Abstract
Keywords
Approximately 15.4 million adults in the United States have a serious mental illness (SMI, e.g., schizophrenia, bipolar, major depression; SAMHSA, 2023). People with SMI experience health inequities that result in a 25-year mortality gap with the general population, and diseases related to tobacco use are the leading cause of death (Correll et al., 2022; Walker et al., 2015). While smoking cigarettes decreased in the general population from 42% in 1964 to 11.5% in 2021, the prevalence among people with SMI has remained around 40% to 80%, depending on the sample and psychiatric diagnosis (Callaghan et al., 2014; Cornelius et al., 2023; Dickerson et al., 2018). In addition to the physical health risk posed by smoking, people with SMI have a higher risk of cardiovascular disease (CVD) due to the cardiometabolic effects of antipsychotic medications (e.g., weight gain), high-calorie, low-nutrient diets, and low levels of physical activity (Firth et al., 2019; Hawes et al., 2020).
Healthy lifestyle interventions are one approach used to reduce the prevalence of chronic health conditions in people with SMI (Naslund et al., 2017). These interventions offer a package of services aimed at improving physical activity, dietary habits, and engagement in health-promoting activities (Cabassa et al., 2010). Evidence indicates that 30% to 40% of people with SMI participating in healthy lifestyle interventions can achieve clinically significant weight loss (i.e., 5% weight loss from baseline; Cabassa et al., 2010; Naslund et al., 2017). Most of these interventions do not provide smoking cessation treatment (Cabassa et al., 2010; Naslund et al., 2017). While these interventions offer promise in helping people with SMI improve their health, a significant portion of participants still do not achieve clinically significant weight loss (Speyer et al., 2019). Understanding which subgroups of people with SMI are not benefiting clinically from these interventions is crucial for developing tailored interventions that meet the needs of all participants.
Studies examining subgroup differences among people with SMI participating in healthy lifestyle interventions are limited, and the findings are inconsistent. For example, Alexander et al. (2019) examined whether the effectiveness of a healthy lifestyle intervention for people with SMI in community psychiatric rehabilitation programs differed by subgroups (e.g., age, psychiatric diagnosis, history of substance use) and did not find any differences. Conversely, the parent study of the current research, a peer-led healthy lifestyle intervention (PGLB) for people with SMI living in supportive housing, examined whether age, race/ethnicity, or gender moderated the effectiveness of the intervention and found that the intervention was most effective for participants aged 49 and younger and for racial/ethnic minoritized participants (e.g., Black, Latinx; Cabassa et al., 2022).
While these studies provide important information about who benefits from healthy lifestyle interventions, gaps remain. No study to date has examined whether tobacco use moderates the effectiveness of healthy lifestyle interventions among people with SMI living in permanent supportive housing (PSH). Smoking cigarettes could impact the intervention in several ways. Tobacco use among people with SMI is associated with higher mental illness severity, more positive symptoms (e.g., delusions), impaired cognition and social interaction skills, and worse functional outcomes (e.g., personal care skills), all of which could impact the ability to engage in healthy lifestyle interventions (Depp et al., 2015; Dickerson, Stallings, et al., 2013; Oluwoye et al., 2019). Particularly relevant to health lifestyle intervention outcomes, smoking can impact body weight, and limited evidence suggests that people with SMI who smoke cigarettes tend to engage in less physical activity while consuming diets less healthy than people with SMI who do not smoke (Bobes et al., 2010; Chiolero et al., 2008; Dipasquale et al., 2013; Tian et al., 2015). There is a need to examine the impact of tobacco use on healthy lifestyle intervention outcomes (e.g., weight change). This is a critical step in developing tailored multifaceted interventions to reduce CVD-risk factors for people with SMI.
Current Study
The current study addressed these gaps using secondary data from an NIMH-funded effectiveness trial of a PGLB for people with SMI between 2015 and 2018 (Cabassa et al., 2015). The PGLB trial results have been published elsewhere, but briefly, the trial found that a larger proportion of participants randomized to the PGLB intervention compared to UC achieved clinically significant weight loss (32% vs. 31%), but this difference was not statistically significant. The proportion of participants who achieved clinically significant weight loss is similar to other U.S.-based healthy lifestyle trials among people with SMI (Bartels et al., 2015; Daumit et al., 2013; Naslund et al., 2017). The current study has two aims: (a) examine the sociodemographic, dietary, physical activity, and physical and behavioral health factors associated with smoking cigarettes at baseline; (b) examine whether smoking cigarettes at baseline moderated the impact of the PGLB healthy lifestyle intervention in helping participants lose weight. Conducting moderation analysis on intervention studies that do not find an intervention effect can provide key information (MacKinnon, 2011). For instance, moderation analyses can identify subgroups that benefited from the intervention even if there was no whole group effect (MacKinnon, 2011). In the current study, weight change over the course of the intervention may have differed based on baseline tobacco use. If so, this would provide evidence for the need to tailor the intervention based on smoking status.
Method
Study Design
The PGLB trial is registered in clinicaltrials.gov (NCT02175641) and a detailed description can be found in the PGLB study protocol (Cabassa et al., 2015) and in a prior publication examining implementation outcomes (Tuda et al., 2023). The trial was conducted in three PSH agencies in two U.S. cities between 2015 and 2018. PSH provides access to subsidized housing and supportive services to people with a history of chronic homelessness and disability (Tsemberis et al., 2004). One agency uses a Housing First model, and the other two use a treatment-first model. Housing First provides low-threshold access to housing without preconditions of abstinence or the need to engage in supportive services (Stefancic & Tsemberis, 2007). Treatment-first models require participants to participate in substance use or psychiatric treatment before they become eligible for housing (Padgett et al., 2011). All participants gave written informed consent, and the institutional review boards of Columbia University and the Philadelphia Department of Public Health approved the study.
Participants
Eligibility was broad to capture a sample that resembled the racially/ethnically diverse people with SMI living in PSH. Inclusion criteria were: being a resident at the PSH site, male or female, ages 18 or older, any race/ethnicity, English or Spanish speaking, a chart diagnosis of an SMI, and a BMI ≥ 25 (kg/m2) at the time of screening (Cabassa et al., 2015). Exclusion criteria were: needing substance use detoxification services at the time of recruitment, posing a danger to themselves or others, failing a capacity-to-consent questionnaire, or self-reported medical conditions that contraindicated their participation (e.g., active cancer treatment; Cabassa et al., 2015; Zayas et al., 2005). Participants were recruited (e.g., flyers, staff referrals) between June 2015 and January 2018 (Cabassa et al., 2015). Research assistants screened 448 people; 340 were eligible to participate, and 314 participants were enrolled and then randomized to UC (N = 157) or the PGLB intervention (N = 157) after they completed their baseline interview. The most common reasons people were excluded from the trial were not having a BMI ≥ 25 (kg/m2), no SMI diagnosis, and having a medical condition that was contraindicative for a weight loss intervention (Cabassa et al., 2015). Randomization was conducted at the participant level in blocks of four and stratified by site. Data was collected at baseline, 6-, 12-, and 18-months postrandomization. The sample for the current study includes all 314 participants enrolled in the PGLB trial.
Intervention
The PGLB program is a 12-month, 22-session, manualized program consisting of 3 months of weekly sessions, followed by 3 months of bi-weekly sessions, and monthly sessions for the remaining 6 months. Sessions were delivered in group and one-on-one formats, lasted approximately one hour, and consisted of physical activity recommendations, nutrition education (e.g., reading food labels), and behavioral strategies (e.g., self-monitoring). Participants received a bathroom scale, pedometer, food log, and calorie reference book. PGLB was delivered by peer specialists (i.e., people with lived experiences recovering from SMI) who were employed by their respective housing agencies and trained and supervised by the study (Cabassa et al., 2015). Additional details about the PGLB intervention and the peer training and supervision approach have been published elsewhere (Cabassa et al., 2015; Stefancic et al., 2021).
Usual Care
All participants, including the intervention group, received UC services throughout the trial. UC services consisted of health promotion groups (e.g., cooking groups, understanding diabetes), linkages to medical care, and community resources (e.g., gyms). Health promotion groups were not manualized interventions and focused on health education. Agency staff at study sites helped clients connect with medical care as needed.
Measures
Outcomes and Moderators
Tobacco Use. Current cigarette smoking at baseline was the Aim 1 outcome and Aim 2 moderator. Current smoking was assessed at baseline using a single question asking participants if they currently smoke cigarettes.
Weight Change. The Aim 2 mixed-effects models’ outcome was weight change at 6, 12, and 18 months. Weight change was calculated by subtracting participants’ weight at each time point from their baseline weight. Research assistants measured participants’ body weight (in pounds) at baseline, 6, 12, and 18 months using a calibrated digital scale. Participants wore indoor clothing without shoes.
Independent Variables
All independent variables were collected during structured face-to-face interviews at baseline before randomization. Demographic correlates were self-reported age, years of education, gender, and racial/ethnic minoritized status.
Participants’ physical health conditions were collected by asking participants if their doctor had ever told them that they had a diagnosis of high cholesterol, diabetes, CVD, or cancer. Our CVD variable was defined as anyone who had a diagnosis of coronary heart disease, stroke, arteriosclerosis, heart attack, or congestive heart failure (Cabassa et al., 2017; Goodwin et al., 2009). Cardiorespiratory fitness was assessed with the 6-min walking test (6MWT; Cabassa et al., 2015). Hypertension was assessed using blood pressure (BP) measurements taken at baseline. Participants’ mental health conditions were collected by asking participants if their doctor had ever told them that they had an SMI diagnosis of depression, schizophrenia/schizoaffective disorder, or bipolar disorder.
Total minutes of physical activity and sitting minutes per day were assessed using the International Physical Activity Questionnaire-Short Form (IPAQ), a self-report measure that captures participants’ levels of PA (Craig et al., 2003). The IPAQ asks questions such as, “During the last 7 days, on how many days did you do moderate physical activities for at least 10 min at a time?,” and “how much time did you usually spend doing moderate physical activities on one those days?” Daily fruit and vegetable servings were measured using the block fruit and vegetable screeners (Block et al., 2000). Sugar-sweetened beverage servings per day were measured using questions from the 2013 Centers for Disease Control and Prevention Behavioral Risk Factors Surveillance System Questionnaire (CDC, 2013).
Physical and mental health quality of life was measured using the Short-Form Health Survey (SF12; Ware et al., 1996). Scores on the SF12 composite measures for physical and mental health range from 0 to 100, with higher scores indicating better physical or mental health-related quality of life. Any past 30-day substance use (i.e., alcohol and illicit drugs) was measured with a subset of questions from the Addiction Severity Index (ASI; Cacciola et al., 2007).
Control Variables
Study site was included as a covariate in all analyses since there were statistically significant differences in tobacco use by site, and baseline weight was included since there were statically significant differences in baseline weight between the intervention and UC conditions at baseline (Cabassa et al., 2021). We published a detailed analysis of site differences elsewhere (Tuda et al., 2023).
Data Analysis
All analyses used an intent-to-treat approach. We conducted Aims 1 and 2 using Stata 17.0 (Stata Corp LP, College Station, TX, USA). We checked the distributions of continuous variables for normality. Tests were 2-sided with a critical value α 0.05. All models included a categorical indicator for study site and continuous baseline weight as control variables since tobacco use differed significantly by site, and baseline weight differed significantly between the intervention and UC conditions.
Frequencies, percentages, and measures of central tendencies and dispersions were used to describe sample characteristics. Multiple logistic regression was used to examine the associations of baseline sample correlates with baseline cigarette smoking. Demographic, physical health, behavioral health, dietary, physical activity, and quality of life indicators were chosen based on the study aims. The link test was used to detect specification error. A significant linear predicted value and nonsignificant linear predicted value squared indicated a correctly specified link function and that the relevant variables were included in both logistic regression models (Long & Freese, 2014). We used Hosmer–Lemeshow methods to test model fit. A nonsignificant statistic indicated a good model fit for both models. VIF with the excess of 10 was used to detect any multicollinearity problems. No problems with multicollinearity were detected in either model. Pregibon's dbeta, which provides summary information of the influence on parameter estimates of each case, was used to identify influential cases. No influential cases were detected. Two-sided p-values of .05 were used to identify statistical significance.
We used an intent-to-treat approach to examine how participants’ baseline smoking status moderated the impact of receiving either PGLB or UC on weight change throughout the trial. Given the longitudinal data structure, meaning the same subjects were observed across four time points, the association between the continuous outcome (weight change) and its predictors were analyzed using mixed-effects models (Hedeker & Gibbons, 2006). Mixed effects models were also used since they employ random effects to accommodate extra heterogeneity. We ran two models. Model l examined the main effects of site, baseline weight, the two treatment conditions (PGLB vs. UC), time (e.g., 6, 12, and 18 months), and baseline cigarette smoking. Model 2 included the main effects plus the following two-way interaction term: Baseline Cigarette Smoking × Treatment Condition.
Hypotheses
Results
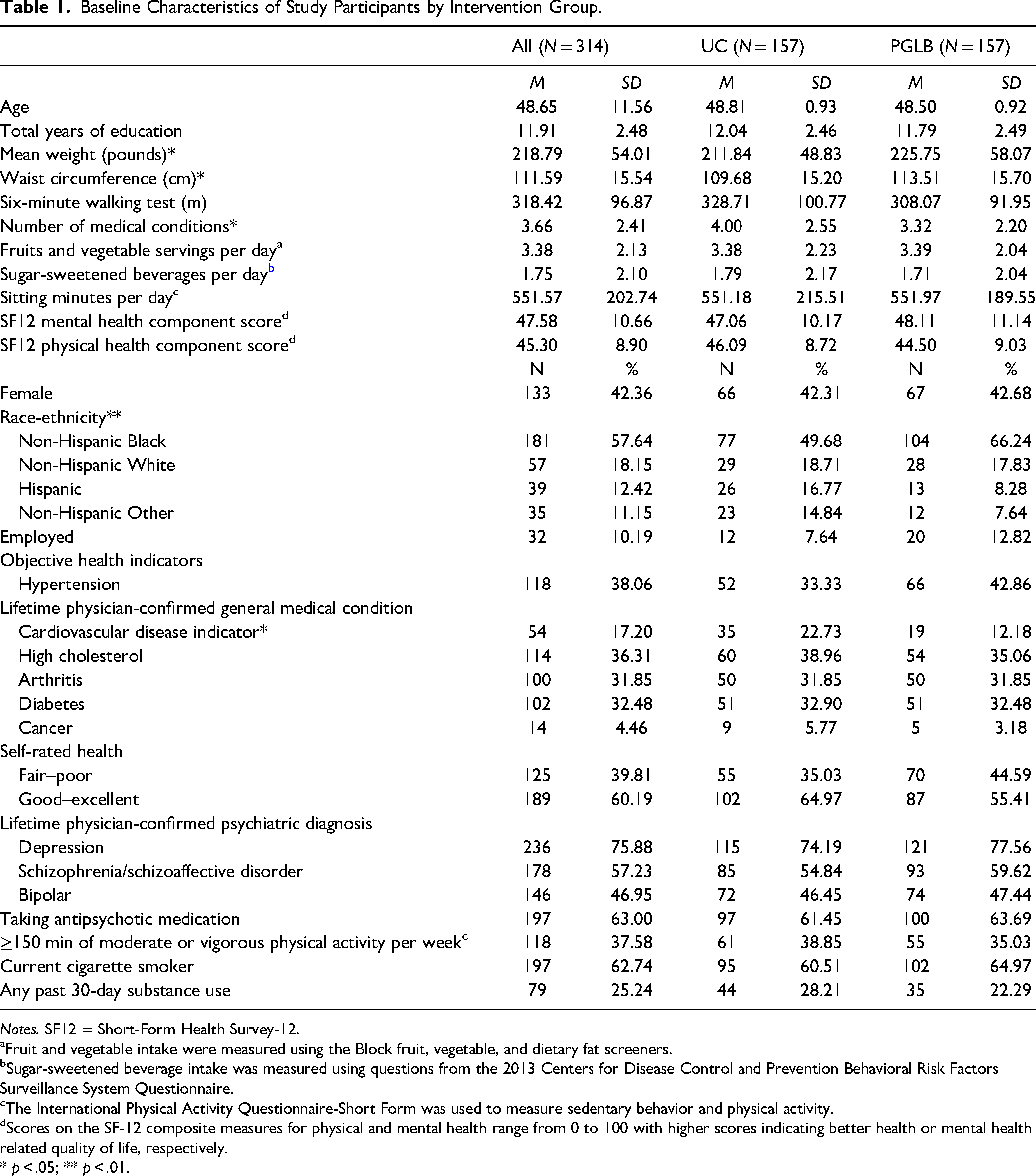
Sociodemographic characteristics. Participant baseline characteristics are presented in Table 1. The mean participant age was 48.65 (SD = 11.56). More than half were male (57.32%), and most were racial/ethnic minoritized groups (81.21%), particularly non-Hispanic Blacks (57.64%). The average years of education was 11.91 (SD = 2.48), and only 10.19% were employed.
Baseline Characteristics of Study Participants by Intervention Group.
Notes. SF12 = Short-Form Health Survey-12.
aFruit and vegetable intake were measured using the Block fruit, vegetable, and dietary fat screeners.
Sugar-sweetened beverage intake was measured using questions from the 2013 Centers for Disease Control and Prevention Behavioral Risk Factors Surveillance System Questionnaire.
The International Physical Activity Questionnaire-Short Form was used to measure sedentary behavior and physical activity.
dScores on the SF-12 composite measures for physical and mental health range from 0 to 100 with higher scores indicating better health or mental health related quality of life, respectively.
* p < .05; ** p < .01.
Mental health characteristics. The most common reported lifetime mental health diagnoses were depression (75.88%), schizophrenia/schizoaffective disorder (57.23%), and bipolar disorder (46.95%). Most participants (63%) were taking an antipsychotic medication. The mean mental health quality of life SF12 component score was 47.58 (SD = 10.66).
Physical health characteristics. Participants’ mean weight was 218.79 pounds (SD = 54.01), and the mean waist circumference was 111.59 cm (SD = 15.54). Participants reported having an average of 3.66 (SD = 2.41) medical conditions. Based on BP measurements taken at baseline, 38.06% of participants had hypertension. The most common reported lifetime physical health diagnoses were CVD (17.20%), high cholesterol (36.31%), arthritis (31.85%), diabetes (32.48%), and cancer (4.46%). More participants rated their health as good–excellent (60.19%) than fair–poor (39.81%). The mean physical health quality of life SF12 component score was 45.30 (SD = 8.90).
Diet and physical activity characteristics. On average, participants reported eating 3.38 (SD = 2.13) servings of fruits and vegetables per day and drinking 1.75 (SD = 2.10) sugar-sweetened beverages daily. Participants walked an average of 318.42 m (SD = 96.87) during 6MWT. On average, participants spent 551.57 (SD = 202.74) min/day sitting, and 37.58% reported getting 150 min or more per week of moderate or vigorous physical activity.
Tobacco and substance use characteristics. More than half of the participants were current smokers (62.74%), and 25.24% reported substance use (i.e., alcohol or illicit drugs, not including marijuana) in the past 30 days.
Aim 1 Results
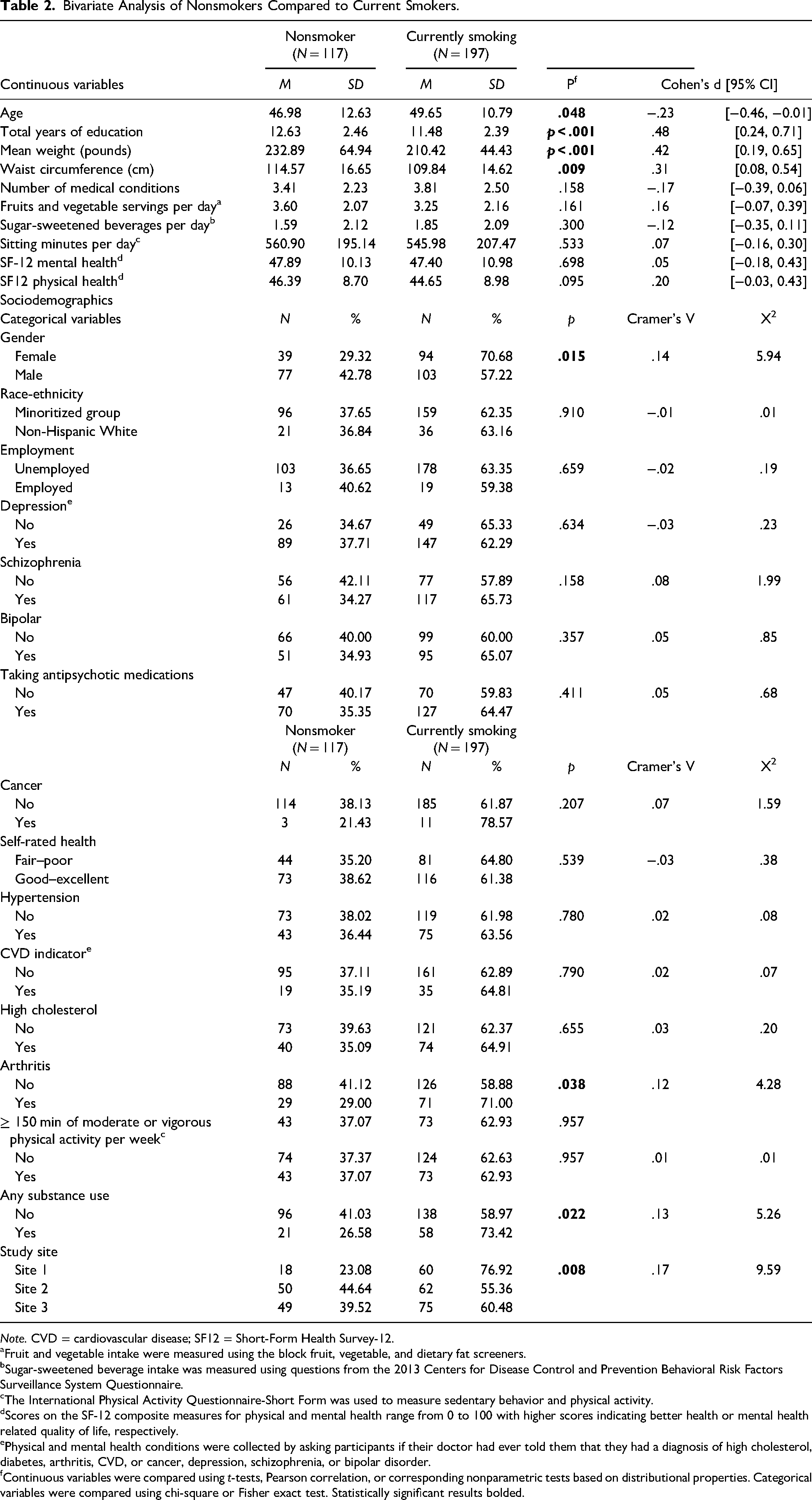
Bivariate results. Full results are presented in Table 2. We calculated effect sizes using Cohen's d for t-tests and Cramer's V for chi-square and Fisher exact tests. Effect sizes for significant findings were small to moderate. We found that being a current smoker was associated with younger age (Cohen's d = −.23, 95% CI [−0.46, −0.01], p = .048), female gender (Cramer's V = .14, X2 = 5.94, p = .015), and fewer years of education (Cohen's d = .48, [0.24, 0.71], p < .001). Smoking was also associated with lower baseline weight (Cohen's d = .42, [0.19, 0.65], p < .001) and with having a lifetime diagnosis of arthritis (Cramer's V = .12, X2 = 4.28, p = .038). In addition, 73.4% of people who reported past 30-day substance use (i.e., alcohol or illicit drugs) smoked cigarettes compared to 58.9% of people who reported no past 30-day substance use (Cramer's V = .13, X2 = 5.26, p = .022). There were no statistically significant differences between current smokers and nonsmokers in race/ethnicity, employment, medical conditions, self-rated health, mental health diagnoses, dietary intake, physical activity, or quality of life.
Bivariate Analysis of Nonsmokers Compared to Current Smokers.
Note. CVD = cardiovascular disease; SF12 = Short-Form Health Survey-12.
aFruit and vegetable intake were measured using the block fruit, vegetable, and dietary fat screeners.
bSugar-sweetened beverage intake was measured using questions from the 2013 Centers for Disease Control and Prevention Behavioral Risk Factors Surveillance System Questionnaire.
cThe International Physical Activity Questionnaire-Short Form was used to measure sedentary behavior and physical activity.
dScores on the SF-12 composite measures for physical and mental health range from 0 to 100 with higher scores indicating better health or mental health related quality of life, respectively.
ePhysical and mental health conditions were collected by asking participants if their doctor had ever told them that they had a diagnosis of high cholesterol, diabetes, arthritis, CVD, or cancer, depression, schizophrenia, or bipolar disorder.
fContinuous variables were compared using t-tests, Pearson correlation, or corresponding nonparametric tests based on distributional properties. Categorical variables were compared using chi-square or Fisher exact test. Statistically significant results bolded.
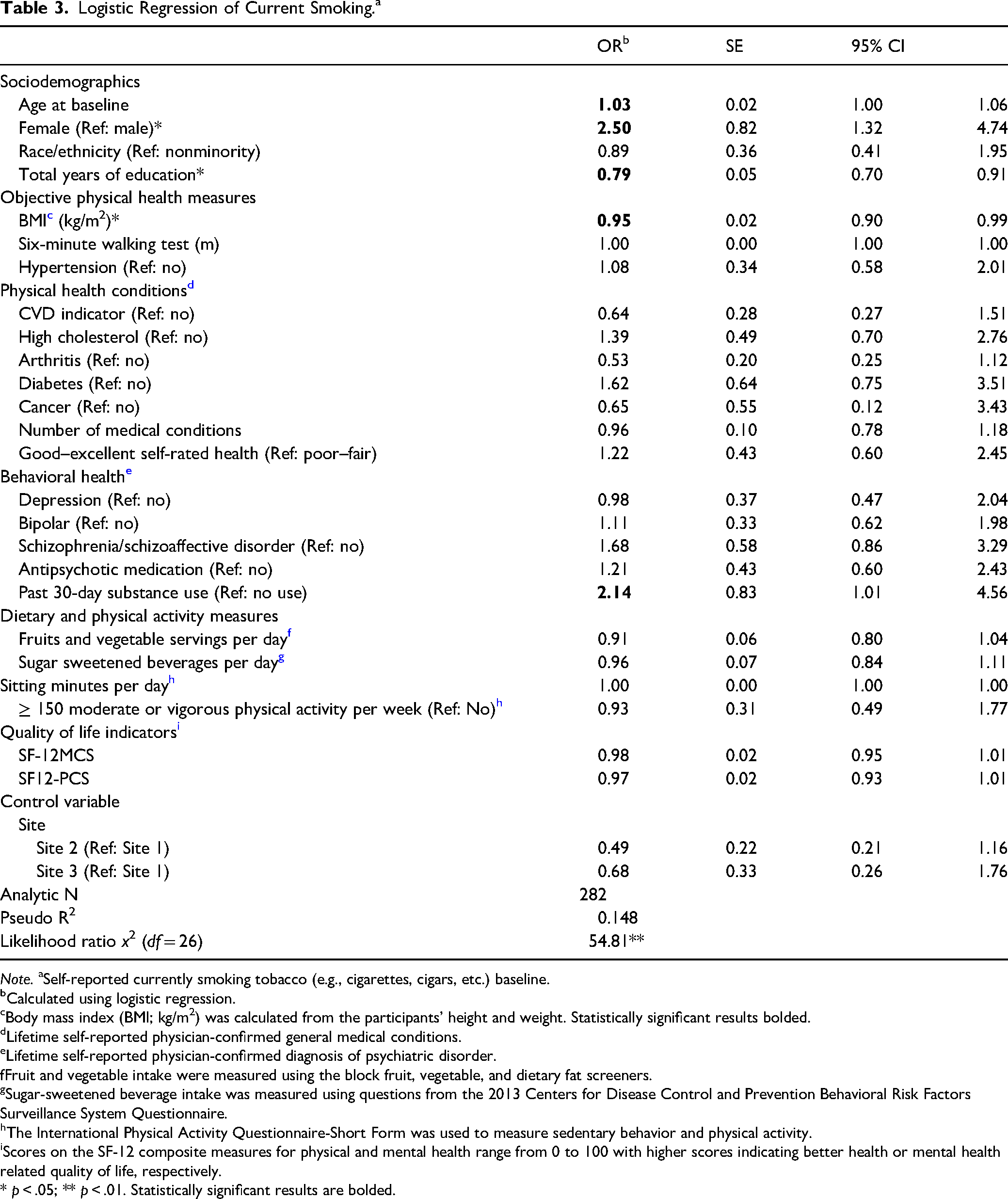
Multiple regression results. We used logistic regression to identify correlates of being a current smoker at baseline (see Table 3). The model had a good fit to data (Hosmer–Lemeshow x2 = 5.67, p = .684), as shown by a model
Logistic Regression of Current Smoking.a
Note. aSelf-reported currently smoking tobacco (e.g., cigarettes, cigars, etc.) baseline.
Calculated using logistic regression.
Body mass index (BMI; kg/m2) was calculated from the participants’ height and weight. Statistically significant results bolded.
Lifetime self-reported physician-confirmed general medical conditions.
Lifetime self-reported physician-confirmed diagnosis of psychiatric disorder.
Fruit and vegetable intake were measured using the block fruit, vegetable, and dietary fat screeners.
Sugar-sweetened beverage intake was measured using questions from the 2013 Centers for Disease Control and Prevention Behavioral Risk Factors Surveillance System Questionnaire.
The International Physical Activity Questionnaire-Short Form was used to measure sedentary behavior and physical activity.
Scores on the SF-12 composite measures for physical and mental health range from 0 to 100 with higher scores indicating better health or mental health related quality of life, respectively.
* p < .05; ** p < .01. Statistically significant results are bolded.
Aim 2 Results
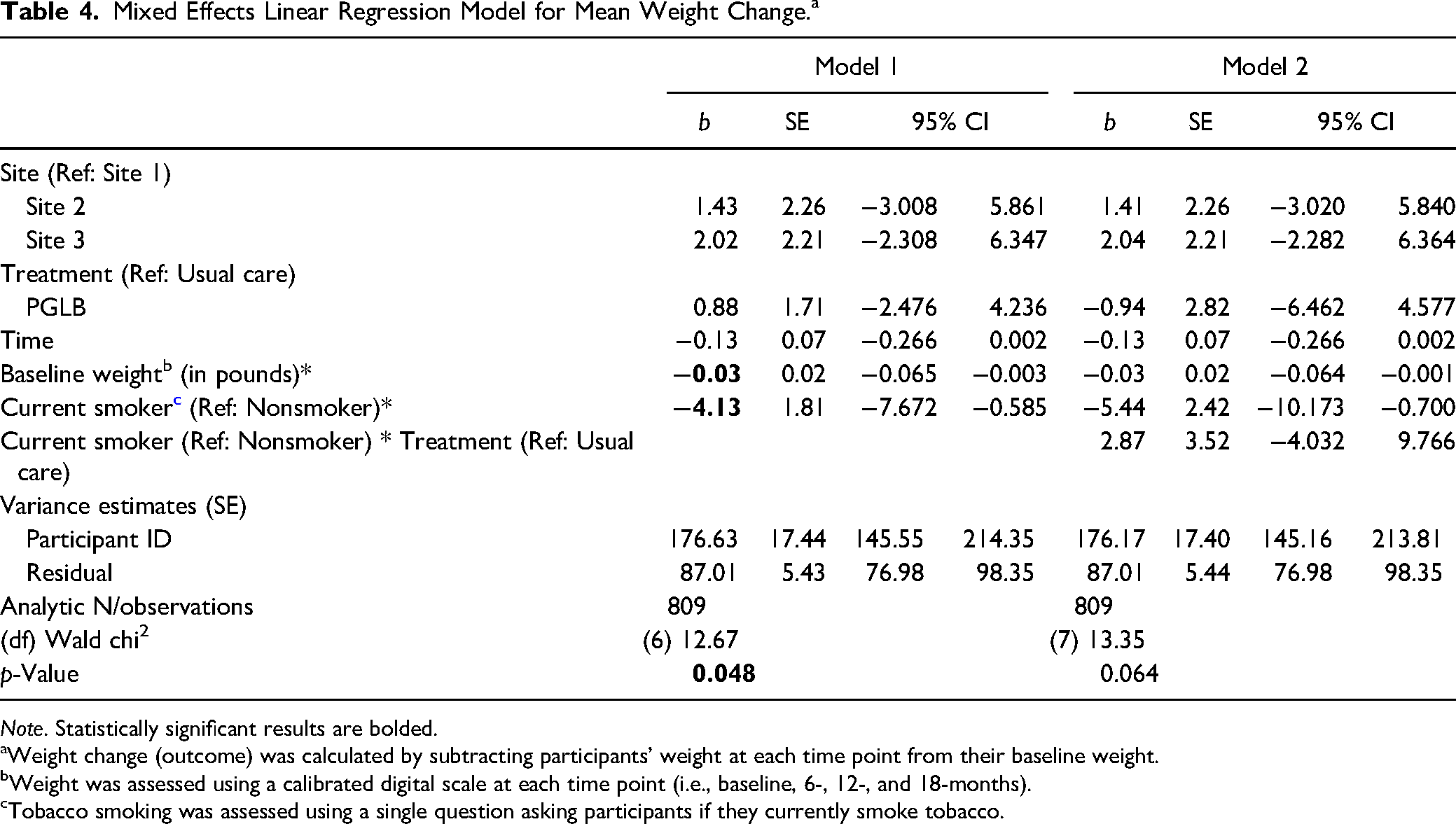
Moderating effects. The mixed effects regression model results are summarized in Table 4. Model 1 tested the direct effects and indicated that baseline weight and smoking status predicted mean weight loss during the trial. For smokers, the predicted weight loss over the course of the trial is 4.13 pounds more than for nonsmokers (95% CI [−7.672, −0.585]). In addition, baseline weight was negatively associated with weight change over the trial (b = −0.03 [−0.065, −0.003]), indicating that participants who started at a higher baseline weight lost more weight during the trial. Model 2 found that participants’ smoking status did not moderate the impact of receiving PGLB or UC on weight loss throughout the trial, indicating that our hypothesis was not confirmed.
Mixed Effects Linear Regression Model for Mean Weight Change.a
Note. Statistically significant results are bolded.
aWeight change (outcome) was calculated by subtracting participants’ weight at each time point from their baseline weight.
bWeight was assessed using a calibrated digital scale at each time point (i.e., baseline, 6-, 12-, and 18-months).
Tobacco smoking was assessed using a single question asking participants if they currently smoke tobacco.
Discussion and Applications to Practice
This study described the health profile of people with SMI living in PSH who smoke cigarettes and examined the impact of tobacco use on weight loss during the PGLB healthy lifestyle intervention. The high prevalence of smoking among people with SMI makes it essential to understand the effect of tobacco use on healthy lifestyle interventions. Tobacco use can potentially impact weight loss, physical activity, and dietary practices, which are targets in healthy lifestyle interventions (Bobes et al., 2010; Dipasquale et al., 2013; Tian et al., 2015).
Overall, the multiple logistic regression findings did not confirm our hypothesis that smoking would be related to worse physical and mental health, less fruit and vegetable consumption, and lower physical activity levels. Although not statistically significant, the bivariate analysis indicated that participants who were current smokers had a slightly higher number of physical health conditions, lower mean fruit and vegetable consumption, higher intake of sugar-sweetened beverages, and less physical activity compared to nonsmokers at baseline. This is similar to a bivariate study by Dickerson, Yu, et al. (2013) that found people with SMI who use tobacco had a higher prevalence of physical health conditions and less healthy dietary intake.
Consistent with current literature, the multiple logistic regression findings confirmed that fewer years of education, lower BMI, and past 30-day substance use were associated with current smoking (Hickling et al., 2018; Hiscock et al., 2012; Tian et al., 2015). In addition, females in our study were more likely to smoke cigarettes than males (baseline prevalence, 71% and 57%, respectively), which is contrary to findings in the general population and among people with SMI that indicate males are more likely to smoke than females (Dickerson et al., 2018; Wang et al., 2018). However, our sample is among people who transitioned from homelessness into PSH, and prior studies have found that 75% of women experiencing homelessness and living in unstable housing use tobacco (Riley et al., 2022). Women experiencing homelessness report high rates of violence and trauma, which could explain higher rates of tobacco use since trauma is highly correlated with smoking (Rubin et al., 2021). This could indicate a need for smoking cessation programs that are tailored for women with a history of homelessness and trauma.
The hypothesis that tobacco use would moderate the impact of receiving either the PGLB or UC on weight change throughout the trial was not confirmed. However, we did find main effects for baseline weight and cigarette smoking as predictors of weight loss over the course of the trial. Participants with higher baseline weights lost more weight throughout the trial than people who started at lower weights. Higher baseline body weight has been shown to predict a greater rate of weight loss in general population studies (Finkler et al., 2012). Importantly, people who smoked cigarettes at baseline lost more weight over the course of the trial compared to those who did not smoke. This is concerning since tobacco use is not the intended mechanism of weight loss in healthy lifestyle interventions. It is possible that the physiological effects of smoking (e.g., increased metabolism, reduced appetite) caused participants to lose more weight throughout the trial compared to those who did not smoke (Bush et al., 2016; Fulkerson & French, 2003; Seeley & Sandoval, 2011). This indicates a need to identify people using tobacco during healthy lifestyle interventions and offer them additional support, including smoking cessation treatment options.
Healthy lifestyle interventions focused on helping people achieve a healthy weight may be an ideal place to assess and treat tobacco dependence. Some people use tobacco as a weight control mechanism, so when these participants stop smoking, they may need additional support and counseling to help counteract the associated weight gain (Bush et al., 2016). There is also concern that weight loss associated with tobacco use could mask the metabolic effects of antipsychotic medications, such as weight gain, further highlighting the need to provide evidence-based tobacco treatment (i.e., combination counseling and pharmacotherapy) for all people with SMI who smoke (Das & Prochaska, 2017; Hawes et al., 2021).
We know of three studies among people with SMI that provided comprehensive cardiovascular risk reduction interventions, including tobacco cessation, dietary, and physical activity components (Baker et al., 2018; Daumit et al., 2020, 2023). Daumit et al. (2023) provided a combination smoking cessation and weight management intervention and found the intervention group achieved significantly greater smoking abstinence rates without significantly increasing body weight compared to the control condition. The other two studies did not assess weight change among those who reported abstinence, nor did they report weight change by smoking status (Baker et al., 2018; Daumit et al., 2020). These studies provide a guide on how social workers can integrate tobacco cessation into healthy lifestyle interventions for people with SMI. Successfully reducing CVD among people with SMI will require increasing access to tobacco cessation treatment along with support for improving diet and physical activity.
Limitations. Several study limitations need to be considered. First, self-report measures were used to evaluate levels of physical activity, diet, smoking, substance use, and other correlates. These measures tend to over- or underestimate the prevalence of variables and are subject to recall bias (Duncan et al., 2017; Lee et al., 2011). In addition to recall bias, self-report physical activity and dietary measures are imprecise. Future studies should try to use objective measures such as a 24-hr food recall to measure dietary intake and activity trackers to capture physical activity. Future studies should also consider employing biochemically verified 7-day point prevalence abstinence combined with a 30-day self-report measure of cigarette smoking (Hughes et al., 2003). Nevertheless, while self-report measures are imperfect, they are commonly used in large community studies due to their feasibility and are used extensively in population-based studies for examination and surveillance of health (Duncan et al., 2017). In addition, the findings of the moderator analysis should be interpreted with caution due to the relatively small sample size used in this trial to investigate treatment moderators. Future studies should use larger samples with greater statistical power to examine moderation effects. This will enhance our understanding of which individuals benefit most from these interventions and the reason for their effectiveness. Also, most of the participants belonged to racial/ethnic minoritized groups, particularly non-Hispanic Blacks. It will be important that studies include larger samples of diverse racial/ethnic groups so that differential treatment effects can be examined. Lastly, this study is based on people with SMI living in supportive housing in New York City and Philadelphia. While this is an important group in need of services, these findings cannot be generalized to all people with SMI who smoke cigarettes. The experiences of the current sample could be very different from people with SMI living in the community (i.e., nonsupportive housing) or rural areas. Future studies need to include representative samples of people with SMI living in a variety of community settings. This will provide a more comprehensive understanding of how tobacco use impacts healthy lifestyle intervention mechanisms of change and outcomes among people with SMI.
Conclusions
To our knowledge, no studies among people with SMI living in PSH have explored the moderating effect of tobacco use on weight loss during a healthy lifestyle intervention. Our findings indicate that baseline smoking may significantly impact weight loss during healthy lifestyle interventions, regardless of intervention group. Participants in healthy lifestyle interventions may need more support to give them the confidence to quit smoking while still achieving their weight loss goals. Further, healthy lifestyle interventions may be the ideal place to offer tobacco cessation treatment since participation indicates a motivation to improve health. Offering treatment for tobacco use at all points of service contact provides an opportunity to reduce high rates of premature mortality among people with SMI.
Footnotes
Acknowledgments
The authors would like to thank the study participants and community partners for their engagement in this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by the National Institute of Mental Health (NIMH) under award numbers: R01MH104574 and T32MH019960.
