Abstract
Concerns about chemical exposure in the electronics manufacturing industry have long been recognized, but data are lacking in Southeast Asia. We conducted a study in Batam, Indonesia, to evaluate chemical exposures in electronics facilities, using participatory research and biological monitoring approaches. A convenience sample of 36 workers (28 exposed, 8 controls) was recruited, and urine samples were collected before and after shifts. Five solvents (acetone, methyl ethyl ketone, toluene, benzene, and xylenes) were found in 46%-97% of samples, and seven metals (arsenic, cadmium, cobalt, tin, antimony, lead, and vanadium) were detected in 60%-100% of samples. Biological monitoring and participatory research appeared to be useful in assessing workers’ exposure when workplace air monitoring is not feasible due to a lack of cooperation from the employer. Several logistical challenges need to be addressed in future biomonitoring studies of electronics workers in Asia in factories where employers are reluctant to track workers' exposure and health.
Keywords
Introduction
The manufacture of electrical and electronic products has increased dramatically over the past several decades and includes rapid growth in contract manufacturing, which takes place through a complicated chain of subcontractors who are often located in Asia and provide products for major multinational companies such as Samsung Group, Apple, Tesla, Hon Hai, Compal, AMD, Infineon, Intel, Texas Instruments, STMicroelectronics, and others. The industry comprises a wide spectrum of manufacturing and non-manufacturing sectors, ranging from the semiconductor industry and thin-film-transistor liquid crystal display (TFT-LCD) manufacturing to inter-business e-commerce, video gaming, and music streaming sectors. The manufacturing sectors, in particular the semiconductor industry, tend to be chemical-intensive in the production and assembly processes such as in wafer fabrication, printed circuit board fabrication, and assembly processes for semiconductors and printed circuit boards. 1 Numerous chemicals are used in these processes often in highly controlled environments that are designed to protect the quality of the products rather than the health of the workers. 1 As a result, workers in this industry have been exposed to a variety of chemicals including but not limited to toxic gases (e.g., arsine, phosphine, diborane, ammonia, and chlorine), strong acids (e.g., hydrofluoric and hydrochloric acids), caustic solutions, photoactive chemicals, as well as solvents and metals.1–3 The potential health impact is significant because many of these chemicals are known or probable human carcinogens, such as arsenic compounds, cadmium and cadmium compounds, benzene, methylene chloride, and trichloroethylene. Others are endocrine disruptors, reproductive toxicants, and/or developmental toxicants such as bisphenol A, mercury, toluene, and dioxins.
As a result, concerns have been expressed for many years about possible increased risks of cancer and adverse reproductive outcomes among workers in semiconductor manufacturing facilities. 4 From the mid-1980s to the early 2000s, multiple epidemiologic studies were conducted in the United States, the United Kingdom, South Korea, and Taiwan to investigate risks of non-Hodgkin's lymphoma, brain cancer, leukemia, lung cancer, and breast cancer in the semiconductor industry.4–15 Excess risks were observed in some cases.5,9,10,12 However, the studies, in general, found little evidence to support the association between specific workplace exposures and cancer. Frequently, the associations initially identified were not significant in the follow-up studies.5–8,14–17 The negative results could possibly be attributed to the healthy worker effect, selection bias, and/or exposure misclassifications. 11 On the other hand, consistent epidemiologic evidence, published between the 1980s and mid-1990s, strongly suggested a causal association between chemical exposures in the semiconductor industry and the risk of spontaneous abortion (miscarriage). The main causal agents identified were ethylene glycol ethers (EGE), although the studies could not rule out xylene or n-butyl acetate as responsible agents.18–24
While these studies used relative exposure estimates, rather than measured, modeled, or estimated concentrations,23,25,26 typical EGE exposures in the electronics industry at the time when the studies were conducted were well below regulatory exposure limits at that time, suggesting that the limits were not adequately protective. For example, Paustenbach 26 reported measurement results of EGE compounds from approximately 400 industrial hygiene air monitoring samples collected in the 1980s. These include sample results received in an industry-wide survey conducted by the Semiconductor Industry Association in 1984-1985. Seven semiconductor companies participated in the survey and provided approximately 300 samples that were estimated to represent about 60% of all the industrial hygiene personal monitoring data collected by these firms during that time. The rest of the samples were collected in 1982 by the National Institute for Occupational Safety and Health (NIOSH), a division of the Centers for Disease Control and Prevention, located in the U.S. Public Health Service, as part of their Health Hazard Evaluation Program of the semiconductor industry. 26 Together, these samples were thought to be representative of the industry. 26 Table 1 shows the geometric mean concentrations of personal samples (among all compiled samples) for 2-ethoxyethanol (2-EE), 2-ethoxyethyl acetate (2-EEA), 2-methoxyethanol (2-ME), and 2-methoxyethyl acetate (2-MEA). 26 At the time of the study, the U.S. Department of Labor Occupational Safety and Health Administration (OSHA) had adopted permissible exposure limits (PELs) at 25 ppm for both 2-ME and 2-MEA, 100 ppm for 2-EEA, and 200 ppm for 2-EE. These limits have been unchanged since then. The American Conference of Governmental Industrial Hygienists (ACGIH®) threshold limit value (TLV) was 5 ppm for all four compounds in 1985 and 1986. 26 At the time of data collection and semiconductor studies, the monitoring data suggested compliance with regulatory standards and guidelines; however, health effects were observed. Subsequently, ACGIH has revised the TLVs for 2-ME and 2-MEA downward.
Comparison of Occupational Exposure Limits With Measured Concentrations of Chemicals in Semiconductor Manufacturers.

The geometric means of personal samples of these chemicals. 26
Permissible exposure limit, https://www.osha.gov/annotated-pels/table-z-1 (accessed November 9, 2023).
Recommended exposure limits, https://www.osha.gov/annotated-pels/table-z-1 (accessed November 9, 2023).
Threshold limit value.
Threshold limit value (1985-1986 values). 26
2-Ethoxyethanol, https://www.acgih.org/2-ethoxyethanol/ (accessed November 9, 2023).
2-Ethoxyethyl acetate, https://www.acgih.org/2-ethoxyethyl-acetate/ (accessed November 9, 2023).
2-Methoxyethanol, https://www.acgih.org/2-methoxyethanol/ (accessed November 9, 2023).
2-Methoxyethyl acetate, https://www.acgih.org/2-methoxyethyl-ace (accessed November 9, 2023).
Upon comparison, the measured exposures reported by Paustenbach in the 1980s were consistently 1-2 orders of magnitude below the 2023 Cal/OSHA PELs (enforceable in California) and 3-4 orders of magnitude below the outdated OSHA PELs established over 50 years ago. In addition, the geometric mean concentration of 2-MEA, measured in the 1980s was an order of magnitude below the NIOSH recommended exposure limit (REL) and the ACGIH® TLV as they are in 2023. Additionally, the geometric means for 2-EE and 2-EEA, measured in the 1980s were an order of magnitude below the 2023 TLVs and slightly below the 2023 RELs. Notably, the geometric mean of 2-ME equaled both the TLV and the REL, indicating that higher exposures exceeded these subsequently established non-mandatory limits. Despite the reduction in some occupational exposure limits over the past 50 years, there is still concern that the lowered standards may not provide adequate protection. However, few studies have been published since the late 1990s, 11 possibly due to limited access to workplaces for exposure monitoring.
Meanwhile, the globalization has shifted mass production from developed countries to developing countries since the 1990s. Many multinational corporations moved their electronics production and assembly operations to lower-wage countries such as those in the Pacific Rim. The number of electronics industry workers throughout the supply chain is expected to grow significantly over the next decade, as the global electronic chemicals and materials market is projected to grow from $59.88 billion in 2021 to $89.82 billion in 2028 at a compound annual growth rate of 6.0%.
27
The rapid growth of the industry has been accompanied by increased use of toxic chemical substances.
28
This raises concerns about workers’ exposure to chemicals in these newly industrialized countries, as workers could be exposed to chemicals through outdated processes while using equipment and material that is no longer used in developed countries,
29
as well as chemicals that are newly introduced into the manufacturing processes. According to the American Public Health Association, compounding issues include a lack of information about the hazardous substances used by the workers, weak occupational exposure limits for hazardous materials, lack of uniform protective exposure standards for workers, and often the complete absence of tracking and reporting of patterns of disease associated with the electronics sector.
30
In response to industry globalization and associated occupational health challenges, civil society also began to organize global networks to alert and train local residents and workers about the hazards that had been uncovered initially in Silicon Valley. The International Campaign for Responsible Technology was formed at the 20th anniversary of the Silicon Valley Toxics Coalition when activists from more than 15 countries that were experiencing high-tech manufacturing growth attended a conference in San Jose and adopted the following mission statement: We are an international solidarity network that promotes corporate and government accountability in the global electronics industry. We are united by our concern for the life cycle impacts of this industry on health, the environment, and workers’ rights. By sharing resources, we seek to build the capacity of grassroots organizations, local communities, workers and consumers, to achieve social, environmental, and economic justice.
Other networks came together to address occupational and environmental hazards, including the Asian Network for the Rights of Occupational and Environmental Victims (ANROEV), the GoodElectronics Network, International Persistent Organic Pollutants (POPs) Elimination Network (IPEN), and Electronics Watch, among others. All these networks adopted strong policy and advocacy positions on worker and community right to know and insisted on greater transparency in opposition to the prevailing position of most electronics companies, which treated chemical information with trade secrecy as confidential business information. Each of these networks also focused on worker and community training to alert people to the hazards in electronics and to help equip them to better protect themselves. Furthermore, due to rapid changes in the industry, manufacturing processes and chemicals used may change quickly, calling for frequent hazard assessments and monitoring in the electronics manufacturing industry, especially in these outsourced manufacturing facilities. Dr. Myron Harrison, a former occupational physician for IBM, articulated a profound insight in a 1992 article in Hazardous Materials Toxicology: Professionals associated with [semiconductor manufacturing] have invariably commented on the rapid pace of change in tools and materials and on the fact that adequate toxicological assessment of chemicals almost never precedes their introduction into manufacturing settings. The pace of change is quickening under the pressure of severe economic competition. As recently as 3-4 years ago, a typical schedule of a new technology from research and development to pilot lines to full manufacturing was 6-8 years. Executives who manage micro-electronics businesses are now demanding the schedule be compressed into a 2-3 year time frame. Engineers are not evaluated nor rewarded on their ability to understand new or unusual health hazards. This task is the responsibility of health and safety professionals. Unfortunately, the opportunities for the professionals to be involved before these new processes arrive at the manufacturing floor are being diminished by the quickening pace of technological change. Any large semiconductor facility uses several thousand chemicals. An attempt to review the toxicology of all these materials is doomed to be superficial and of little value.
28
In 1999, a group of environmental, labor, and health activists from the United States and Europe met in Soesterberg, Netherlands to develop a new vision of sustainability to track the dynamism of Moore's law, which is named after Gordon Moore, co-founder of Intel. Moore's law posits that the processing power and capabilities of computer chips will approximately double every 2 years, leading to exponential growth in computing performance and efficiency over time. The new vision states: Each new generation of technical improvements in electronic products should include parallel and proportional improvements in environmental, health and safety, as well as social justice attributes. (Adopted by the Trans-Atlantic Network for Clean Production, May 16, 1999)
Despite these clear warnings, the lack of regulations and industrial hygiene support has meant that exposure monitoring has rarely been conducted in electronics production. Consequently, there has been little exposure data available in published literature since the early 2000s. While government agencies may have authority to inspect the workplace and collect samples to monitor workers’ exposure to hazardous materials, they usually have a very limited number of inspectors to conduct monitoring and inspections. Meanwhile, access to factories has been very limited for non-government parties including trade unions. There is no legal right of such access and employers have been unwilling to grant it voluntarily. The Ministry of Manpower of Indonesia is a government ministry responsible for the workers and labor laws of Indonesia. In Batam, Indonesia specifically, the local office of the Ministry of Manpower is responsible for the implementation of state policies on the labor sector including conducting chemical monitoring. However, the local Manpower Office conducts inspections and monitoring only at factories where potential violations are reported, even though high exposure to hazardous chemicals could happen in any electronics manufacturing companies including those where no violations have been reported. The local Manpower Office does not have a clear understanding of the need to monitor chemicals that can cause chronic occupational illness. Monitoring of chemicals in the workplace is absent.
Biomonitoring is a valuable tool that has rapidly developed within the last two decades. It can potentially be used to screen for workers’ exposures, especially in the workplace where air monitoring is difficult to conduct. Our goal in this study was to explore the feasibility of conducting a worker-participatory biomonitoring project to assess chemical exposures in the electronics manufacturing industry in Southeast Asia. Due to the long-term concern about occupational chemical exposures in the region, several organizations initiated the study. These included the Federation of Indonesia Metalworkers Union (FSPMI), the Sedane Labour Resource Centre (Lembaga Informasi Perburuhan Sedane, LIPS), and the International Campaign for Responsible Technology. The project was funded by the GoodElectronics Network, an international environmental and occupational health network. The study took a participatory research approach, with the union, volunteer workers, the non-profit organizations, and a representative of the funding agency deeply involved in the project from research question formulation to study design, field sampling strategy development, sample collection and handling, and results dissemination (Figure 1). The coalition developed several strategic goals:
Test out a model that could identify hazards/exposures even without the cooperation of the management; Do the monitoring project in a place where there were workers organized into a union in factories that used toxic chemicals. Earlier research by LIPS had identified several factories using large numbers of chemicals in Batam;
31
Focus on Batam and leverage the occupational health and safety training that had previously been done with ANROEV; Work closely with the local unions organized by FSPMI; and Link the advocacy to the technical support of experts. A research group at Purdue University was selected to be the academic partner based on their knowledge and interest.
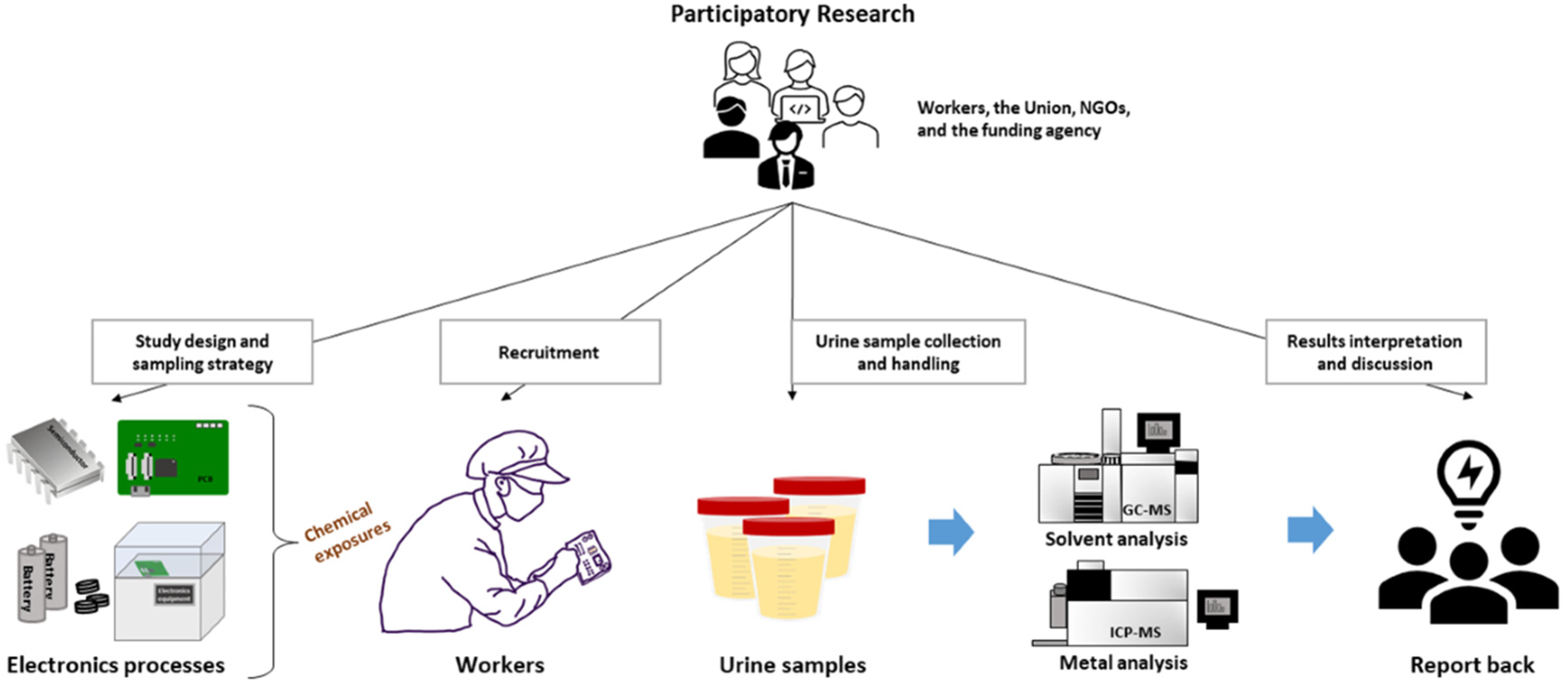
An illustration showing a participatory research blueprint. Participation research includes involving all the parties in study design, sampling strategy development, participant recruitment, sample collection, and results interpretation and dissemination.
This article describes the study process and reports urinary solvent and metal concentrations among workers in the electronics manufacturing facilities in Batam, Indonesia. Although the results of this feasibility study have limited generalizability due to small sample size and lack of air monitoring data at the workplace, the biomonitoring data and the participatory research approach reported can be useful to industrial hygiene professionals and used to inform future studies investigating chemical exposures in the electronics manufacturing industry in Southeast Asia and elsewhere. In addition, the biomonitoring data and the participatory research approach can also be used in future policy advocacy to improve worker safety and prevent adverse health outcomes in manufacturing and non-manufacturing sectors.
Materials and Methods
Study Site and Chemicals of Interest
Batam is an island located on the Malacca Strait that is 20 km off Singapore’s South coast. It is an industrial boomtown and part of a free trade zone in the Indonesia-Malaysia-Singapore Growth Triangle. Electronics manufacturing is one of the major industries on the island with about 50 electrical and electronics subsidiary firms operating in 2012. 32 It was estimated that the number of electronics workers in Batam was approximately 65,000 in 2016. 31 LIPS conducted preliminary research in 2016 in collaboration with the GoodElectronics Network and reported chemical usage, workers’ health conditions, and workers’ concerns as case studies in several electronics manufacturing facilities in Batam. 31 In the current study, researchers and representatives of the union and the Non-Governmental Organizations (NGOs) met several times after the project was funded to develop the research plan and field sampling strategy. After reviewing chemicals used in electronics facilities in Batam as summarized in the aforementioned report, 31 the team decided to focus on solvents and metals due to their known potential health effects, the feasibility of sample analysis, and the availability of occupational exposure guidelines [the ACGIH Biological Exposure Index (BEI)] (Table 2).
Chemicals to Be Analyzed in Urine Samples That Have BEI.

BEI, biological exposure indices—expressed as concentrations in urine samples, developed by the American Conference of Governmental Industrial Hygienists (ACGIH); TLV, threshold limit value—expressed as concentration in the air, developed by the ACGIH.
Workshop Training, Recruitment, and Urine Sample Collection
With the support of a training grant from the Developing World Outreach Initiative, an initiative of the Northern California Section of the American Industrial Hygiene Association, the project team co-conducted an occupational safety and health (OSH) workshop with Asia Monitor Resource Centre in June 2019 in Batam. Workers were invited to the workshop to participate in the training and learn about the participatory research project. About 50 workers from electronics facilities participated in the training that covered an overview of OSH issues (in particular chemical hazards), right to know, hazard mapping, body mapping, and health effects of chemical exposures, and the Chemical Hazard and Alternatives Toolbox (ChemHAT). 33 The training included an explanation of what biomonitoring is and why it is done. The study's informed consent document for human subject research including biomonitoring was explained and discussed. The training lasted for about 6 h.
Following the workshop, the attendees were invited to participate in the research project. Participation was voluntary. The local collaborators also promoted the project outside the training workshop, leading to recruitment of some study participants who had not attended the workshop. As a result, a convenience sample of 36 workers was recruited to participate in the study, including 28 exposed workers (n = 28) and eight unexposed workers (“controls” hereafter) (n = 8). One of the selection criteria for participants was that both exposed workers and controls had to have been at their current job for at least 6 months prior to participating in the study. Written informed consents were obtained from all participants before the enrollment. Exposed workers were factory floor workers from five electronics manufacturing facilities, whereas controls were union representatives or office workers from the same or similar facilities. Union representatives were full-time employees of the union working in an office located outside of the factories. Hence control workers did not work on the production lines. They were not known to have direct contact with chemicals used in the manufacturing processes. However, they live on the same island as exposed workers. They may live near industrial facilities. A questionnaire was administered by a trained researcher to collect information on demographics, work history, and co-variates such as cigarette smoking.
Two spot urine samples were collected from each worker, one before and one right after the work shift on the same day. The work shift was 8 h, which did not vary from day to day nor from worker to worker. Urine samples were collected by filling in a 90-mL sterile polypropylene straight-sided vial with polyethylene closure (Fisher Scientific, Waltham, MA) leaving minimal headspace. All samples were refrigerated immediately after the collection. An aliquot of each sample was analyzed at a local laboratory for urinary creatinine concentrations. Samples were packed in an insulated container with bagged dry ice for shipping to a laboratory at Purdue University where they were stored at −80°C prior to the analyses. The sample collection and handling followed the Centers for Disease Control and Prevention (CDC) guidelines for urine sample collection, transportation, and storage. 34 Union representatives from the FSPMI, members of the LIPS, and volunteer workers were trained on urine sample handling and coordinated the sample collection, storage, and transportation. The procedure followed a study protocol approved by the Internal Review Board at Purdue University (IRB Protocol #1903021912). The study team was not able to obtain permission from the factory management to collect personal or area air samples or bulk material samples at the workplace, which is a common challenge to face in the region (more in the discussion session).
Sample Analysis
Solvents in urine samples were analyzed by headspace solid phase microextraction (SPME) followed by gas chromatography–mass spectrometry a (GC-MS). 35 Besides targeted compounds listed in Table 2 [acetone, methyl ethyl ketone (MEK), and toluene], we also analyzed samples for benzene, ethyl benzene, and xylenes. Although these three compounds do not have BEI, they were included because of their usage in the facilities in Batam.
Metals in urine samples were analyzed by inductively coupled plasma mass spectrometry (ICP-MS). b Since ICP-MS can analyze multiple elements simultaneously, we initially screened for 12 elements, based on their use/possible use in the facilities. Besides targeted metals listed in Table 2 [arsenic (As), cadmium (Cd), and cobalt (Co)], which have BEI, we also included antimony (Sb), beryllium (Be), chromium (Cr), indium (In), lead (Pb), manganese (Mn), nickel (Ni), tin (Sn), and vanadium (V). Three elements, Be, In, and Ni were not detected in any of the urine samples. The levels of Cr and Mn were also too low to be quantified. Therefore, the subsequent analyses were focused on As, Co, Cd, Pb, Sn, Sb, and V. Analyses were conducted at Purdue University's Campus-wide Mass Spectrometry Center. Quality control measures were routinely taken in the labs to ensure the accuracy and precision of the sample results.
Urinary creatinine level was analyzed for each sample and was used to normalize concentrations of solvents and metals in urine samples. Creatinine-corrected concentrations (µg/g creatinine) were used to compare results before and after the work shift as creatinine correction accounts for the dilution of urine caused by water or liquid consumption. This means if two workers had the same chemical exposure, but one drank twice as much water, both chemical and creatinine concentrations would reduce. The creatinine correction would count for the water consumption and show that these two workers’ exposure were similar. Uncorrected concentrations (µg/L) were used when comparing the results with exposure limits if the limits were set as uncorrected concentrations.
Statistical Analysis
All statistical analyses were performed using SAS system software (SAS Institute, Cary, NC) using a significance level of 0.05 (two-tailed). Considering the highly skewed distributions, the analyses were carried out using (natural) logarithmic transformation of concentrations. Mann-Whitney U tests (Proc NPAR1Way of SAS with Wilcoxon option) were performed to determine whether metal or solvent concentrations differed between exposed workers and controls. Paired t-tests were used to test for differences in the concentrations of the chemicals between, before, and after the work shift.
Results
Study Population
Twenty-eight exposed workers were from five different facilities. Controls were union representatives or office workers from the same/similar facilities. A summary of demographic data (age, gender, and workplace) for exposed workers and controls is listed in Table 3. The exposed workers worked specifically in plating, washing, soldering, and rework processes. The job titles and number of workers recruited from these jobs are listed in Table 4.
Cohort Demographics and Exposure Summary.
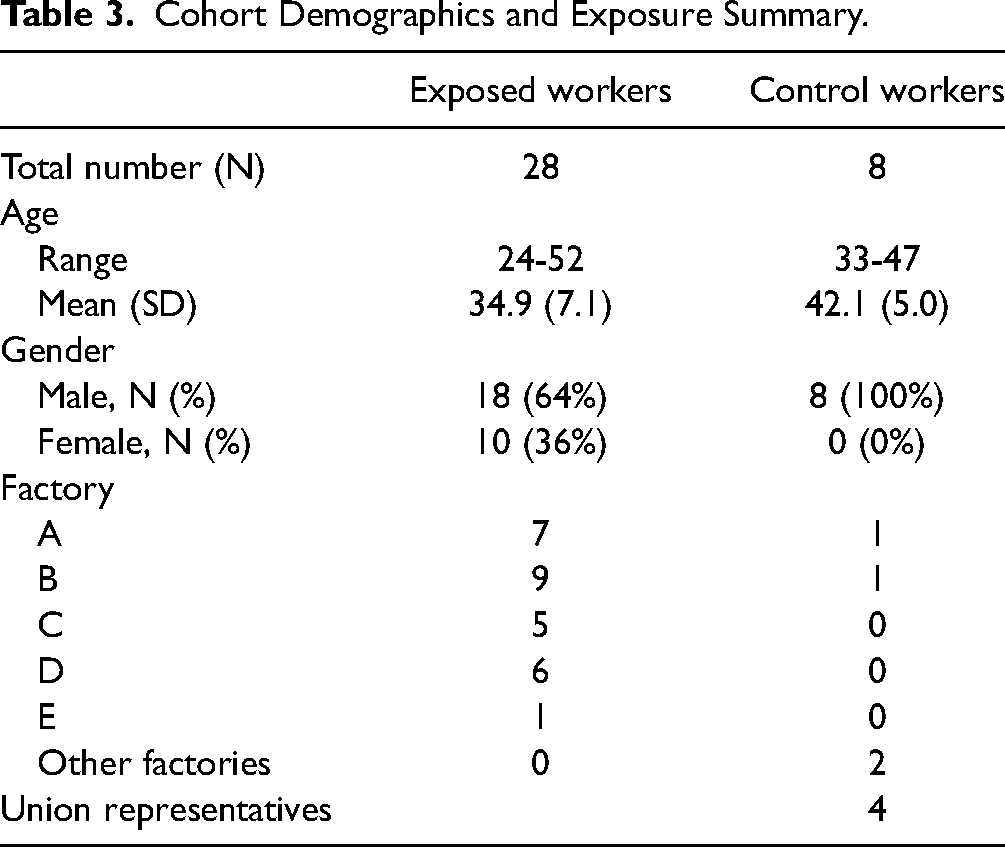
Job Titles Held by Exposed Workers.
Solvent Exposure
Solvent Concentrations in Urine Samples
Toluene and xylenes were detected in almost all the samples (97% for toluene and 93% for xylenes), whereas benzene and MEK were detected in most of the samples (85% for benzene and 72% for MEK). In contrast, acetone and ethyl benzene were only detected in 46% and 6% of the samples, respectively. Ethyl benzene was not included in the subsequent analysis because it was only detected in 4 samples. The concentrations of these solvents (average, median, and range) are listed in Table 5. These concentrations were not corrected for creatinine concentrations so that the values could be compared to their BEI. Concentrations of acetone, methyl ethyl ketone, and toluene were much lower than their BEI (Table 5).
Concentrations (µg/L) of Solvents Detected in the Urine Samples From All Participants (n = 36) Concentrations Are Not Corrected for Creatinine.
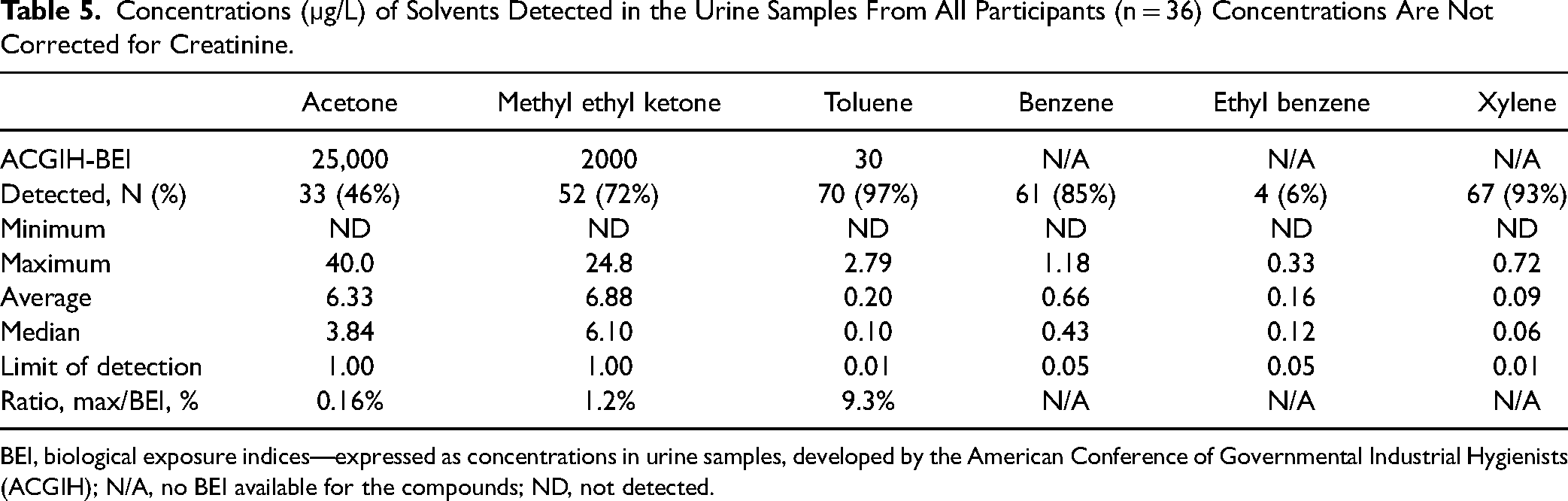
BEI, biological exposure indices—expressed as concentrations in urine samples, developed by the American Conference of Governmental Industrial Hygienists (ACGIH); N/A, no BEI available for the compounds; ND, not detected.
Solvent Concentrations Before- and After-Shift
When comparing the urinary concentrations of solvents before and after the work shift among the exposed workers, we observed increases in the concentrations of acetone, benzene, toluene, and xylenes from before to after their shift (Figure 2). However, none of these increases were statistically significant. Surprisingly, the concentrations of these solvents also increased among the controls from before to after the work shift, indicating either a phenomenon of bystander exposure or the existence of non-work-related exposures during the work day (e.g., cigarette smoking). Solvent concentrations were corrected for creatinine concentrations to compare the concentrations before and after the shift.
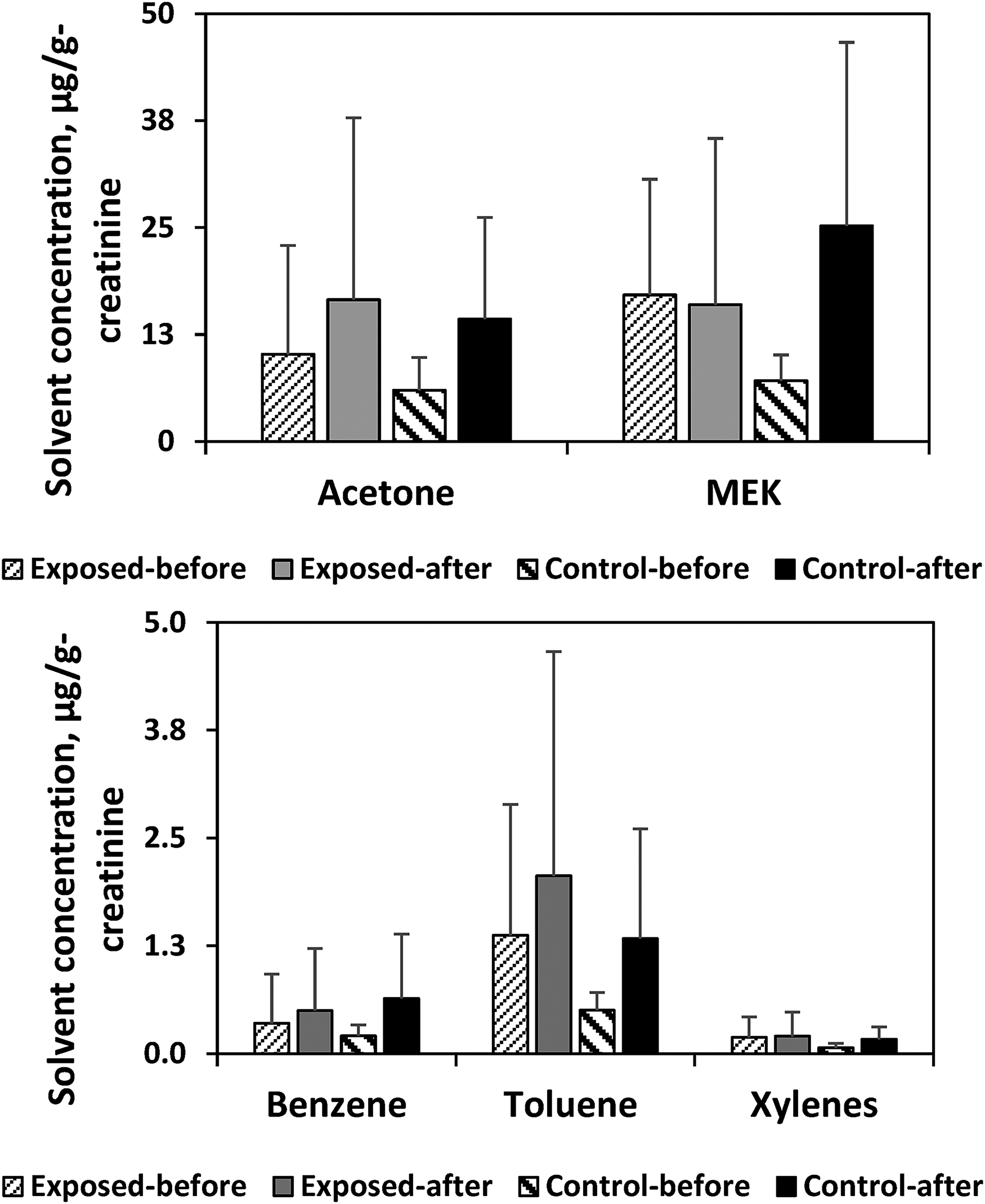
Solvent concentrations for exposed (n = 28) and control (n = 8) workers before and after the work shift. Concentrations were corrected for creatinine.
Difference Between Exposed Workers and Controls
When the concentrations of solvents among exposed workers were compared with those of controls, we observed higher concentrations among exposed workers for all five solvents before shift (Figure 2). However, none of these differences were statistically significant. Furthermore, none of these differences increased after the work shift ended, suggesting no evidence of work-related contributions to the initial differences in urinary solvent concentrations before the shift. Chemical exposure typically varies by processes and job titles held by the workers. We were not able to analyze exposure by job titles due to the small sample size. However, we provided the urinary solvent concentrations by job titles in Supplemental Information (S.1).
Metal Exposure
Metal Concentrations in Urine Samples
Arsenic, Cd, Co, and Pb were detected in >90% of the samples, whereas Sb, Sn, and V were detected in 61%-68% of the samples. Their concentrations are listed in Table 6. These concentrations were not corrected for creatinine concentrations so that the values could be compared with their BEI. The average concentration of As in the urine samples was 58.6 µg/L, exceeding the BEI of 35 µg/L. The maximum concentration of Cd was 3.03 µg/g creatinine, which reached 61% of its BEI of 5 µg/g creatinine. Concentrations of Co were much lower than its BEI.
Concentrations of Metals Detected in the Urine Samples From All Participants (n = 36), Not Corrected for Creatinine Levels.
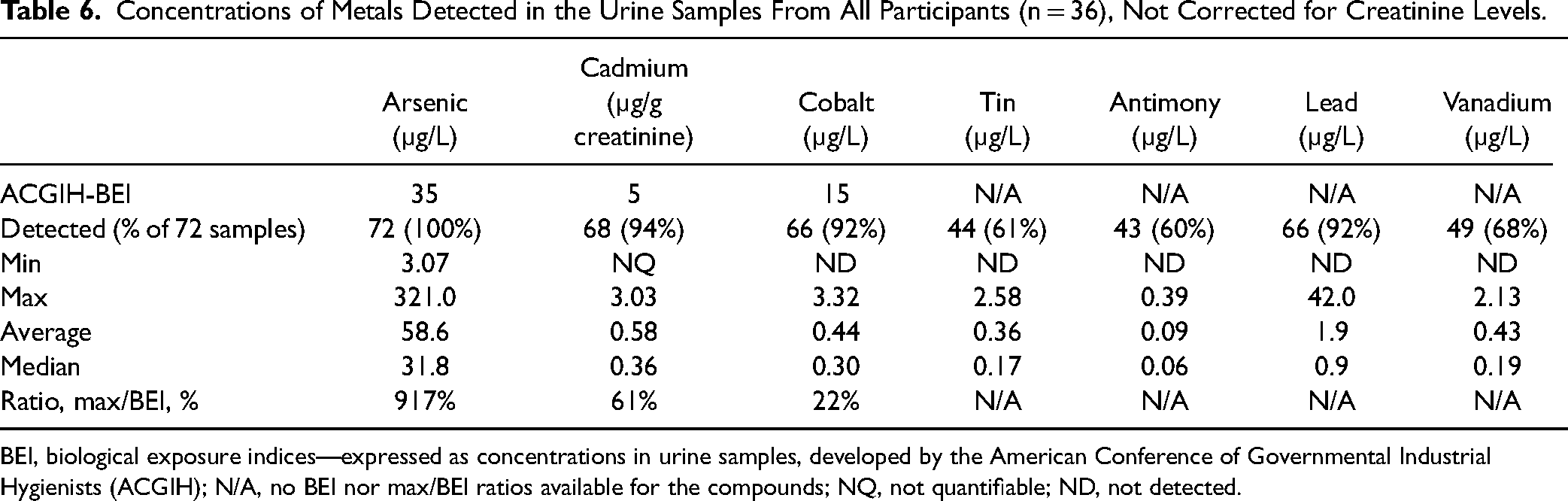
BEI, biological exposure indices—expressed as concentrations in urine samples, developed by the American Conference of Governmental Industrial Hygienists (ACGIH); N/A, no BEI nor max/BEI ratios available for the compounds; NQ, not quantifiable; ND, not detected.
Metal Concentrations Before- and After-Shift
When comparing the concentrations of metals before and after the work shift among the exposed workers, we observed small increases in the concentrations of Cd and V, but these increases were not statistically significant. We did not observe any increases in the concentrations of Sn, Sb, Pb, Co, or As (Figure 3). Again, it is surprising to see that concentrations of metals increased among the controls from before to after work shift for six out of seven metals, indicating either bystander exposure or non-work-related exposures during the work day (e.g., cigarette smoking or dietary intake). Metal concentrations were corrected for creatinine concentrations to compare the concentrations before and after the shift.
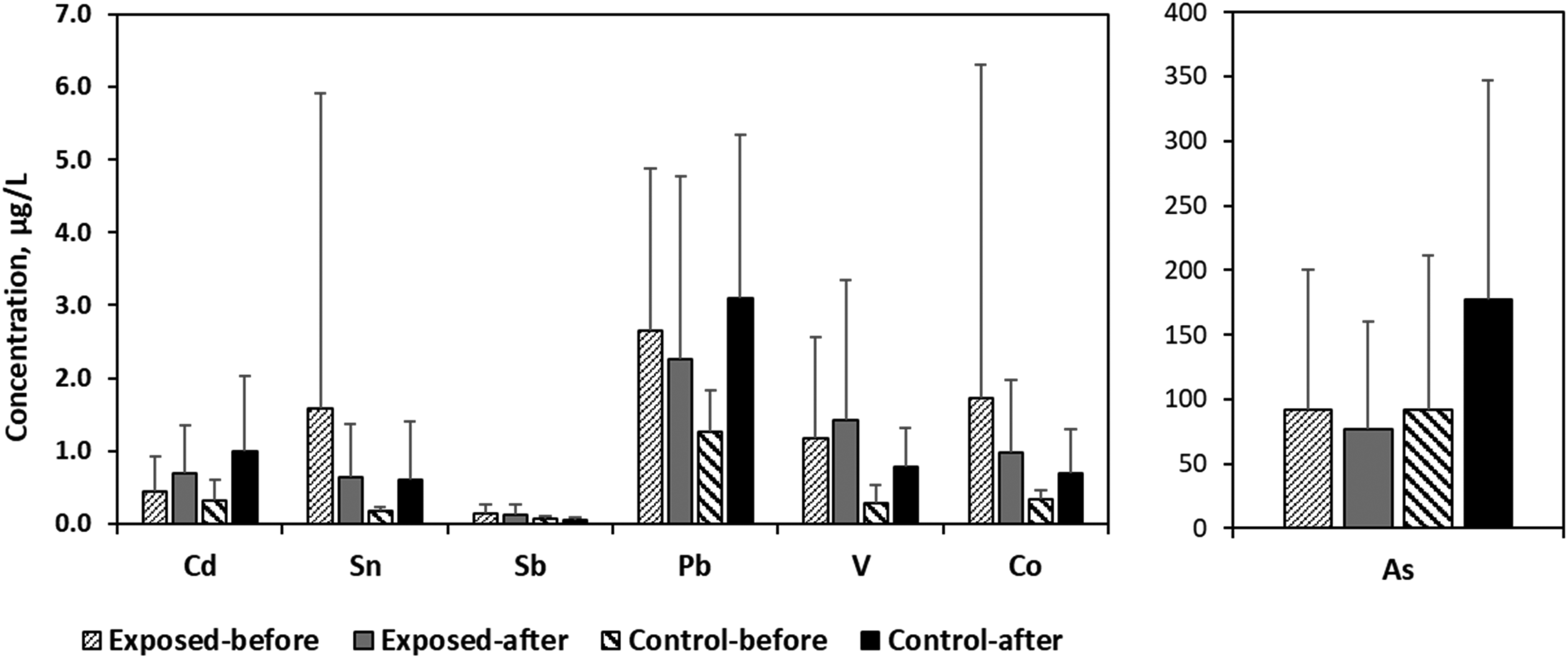
Element concentrations for exposed (n = 28) and control (n = 8) workers before and after the work shift. Concentrations were corrected for creatinine.
Difference Between Exposed Workers and Controls
When the concentrations of metals among exposed workers were compared with those of controls, we observed higher concentrations among the exposed workers than those among the controls before the work shift for all the metals (Figure 3). However, none of these differences were statistically significant. Unexpectedly the contrasts became smaller after the shift, mainly because of the increases in the metal concentrations in the controls during the work day, suggesting non-work-related sources of exposure mainly among the controls. We provided the urinary metal concentrations by job titles in Supplemental Information (S.2).
Discussion
To determine whether observed urinary chemical concentrations were related to workplace exposure in the electronics manufacturing facilities, we compared urinary chemical levels among the exposed workers before and after the shift, and between exposed workers and controls before and after the shift. We observed increases in concentrations among the exposed workers from before to after shift, such as for acetone, benzene, toluene, xylenes, Cd, and V (Figures 2 and 3). We also observed higher concentrations of all chemicals among the exposed workers than among the controls before the work shift. However, we could not conclude that these increases or differences were related to workplace exposures, because firstly, none of them were statistically significant; and secondly, urinary concentrations of these chemicals also increased among the controls from before to after shift, and none of the differences between the exposed and the controls before the shift increased after the work shift.
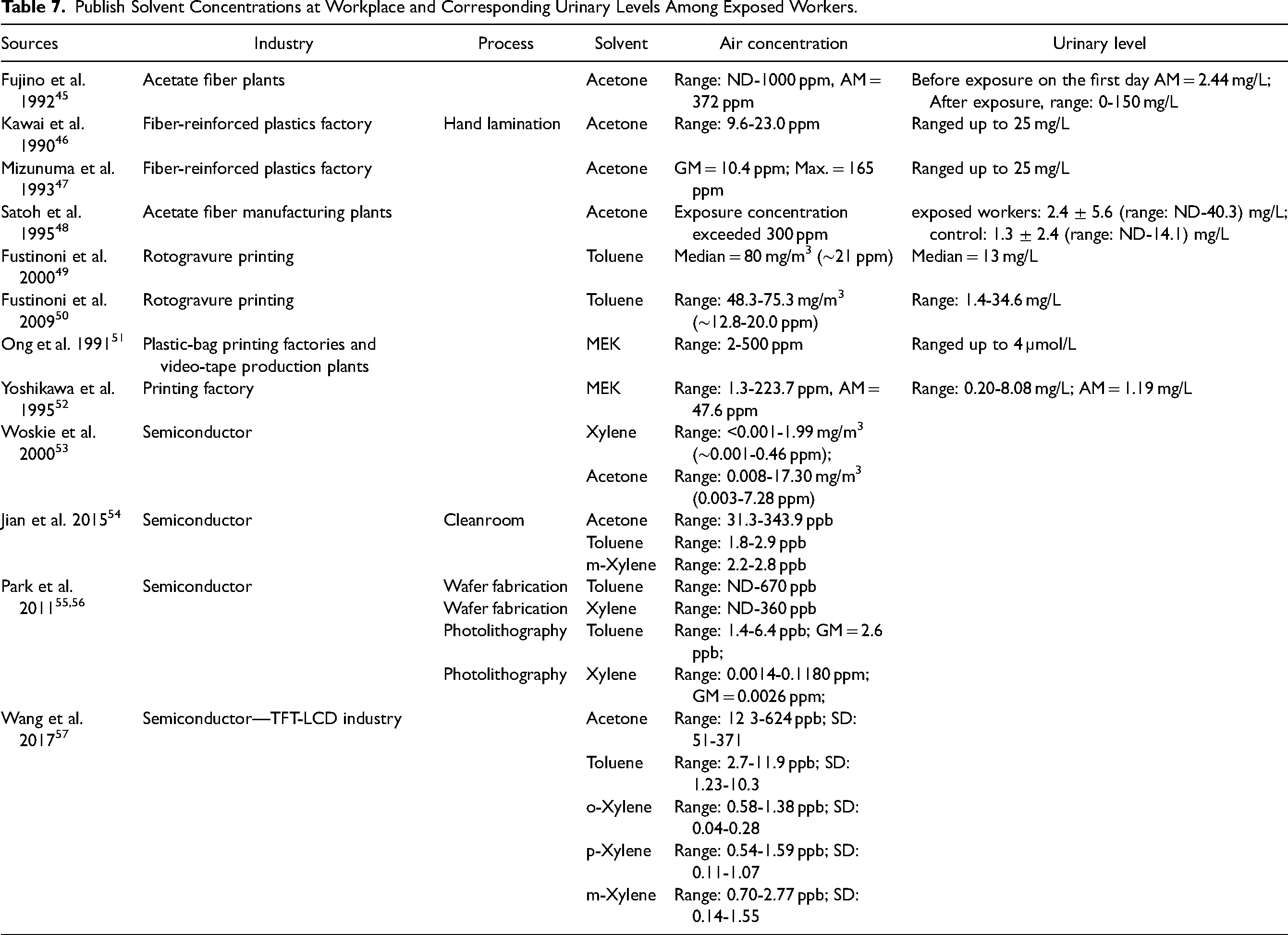
Other studies have reported urinary concentrations of acetone, toluene, MEK, and xylenes for individuals who worked in industries where solvent exposures were high, such as in acetate fiber plants and fiber-reinforced plastics factories. The reported exposure levels as air concentrations were generally ranged from ≥1 ppm to <1000 ppm. (Table 7). Corresponding urinary concentrations of solvents among exposed workers were also high, e.g., in the low ppm range (Table 7). Several more recent studies reported urinary concentrations of benzene, toluene, xylene, and ethylbenzene in petrochemical plants, 36 toluene and methyl ethyl ketone in a rubber shoe factory, 37 toluene in paint factories,38,39 bisphenol in plastic industry, 40 toluene in automotive garage, and urinary metabolites of the solvents in electronics manufacturing and the e-waste industry.41–43 However, none of these recent studies directly measured the solvents (parent compounds) in urine samples from workers in the electronics manufacturing industry. As indicated in Table 7, recent data on urinary concentrations of solvents and metals among workers in the electronics manufacturing industry are still limited. Hence, there is a need for monitoring data collection. 44 Based on air concentrations reported in semiconductor cleanrooms or TFT-LCD facilities, which were generally lower than what has been observed in the traditional manufacturing industries (e.g., typically ranged from ≥1 ppb to <1000 ppb.) (Table 7), we anticipate that urinary solvent concentrations among workers in electronics manufacturing facilities would also be lower, e.g., 100-1000 times lower than what reported in the aforementioned traditional manufacturing industries. Our results in general are consistent with this expectation.
Publish Solvent Concentrations at Workplace and Corresponding Urinary Levels Among Exposed Workers.
There were several challenges that we faced in the study. The small sample size limits the statistical power to detect potential differences in chemical urinary concentrations between exposed and controls and therefore limits the generalizability of the results. In this situation, many factors may have influenced the contrast in the urinary concentrations of chemicals between exposed workers and controls. These factors might include variations in day-to-day exposure, exposure misclassification, dietary intake, cigarette smoking or secondhand smoking exposures, gender effects, and potential sample loss during sample shipment.
Exposure Misclassification
We used self-reported exposure status to recruit exposed workers and controls. Some exposed workers might have low exposure levels that were not distinguishable from that of the controls. It is possible that office workers were exposed to fugitive factory emissions or to “cross-contamination” from poor or inappropriately designed ventilation or contaminated drinking water. Future biomonitoring studies in the electronics supply chain should include more exposed workers as well as controls who do not work in the same facility to increase the validity of the study.
Smoking
Given the prevalence of active smoking in Indonesia (∼30%) and potential exposure to secondhand smoking, interference of cigarette smoking could be a limitation of our study. In our survey, we inquired about the current smoking status of the participants which revealed that approximately 52% of the exposed workers and 47% of the controls were smokers. Stratified analysis did not reveal any significant effect of smoking status on the comparison of exposure before and after shift between exposed and control workers. This could be due to different smoking patterns among workers and controls as well as exposure to secondhand smoke.
Gender
There were no females among our controls, as few women worked as union representatives or office workers in the electronics industry in Batam, Indonesia. Exposure to certain chemicals such as toluene and lead could lead to gender-specific health outcomes. This pilot study focused on evaluating exposure and testing the feasibility of conducting a participatory biomonitoring project. We did not observe gender-specific differences in exposure among exposed workers. However, the lack of female controls resulting from the shortage of female union representatives may have effects on the comparison of exposure before and after shift between exposed and control workers. Promoting female workers to union staff could help improve the gender diversity in the union leadership and promote occupational health issues specific to female workers.
Shipment Problems
The samples were shipped via FedEx Overnight from Singapore to Purdue University, the fastest shipping mechanism identified at the time of the study. However, the shipment was held up at customs in Memphis for several days and the dry ice had to be replaced. The samples arrived with adequate remaining dry ice, but this factor raised a concern about potential impact on solvent analysis.
Nonetheless, our study was the first to report biomonitoring data on chemical exposure in electronics manufacturing facilities in Southeast Asia. Although the evidence is insufficient to meet the rigorous test of “statistical significance” due to the small sample size of the study, the observed higher urinary concentrations among the exposed workers compared to the controls before the work shift pointed to potential cumulative workplace exposures. However, due to the short biological half-lives of the chemicals studied (most within 24 h), “cumulative exposure” refers to accumulation over less than a week, meaning that neither for the exposed group nor the controls are the results likely to reflect exposure at previous jobs. Biomonitoring and workers’ participatory approaches allowed us to collect urine samples outside the workplace without needing the cooperation and participation of the management. That is clearly one of the main benefits of this strategy since there is so little exposure monitoring and medical monitoring going on currently in the electronics global supply chain. Access to factories has been very limited in the factories in the Southeast Asian region even for trade unions (and unions are rare—often non-existent—throughout the region, which makes this work even more difficult).
As to the occupational participatory research, this pilot study was a very challenging project with many moving parts to coordinate and with very few resources. Workers’ active participation and involvement of the union and NGOs played essential roles in study planning, securing the funding, conducting the workshop, recruitment, sample collection, and sample handling. Attempting to conduct such a study in a non-union facility would be much more difficult, and since most of the global electronic industry is non-union, the possibility of conducting more biomonitoring studies without the cooperation and support of management is limited. The workshop conducted at the beginning of the study increased awareness of chemical exposure and motivated workers’ participation in the study. It was an incredible team effort by many organizations and individuals. We plan to develop a research brief for this study that can be published on the web sites of the participating organizations. The unions, NGOs, and researchers involved in this study plan to continue the collaboration and conduct further biomonitoring investigations in the region. If electronics workers and their allies are going to consider additional biomonitoring projects, we need to learn the lessons from this historic first case and do the following:
It is essential to do this work in places that have strong unions of electronics workers to assure robust worker recruitment, training, and participation in sample collection—FSPMI was key to this project and is so important, particularly since there are very few places in the electronic global supply chain that have strong unions. Increase the time from the first sample to the second sample—such as the first sample taken on Monday morning before going to work and the second sample taken on Friday afternoon at the end of the shift—that will provide a more accurate picture of the actual exposures to the workers. Increase the sample size to include more workers participating in the study and also increase the exposure contrast by recruiting highly exposed workers and “controls” who are clearly not exposed. Passive samplers can be considered for air monitoring at the workplace. Additional research should include a more detailed investigation of how participatory approaches to biomonitoring can contribute to system building for exposure assessment and occupational health risk reduction. To develop an effective system for chemical exposure assessment, it is important to coordinate various components into the system, including sample collection, sample analysis, results interpretation, and results dissemination. Capacity building for local laboratories and establishing connections with the existing data collection mechanisms should be given thorough consideration within the framework of the participatory approach. These issues should be addressed systematically and considered in advance during the initial feasibility assessment phase of future participatory research projects.
A report-back meeting was conducted in December 2022, organized by the union, the participating NGOs, and the funding agency, and was participated by the representatives of these organizations and volunteer workers. The long history of toxic chemical exposures to workers in Silicon Valley going back to the 1970s was presented to attendees who then discussed it. Participants also learned that the legal exposure limits for workers are not health-protective. Just because exposure is “legal” does not mean it is safe. Therefore, increasingly workers, unions, and NGOs have been advocating for the elimination and substitution of hazardous chemicals throughout the global electronics supply chain. Our research can contribute to the improvement of working conditions through building capacity among workers, unions, and NGOs to participate in, to co-design, and to implement investigative monitoring projects. The experience can be used to inform the development of collaborative partnerships, and participatory research guidelines, as well as continuous monitoring and improvement.
Conclusions
We analyzed six solvents (acetone, methyl ethyl ketone, toluene, benzene, ethyl benzene, and xylenes) and seven metals (As, Co, Cd, Sn, Sb, Pb, and V) in urine samples collected from exposed workers (n = 28) and controls (n = 8) in electronics manufacturing facilities located in Batam, Indonesia. Among the chemicals that have BEI to compare with, the average concentration of As in the urine samples exceeded its BEI; the maximum concentration of Cd and Co reached 61% and 22% of their BEI; and concentrations of acetone, methyl ethyl ketone, and toluene were much lower than their BEI. We observed that urinary concentrations of several chemicals increased among exposed workers from before to after the shift and were higher among exposed workers than among the controls at the beginning of the work day. The participatory research approach in combination with biomonitoring worked effectively in generating occupational exposure-related data in places where industrial hygiene support and recourses ma limited to conduct air monitoring at the workplace.
Policy Implications
In Asia, factory management tends to be highly resistant to biomonitoring, air sampling, and workplace inspection by external parties. The LIPS study 31 exposed the weak role of the Indonesian government in regulating the influx of hazardous and toxic chemicals and the lack of workplace chemical monitoring. LIPS found five different regulations on the use of chemicals in the workplace. However, the regulations are not strictly enforced nor regularly monitored by the government. In addition, the existing regulations are not updated regularly, even though, as Harrison pointed out in 1992 “The pace of change is quickening under the pressure of severe economic competition.” If this was true then, how much more must it be true now? As a result, it is necessary constantly to update regulations to protect workers’ health and safety and the environment. In the absence of government support, the lack of effective unions (or no unions at all), and the constant pressure from management in most of the lower-tier electronics suppliers to reduce the costs of production, worker health, and safety is rarely a priority. Hence, it is necessary to put pressure on the brands to insist that their suppliers provide a workplace where workers have decent working conditions and basic health and safety protections, including training on chemical hazards in their workplaces and regular industrial hygiene and medical monitoring testing to assure a safe and healthy workplace. The support of civil society under these circumstances is critical. In addition to providing workers with research and financial support, it is necessary to organize socially responsible investors as well as large electronics buyers, such as governments and universities to apply that pressure.
Many multinational companies with factories in Southeast Asia must take responsibility for the health and safety of all their workers regardless of their location. They should adopt a global approach to occupational health and safety and ensure that all their suppliers and contractors meet health-protective standards for chemical exposure monitoring and control. This will require the development of an effective management system that prioritizes the safety and health of workers and a new perspective from management to empower workers, especially ensuring that workers have access to the information on the materials they work with and are trained to understand the risks associated with chemical exposure. Investors and public buyers should pressure the brands to make sure their suppliers develop such management systems.
One example of movement in this direction is the Clean Electronics Production Network (CEPN), 58 a multi-stakeholder initiative that has set a goal of zero exposure to hazardous solvents in the global electronics supply chain. Members include multinational corporations such as Apple, HP, Dell, Intel, and Seagate, as well as NGOs, academics, and government agencies. Many NGOs participate as advisors. CEPN has launched an initiative called Toward Zero Exposure in which members agree to a series of commitments such as phasing out the most hazardous solvents and substituting safer ones. 59 CEPN is identifying the most hazardous solvents used in the supply chain and targeting them for removal and substitution with safer alternatives. Two of the chemicals that were identified in the workers’ urine in our study—benzene and toluene—are on the first list of target chemicals that CEPN published. CEPN will soon publish an additional list of chemicals. We urge CEPN to play a role in advocating workers’ right to know and access to Safety Data Sheets (SDS), easier workplace access for chemical exposure monitoring and data collection, and monitoring waste discharge from electronics manufacturing facilities to protect surrounding communities and the environment.
A coalition of socially responsible investors and large electronics buyers could demand the following:
Workers’ right to know and access to SDS. The electronics industry has faced the fundamental demand for the right to know since its inception in Silicon Valley, where local city councils passed ground-breaking laws in the early 1980s. These laws required companies to disclose their chemical inventories to the public. These provisions were later incorporated into state laws and federal laws. Unfortunately, countries in Asia have been much more resistant to toxic chemical transparency for workers or communities. Many observers believe that this is one of the important reasons that many companies have moved production to Asia. While the right to know has been widely adopted in many parts of the world, government support is still sadly lacking in Asia. Because many workers in Asia lack both right to know and union representation, it is extremely difficult for Asian workers to enjoy the same protections that are almost taken for granted in many places. It continues to be one of the great ironies that the electronics industry, which makes information technology, continues to resist disclosure of hazardous chemicals information to its own work force. Workers’ right to know and access to SDS are especially important, given what Dr. Myron Harrison articulated in the article in Hazardous Materials Toxicology about the rapid pace of change in tools and materials and the inability conducting adequate toxicological assessment to proceed the introduction of hazardous material into manufacturing settings;
28
Easier workplace access for chemical exposure monitoring data collection. Unions and other worker representatives should have authority to arrange for monitoring data collection when government agencies lack the resources and/or the will to inspect and monitor workplaces; Incorporate biomonitoring data in the occupational health policy making in Southeast Asian countries; to do so, government agencies, such as the Manpower Office in Batam, academic institutions, industry representatives, and labor groups should collaborate to develop and implement protocols for biomonitoring data collection, analysis, and reporting. These protocols should be consistent with international best practices. There should be a centralized database for biomonitoring data, which can be accessed by stakeholders including government agencies, researchers, and the general public. Investors and public buyers should encourage management to participate in biomonitoring. Study results need to be disseminated to the management in order to generate meaningful changes. When seeking to engage management in participatory biomonitoring initiatives, potential concerns may arise regarding the impact of research results on regulatory requirements and associated costs related to enhancing workplace safety and environmental practices. These concerns may manifest as short-term cost increases and potential pressure from investors to abstain from participation. Additionally, increased costs may worsen a company’s competitiveness in global markets. To encourage management participation in these situations, it may be necessary to provide management with information about the benefits of participatory biomonitoring and highlight how management involvement can lead to improved workplace safety, regulatory compliance, and enhanced corporate social responsibility. Also, providing regular reports on the progress of the program and its impact on exposure reduction and health outcomes would be a good strategy, highlighting the impact they may have on local community settings. Additionally, researchers can highlight that management’s participation in occupational health and safety initiatives, including biomonitoring, is consistent with legal and regulatory requirements. Monitoring waste discharges from electronics manufacturing facilities has not been regulated but is alarmingly important to investigate as several cases of electronics waste being dumped have emerged. This can be done by activists and scientists without support of governments and employers; Activists and scientists can play an important role in raising awareness, conducting preliminary research, and advocating for improved waste management practices. Activists and scientists can make a valuable impact by raising public awareness, and pressuring employers to take responsible action in managing waste from electronics manufacturing. Investors and public buyers can demand that the brands require that wastewater be monitored, and the data be made public as a condition of doing business. Promoting and supporting collaborations of the unions, NGOs, and researchers and building local capacities in conducting workers’ participatory research projects to identify workplace exposures, and generate data more efficiently to promote policy making to reduce exposure and protect workers’ health.
Supplemental Material
sj-docx-1-new-10.1177_10482911231218478 - Supplemental material for Biomonitoring of Exposures to Solvents and Metals in Electronics Manufacturing Facilities in Batam, Indonesia
Supplemental material, sj-docx-1-new-10.1177_10482911231218478 for Biomonitoring of Exposures to Solvents and Metals in Electronics Manufacturing Facilities in Batam, Indonesia by Jung Hyun Lee, Vesara Ardhe Gatera, Ted Smith, Fahmi Panimbang, Alejandro Gonzalez, Rizky Abdulah, Connie Bonham, Alaina Kateri Bryant and Sa Liu in NEW SOLUTIONS: A Journal of Environmental and Occupational Health Policy
Footnotes
Data Availability
Data are contained within the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the GoodElectronics Network and supported by a training grant from the Developing World Outreach Initiative, an initiative of the Northern California Section of the American Industrial Hygiene Association.
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Purdue University (Protocol #1903021912 and approval date: May 14, 2019) for studies involving humans.
Informed Consent
Informed consent was obtained from all subjects involved in the study.
Supplemental Material
Supplemental material for this article is available online.
Notes
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
