Abstract
Keywords
Atopic dermatitis (AD) is a chronic, relapsing inflammatory skin condition that commonly emerges in early childhood and presents with pruritus, eczematous lesions, and systemic immune dysregulation. It is characterized by a complex interplay of genetic predisposition, environmental triggers, and immunological factors, making it one of the most prevalent childhood atopic diseases (Otsuka et al., 2017). Global prevalence estimates suggest that AD affects between 15% and 30% of children (Kowalska-Olędzka et al., 2019; Silverberg et al., 2021). In certain regions, rates are even higher, for example, an epidemiological study found a 41.6% prevalence among primary school children in Hong Kong (Chan et al., 2022; Ng et al., 2023). This elevated prevalence has been attributed to factors such as dense urban living, air pollution, and subtropical climate conditions, all of which may contribute to skin barrier disruption and heightened allergen sensitivity.
Beyond its cutaneous symptoms, AD significantly impairs quality of life, affecting physical, emotional, behavioral, and social functioning and contributes to diminish self-esteem in children (Barilla et al., 2017; Weidinger et al., 2024). A meta-analysis further showed that children with AD are at a significantly higher risk of developing mental health disorders than those without the disease (Lam et al., 2024; Xie et al., 2019). Given its high prevalence, chronicity, psychosocial impact, and economic costs, AD is increasingly recognized as a global burden of disease, warranting comprehensive and multidisciplinary strategies for prevention, treatment, and support (Laughter et al., 2021).
Despite therapeutic advancements, the pathways by which immune dysregulation drive the progression and severity of pediatric AD remain insufficiently understood. Most biomedical research has focused on isolated cytokine levels or narrow immunological profiles, often overlooking the complex interplay between inflammatory mediators in real-world settings (Criton & Brahmakulam, 2024; Pareek et al., 2024). Moreover, these studies have rarely been integrated with psychosocial or holistic intervention research, limiting our ability to fully evaluate how supportive care approaches influence physiological systems. This represents a critical gap in outcome-based social work research, particularly as the field increasingly embraces integrative and interdisciplinary approaches to well-being (Yosipovitch et al., 2024).
This study contributes to emerging interdisciplinary research by exploring how immunological indicators can be meaningfully integrated into the evaluation of psychosocial care. It reflects a growing recognition within social work that physiological outcomes such as inflammatory responses can serve as objective complements to conventional self-report and behavioral measures. The use of biomarkers in this context represents a contemporary direction in social work efficacy research, offering insights into how supportive interventions may influence biological pathways linked to chronic disease. By bridging psychosocial and physiological domains, this approach supports more holistic, evidence-informed models of care that align with the field's commitment to integrative, person-centered social work practice.
Cytokine Imbalance and Immune Dysregulation in Pediatric AD
Cytokines, both pro-inflammatory and anti-inflammatory, are key immunological biomarkers that reflect disease activity and severity (Kiwan et al., 2022; Krupka-Olek et al., 2024). Among the pro-inflammatory cytokines, interleukin (IL)-1β and IL-6 are particularly significant for their roles in promoting inflammation that leads to tissue damage, edema, epidermal degradation, and pruritus. Elevated serum levels of IL-1β and IL-6 are consistently observed in AD patients, establishing these cytokines as markers of active inflammatory processes (Huang et al., 2023; Hunter & Jones, 2015; Lee et al., 2020; Lyubchenko et al., 2021; Matarazzo et al., 2022).
The acute phase of AD is marked by a predominance of T-helper 2 (Th2) cell responses, triggered by impaired skin barrier function and enhanced antigen presentation. The Th2-associated cytokines, particularly IL-4, are upregulated and contribute to allergic inflammation by promoting IgE class switching and activating eosinophils (Fania et al., 2022; Zhang et al., 2021). In contrast, regulatory cytokines, including IL-10 and transforming growth factor-beta 1 (TGF-β1), primarily secreted by regulatory T cells (Tregs), act to suppress Th2-mediated inflammation and restore immune homeostasis (Palomares et al., 2014). Notably, IL-10, a cytokine with pleiotropic functions, also exerts potent anti-inflammatory effects when produced by regulatory B (Breg) cells. Recent evidence shows that IL-10 from Breg cells can directly inhibit eosinophil activation and infiltration into the skin, thereby alleviating AD symptoms and reducing eosinophil-mediated tissue damage (Lee et al., 2024). The TGF-β1, while generally viewed as an anti-inflammatory cytokine, may exhibit pro-fibrotic and immune-suppressive effects, particularly in the context of chronic inflammation and tissue remodeling processes (Weissler & Frischmeyer-Guerrerio, 2019). Therefore, for the purposes of this study, cytokine classification was guided by their dominant functional roles in acute pediatric AD, with IL-1β, IL-6, and IL-4 treated as pro-inflammatory markers, and IL-10 and TGF-β1 as anti-inflammatory markers.
Immunological studies in infants and children with AD consistently show an altered cytokine profile compared to healthy controls, but findings remain variable across studies. For example, Gürkan et al. (2016) observed elevated levels of IL-5 and IL-35, alongside decreased IL-13 and TGF-β1 in affected infants. A recent study in children with atopic eczema/dermatitis syndrome reported elevated IL-17 and IL-23 and reduced IL-10 levels, particularly in atopic cases, with all three cytokines correlating with disease severity (Leonardi et al., 2015). Another study found that children with certain IL-4 gene variants had higher IL-4 levels and more severe AD, suggesting a genetic link to stronger allergic inflammation (Shang et al., 2016). These findings highlight the complex immunological profile of pediatric AD, suggesting that cytokine imbalances contribute to the variability in disease severity and clinical presentation among affected children.
The dynamic balance between pro-inflammatory and anti-inflammatory cytokines is crucial for regulating immune responses and preventing excessive tissue injury. In the context of AD, disruption of this balance is closely associated with increased disease severity (Cicchese et al., 2018). Research suggests that the ratio of pro-inflammatory to anti-inflammatory cytokines (e.g., IL-1β/IL-4 or IL-6/TGF-β1) may be a more reliable indicator of immune imbalance than absolute cytokine levels alone (Kim et al., 2007; Liang et al., 2015). Building on this evidence, the current study adopts six cytokine ratios (i.e., IL-6/IL-4, IL-6/TGF-β1, IL-1β/IL-4, IL-1β/TGF-β1, IL-10/IL-4, and IL-10/TGF-β1) as indicators of the inflammatory balance in children with AD, which are shown in Table 1.
Summary of the Cytokine to be Studied in the Current Study.

Psychosocial Interventions for Children with AD
Children with AD often experience not only chronic physical discomfort but also substantial emotional distress. In addition to symptoms such as persistent itching and skin lesions, children often report heightened anxiety, difficulties in social functioning, and diminished quality of life (Ersser et al., 2014; Hashimoto et al., 2017; Hua et al., 2023). Several recent systematic reviews and meta-analyses have systematically supported the effects of nonpharmacological interventions on patient-reported disease severity, but mixed effects on quality of life and psychological outcomes (Mitchell et al., 2025; Mostafa & Smith, 2023). While educational interventions have been shown to improve symptom management and daily functioning (Lu et al., 2018), most interventions have primarily focused on symptoms and lifestyle management. In contrast, comparatively fewer programs address the psychosocial dimensions of pediatric AD, despite growing evidence that psychological stress can dysregulate immune function and exacerbate inflammatory conditions (Ferat-Osorio et al., 2024; Shields et al., 2020).
To address this gap, our team developed the Integrative Body–Mind–Spirit (IBMS) psychosocial group intervention, a theory-driven and evidence-based program designed specifically to support the emotional, cognitive, and psychosocial well-being of children with AD (Fung et al., 2020; Xie, Chan, Lau et al., 2020). The IBMS framework draws Eastern spiritual health perspectives, which emphasize internal balance, the integration of body and mind, and the cultivation of meaning and connectedness in adversity. These are complemented by Western therapeutic principles, including emotion-focused coping, stress regulation, and strengths-based support (Chan et al., 2001; Lee et al., 2018). The IBMS program incorporates experiential methods such as mindfulness-based relaxation, guided imagery, storytelling, expressive arts, and peer support. These components are designed to cultivate emotional awareness, self-regulation, and resilience, while also fostering a supportive group environment that promotes connectedness and reduces feelings of isolation.
The IBMS model is grounded in over two decades of social work intervention research. Previous randomized controlled trials (RCTs) have shown that participation in IBMS is associated with reduced psychological distress, improved emotional regulation, and enhanced psychosocial functioning in both youth and adult clinical populations (Chan et al., 2001; Chan et al., 2006; Chan et al., 2012; Chan et al., 2019; Chan, Ji & Chan et al., 2017; Chan, Ji, Chan et al., 2017; Lee et al., 2018). In a previous RCT, children who received the IBMS intervention demonstrated significantly lower AD severity, reduced generalized anxiety and social phobia, and enhanced emotional functioning relative to those in a waitlist-control group (Xie, Chan, Lau et al., 2020).
Cytokines as Outcome Markers in Psychosocial Research
Emerging research indicates that psychosocial interventions can influence immune function, particularly through modulation of inflammatory cytokines. A meta-analysis of 56 randomized clinical trials involving over 4,000 participants found that psychosocial interventions were associated with improvements in immune functioning, including a 14.7% increase in beneficial responses and an 18.0% reduction in harmful pro-inflammatory markers. These effects were most consistently observed in cognitive-behavioral therapy (CBT) and multimodal approaches incorporating psychoeducation, relaxation training, mindfulness, or group-based support, especially when studies assessed changes in pro-inflammatory cytokines (Shields et al., 2020). This supports the use of cytokines as biological indicators in evaluating psychosocial interventions.
Beyond CBT, other psychosocial modalities have shown immunomodulatory effects. Mindfulness-based stress reduction programs have reduced IL-4, IL-6, and IL-10 in cancer patients (Witek-Janusek et al., 2008), and meaning-centered interventions have lowered IL-1β and IL-4 (Pang et al., 2021). Creative arts-based therapies like group drumming increased IL-4 (Fancourt et al., 2016), while yoga-based programs have reduced IL-6 and increased TGF-β among individuals with rheumatoid arthritis (Gautam et al., 2020). These findings suggest that diverse psychosocial modalities may modulate immune function across clinical populations. Complementing these findings, large-scale observational studies have established the broader immunological relevance of psychosocial factors. A meta-analysis of 41 studies including over 73,000 participants demonstrated that higher levels of social support and integration were significantly associated with lower systemic inflammation, particularly reduced levels of IL-6 and C-reactive protein (Uchino et al., 2018). While not derived from intervention studies, this body of evidence highlights the importance of social support as a protective factor in immune regulation. All these findings provide a compelling rationale for incorporating biomarkers into psychosocial and behavioral health research, particularly when examining the physiological effects of interventions that target social connectedness and emotional well-being.
Although prior research has demonstrated encouraging psychosocial benefits of the IBMS intervention, its potential to influence immune-related biological processes, such as cytokine regulation, remains underexplored in pediatric populations. The IBMS model emphasizes psychological processes such as somatic awareness, emotional regulation, meaning-making, and social connectedness, which are factors that are theorized to modulate physiological stress responses and support immune balance. In our earlier studies with adults, similar IBMS-based protocols led to significant reductions in pro-inflammatory cytokines such as IL-1β and IL-6 (Chan, Ji et al., 2017; Ng et al., 2022), suggesting that these interventions may exert measurable effects on immune functioning. Building on this evidence, the present study investigates whether comparable immunological changes accompany psychosocial improvements in children with AD who receive the IBMS intervention.
Research Gaps and Significance of the Study
These findings support the integration of inflammatory biomarkers as an outcome of psychosocial intervention research. While self-report measures and behavioral assessments remain essential in evaluating psychosocial interventions, they are subject to limitations such as recall bias, social desirability effects, and variability in interpretation, particularly among children or individuals with emotional or cognitive challenges (Althubaiti, 2016; Logan et al., 2008). These methodological limitations can obscure the full impact of an intervention. By incorporating biomarkers like cytokines, researchers can complement subjective and behavioral data with objective indicators of physiological change. This is especially valuable in populations affected by chronic inflammation or stress-related conditions, such as individuals with cancer, autoimmune disorders, or pediatric AD, where psychosocial well-being and biological health are deeply interconnected (Ferat-Osorio et al., 2024; Jurgens et al., 2023; Montgomery et al., 2024).
Despite growing interest, the application of biomarker-based outcome measures remains rare in social work research, particularly in pediatric populations. Children with AD often experience both physical discomfort and psychosocial challenges, yet little is known about how psychosocial interventions might influence their immune function. Building on our prior studies using the IBMS model, this research extends our investigation into how psychosocial care may impact inflammatory cytokine profiles in children with AD (Fung et al., 2019; Fung et al., 2020; Xie, Chan, Lau et al., 2020). By integrating biological, emotional, and behavioral data, this study contributes to a more holistic and evidence-informed framework for evaluating psychosocial interventions in social work practice.
Objectives of the Study
Although many studies have assessed the symptomatic and psychosocial outcomes of interventions for children with AD, their potential impact on immune function remains largely unexplored. Building on prior research demonstrating the efficacy of the IBMS intervention in improving emotional outcomes among children with AD (Xie, Chan, Lau et al., 2020), the present RCT aims to extend this work by incorporating biological outcome measures. Specifically, this study investigates whether participation in the IBMS intervention can influence inflammatory cytokine profiles, in addition to improving clinical and psychosocial outcomes. The study tests the following hypotheses:
H1: Children who receive the IBMS intervention will show a significantly greater reduction in pro-inflammatory cytokines (IL-6, IL-1β, and IL-4) than those in the waitlist control group. H2: Children in the IBMS intervention group will show a significantly greater increase in anti-inflammatory cytokines (IL-10 and TGF-β1) compared to the waitlist control group. H3: The IBMS intervention will produce a more favorable balance between pro- and anti-inflammatory cytokines relative to the waitlist control group. H4: The IBMS intervention will result in a significantly greater reduction in AD symptom severity than the waitlist control group. H5: Children receiving the IBMS intervention will report significantly greater improvements in quality of life and physical and emotional impacts compared to those in the waitlist control group.
Method
Study Design
This study employed a two-arm randomized, waitlist-controlled trial design to evaluate the efficacy of the IBMS psychosocial group intervention. The trial adhered to CONSORT guidelines for randomized studies and was approved by the Human Research Ethics Committee of the University of Hong Kong (EA1612023). It was registered at the Hong Kong Clinical Trials Registry [HKUCTR-2234].
Participants
Participants were children diagnosed with AD. Inclusion criteria for children were: (1) aged 6–12 years, (2) diagnosis with AD (ICD-10 codes L20-L30) confirmed by a pediatrician on the research team, (3) ability to communicate in Chinese (Cantonese), and (4) provision of informed parental consent. Children were excluded if they had a diagnosis of autism spectrum disorder or attention-deficit/hyperactivity disorder.
Sample Size
The sample size was calculated using G*Power (V.3.1.9), targeting a moderate effect size (f = 0.25) with 80% power and α = 0.05. A total sample of 86 children was required. Allowing for a 10% attrition rate, a minimum of 96 children (48 per group) was set as the recruitment target.
Recruitment and Study Procedure
Participants were recruited through convenience sampling from various sources across Hong Kong, including social media platforms, non-governmental organizations, and outpatient clinics displaying study materials. A total of 336 children were enrolled and initially screened for eligibility. All participants underwent a pre-intervention medical evaluation conducted by a pediatrician, who confirmed the diagnosis of AD based on ICD-10 criteria. This assessment led to the exclusion of 195 children who did not meet eligibility requirements. When appropriate, ineligible families were referred to other medical services.
Following baseline assessment, a total of 141 children were randomly assigned in a 1:1 ratio to either the IBMS psychosocial intervention group (n = 71) or the waitlist-control group (n = 70). Randomization was performed by an independent team member using a computer-generated sequence. Group allocation was concealed from laboratory staff and data analysts to minimize bias. However, due to the nature of the intervention, participants, social workers, and intervention facilitators were not blinded.
Data Collection
Figure 1 presents the participant flow diagram. The data collection procedure is demonstrated in Figure 2. Eligible children (n = 141) were invited to participate in a semistructured baseline interview conducted by trained research assistants. Informed consent was obtained from parents or legal guardians, and child assent was also secured. During this session, participants completed a standardized set of psychosocial measures (T0) either independently or with assistance from trained research assistants. Additionally, with parental consent, blood samples were collected from the children by a pediatrician for laboratory analysis of pro- and anti-inflammatory cytokines.
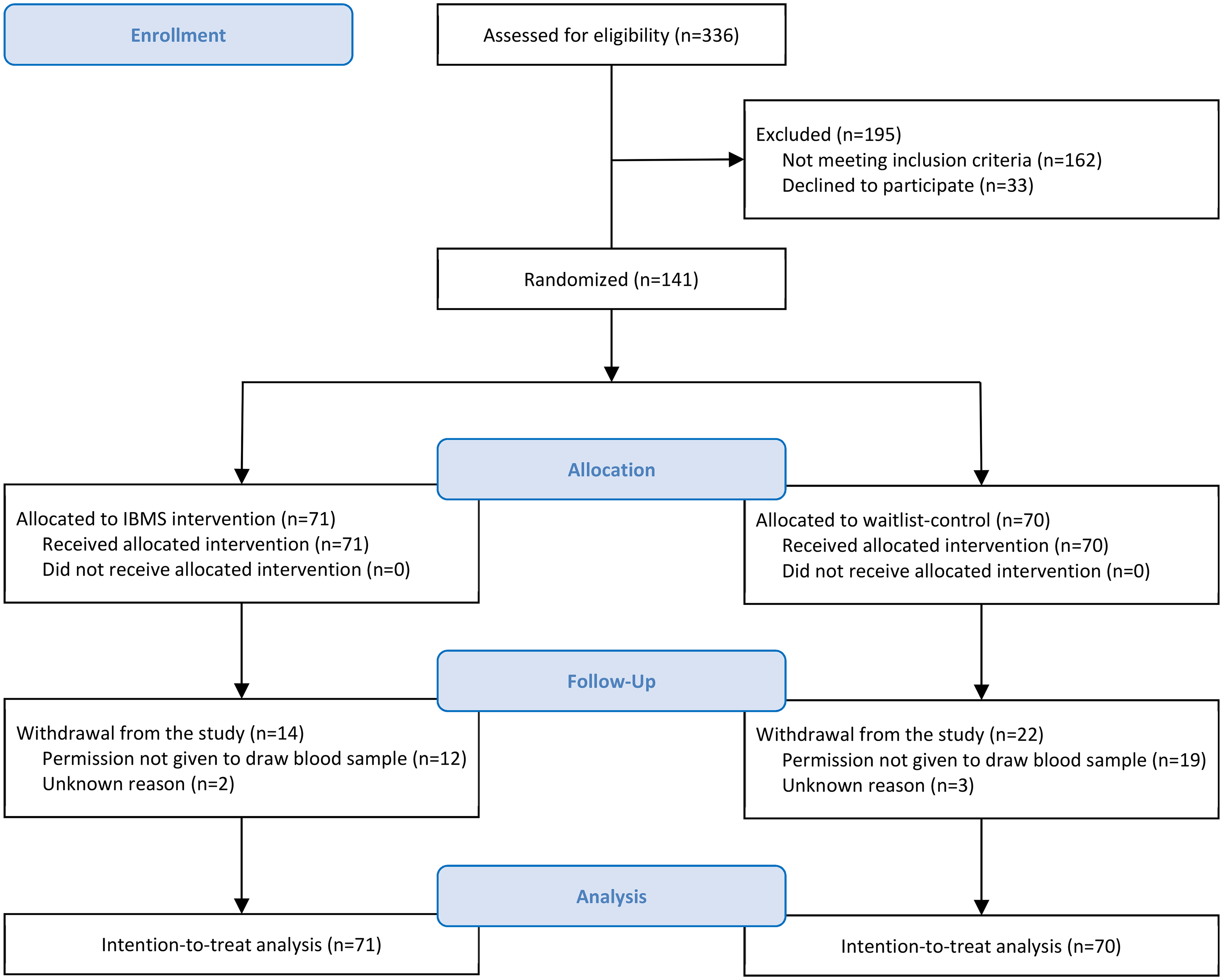
CONSORT flow diagram.
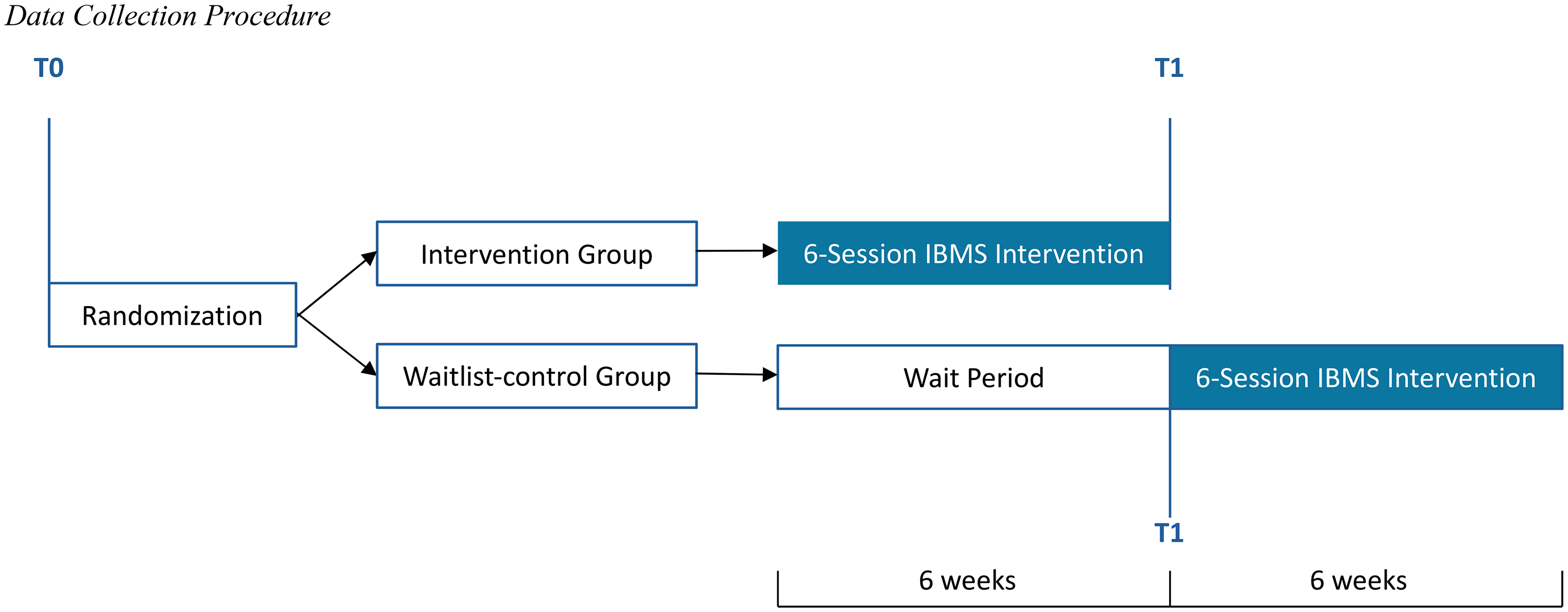
Data collection procedure.
Children assigned to the IBMS intervention group began the 6-week group program one week after randomization. At the conclusion of the program, they completed follow-up assessments (T1), including psychosocial questionnaires and a second round of blood sampling for cytokine analysis. Children in the waitlist-control group underwent the same follow-up assessments (T1) 6 weeks after randomization, immediately prior to beginning the delayed IBMS intervention. All blood samples were processed and analyzed in the laboratory by researchers blinded to participant groupings.
Intervention Design: The IBMS Psychosocial Group Intervention
The IBMS psychosocial group intervention is a structured, theory-driven program developed by the first author and her research team, with its conceptual foundation and clinical applications well established across diverse health and mental health populations (e.g., Chan et al., 2001; Chan, Ji, Chan et al., 2017; Xie, Chan, Lau et al., 2020). The current version was adapted from these validated IBMS protocols to address the developmental needs of children with eczema and their families. It was designed to promote holistic well-being by fostering balance across the domains of body, mind, and spirit, and to enhance children's physical awareness, emotional regulation, and meaning-making in relation to their experience of AD. The intervention manual was refined by a multidisciplinary research team, including pediatricians and social workers trained in the IBMS model, and pretested in pilot sessions with a small group of children to ensure developmental appropriateness and cultural relevance prior to full implementation.
The finalized program was delivered in 6 weekly group sessions, each lasting approximately 3 hours, to small groups of 8–10 children. Each session followed a developmentally appropriate protocol guided by clearly defined therapeutic goals. The sessions used interactive, experiential methods, including guided imagery, creative expression (e.g., art and storytelling), somatic awareness exercises, and peer-based activities to encourage self-discovery and emotional processing. Table 2 illustrates the six-session protocol progressed through IBMS core thematic components.
Integrative Body–Mind–Spirit (IBMS) Intervention Session Framework.

Fidelity and Trial Monitoring
To ensure the quality and consistency of the intervention, multiple strategies were implemented to monitor fidelity and trial procedures. All sessions were delivered by social workers trained and certified in the IBMS psychosocial group intervention model. Prior to the study, group facilitators participated in structured training that covered the model's theoretical foundations, delivery protocols, and session-specific techniques. A comprehensive facilitator manual outlining standardized procedures, session goals, therapeutic activities, and timing was provided to support adherence to the intervention protocol.
Throughout the trial, group facilitators completed structured checklists after each session to document which components were delivered and whether any deviations occurred. Session logs were also maintained to record qualitative observations, participant engagement, and any implementation challenges. Weekly supervision meetings were held with the principal investigator to review fidelity reports and address procedural concerns. In addition, random sessions were observed by independent researchers not involved in the intervention delivery, using a standardized observation form to assess adherence and facilitator competence. A trial coordinator was responsible for overseeing scheduling, data collection, and participant follow-up, as well as for documenting any protocol deviations or adverse events. These layered strategies ensured rigorous implementation of the intervention and enhanced the internal validity and reproducibility of the study.
Measurement
Biological Outcomes
Inflammatory Cytokines
To assess immune function, inflammatory cytokine levels were measured from whole blood serum samples collected after overnight fasting. A 5 ml venous blood sample was drawn into EDTA-containing vacutainer tubes (BD Vacutainer PLUS, Franklin Lakes, NJ), centrifuged at 1,000 g for 15 min at 4 °C, and the resulting supernatant stored at −80 °C until analysis. Cytokine concentrations (IL-6, IL-1β, IL-4, IL-10, and TGF-β1) were quantified using the BD Cytometric Bead Array system (BD Biosciences Pharmingen, USA). The latent form of TGF-β1 was activated and assessed using a single-plex assay, while the remaining cytokines were analyzed using a multiplex format, following the manufacturer's protocols.
Clinical Outcomes
Atopic Dermatitis Symptom Severity
Children's eczema severity was assessed using the Patient-Oriented Eczema Measure (POEM) – Parent Form, a 7-item scale evaluating the frequency of AD symptoms over the past week (Charman et al., 2004; Charman et al., 2013). Each item is scored on a 5-point Likert scale ranging from 0 (no days) to 4 (every day), with higher scores indicating more severe symptoms. It has demonstrated sufficient internal consistency, with Cronbach's alpha ranging from 0.86 to 0.88 (Li et al., 2022).
Psychosocial Outcomes
Quality of Life
The Children's Dermatology Life Quality Index (CDLQI) was used to assess the impact of skin conditions on quality of life over the previous week. This 10-item self-report measure evaluates the effects of AD on emotions, school, leisure, relationships, sleep, and treatment burden (Lewis-Jones & Finlay, 1995). Items are scored on a 4-point Likert scale (0 = not at all to 3 = very much). The Chinese version of the CDLQI shows good psychometric properties, with Cronbach's alpha for internal consistency of 0.83 (Chuh, 2003).
Physical and Emotional Impact
The Pediatric Allergic Disease Quality of Life Questionnaire (PADQLQ) is a 26-item self-administered scale measuring the health-related impact of allergic disease on the child's quality of life over the past week (Roberts et al., 2003). It consists of three elements: activity problems (8 items), symptoms (15 items), and emotional problems (3 items). Each item is rated on a 7-point Likert scale, ranging from 0 (not troubled) to 6 (extremely troubled). The Chinese version of the PADQLQ has demonstrated high internal consistency, with a Cronbach's alpha of 0.92 (Ng et al., 2011).
Statistical Analysis
Between-group differences in participant characteristics were examined using Mann–Whitney U tests for continuous outcomes and chi-square tests for categorical variables. Cytokine data were log-transformed. Efficacy analyses followed an intention-to-treat (ITT) approach, with last observation carried forward for missing data. Within-group differences were compared using paired-sample t-tests for normally distributed outcomes and Wilcoxon signed-rank tests for non-normally distributions. Linear mixed models (LMM) were used to estimate between-group differences in normally distributed outcomes, and generalized LMMs (GLMM) were applied for non-normally distributed outcomes. Both models included random intercepts and used restricted maximum likelihood estimation to assess changes in continuous outcomes from baseline to postintervention.
The main effect of the intervention, assessment time point, and their interaction effect (group × time) were treated as fixed effects, with repeated measures treated as random effects. Cohen's d was calculated by dividing the mean chance difference in change by the pooled standard deviation. As LMM and GLMM followed ITT principles, they accommodated missing data without with need for explicit imputation. Statistical significance was set at a two-sided p < 0.05.
Children's ages (below 10 years old/ 10 years or above), gender (male/female), and the ratio of IL-6/IL-4 were considered as subgroups. The ratio of pro-inflammatory and anti-inflammatory cytokines is commonly used to indicate an individual's inflammation balance (Kim et al., 2007; Liang et al., 2015). Following Gori et al. (2009), children were categorized into three subgroups based on IL-6/IL-4 ratios: (1) “Anti-” subgroup where IL-6 was in the first or second tertile, and IL-4 was in the third tertile indicating that the anti-inflammatory cytokine IL-4 overwhelmed the pro-inflammatory cytokine IL-6; (2) “Balance” subgroup where IL-6 and IL-4 were in the same tertile; and (3) “Pro-” subgroup where IL-6 was in the third tertile, and IL-4 was in the first or second tertile indicating that the pro-inflammatory cytokine IL-6 was not compensated by the anti-inflammatory cytokine IL-4. Generalized LMM was used for subgroup analysis, with a significant subgroup interaction effect defined at p < 0.1 (van Oers et al., 2016). All analyses were conducted using SPSS (version 23.0), and R Studio was used to generate a forest plot for subgroup analysis on cytokine balance changes.
Adverse Events
No serious adverse events were reported during the study.
Results
Baseline Characteristics
Of the 141 children that participated in the study, 58 in the intervention group and 56 in the waitlist-control group completed the post-intervention assessment (T1). Blood serum samples were successfully collected from 57 children in the intervention group and 43 children in the waitlist-control group 6 weeks after their baseline assessment. The mean age of children was 8.10 years (SD = 1.87), with 53.9% as female. Nearly half (46.1%) reported experiencing AD symptoms for more than one year. According to POEM scores, 65.3% of participants presented with moderate to severe eczema. Baseline comparisons revealed no significant group differences in demographic or clinical characteristics (all p > 0.05), as summarized in Table 3.
Characteristics of the Child Participants.
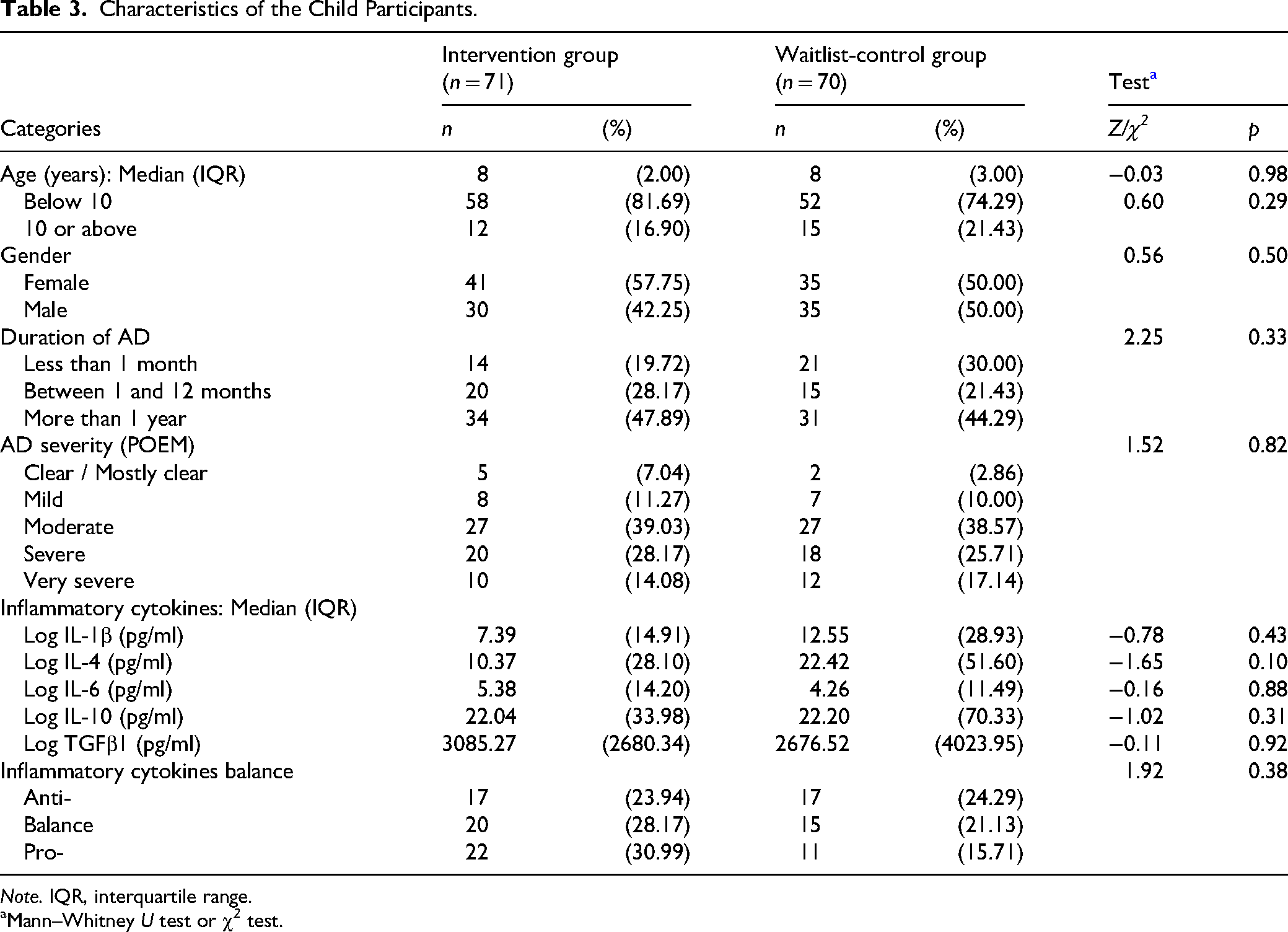
Note. IQR, interquartile range.
Mann–Whitney U test or χ 2 test.
Effects of the IBMS Intervention on Inflammatory cytokines and cytokine ratios
Hypothesis 1 stated that children who receive the IBMS intervention will show a significantly greater reduction in pro-inflammatory cytokines (IL-6, IL-1β, and IL-4) than those in the waitlist control group. Within-group analyses revealed significant reductions in pro-inflammatory cytokines among children who received the IBMS intervention after the 6-week program, including IL-1β (p < 0.01) and IL-6 (p < 0.05) (Table 4). The IL-4 levels remained stable across the intervention period. In contrast, the waitlist-control group showed a modest reduction only in IL-1β (p < 0.05), with no significant changes detected in in other cytokines. However, no statistically significant Group × Time interaction effects were observed for any individual cytokines, indicating that between-group differences in cytokine trajectories did not reach statistical significance over the intervention period. Therefore, Hypothesis H1 is only partially supported: the intervention significantly reduced IL-1β and IL-6 within the intervention group, but not IL-4, and between-group changes did not reach statistical significance.
Integrative Body–Mind–Spirit (IBMS) Group Intervention Efficacy (Intention-to-Treat Analysis).
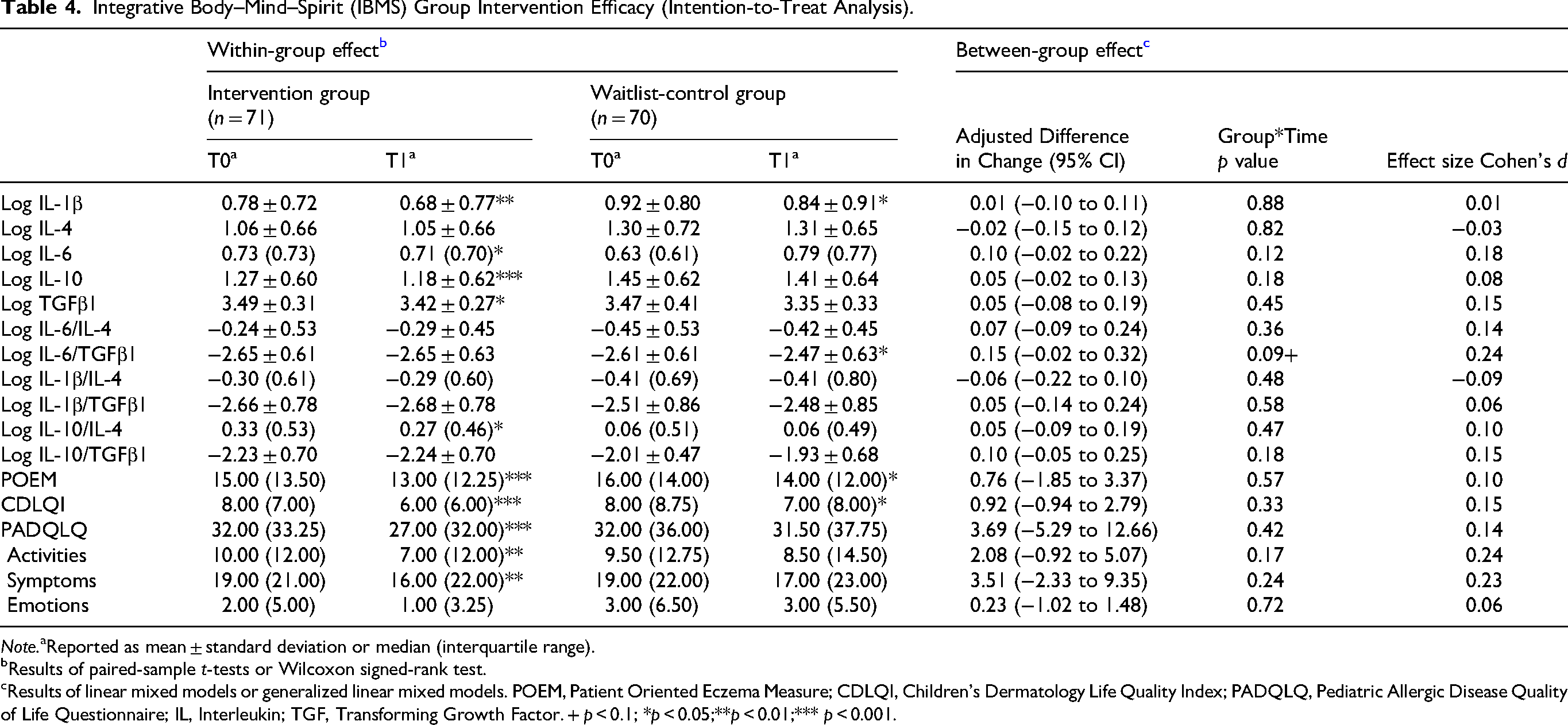
Note.aReported as mean ± standard deviation or median (interquartile range).
Results of paired-sample t-tests or Wilcoxon signed-rank test.
Results of linear mixed models or generalized linear mixed models. POEM, Patient Oriented Eczema Measure; CDLQI, Children's Dermatology Life Quality Index; PADQLQ, Pediatric Allergic Disease Quality of Life Questionnaire; IL, Interleukin; TGF, Transforming Growth Factor. + p < 0.1; *p < 0.05;**p < 0.01;*** p < 0.001.
Hypothesis 2 posited that children in the IBMS intervention group will show a significantly greater increase in anti-inflammatory cytokines (IL-10 and TGF-β1) compared to the waitlist control group. Contrary to the hypothesized increase, both anti-inflammatory cytokines showed reductions in the IBMS intervention group: IL-10 (p < 0.001) and TGF-β1 (p < 0.05). The waitlist-control group showed no significant changes in either IL-10 or TGF-β1. No statistically significant Group × Time interaction effects were observed for IL-10 or TGF-β1, indicating that between-group differences did not reach statistical significance. Therefore, Hypothesis H2 was partially supported.
Hypothesis 3 predicted that the IBMS intervention would produce a more favorable balance between pro- and anti-inflammatory cytokines relative to the waitlist control group. To further assess immune modulation, cytokine ratios were examined as indicators of the balance between pro- and anti-inflammatory activities. Within-group analyses showed that the IL-10/IL-4 ratio significantly increased in the IBMS group (p < 0.05), reflecting a shift toward a more anti-inflammatory profile. In contrast, the waitlist-control group showed a significant increase in the IL-6/TGF-β1 ratio (p < 0.05), suggesting a pro-inflammatory trajectory. Between-group comparisons revealed a marginally significant Group × Time interaction effect for the IL-6/TGF-β1 ratio (p = 0.09), with the ratio rising in the control group but remaining stable in the intervention group (Table 4). Although this trend suggests a potential protective immunoregulatory effect of the IBMS intervention on cytokine balance, the difference did not reach conventional levels of statistical significance (p < 0.05). Hypothesis 3 received partial support.
Improvements in Clinical Symptoms and Quality of Life
Hypothesis 4 stated that the IBMS intervention will result in a significantly greater reduction in AD symptom severity than the waitlist control group. Significant within-group improvements in AD symptom severity were observed in the IBMS intervention group (POEM, p < 0.001). The waitlist-control group also showed modest improvements in POEM (p < 0.05). Despite the substantial within-group improvement in the intervention group, no significant Group × Time interaction effect was found for POEM, indicating that the improvement in AD symptom severity, while evident within the intervention group, was not statistically greater than that observed in the control group over the same period. Hypothesis 4 was therefore partially supported.
Hypothesis 5 predicted that children receiving the IBMS intervention will report significantly greater improvements in quality of life and physical and emotional impacts compared to those in the waitlist control group. Significant within-group improvements were observed in the IBMS intervention group for dermatology-specific quality of life (CDLQI, p < 0.001) and allergy-related quality of life (PADQLQ: Total p < 0.001; Activities p < 0.01; Symptoms p < 0.01). The waitlist-control group showed modest improvements in CDLQI (p < 0.05), but no significant changes in any PADQLQ domains. No significant Group × Time interaction effects were found for any of the quality of life outcomes (CDLQI or PADQLQ domains). Hypothesis 5 was therefore partially supported.
Subgroup Analysis on the Intervention Effects
Cytokine Response by Inflammatory Balance Subgroup
Subgroup analyses revealed differential patterns of change in inflammatory markers based on baseline cytokine profiles, gender, and age (Table 5). It is important to note that Table 5 presents only statistically significant subgroup findings. A significant three-way interaction (group × time × balance-subgroup) was observed for TGF-β1 levels (p = 0.022), indicating variation in response depending on cytokine balance profiles. Children in the “balance” subgroup who received the IBMS intervention exhibited a slight but stable TGF-β1 level (M = 3.47 to 3.42), whereas the same subgroup in the waitlist-control group showed a significant reduction (M = 3.67 to 3.36, p < 0.01), contributing to a differential group effect.
Subgroup Analysis on the Intervention Effects.
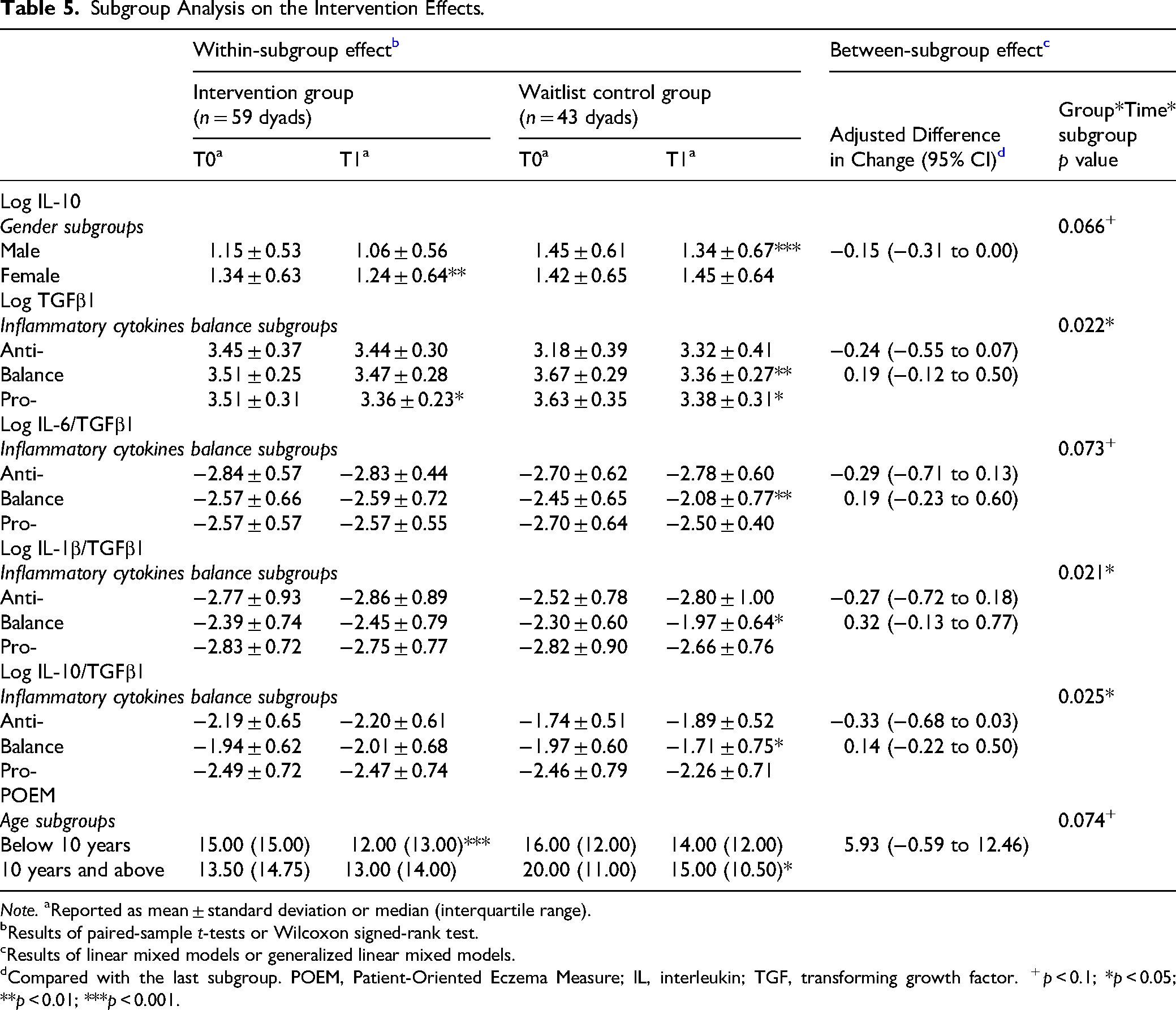
Note. aReported as mean ± standard deviation or median (interquartile range).
Results of paired-sample t-tests or Wilcoxon signed-rank test.
Results of linear mixed models or generalized linear mixed models.
Compared with the last subgroup. POEM, Patient-Oriented Eczema Measure; IL, interleukin; TGF, transforming growth factor. + p < 0.1; *p < 0.05; **p < 0.01; ***p < 0.001.
In addition, significant group × time × subgroup interactions were found for cytokine ratios including IL-1β/TGF-β1 (p = 0.021), IL-10/TGF-β1 (p = 0.025), and marginally for IL-6/TGF-β1 (p = 0.073). For the balance subgroup, the intervention group demonstrated significant reductions in both IL-1β/TGF-β1 and IL-6/TGF-β1, while these ratios significantly increased in the control group. The pooled standardized mean difference across these three ratios in the balance subgroup was −0.37 (95% CI: −0.61 to −0.12), reflecting a moderate immunomodulatory effect (Figure 3).
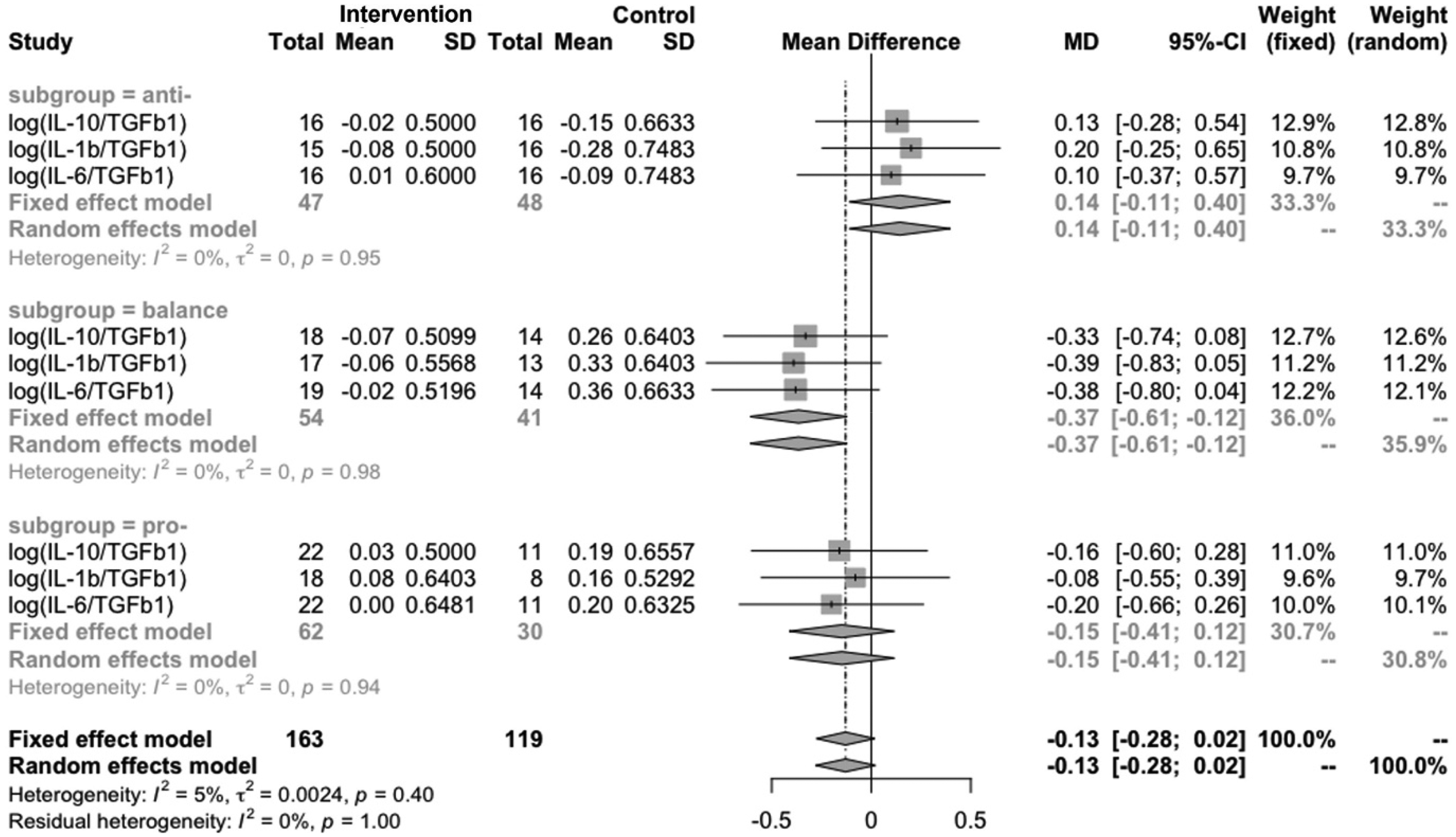
Forest plot of subgroup analysis on the balance change of inflammatory cytokines.
Gender Differences in Cytokine Response
A marginally significant group × time × gender interaction was detected for IL-10 (p = 0.066). Girls in the intervention group showed a significant decline in IL-10 levels (M = 1.34 to 1.24, p < 0.01), while boys in the control group experienced a significant increase (M = 1.45 to 1.34, p < 0.001), suggesting gender-specific response patterns.
Age Differences in Clinical Outcomes
Lastly, for clinical outcomes, a marginally significant interaction (p = 0.074) was observed in POEM scores by age subgroup. Children under 10 years old in the intervention group exhibited greater symptom improvement (M = 15.00 to 12.00, p < 0.001) compared to older children (M = 13.50 to 10.50, p < 0.05), indicating potentially greater responsiveness among younger participants.
Discussion
This randomized waitlist-controlled trial is among the first to evaluate a psychosocial intervention for children with AD using both biological (inflammatory cytokines) and psychosocial outcomes. The study extends beyond traditional symptom management by incorporating immunological markers indicators of intervention effects. This dual-outcome approach offers robust evidence for the potential of the psychosocial group intervention to influence underlying physiological processes associated with AD in children. Notably, children in the intervention group demonstrated significant within-group reductions in pro-inflammatory cytokines and improvements in clinical and QoL measures. These results highlight the relevance of integrative, psychosocial models in addressing both the physical and emotional burden of pediatric AD and support the inclusion of biomarker-based outcomes in future child-focused intervention research.
Modulation of Inflammatory Markers
Within-group analyses revealed significant reductions in the pro-inflammatory cytokines IL-1β and IL-6 among children who participated in the IBMS intervention, both of which are known to play pivotal roles in the pathophysiology of eczema (Huang et al., 2023; Yeung et al., 2021). These reductions suggest a downregulation of inflammatory pathways, potentially contributing to symptom improvement. These findings are consistent with earlier research showing that psychosocial interventions can attenuate systemic inflammation by modulating stress-related immune pathways (Ng et al., 2022; Witek-Janusek et al., 2008). However, IL-4, another cytokine central to Th2-mediated allergic responses, did not change significantly in either group, and between-group differences in individual cytokine changes were not statistically significant over the 6-week period. This suggests that while the IBMS intervention may modulate specific inflammatory markers such as IL-1β and IL-6 within participants, these effects did not translate into significant between-group differences within the study time frame.
With regard to anti-inflammatory markers, a significant within-group reduction in IL-10 levels was observed in the intervention group. While IL-10 is typically characterized as an anti-inflammatory cytokine, its elevated presence in AD is often interpreted as a compensatory response to sustained immune activation (Lesiak et al., 2014). Thus, the observed reduction may reflect a normalization of immune regulation, indicative of decreased inflammatory burden rather than a loss of immunosuppressive control. TGF-β1, another critical cytokine involved in immune tolerance and regulatory function, did not show a significant increase over the intervention period in either group. Furthermore, no significant between-group differences were found in IL-10 or TGF-β1. Taken together, these results suggest that while the intervention may have contributed to immunomodulatory shifts within the intervention group, it did not produce significantly greater increases in anti-inflammatory cytokines relative to the control group.
Modulation of Cytokine Ratios and Immune Balance
Between-group comparisons offer the most compelling evidence of intervention effectiveness. In this study, a marginally significant Group × Time interaction was observed for the IL-6/TGF-β1 ratio (p = 0.09), which increased significantly in the waitlist-control group but remained stable in the intervention group. This emerging divergence suggests a potential protective effect of the IBMS intervention in preserving immune balance over the course of the program. The IL-6 is a pro-inflammatory cytokine closely linked to systemic inflammation and disease activity in AD, while TGF-β1 serves a key role in maintaining immune regulation and tolerance. An increased IL-6/TGF-β1 ratio is frequently associated with chronic inflammatory states, highlighting the clinical importance of this trend. Findings indicated that children in the waitlist-control group exhibited a significant rise in this ratio, suggesting a shift toward a pro-inflammatory immune profile. Although the between-group difference did not reach conventional significance, the consistency in direction and significant within-group change in the control group reinforce the relevance of this pattern.
These findings are particularly notable given that IL-6 and TGF-β1 together represent a critical axis of immune balance, and their ratio is increasingly recognized as a sensitive biomarker for immune dysregulation in allergic and inflammatory diseases. That the intervention group maintained stability in this ratio over time suggests that the IBMS program may buffer against inflammatory escalation and help sustain immune homeostasis in children with AD. This interpretation aligns with prior evidence that psychosocial factors, particularly stress reduction and improved emotion regulation, can modulate cytokine activity and immune signaling (Kim et al., 2007; Liang et al., 2015).
Another indicator of immune modulation, the IL-10/IL-4 ratio, also demonstrated favorable changes in the intervention group, with a significant within-group increase observed post-intervention. The IL-10 is a regulatory cytokine that suppresses Th2-driven inflammation, while IL-4 promotes allergic responses characteristic of AD. An increased IL-10/IL-4 ratio thus reflects a shift away from Th2 dominance and toward immune regulation. While between-group differences for this ratio were not statistically significant, the improvement in the intervention group, alongside a stable trajectory in the control group, provides further support for Hypothesis H3: that the IBMS intervention enhances the balance between pro- and anti-inflammatory cytokines. Together, these ratio-based findings emphasize the biological plausibility of psychosocial interventions in influencing immune homeostasis through holistic, stress-informed mechanisms.
Quality of Life Improvements with IBMS Intervention
Significant within-group improvements across all measured quality-of-life indices, including POEM, CDLQI, and PADQLQ (Activities and Symptoms domains), highlights the psychosocial and clinical benefits of the IBMS intervention. The POEM reductions were particularly notable (p < 0.001), suggesting meaningful improvements in day-to-day eczema symptoms. While the waitlist-control group also exhibited a modest decrease in POEM (p < 0.05), the magnitude of change was substantially smaller, and between-group differences did not reach statistical significance.
These trends are consistent with Hypothesis H5, which proposed that IBMS would enhance quality of life more effectively than usual care. The CDLQI and PADQLQ improvements in the intervention group reflect a broader enhancement in well-being, not limited to physical symptoms but encompassing social, emotional, and activity-related functioning. By fostering awareness of body-mind interconnectedness, the IBMS framework equips children with practical tools to recognize and manage emotional triggers that exacerbate eczema. These techniques may underlie the observed improvement in psychosocial domains of PADQLQ and CDLQI. This interpretation aligns with prior evidence suggesting that psychological interventions can significantly enhance dermatology-related quality of life in children with AD (Hua et al., 2023; Xie, Chan, Lau et al., 2020).
Biologically, the observed improvements may be interpreted through the framework of the Psycho-Neuro-Endocrine-Immunology model, which highlights the potential interplay between psychological processes, neuroendocrine function, and immune responses. Chronic psychological stress is known to activate the hypothalamic–pituitary–adrenal (HPA) axis, leading to the release of glucocorticoids that may influence both immune activity and skin barrier function (França & Lotti, 2017). Additionally, stress-induced neuroendocrine alterations have been implicated in cytokine dysregulation and impaired skin homeostasis in inflammatory skin conditions (Jafferany & Patel, 2020). While direct mechanistic pathways were not assessed in this study, the IBMS intervention's emphasis on emotional regulation and stress reduction may have contributed indirectly to improvements in skin-related outcomes by reducing psychophysiological stress responses. These interpretations remain speculative but are consistent with emerging biopsychosocial models of chronic inflammatory disease.
Improvements observed in the waitlist-control group, particularly in POEM and CDLQI, may partly reflect seasonal and contextual influences rather than intervention effects. The study was conducted during the summer holiday period in Hong Kong, a time when children typically experience reduced academic stress and greater flexibility in daily routines, both of which can facilitate better symptom management. Additionally, participation in the study may have heightened caregiver and child awareness of symptoms, promoting increased self-monitoring and adherence to existing medical treatment protocols. These factors could have contributed to observed gains even in the absence of psychosocial intervention. Such findings highlight the importance of accounting for external environmental and behavioral influences when interpreting outcomes in psychosocial intervention research.
Differential Cytokine Responses by Baseline Immune Balance
Subgroup analysis based on baseline cytokine balance revealed that children with moderately immune profiles (i.e., the “balanced” subgroup) exhibited more favorable immunological responses to the IBMS intervention. This suggests that a relatively stable immune environment may enhance receptivity to psychosocial and mind–body therapies. Specifically, the maintenance of TGF-β1 levels and reduction in pro-/anti-inflammatory cytokine ratios, such as IL-1β/TGF-β1, IL-6/TGF-β1, IL-10/TGF-β1, point to a stabilizing effect on immune regulation. These effects were observed exclusively in the intervention group and suggest that the IBMS program may support homeostatic immune functioning, particularly in children whose immune systems retain adaptive flexibility.
This pattern aligns with emerging evidence that therapeutic responsiveness is partially conditioned by initial immune plasticity, where individuals with moderate levels of baseline inflammation are more amenable to intervention-induced modulation (Marsland et al., 2017). In contrast, children in the control group with comparable immune profiles showed significant increases in the same cytokine ratios, indicating a shift toward heightened inflammatory activity. Such divergence highlights the potential risks of leaving psychosocial stress and low-grade inflammation unaddressed.
These findings highlight the importance of accounting for baseline inflammatory status when evaluating intervention effects. They also suggest that integrative approaches like IBMS may be particularly effective for children with balanced or subclinical immune dysregulation. The consistent changes across multiple cytokine ratios provide converging evidence for the IBMS intervention's capacity to influence systemic immune balance, especially in those not yet exhibiting chronic immune dysfunction.
Gender-Specific Responses to the IBMS Intervention
The observed reduction in IL-10 among girls in the IBMS intervention group points to potential gender-based differences in biological sensitivity to psychosocial interventions. Girls in the IBMS intervention group demonstrated a significant reduction in IL-10, a cytokine often elevated in chronic inflammatory states as a compensatory mechanism. Its significant decline may indicate a more effective resolution of inflammation in girls, possibly due to greater responsiveness to emotional and physiological stress regulation. This is consistent with research showing that females tend to exhibit stronger reactivity in neuroendocrine-immune pathways, including heightened HPA axis activity and emotional processing under stress (Goel et al., 2014; Heck & Handa, 2019).
Moreover, previous studies suggest that girls generally experience more intense emotional and physiological stress responses compared to boys, which may make them more susceptible to stress-related immune dysregulation (Ordaz & Luna, 2012; Roberts & Lopez-Duran, 2019; Verma et al., 2011) and, in turn, more responsive to psychosocial stress-reducing interventions. Such heightened sensitivity may contribute to more pronounced immune modulation when stress is reduced, as in psychosocial group settings.
In contrast, boys in the control group experienced a significant increase in IL-10, which may reflect heightened immune activation in the absence of targeted stress regulation. This pattern could suggest that without structured psychosocial support, boys may be more vulnerable to persistent or reactive inflammatory processes. These findings point out the importance of considering gender as a potential moderating factor in the design of psychosocial interventions. Tailoring content and delivery to address sex-specific developmental and emotional needs may improve intervention effectiveness, particularly during critical periods of biopsychosocial development in childhood.
Age-Related Variations in Clinical Improvement
Age appeared to moderate the effectiveness of the IBMS intervention, with children under 10 years showing significantly greater reductions in AD symptom severity. Younger children may be more receptive to psychosocial and mind–body strategies, as they are still developing core emotional regulation skills and stress–response patterns. Interventions like IBMS, which promote body–mind wellness and emotional literacy, may therefore be particularly impactful during this developmental stage.
Moreover, previous studies suggest that AD severity tends to increase with age, making symptom management more challenging for older children (Ballardini et al., 2013; Evans et al., 2013; Silverberg & Simpson, 2014; Yang et al., 2020). The relatively milder or more adaptable immune profiles of younger children may enhance their responsiveness to psychosocial modulation. These findings underscore the value of introducing integrative approaches early in the course of chronic disease management, when children may be more open to acquiring adaptive coping mechanisms and behavior change.
Discussion and Applications to Practice
The findings support translating the IBMS model into a concise, manualized toolkit for child- and family-focused social work practice, detailing core elements (e.g., emotion-regulation practices, somatic-awareness exercises, meaning-making activities), practical session guides, caregiver take-home practice sheets, and a brief training-and-fidelity checklist for frontline staff working with children with AD. Such a toolkit would enable consistent delivery across clinic and community settings while preserving flexibility for developmental adaptation.
Culturally sensitive implementation requires careful consideration of the intervention context, particularly the alignment between family-centered values prevalent in East Asian settings (such as collective decision-making, defined family roles, and somatic expressions of distress) and Western-derived skills training approaches (such as cognitive reframing and explicit emotion-coaching). The goal is to ensure that interventions honor family norms while promoting transferable self-regulation skills. In practice, this entails codesigning materials with families and offering flexible delivery formats to enhance acceptability, feasibility, and equity across diverse service environments.
Study Limitations
This study has several limitations that warrant consideration. First, assessments were limited to baseline and postintervention time points to minimize delays for the waitlist-control group, who subsequently received the IBMS intervention. While this design facilitated ethical access to treatment, it restricts conclusions about the durability of intervention effects. Without longer-term follow-up, it remains unclear whether observed improvements in symptom severity, quality of life, or immune balance are sustained over time. Future studies should incorporate multiple follow-up assessments to evaluate the longevity of intervention benefits.
The absence of an active control condition limits interpretability of the IBMS intervention's specific effects. Nonspecific therapeutic factors such as group interaction, attention from facilitators, and expectancy effects may have contributed to positive outcomes in both groups. Participants may also have modified behavior due to increased symptom awareness or self-monitoring. Including an active comparison group such as psychoeducation or supportive therapy, would strengthen causal inferences regarding the unique efficacy of the IBMS model by controlling for these common intervention effects.
One notable limitation concerns the timing and setting of the intervention, which was conducted during the summer holiday period. This scheduling allowed children to attend six 3-h sessions with fewer academic demands, reducing stress and improving attendance and engagement. Additionally, the warm and humid climate in Hong Kong during summer may have contributed to more favorable skin conditions due to warm and humid climate, which could support symptom improvement independently of the intervention. These contextual factors may partly explain the significant within-group improvements observed in the intervention group. However, they also limit the generalizability of findings to other times of the year, particularly during school terms when children face increased stress and environmental conditions are less supportive of symptom control.
A final limitation is self-selection bias arising from our use of convenience sampling. Consequently, our findings are most applicable to children living in social contexts similar to Hong Kong (e.g., Mainland China and Singapore), which limits their generalizability. Future research should employ more rigorous, probability-based sampling strategies to evaluate the intervention's effectiveness across diverse cultural settings.
While this study included several important immune markers, it did not capture the full complexity of immune processes in AD. Key cytokines like IL-33, which play multiple roles in allergic inflammation and skin signaling, were not assessed. Including a broader range of markers would offer a more complete understanding of how psychosocial interventions like IBMS affect immune function. Additionally, cytokine levels were measured only at baseline and immediately post-intervention, which may have missed important fluctuations. Immune responses can change quickly or emerge over time, so future studies should include multiple time points to better track how immune markers respond throughout and after the intervention.
Conclusion
This study provides evidence that the IBMS psychosocial group intervention may contribute to improvements in both clinical symptoms and systemic immune balance among children with AD. Significant within-group reductions were observed in key pro-inflammatory cytokines (IL-1β and IL-6), alongside shifts in cytokine ratios such as IL-10/IL-4 and IL-6/TGF-β1. These patterns suggest that psychosocial interventions may support immunological regulation and symptom management in pediatric populations. Although between-group effects were limited, with only one cytokine ratio showing a marginally significant difference, the directionality and internal consistency of findings offer support for the potential benefits of psychosocial interventions.
By incorporating immune biomarkers alongside clinical and quality of life outcomes, this study contributes to advancing evidence-based psychosocial research. It supports the growing recognition that psychosocial interventions can have measurable physiological effects and that immune markers can serve as valuable tools for evaluating therapeutic impact beyond self-reported outcomes. The findings also indicate that individual factors such as baseline immune profile, age, and gender may shape intervention responsiveness. This highlights the importance of considering developmental and biological context in the design and evaluation of psychosocial interventions for pediatric populations. Further research using broader immune profiling, multiple follow-up points, and active control conditions is recommended to clarify the mechanisms involved and to strengthen the empirical foundation for biologically informed psychosocial care in chronic pediatric conditions like AD.
Footnotes
ORCID iDs
Ethical Considerations
The trial adhered to CONSORT guidelines for randomized studies and was approved by the Human Research Ethics Committee of the University of Hong Kong (EA1612023). It was registered at the Hong Kong Clinical Trials Registry [HKUCTR-2234].
Consent to Participate
Written consent for participation and publication was obtained from the participants and their guardians.
Authors’ Contributions
Celia Hoi-Yan Chan developed the conceptualization and design of the study. Celia Hoi-Yan Chan, Margaret Xi-Can Yin, and Dan-Yuan Guo wrote the original draft, with Celia Hoi-Yan Chan also leading the review and editing. Celia Hoi-Yan Chan, Yat Lui Fung, Kam Lun Ellis Hon, and Cecilia Lai Wan Chan contributed to the study methodology. Kam Lun Ellis Hon, David Chi Kong Lu, James Wesley Ching Hei Cheng, Ashleigh Ka Ying Chu, Michelle Yuk Ping Tsang, and Sam Ying Yin Lam carried out patient screening and blood serum collection. Yat Lui Fung, Christina Sum Yi Ng, Martha Sin Ki Luk, Georgina Yuen Kiu, Priscilla Sin Ying Chau, Irene Kit Man Cheung, and Cecilia Lai Wan Chan conducted intervention groups. Margaret Xi-Can Yin, Charmaine Jeng Sum Kung, Victoria Ka Ying Hui, and Irene Kit Man Cheung performed data analysis. Celia Hoi-Yan Chan and Margaret Xi-Can Yin managed data curation. Celia Hoi-Yan Chan and Yat Lui Fung supervised the study. Celia Hoi-Yan Chan and Cecilia Lai Wan Chan acquired funding.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research study was funded by UBS Optimus Foundation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data cannot be shared openly but is available on request from authors.
