Abstract
Reducing the burden of mutant Huntingtin (mHTT) protein in brain cells is a strategy for treating Huntington’s disease (HD). However, it is still unclear what pathological changes can be reproducibly reversed by mHTT lowering and whether these changes can be measured in peripheral biofluids. We previously found that lipid changes that occur in brain with HD progression could be prevented by attenuating HTT transcription of the mutant allele in a genetic mouse model (LacQ140) with inducible whole body lowering. Here, we tested whether intrastriatal injection of a therapeutic capable of repressing the mutant HTT allele with expanded cytosine-adenine-guanine (CAG) can provide similar protection against lipid changes in HD mice with a deletion of neo cassette (zQ175DN). Wild-type or zQ175DN mice were injected with adeno-associated virus 9 (AAV9) bearing a cDNA for a zinc finger protein (ZFP), which preferentially targets mutant HTT (ZFP-HTT) to repress transcription. Proteins from brain tissues were analyzed using western blot, capillary electrophoresis, and nitrocellulose filtration methods. Lipid analyses of brain tissue and plasma collected from the same mice were conducted by liquid chromatography and mass spectrometry (LC-MS). Somatic instability index was assessed using capillary gel electrophoresis of PCR products and was shown to be impeded by ZFP-HTT. Lowering mHTT levels by 43% for 4 months prevented loss of total lipid content including the subclasses sphingomyelin, ceramide, phosphatidylethanolamine and others of caudate-putamen in zQ175DN mice. Moreover, LC-MS analysis of plasma demonstrated total lipid increases and lipid changes in monogalactosyl monoacylglyceride and certain phosphatidylcholine species were reversed with the therapy. In summary, our data demonstrate that analyzing lipid signatures of brain tissue and peripheral biofluids are valuable approaches for evaluating potential therapies in a preclinical model of HD.
INTRODUCTION
A heritable mutation causing abnormal expansion of a normal cytosine-adenine-guanine (CAG) repeat sequence in the HTT gene causes Huntington’s disease (HD), 1 a neurodegenerative disease in which patients suffer progressive cognitive, psychiatric, and motor symptoms, and ultimately death. 2 Further CAG repeat expansion can happen in individual cells with age (somatic instability, SI), and genes involved in DNA mismatch repair were identified as modifiers of age of onset in human genome-wide association studies. 3,4
Because mutant HTT gene products may be involved in diverse pathological mechanisms, gene therapy approaches to block the expression of HTT gene products may be superior to other therapies targeting downstream mechanisms. One approach is to target the HTT gene directly at the level of transcription using proteins engineered to bind the mutant allele, such as a zinc finger protein (ZFP), which has been shown to selectively repress transcription from the mutant HTT allele containing high CAG repeats. 5 This ZFP fusion protein has the advantage of blocking all gene products, 5 including the small HTT1a exon1-intron1 readthrough transcript. 6
Several mouse models of HD are available for preclinical testing. The R6/2 model overexpresses a transgene of HTT exon1 with a highly expanded CAG tract (180 CAGs) resulting in profound motor abnormalities and death around 16 weeks of age. 7 In contrast, knock-in models such as Q140 mice express near endogenous levels of mutant HTT protein (mHTT) and have a more protracted sequelae of neuropathology progressing in a similar path to HD patients; 8 however, appreciable motor symptoms are mild and most do not present until 6 months to 1 year. The zQ175 mouse line was isolated as a natural variant from the Q140 line in which the CAG tract had expanded and it displays transcriptomic changes and electrophysiology changes by 6 months; 5,9 subsequent excision of the neo resistance gene (delta neo [DN]) from zQ175 mice was found to boost expression levels of mutant HTT and produce more robust phenotypes including white matter changes. 10 zQ175DN mice were chosen for our lipidomic studies both due to the long window for therapeutic intervention and the potential to measure changes in white matter pathology.
Many of the experimental readouts reported in studies using HD mouse models are semi-quantitative, have high variability requiring numerous mice, and are labor intensive. The readouts used to assess the effects of HD gene lowering include mRNA levels for striatal genes known to change in HD (PDE10a and DARPP32), 11 immunofluorescence for DARPP32, ligand binding to PDE10a, dopamine receptors 1 and 2, 5 brain mass, volume of caudate-putamen, the presence of inclusions formed by mHTT, and behavioral and cognitive measures all assessed at one or two time points post treatment. 12 –14
RNAseq or microarray analysis are quantitative and give strong, reproducible phenotypes in HD mouse models 9,15 and have become an essential quantitative readout for lowering therapies. However, there was limited improvement in reversing changes in levels of mRNA transcripts specific to striatal medium spiny neurons in brains treated with an antisense oligonucleotides targeting HTT. 11 Only partial reversal of the transcriptomic signature occurred with a genetic inducible model of mHTT lowering, which lowered in all cell types (LacQ140). 16
Lipids constitute about half the dry weight of brain and are enriched in white matter. 17,18 HD patients suffer early and ongoing loss of white matter 19 –33 and degeneration of striatal and cortical neurons, cells with a vast surface area of membranes. 34 In a study of biopsy material from frontal cortex of HD patients, lipofuscin and organelles with the ultrastructure and electron density of lipid droplets were dominant features; these organelles were notable in neurons, and present in astrocytes and microglia, 35 suggesting widespread lipid derangements. Changes in lipids including in acyl carnitines, cholesterol ester, and sphingolipids have been identified from autopsy tissue of HD patients. 36 –38 Lipids or their metabolites might be released into peripheral circulation and lipid alterations have been detected in plasma of HD patients. 39 –41 Lipid changes have been described in brain from several HD mouse models, 42 –44 including in zQ175. 45,46 Thus, leveraging lipid surveys may be useful in evaluating potential treatments in preclinical models and have potential for development as biomarkers.
Using the highly accurate and quantitative method of liquid chromatography and mass spectrometry (LC-MS), we recently demonstrated in the LacQ140 mice that significant changes in levels of numerous lipids occurred, and reducing mHTT levels was protective. 47 Here, we tested whether the benefits of an injected therapeutic could be evaluated using LC-MS to quantitatively track global lipid changes in the brain and in a peripheral biofluid in the zQ175DN HD mouse model.
METHODS AND MATERIALS
Animal welfare statement
All the studies in this article were conducted in compliance with the Institutional Care and Use Committee guidelines of the University of Massachusetts Chan Medical School (docket #202100018).
Adeno-associated virus subcloning and packaging
Details of the adeno-associated virus (AAV) subcloning and packaging can be found in Figure 1A and Supplementary Data S1.
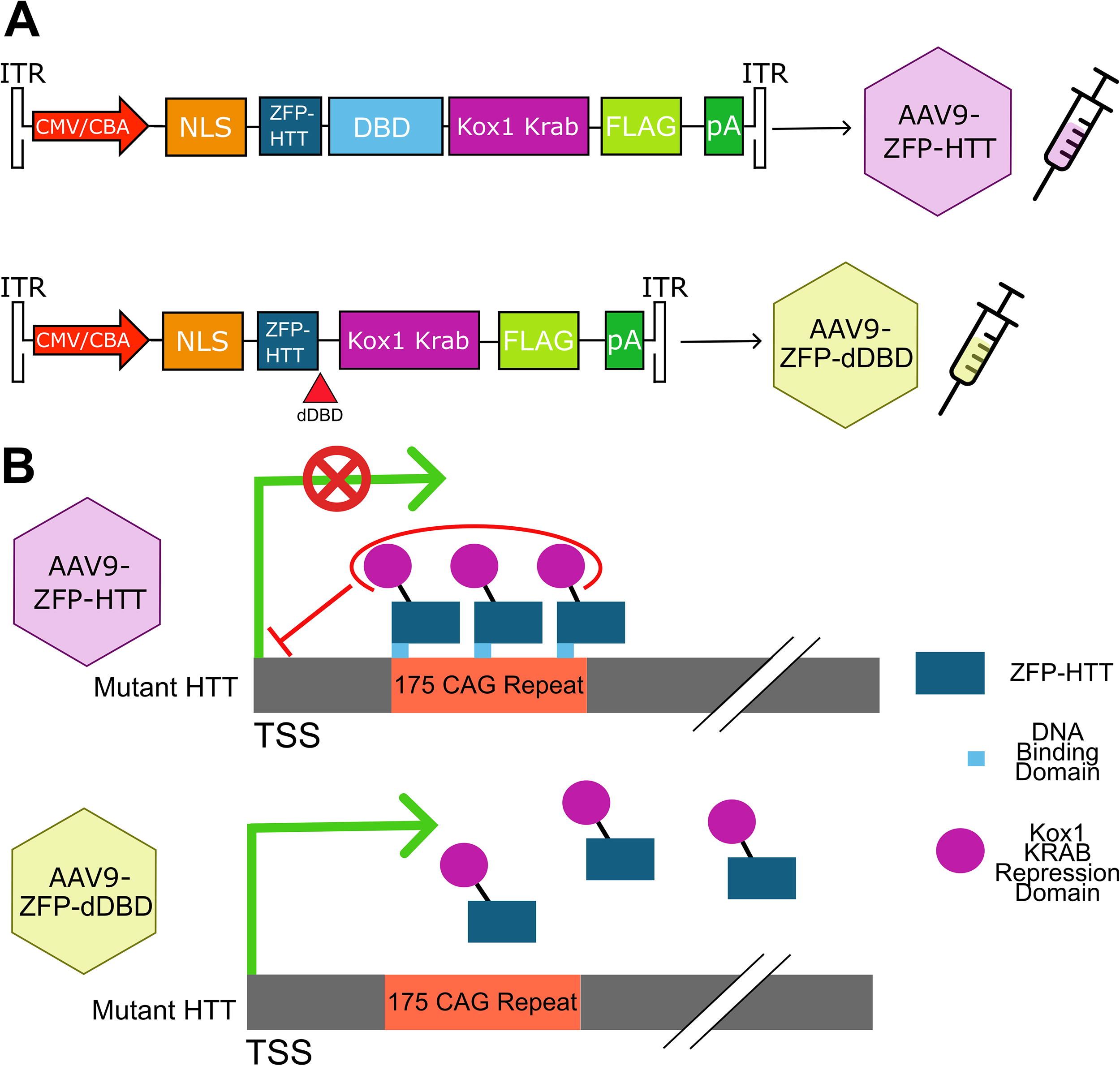
Diagram of (AAV9-ZFP-HTT) and AAV9-ZFP-dDBD constructs.
Mouse intracranial injection surgeries and tissue collection
Heterozygous zQ175DN HD mice (abbreviated as HD in Figures and Tables) and their wild-type (WT) littermate controls (Q7/Q7) were purchased from The Jackson Laboratories (Z_Q175 KI (neo-); (C57BL/6J)-CHDI stock #370832, JAX stock #029928). Briefly bilateral intrastriatal injections of AAV9 virus (3.5 × 1010 viral genomes) were performed at 8 weeks of age. At 6 months of age, brain tissue and plasma were collected. More details can be found in Supplementary Data S1.
Tissue fractionation, SDS-PAGE, Western blot analysis, capillary gel electrophoresis and filter trap assay
Details can be found in Supplementary Data S1.
Lipid extraction, LC-MS, and annotations
Lipids were extracted using the two-phase methyl-tert-butyl ether method as previously described. 48 Details are given in Supplementary Data S1.
Computational analysis/visualization
Pixel intensity quantification for immunoblotting was performed using ImageJ, more details can be found in Supplementary Data S1. To correct for duplicate annotation of lipid ions by LipidSearch, caused by wide elution profiles, all instances of identical lipid ions were summed together. Lipid ions were then filtered using the LipidSearch parameter (Rej. = 0) and lipid ions missing in over 20% of the samples were excluded from analysis. Data was then log-transformed and normalized with Eigen MS. 49 The principal component analysis was performed using FactoMineR (v2.11), data were centered and scaled. The total level of lipid in each sample was determined by summing the intensity of all the lipid ions in each sample. The heatmaps in figures four and five were generated in R using ComplexHeatmaps (v2.20.0.). 50 Statistical significance of lipid subclasses was determined by two-way analysis of variance between sex (male, female) and group (WT+ZFP-dDBD [delta DNA binding domain], HD+ZFP-dDBD, HD+ZFP-HTT), followed by Tukey’s t-test (p value), and the false discovery rate ([FDR], 5%) (q value) was controlled by the two-stage linear step-up procedure of Benjamini, Krieger, and Yekutieli (brain lipid subclass N = 42, brain lipid ions N = 2,398, plasma lipid subclass N = 47, plasma lipid ions N = 2,251). Data were evaluated for a significant effect of the interaction between sex and treatment groups. This interaction had no significant effect on plasma or caudate-putamen lipids or subclasses, so male and female data were combined. A lipid subclass/ion was considered recovered if the sign of the log2-Fold change was the same when comparing WT treated with ZFP-dDBD and zQ175DN mice treated with ZFP-HTT to HD mice treated with ZFP-dDBD, and the q value between the zQ175DN treatment groups was less than 0.05.
Somatic instability index
Details can be found in Supplementary Data S1.
RESULTS
Lowering of mutant HTT protein in zQ175DN HD mice
To test whether a gene therapy capable of allele-specific gene repression could prevent lipid-related neurodegenerative changes in an HD preclinical mouse model, prepared virus for AAV9-ZFP-HTT or AAV9-ZFP-dDBD (Fig. 1B) were introduced into the zQ175DN, HD mouse model, caudate-putamen with bilateral injections at 2 months of age, prior to known behavioral phenotypes in this model. 51,52 Wild-type (WT) Q7/Q7 mice were injected with AAV9-ZFP-dDBD virions as an additional control to obtain baseline levels of genotype-dependent changes in proteins and lipids. All tissues were harvested at 6 months of age when considerable changes in transcription have been described in zQ175/Q7 striatum compared with WT, nuclear and cytoplasmic aggregates are present, and electrophysiological changes can be detected. 5,9
To examine HTT protein levels, we performed capillary gel electrophoresis on crude homogenates of AAV-treated caudate-putamen of HD mice using anti-polyglutamine antibody, MW1, to detect mHTT. Results showed a significant lowering (48%) of mHTT with expression of ZFP-HTT but not with ZFP-dDBD in caudate-putamen of HD mice (Fig. 2A, B). We also performed western blot (WB) assays using the crude homogenates from both WT and HD-treated mice and probed with anti-N-terminal HTT antibody Ab1, which detects both WT HTT and mHTT (Fig. 2C, D). 47,53,54 Using this method, pixel intensity quantification showed that the level of mHTT was 43% lower in HD mice treated with ZFP-HTT compared with those treated with ZFP-dDBD (Fig. 2D). The levels of WT HTT in heterozygous zQ175DN HD were an average of 54 and 64% lower than in WT mice which have two WT alleles, as expected (Fig. 2D). There was no difference in WT HTT levels between the HD treatment groups demonstrating the specificity of the ZFP-HTT repressor protein for the mutant HTT allele. Thus, 4 months after injection, mHTT in crude homogenates was significantly lowered with ZFP-HTT treatment compared with the control virus (ZFP-dDBD) and WT HTT levels were spared.
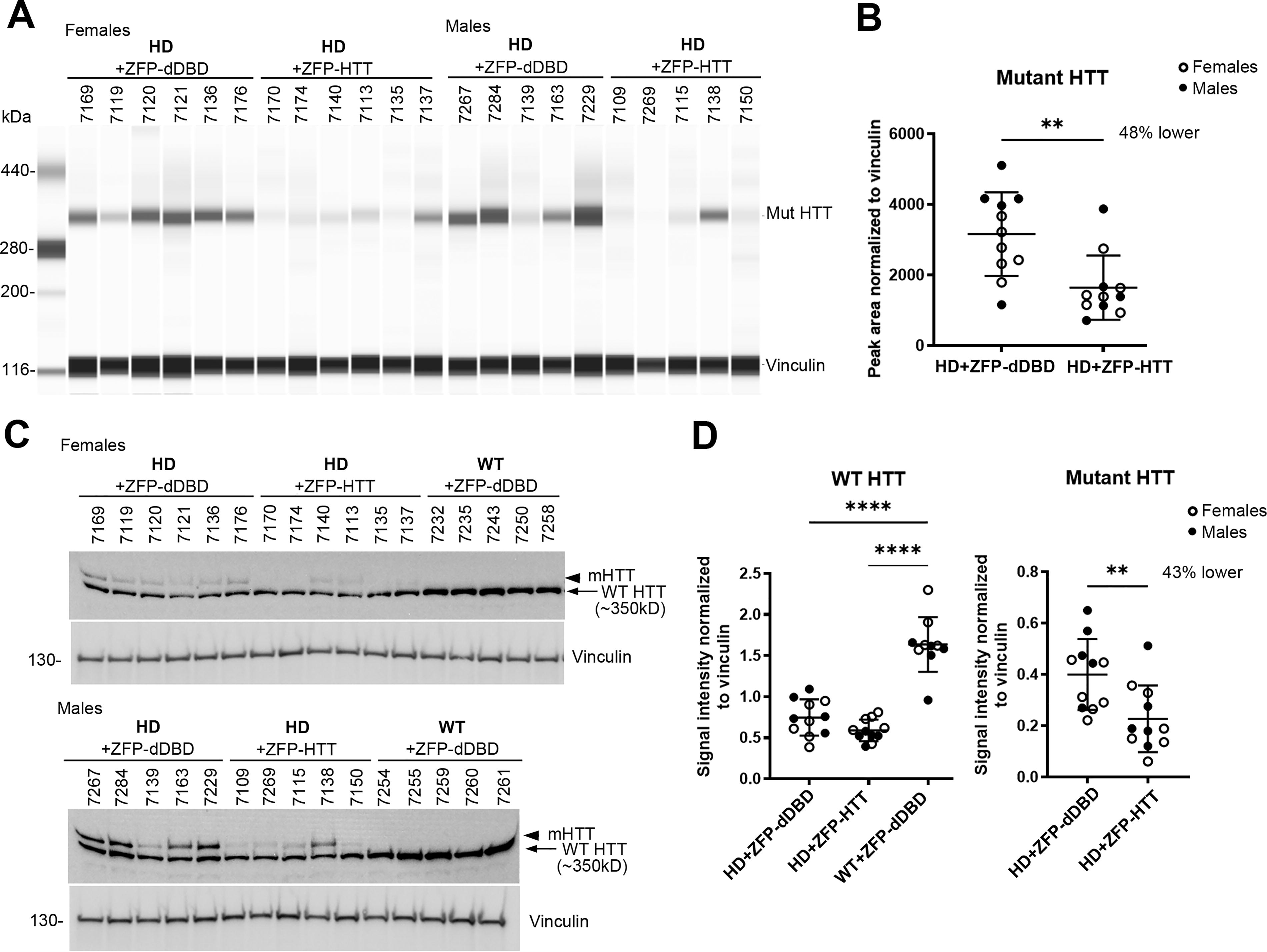
Capillary electrophoresis (WES) analysis and western blot analyses of protein levels of soluble normal and mutant Huntingtin in crude homogenates from zQ175DN caudate-putamen injected with AAV9-ZFP-HTT or AAV9-ZFP-dDBD and WT injected with AAV9-ZFP-dDBD.
A molecular species of mHTT persists with lowering by AAV9-ZFP-HTT
Previously, we showed that a molecular species of mHTT in the crude nuclear pellet fraction (P1) was resistant to lowering in LacQ140 mice. 47 We and others have shown that the anti-HTT antibody S830 detects a slowly migrating species of mHTT that appears as a high molecular weight smear in homogenates from brain tissues containing mHTT. 47,55,56 Using Ab1 on WB, levels of mHTT were 54% lower in P1 from zQ175DN mice treated with ZFP-HTT compared with ZFP-dDBD (Fig. 3A, B). WT HTT levels in the P1 fraction were an average of 50–53% lower in the zQ175DN mice compared with Q7/Q7, as expected for one allele versus two alleles of normal HTT, consistent with findings above in crude homogenates. However, antibody S830 recognized full-length soluble mHTT detected as a resolvable band and no lowering was observed in the AAV9-ZFP-HTT mice compared with those treated with AAV9-ZFP-dDBD; also, no lowering of the high molecular weight smear detected with S830 occurred (Fig. 3A, C). This result is consistent with our findings in LacQ140 striatum where an SDS-soluble, S830-positive species of HTT persists even with transcriptional lowering. 47 Next, a filter trap assay to analyze lowering of aggregated mHTT in P1 was conducted. MW8 antibody, which detects aggregated exon1 HTT, 55 showed a significant reduction of aggregated mHTT in the zQ175DN mice treated with AAV9-ZFP-HTT compared with those treated with AAV9-ZFP-dDBD (Fig. 3D).
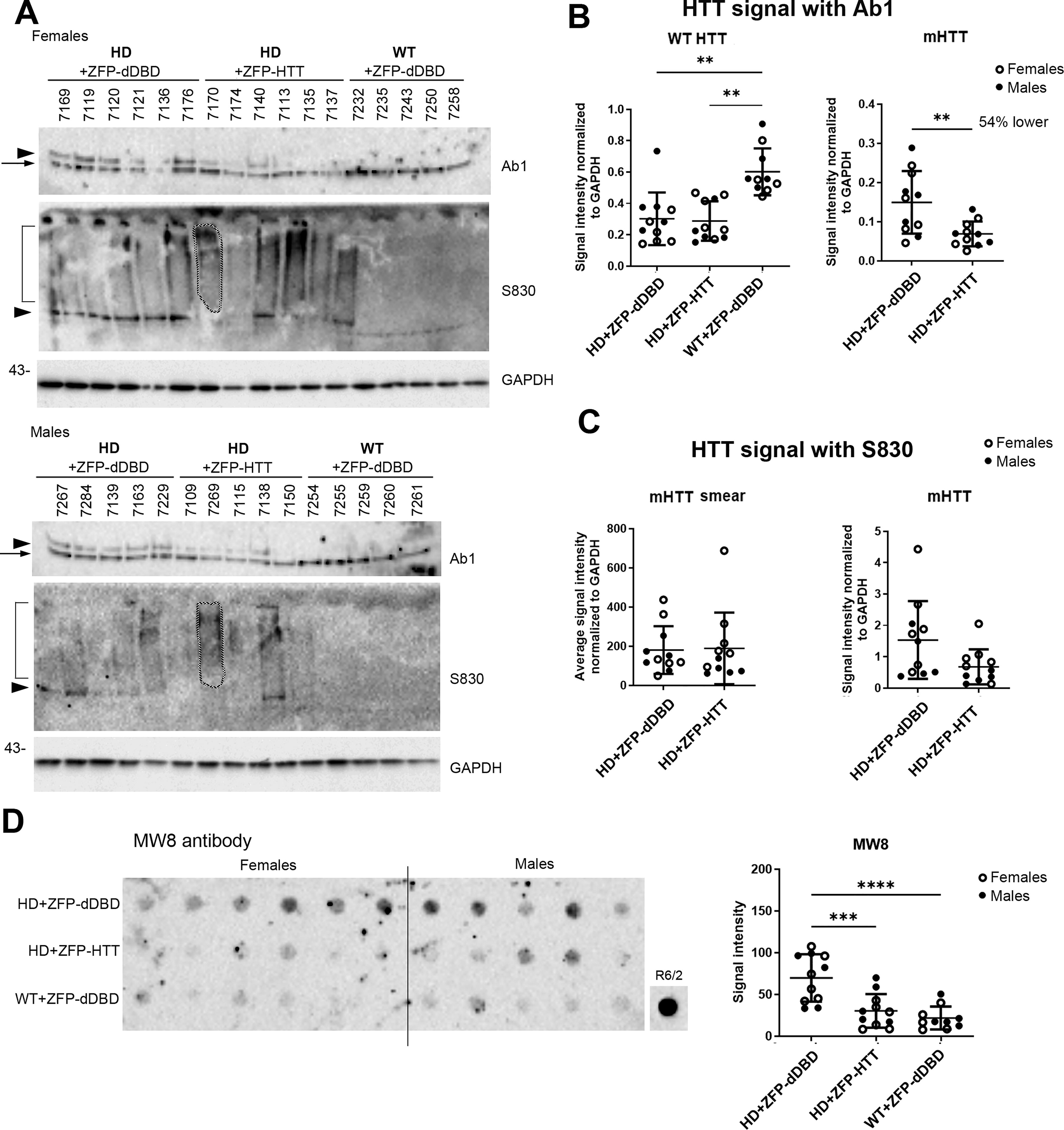
Protein analysis of the crude nuclear P1 fractions from caudate-putamen from mice injected with AAV9-ZFP-HTT or AAV9-ZFP-dDBD and WT injected with AAV9-ZFP-dDBD analyzed by western blot and gel filtration.
All together, these data indicate that reducing mutant HTT transcription with AAV9-ZFP-HTT lowers soluble and aggregated forms of mHTT in crude homogenate and P1 fraction, but that a S830-positive misfolded molecular species of mHTT, which is present in the crude nuclear fraction, resists lowering.
AAV9-ZFP-HTT prevents genotype-dependent lipid changes in zQ175DN HD caudate-putamen
Previously, we found that age and genotype-dependent lipid changes occur in both zQ175/Q7 mice 45 and LacQ140 mice, and these changes could be prevented by early whole body genetic downregulation of mutant HTT transcription in LacQ140 mice. 47 To test whether intrastriatal injection of a therapeutic would have the same protective effects, we surveyed lipids in crude homogenates of our AAV-treated mice by LC-MS. LC-MS analysis of lipids from caudate-putamen of treated WT and zQ175DN mice at 6 months of age yielded high coverage of lipids species with 2,398 unique lipid ions identified across 42 lipid subclasses. To assess the variation within the data, a principal component analysis (PCA) was performed. The projection of the two most informative principal components, PC1 and PC2 are shown in Figure 4A (PC1, 14.3% of variance; PC2, 13.0% of variance). Most of the variation along the first principal component was within each group. Along the second principal component, all groups showed separation from each other. The HD mouse models treated with ZFP-HTT are closer to the WT mice treated with ZFP-dDBD. To confirm the PCA results obtained using two components, we undertook a more in-depth quantitative analysis called k-means computational clustering, which considers changes in all lipid levels (increased or decreased). We queried which two of the groups (k = 2) were most similar using this method with 25 initial starting points employed on the first eight principal components which accounted for 57% of the total variation. zQ175DN mice treated with the control virus clustered into group A (Fig. 4A, blue ellipse) and WT mice treated with the ZFP-dDBD and zQ175DN mice treated with ZFP-HTT clustered into group B (Fig. 4A, red ellipse). This k-cluster analysis showed that zQ175DN mice that had received the therapeutic were more similar to WT mice than zQ175DN mice that had received the control virus.
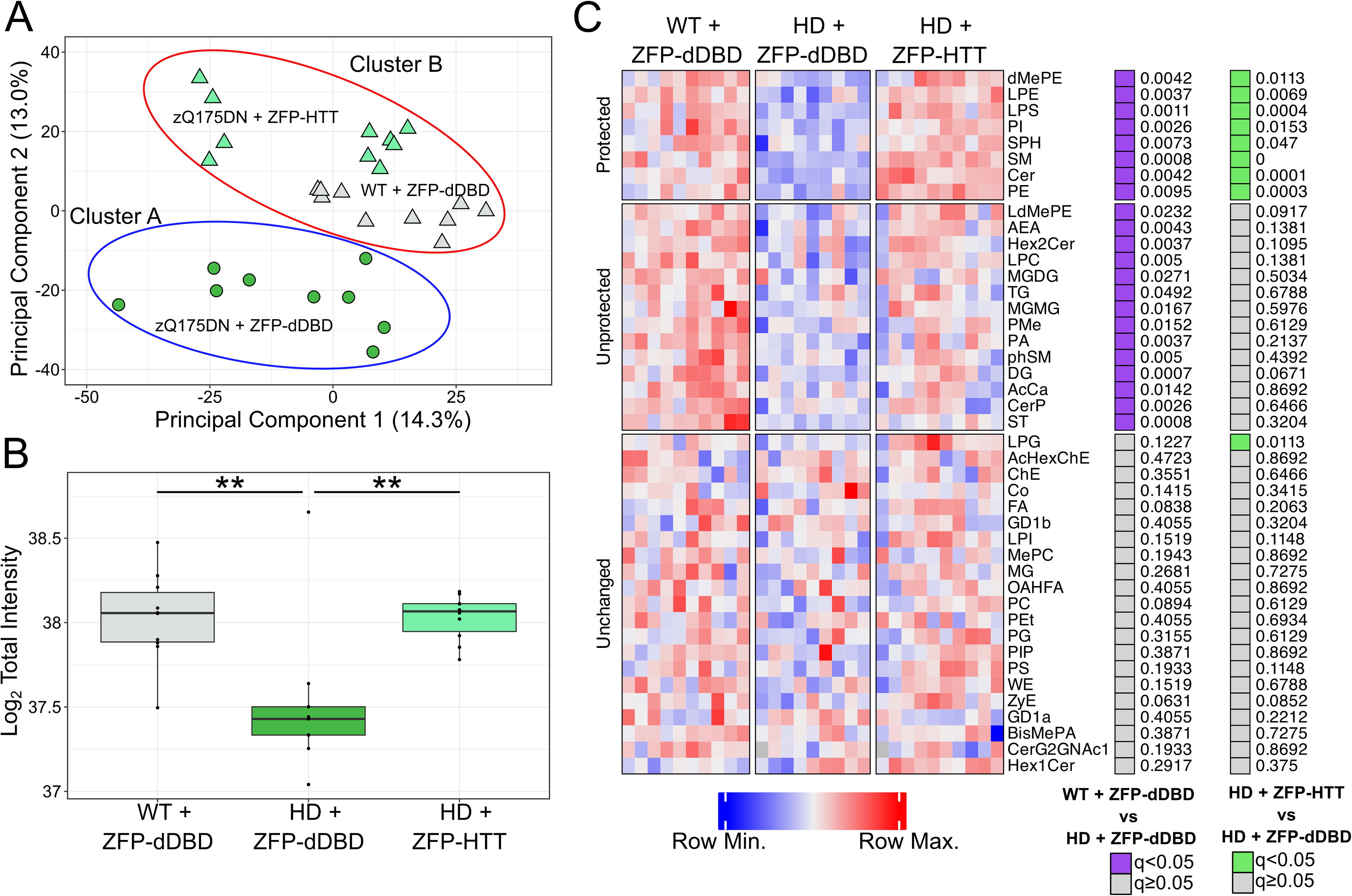
Protection of total lipid levels and subclass levels via lowering of mutant HTT in the caudate-putamen.
An examination of total lipid levels showed a genotype-dependent loss of lipids in HD mouse caudate-putamen (30%) compared with WT mice, each treated with ZFP-dDBD (Fig. 4B), consistent with previous results in LacQ140 mice. Changes in several lipid subclasses were prevented by treatment with AAV9-ZFP-HTT. There were 23 subclasses of lipids that were significantly lower in HD mice compared with WT mice treated with ZFP-dDBD (Fig. 4C and Supplementary Fig. S2). Levels of eight subclasses were corrected with ZFP-HTT treatment (Fig. 4C).
Next we looked at individual lipid species that can have precise functions in signaling such as arachidonic acid signaling 57 and myelin stability. 57,58 ZFP-HTT treatment prevented most genotype-dependent changes in individual lipid species (Supplementary Data S1). When compared with WT mice treated with the control virus, the HD mice treated with the control virus had 270 significantly altered individual lipid ions. Of those, 58% (157/270) were corrected by the ZFP-HTT treatment (Table 1).
Lipid ion changes in Huntington’s disease caudate-putamen and plasma
Protected: Log fold change between HD mice treated with ZFP-HTT and WT mice treated with ZFP-dDBD, compared with zQ175DN mice treated with ZFP-dDBD is the same and the q value of Tukey’s multiple comparison test between the zQ175DN treatment groups is less than 0.05.
HD, Huntington’s disease; WT, wild-type; ZFP-dDBD, zinc finger protein-delta-DNA-binding domain.
AAV9-ZFP-HTT prevents genotype-dependent lipid changes in zQ175DN HD plasma
Lipid alterations have been detected in plasma of HD patients, especially in the phosphatidylcholines and free fatty acids. 39 –41 To determine if lipid levels in plasma could be an effective peripheral readout in a HD preclinical model, plasma was collected from the same mice used for caudate-putamen studies and analyzed by LC-MS. LC-MS analysis offered excellent coverage of plasma lipids with 2,251 lipid ions across 47 subclasses.
There was a genotype-dependent increase in total lipid levels in plasma of zQ175DN mice compared with WT mice, that were treated with ZFP-dDBD (Fig. 5A). Treatment of mice with ZFP-HTT corrected the total lipid levels in zQ175DN mice to WT levels. This is in contrast to the loss in total lipid levels in caudate-putamen in zQ175DN versus WT mice treated with control vector. Of note, inverse changes in total lipids were measured in plasma (increased, Fig. 5A) compared with caudate-putamen (decreased, Fig. 4B) and treatment status, suggesting a relationship between this peripheral biofluid and brain.
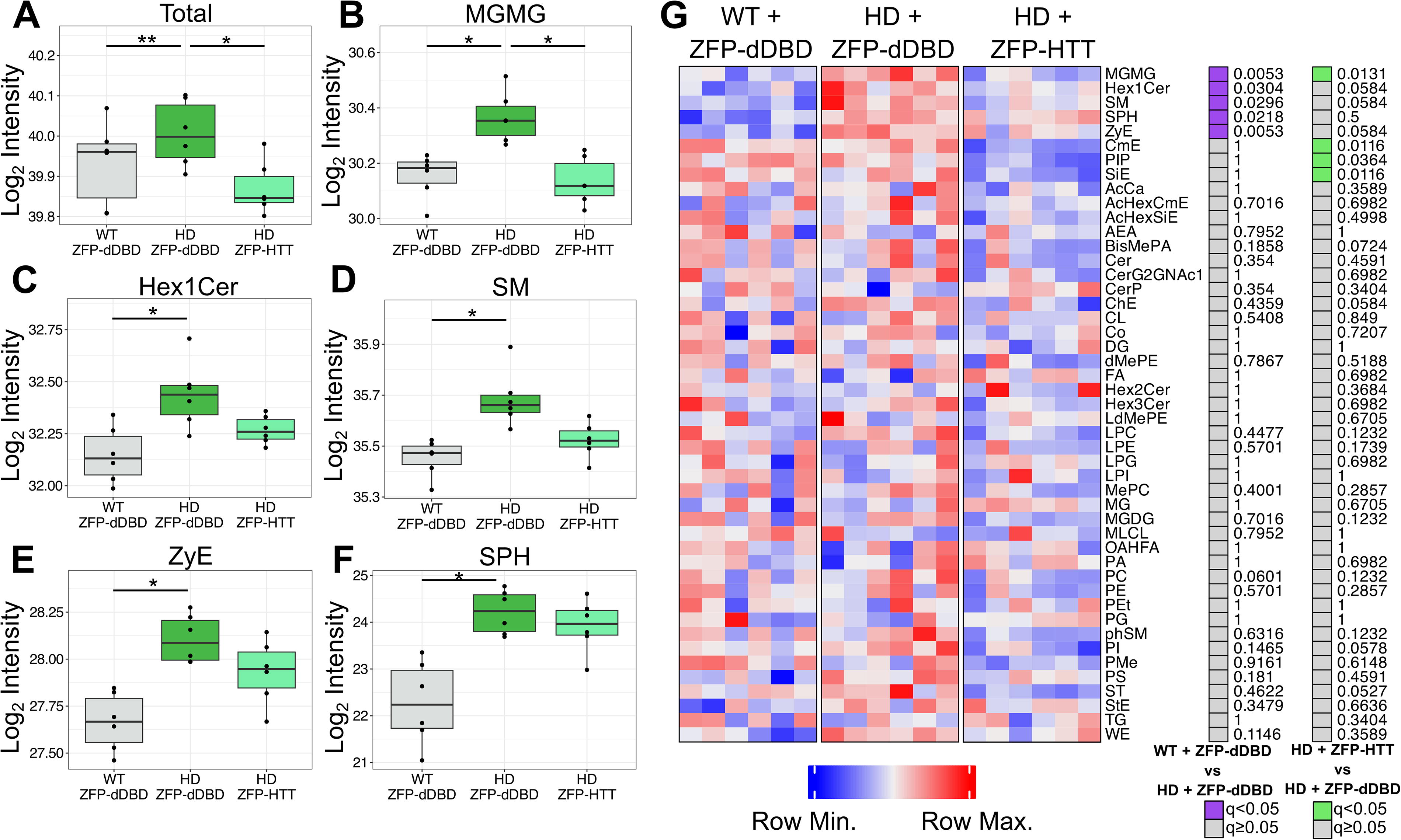
Protection of total lipid levels and subclass levels via intrastriatal lowering of mutant HTT in the plasma.
Treatment with ZFP-HTT prevented changes for lipid subclasses in plasma (Fig. 5G). There were five lipid subclasses significantly higher in zQ175DN mice compared with WT mice each treated with ZFP-dDBD (Fig. 5B–F). Notably, levels of monoacylglyceride (MGMG) and sphingomyelin (SM) were increased in plasma and moved the opposite direction in the caudate-putamen (down). In addition, MGMG was significantly lower in zQ175DN mice treated with ZFP-HTT compared with those treated with ZFP-dDBD, indicating MGMG subclass in plasma is an effective readout for therapy-responsive changes. Three of the other lipid subclasses: Hex1Cer, SM, and ZyE, were trending toward recovery.
Intrastriatal treatment with ZFP-HTT prevented many genotype-dependent changes in individual lipid ions in the plasma (Supplementary Data S2). There were 70 significantly altered lipid ions between WT and zQ175DN mice treated with ZFP-dDBD, including species of PC. Of those, 56% (39/70) were protected by the ZFP-HTT treatment (Table 1).
Overall, these results suggest that monitoring plasma was an effective method to evaluate the protective effects of intrastriatal injection of AAV-ZFP-HTT.
AAV9-ZFP-HTT blocked somatic instability zQ175DN mice
Others have reported that ZFP-HTT can block SI in mice. 6 We found there was a statistically significant decrease in SI index of 64% of the mutant HTT allele in zQ175DN mice treated with ZFP-HTT (2.59 ± 1.42) compared with ZFP-dDBD (7.33 ± 0.877), (unpaired t-test p = 0.014). This demonstrates that the ZFP-HTT can strongly inhibit CAG repeat expansion in the caudate-putamen of zQ175DN mice.
DISCUSSION
In this study, we found numerous changes in lipid species between WT and zQ175DN HD mice at 6 months of age, and preservation of levels for most lipids with intrastriatal injection of a HTT lowering therapy. Lipid changes could be measured in caudate-putamen and complimentary changes could be detected in peripheral biofluid (plasma). We also detected a misfolded species of mHTT resistant to lowering present in the crude nuclear fraction, reproducing earlier results in a different mouse HD model. Our results show that LC-MS offers a quantitative way to track biochemical changes in lipids reflective of neurodegeneration in preclinical models.
Treatment with AAV9-ZFP-HTT resulting in mHTT lowering was highly effective at preventing total lipid changes in caudate-putamen of zQ175DN mice. Thus, while complete reversal of all pathologies, including transcriptional changes, may require either very early mHTT lowering to prevent accrual of mHTT, or actively targeting accumulated mHTT in nuclear compartments, lowering mHTT with AAV9-ZFP-HTT still awarded significant benefits as measured by lipid changes.
Many of the lipid changes found in zQ175DN mouse brain in this study were like those found in synaptic fractions in zQ175/Q7 mice at 6 months of age in our previous study. 45 Lipid changes in the LacQ140 mice, which have a shorter CAG repeat and a more prolonged disease process, were slightly different and changed with age, but also showed a significant loss of total lipids (at 9 months) with heavy losses of lipids important for white matter. Here, we also saw loss of lipids important for white matter health (SM, Hex2Cer, sulfatides), which can be lost with neurodegeneration and aging, 59,60 but lipid profiles were mostly unique to each animal model. The identity of lipids changed in HD mice (measured by us) and measured in HD autopsy tissue 38 are consistent with altered white matter, which reflects processes observed in HD patients and is likely to affect nerve conduction. 61
Genotype- and treatment-dependent changes in lipids were detectable in mouse plasma. These results suggest a relationship between lipid levels in brain and plasma. Lipid alterations have been detected in plasma of HD patients, including in phosphatidylcholine (PC) and free fatty acids. 39 –41 We measured changes in PC in zQ175DN mouse plasma, although the exact species of PC were different than those measured from HD patients. This could be due to species differences (human vs. mouse) or extraction methods. We measured many more changes than prior studies using human plasma, which may be due to improved lipid annotation and increased sensitivity of instrumentation.
Lipid changes caused by mHTT may hinder neuronal function or alter white matter maintenance. mHTT immunoreactivity can be detected with anti-HTT antibodies PHP1 and PHP2 in myelin in zQ175/Q7 mice 62 positioning mHTT to contribute to pathology through post-transcriptional mechanisms such as interfering with membrane-associated protein complexes or changing lipid bilayer organization through direct lipid-mHTT interactions. 63 –65 The impact of mHTT in these compartments may be overcome by mHTT lowering. The misfolded species of mHTT resistant to lowering, which we detected may also impair transcription including lipid metabolizing enzymes.
Total lipid levels, MGMG, SM, and sphingosine (SPH) are all decreased in the caudate-putamen and increased in plasma. We cannot definitively conclude that the lipid changes in the plasma are directly due to the inverse changes in the brain versus arising from peripheral tissues. However, our HTT lowering treatment was introduced by direct intrastriatal injection which should minimize peripheral effects. Thus, the recovery of total lipids, MGMG, and trend of SM toward recovery in plasma support the notion that the lipid changes in plasma are related to the health of the caudate-putamen. Sphingolipids, which have an SPH backbone such as SM are important for white matter integrity. Release of SM and SPH out of the brain into the periphery as white matter integrity is disrupted could explain the inverse changes seen in the caudate-putamen and plasma. While the exact mechanism is unknown, MGMG has been shown to decrease in mouse brains with age. 66 HD has been described as an advanced aging phenotype. 67 One possibility is that MGMG is transported out of the brain across the blood brain barrier into the plasma, 68 possibly by passive diffusion or an active transport mechanism as the advanced aging phenotype manifests.
We found species of mutant HTT resolving as a full-length HTT band and as a high molecular weight smear, could not be lowered with transcriptional repression both in this study and in Shing et al., (2023). 47 We do not know whether the S830 smear is already present at 2 months (time of injection) in zQ175DN mice or whether it is composed of full-length HTT, an N-terminal fragment or both. However, work by Gillian Bates lab showed S830-positive nuclear staining using immunohistochemistry at 2 months in the regular Q175 mice. 69 Since a species of full-length mHTT was detected with S830 and failed to lower, we speculate that this is one of numerous conformations that mHTT adopts but which becomes stabilized perhaps due to an interaction with another protein hindering its turnover. Our system using AAV9 which does not result in mHTT lowering in every cell is inadequate to determining whether this mHTT product cannot be cleared versus cannot be prevented. However, in the LacQ140 mice, for which genetic lowering of mutant HTT was achieved using isopropyl β-D-1-thiogalactopyranoside, even lowering of mHTT at embryonic day 5 was unable to reduce the smear detected with S830 when measured at 12 months suggesting one cannot prevent this species from forming with partial lowering. Since the level of S830-positive protein levels seemed to plateau with age, either a steady state turnover is achieved, or the hypothetical stabilizing interactor protein is rate limiting.
Carroll and colleagues 6 reported that binding of ZFP-HTT to the CAG on the HTT allele may block somatic instability providing additional protection from disease in addition to that afforded by mHTT protein lowering. A CAG repeat of 175 is thought to already exceed the length needed to induce most phenotypes including transcriptional phenotypes without further expansion. 70 Even so, in addition to lowering mHTT levels, impeding somatic instability with ZFP-HTT may be one mechanism that contributes to the protection of lipid levels in our study.
In conclusion, assessment of lipid levels using a quantitative method revealed sweeping positive effects of a lowering therapeutic targeting mutant HTT transcription in a preclinical mouse model of HD.
Lipid abbreviations
Glycerophospholipids
Bis-methyl phosphatidic acid (BisMePA), Cardiolipin (CL), Dimethyl phosphatidylethanolamine (dMePE), Lysophosphatidylcholine (LPC), Lysophosphatidylethanolamine (LPE), Lysophosphatidylglycerol (LPG), Lysophosphatidylinositol (LPI), Lysophosphatidylserine (LPS), Lysodimethyl phosphatidylethanolamine (LdMePE), Methyl-Phosphatidyl Choline (MePC), Mono-lysocardiolipin (MLCL), Phosphatidic acid (PA), Phosphatidylcholine (PC), Phosphatidylethanolamine (PE), Phosphatidylethanol (PEt), Phosphatidylglycerol (PG), Phosphatidylinositol (PI), Phosphatidylinositol-monophosphate (PIP), Phosphatidylmethanol (PMe), Phosphatidylserine (PS).
Glycerolipids
Diacylglyceride (DG), Monoacylglyceride (MG), Monogalactosyldiacylglycerol (MGDG), Monogalactosylmonoacylglycerol (MGMG), Triacylglyceride (TG).
Sphingolipids
Dihexosylceramide (Hex2Cer), Ceramides (Cer), Ceramide phosphate (CerP), Ganglioside (GD1a, GD1b), Hexosylceramide (Hex1Cer), Simple Glc series (CerG2GNAc1), Sphingomyelin (SM), Sphingomyelin phytosphingosine (phSM), Sphingosine (SPH), Sulfatide (ST) Trihexosylceramide (Hex3Cer).
Sterols
AcylGlcCholesterol ester (AcHexChE), AcylGlcCamposterol (AcHexCmE), AcylGlcSitosterol (AcHexSiE), Cholesterol Ester (ChE), Camposterol Ester (CmE), Sitosterol Ester (SiE), Stigmasterol Ester (StE), Zymosterol Ester (ZyE).
Fatty acyls
Acyl Carnitine (AcCa), Fatty Acid (FA), N-acylethanolamine (AEA), (O-acyl) −1- hydroxy fatty acid (OAHFA), Wax ester (WE).
Prenol lipids
Coenzyme (Co).
Footnotes
ACKNOWLEDGMENTS
The authors would like to thank David Howland and Deanna Marchionini at CHDI Foundation for helpful advice and Jonathon Asara at the Beth Israel Deaconess Medical Center Mass Spectrometry Core for help with LC-MS and lipid annotation.
AUTHORS’ CONTRIBUTIONS
A.I. and K.S. extracted lipids and performed computational and statistical analysis. A.B. and C.S. collected plasma and brain tissues. S.L. and E.S. performed protein chemistry. K.C. maintained mouse colonies. R.M. performed stereotaxic injections. A.R.B. subcloned ZFP cDNAs and prepared virus. M.S-E., N.A., M.D., and K.B.K.-G. planned experiments and wrote article.
AUTHOR DISCLOSURE STATEMENT
The authors have nothing to disclose.
FUNDING INFORMATION
This study was supported by the CHDI Foundation (A-6367), Dake Family Fund.
SUPPLEMENTARY MATERIAL
Supplementary Data S1
Supplementary Data S2
Supplementary Figures
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
