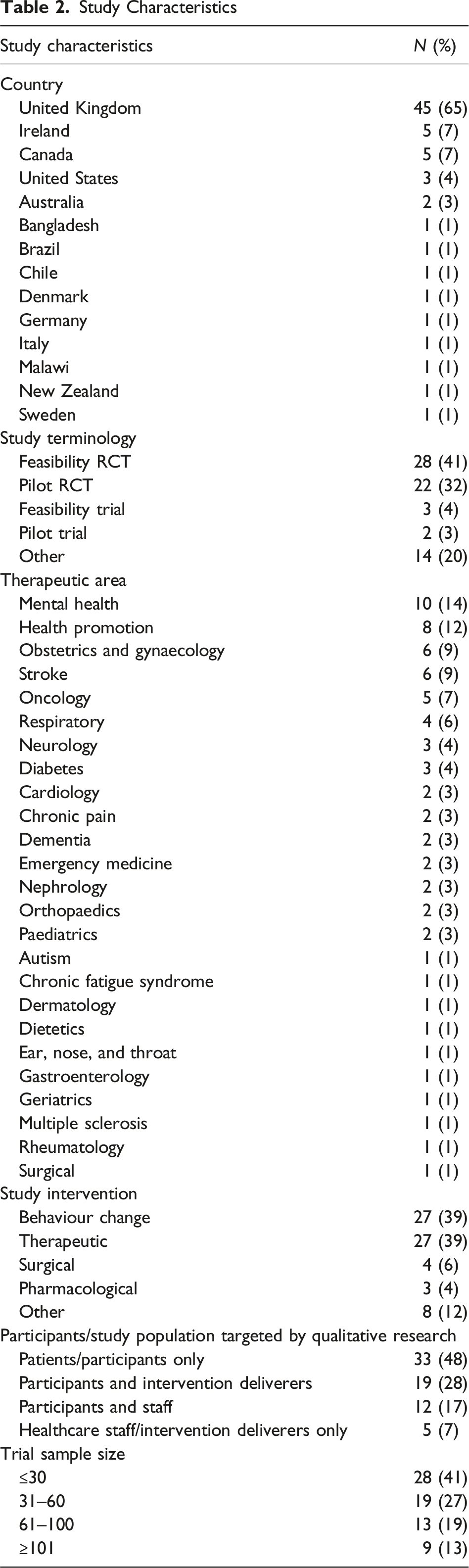
Study Components Assessed Using Qualitative Research Methods
In the included pilot and feasibility RCTs, qualitative research was mainly used to collect data related to the study intervention, such as its acceptability (n = 44, 64%), feasibility (n = 32, 46%), and views about specific intervention components (n = 8, 41%). Qualitative methods were also used to assess participants’ and study staff’s overall views, experiences, and perspectives of having taken part in the study (n = 44, 64%). Other data collected through qualitative methods included the acceptability or feasibility of the recruitment methods (n = 20, 29%), acceptability of outcome data collection methods (n = 13, 19%), randomization (n = 10, 14%), retention (n = 8, 12%), adherence (n = 4, 6%), intervention fidelity (n = 2, 3%), and consent procedures (n = 1, 1%).
Approaches to Decision-Making About Trial Progression Using Qualitative Research
In line with the review eligibility criteria, all included pilot and feasibility RCTs reported using qualitative research to some extent to inform decisions about trial progression. Two approaches to decision-making were evident: (a) use of prespecified progression criteria and (b) use of data integration strategies. These two approaches were not always used together, and data integration was not always explicitly stated as being used to inform trial progression decision-making.
Use of Prespecified Progression Criteria
Domains Included in Prespecified Progression Criteria Within the 18 Studies That Included Them

Feasibility and Pilot RCTs With Combined Quantitative and Qualitative Progression Criteria

In pilot and feasibility RCTs where prespecified progression criteria were used, trial progression decision-making was based on whether those criteria were met or not (e.g., ‘A future RCT using the recruitment strategy reported here is not likely to be feasible. The fundamental problem was in accessing sufficient struggling HA [hearing aid] users at follow-up’ (Watson et al., 2021)). In those studies, the qualitative findings were used to varying degrees to understand why those criteria were (or were not) met. In pilot and feasibility RCTs where progression criteria were assessed using a traffic light system, decisions about trial progression and changes to the trial were more clearly on both quantitative and qualitative findings; one study (Amaefule et al., 2022) found that the quantitative progression criteria for adherence were within the amber zone and through qualitative findings were able to determine what the barrier was and what the participants preferred (i.e., a preference for the intervention supplement to be delivered in a tablet form instead of powder).
Data Integration Strategies
Two categories of integration of quantitative and qualitative data were identified: (1) description and/or discussion of quantitative and qualitative findings in the results or discussion section of the paper and (2) reported use of specific methods to integrate qualitative and quantitative data.
Description of Findings in Results or Discussion
Forty-six (67%) of the included 69 pilot and feasibility RCTs integrated the qualitative and quantitative data to varying degrees through simple description and/or discussion of the findings. Some studies used sequential description and/or discussion (e.g., discussing quantitative findings about study recruitment first and then discussing qualitative findings relating to study recruitment separately). Other studies discussed trial domains of interest (e.g., study recruitment) more generally and referred to both types of data throughout the discussion. Although the qualitative findings were discussed in some capacity, how they impacted the trial progression decision-making was sometimes unclear, particularly in the absence of prespecified progression criteria.
Specific Methods to Integrate Data
Eight (12%) of the 69 pilot and feasibility RCTs reported using specific methods to integrate the qualitative and quantitative data. Reported integration techniques used included data triangulation, a mixed-methods matrix, meshing, comparing and contrasting the data, and concurrent presentation of the data. While these studies reported the method of integration, integrated findings were not always presented in the results section or discussed. For example, Agarwal et al. (2019) reported using meshing to integrate the qualitative and quantitative data: ‘Meshing quantitative and qualitative methods was intended to provide a more multi-dimensional understanding of the feasibility of this study’. However, qualitative findings were presented in their own paragraph in the results section and discussed separately to the quantitative findings (Agarwal et al., 2019).
Only three of the included pilot and feasibility RCTs included both prespecified progression criteria and specific methods to integrate data (4%). Young et al. (2020) used a joint display showing the progression criteria and the qualitative findings relating to them side by side (see Figure 2). Taylor et al. (2022) reported the progression criteria in the main results publication and discussed the integration of qualitative and quantitative data into a single framework in a separate qualitative publication. However, a visual display of integration was not provided. A third study by Dombrowski et al. (2020) reported using triangulation and matrix coding to integrate qualitative and quantitative data but did not explicitly relate data integration to the progression criteria or provide a visual display of the integration. Example of a joint display table by Young et al. BMJ Open 2020; 10: e041227 (Young et al., 2020)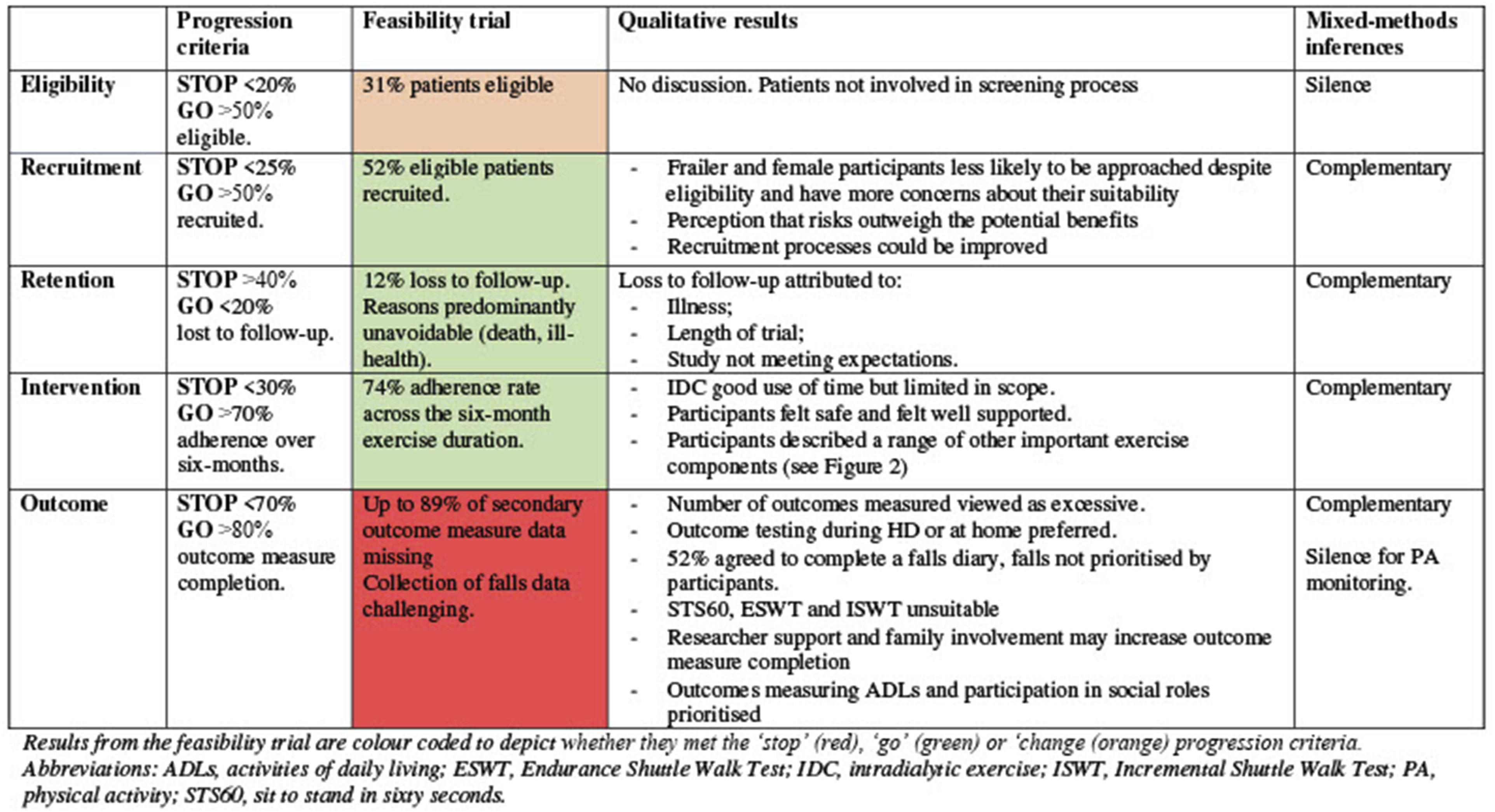
Decision-Making About Trial Progression
Trial Progression Decision-Making Process

Discussion
This scoping review aimed to explore how qualitative research has informed decision-making about progression from pilot and feasibility RCTs to full-scale RCTs. In total, 69 studies were included in the review that were conducted in different countries and tested a variety of interventions in different therapeutic areas. Within the included studies, qualitative research methods were most commonly used to assess intervention acceptability, feasibility, and components of the interventions.
Two broad approaches to using qualitative data in trial progression decisions were identified: the use of prespecified progression criteria and the integration of qualitative and quantitative findings. While progression criteria were more frequently reported, they tended to be quantitative in nature. Integration strategies were used but were often underutilized and rarely made explicit in relation to progression decision-making. Furthermore, the actual processes through which decisions were made about progression to a full-scale RCT were poorly reported.
Only 18 (26%) of the included studies reported prespecified progression criteria. On the one hand, this is surprising given the presence of such criteria in methodological guidance on pilot and feasibility trials (Skivington et al., 2021). On the other hand, this finding reflects prior reviews that also noted a lack of consistent reporting criteria (Mbuagbaw et al., 2019; Mellor et al., 2021). Prespecified criteria serve a critical role as they offer a structured approach for assessing feasibility and acceptability of both trial processes and interventions. However, their largely quantitative nature may limit their ability to capture the complex, context-dependent aspects of trial acceptability that qualitative methods can highlight when meaningfully integrated with those findings (Aschbrenner et al., 2022).
Previous reviews have primarily examined the reporting of progression criteria (Mbuagbaw et al., 2019; Mellor et al., 2021) or whether qualitative research has been reported in pilot and feasibility studies (Baldeh et al., 2020). These reviews consistently found that both remain underreported despite methodological guidance highlighting their importance. What has not yet been sufficiently explored is how qualitative research is used to inform decisions about trial progression, which is what this review addresses. This is a critical area of inquiry, particularly as guidance continues to recommend the use of qualitative research in evaluating complex interventions and informing future study design (Skivington et al., 2021).
In current practice, progression criteria have become closely associated with quantitative thresholds, for example, recruitment rates, retention levels, or adherence measures. However, these kinds of measures may be insufficient for evaluating less distinct but equally important aspects of feasibility, such as how acceptable participants find the intervention or trial procedures (Mellor et al., 2023). In this context, the term ‘progression criteria’ may not adequately reflect the interpretive nature of qualitative data, which offer a different kind of understanding not easily determined through numerical thresholds alone. Five studies in this review demonstrated that qualitative findings contributed to judgements about feasibility or acceptability in ways that influenced the progression decisions. These examples illustrate how qualitative data can play a substantive role when it is considered alongside quantitative data, although it is not clear whether this was due to the inclusion of qualitative progression criteria or explicit data integration methods, or both.
Across the included studies, qualitative research supported a range of contributions to progression decisions, including identifying modifications to intervention delivery (e.g., Amaefule et al., 2022), clarifying barriers to recruitment (Watson et al., 2021), and helping to interpret mixed-methods feasibility outcomes (Dombrowski et al., 2020; Young et al., 2020). These examples show how qualitative findings can shape decisions even in studies without prespecified qualitative criteria or formal integration techniques. This aligns with findings from the QUART study, which identified examples of qualitative research influencing intervention refinement, recruitment approaches, outcome measures, and the interpretation of trial findings (O’Cathain et al., 2014).
Among the studies that included both types of data, the majority (n = 46, 67%) described some form of integration methods, typically through narrative synthesis or descriptive comparison. However, only eight studies (12%) employed formal data integration. This is notable given recent guidance that calls for deliberate and transparent integration strategies to maximize the value of mixed methods in pilot and feasibility studies (Aschbrenner et al., 2022). When used meaningfully, these integration approaches can facilitate deeper understanding of the relationships between feasibility, acceptability, and contextual factors, thereby providing a more holistic foundation for progression decisions.
Notably, 93% of studies did not report the actual process by which progression decisions were made, limiting the field’s understanding of how both qualitative and quantitative findings are used in practice. In the few studies (n = 3) that did use both prespecified progression criteria and specific data integration methods, the influence of qualitative findings on progression decision-making was more explicit. In these cases, integration enhanced transparency and provided clear justifications for whether and how to proceed to a full-scale RCT. Such clarity is essential for research teams, funding bodies, and trial steering committees who evaluated the feasibility for larger-scale trials. Furthermore, these examples serve as potential examples for future trials aiming to embed qualitative research more meaningfully within progression decision-making processes.
The findings from this review suggest that researchers may benefit from clearer guidance on how to plan for, conduct, and report qualitative research in the context of pilot and feasibility study progression decision-making. There remains an important need to better understand why qualitative research is or is not used in these decision-making processes and how its integration with quantitative findings can be made more systematic and transparent. Methodological research to address these issues could support more rigorous and impactful pilot and feasibility studies. Such work could also ensure that the full potential of qualitative inquiry is realized in optimizing intervention development and trial design.
Strengths and Limitations
This review contributes to the literature by offering the first, to our knowledge, focused examination of how qualitative research has been used to inform decisions about whether to progress from pilot and feasibility to full-scale RCTs. It complements and extends prior reviews by examining not just whether qualitative research was used, but how it was applied within the context of trial development and progression decision-making. A particular strength is the inclusion of multiple publications linked to single studies, allowing for a more comprehensive capture of qualitative findings, especially those reported separately from the main trial findings. This approach enabled a more holistic view of how qualitative data contributed to the overall decision-making process.
The review also highlights three exemplar studies that integrated both prespecified progression criteria and structured data integration techniques, demonstrating how mixed-methods approaches can be optimally employed to support progression decision-making. These studies may serve as useful reference points for researchers aiming to better integrate qualitative research into pilot and feasibility studies.
As a scoping review, this study did not synthesize findings across all relevant literature or assess the quality of included studies. Instead, its aim was to map key themes, gaps, and patterns in the use of qualitative research for trial progression decisions. Another limitation is the review’s focus on randomized and controlled pilot and feasibility studies. While this ensured a manageable number of included studies, it excluded other forms of feasibility research. Nonetheless, the focus on qualitative contributions to progression decision-making means that many of the findings here may be transferable to other types of pilot and feasibility studies beyond RCTs.
Restricting the search to four journals may have limited the pool of eligible results papers, as pilot and feasibility RCT findings can be published across a broader range of journals. This may have reduced the diversity of included studies. However, the journals selected were deemed to publish a considerable number of pilot and feasibility RCTs, and the restriction contributed to maintaining a manageable scope for the review. As such, this is unlikely to affect the main conclusions of the review.
Conclusions
This review identified pilot and feasibility RCTs that used qualitative research to help determine whether and how to progress to a full-scale RCT. Approaches to supporting trial progression included prespecified progression criteria, integration of qualitative and quantitative data, or a combination of both. However, most studies lacked transparent reporting of these processes, and the combined use of progression criteria and integration strategies was rare. While guidance for both elements exists, current practice remains inconsistent. The three exemplar studies identified here offer valuable examples for integrating qualitative and quantitative findings to support more robust, transparent, and holistic progression decisions. Embracing such approaches could help ensure that qualitative research contributes more meaningfully to progression decision-making in pilot and feasibility studies. This would ultimately support better-informed intervention development and more effective health research.
Supplemental Material
Supplemental Material - The Role of Qualitative Research in Progression Decision-Making in Mixed-Methods Pilot and Feasibility Randomized Controlled Trials: A Scoping Review
Supplemental Material for The Role of Qualitative Research in Progression Decision-Making in Mixed-Methods Pilot and Feasibility Randomized Controlled Trials: A Scoping Review by Matilda Björklund, Nicola Cornwall, Melanie Ann Holden, and Clare Jinks in Qualitative Health Research
Footnotes
Ethical Considerations
Our study did not require an ethical board approval because it did not directly involve humans or animals.
Author Contributions
MB: Conceptualization, data curation, formal analysis, investigation, methodology, visualization, and writing – original draft. NC: Conceptualization, data curation, formal analysis, investigation, methodology, visualization, supervision, writing – review and editing, and validation. MH: Conceptualization, data curation, formal analysis, investigation, methodology, visualization, supervision, writing – review and editing, and validation. CJ: Conceptualization, data curation, formal analysis, investigation, methodology, visualization, supervision, writing – review and editing, and validation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This PhD was funded by the Faculty of Medicine and Health Sciences Doctoral Fellowship Scheme at Keele University. NC and CJ are part funded by the National Institute for Health Research and Care Applied Research Collaboration West Midlands.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
