Abstract
Spinal deformities in finfish have the potential to impact aquaculture industries and wild populations by increasing morbidity, mortality, and reducing growth rates. Myxobolus acanthogobii has been implicated in causing scoliosis and lordosis in various aquatic species in Japan. We investigated 4 cases of spinal deformity in 2 flathead (Platycephalus) species that were submitted to the Elizabeth Macarthur Agricultural Institute (EMAI) in New South Wales (NSW), Australia, between 2015 and 2021. Flathead are commercially significant species that are popular among Australian consumers, and are also sought-after species targeted by recreational fishers. Gross deformities are concerning to the community and may impact the quality and quantity of specimens available for consumption. Three blue-spotted flathead (P. caeruleopunctatus) and one marbled flathead (P. marmoratus) were submitted, all with marked scoliosis and kyphosis; 1–2-mm cysts were present on the dorsum of the brain, most often over the optic lobe or cerebellum. Cytology and differential interference microscopy of cyst material revealed numerous oval spores, x̄ 14 ± SD 0.75 µm × x̄ 11.5 ± SD 0.70 µm, with 2 pyriform polar capsules, the morphology of which is consistent with a Myxobolus sp. PCR assay and 18S rDNA sequencing of the cyst material identified a Myxobolus sp. with 96% identity to M. acanthogobii. The identification of this Myxobolus sp. confirms the presence of parasites with the potential to cause spinal deformity in significant aquatic species in NSW waterways.
Spinal deformities in both wild and farmed fish have a range of etiologies, including bacterial 16 and parasitic 26 agents, physical trauma, 18 developmental issues, such as the absence of a swim bladder, 8 genetic conditions, 3 or nutritional factors such as deficiencies of vitamin C, vitamin D, 10 or phosphorus, 14 or excess of vitamin A. 20 Water quality parameters can also influence the development of spinal deformities and include factors such as high temperatures, 39 low dissolved oxygen content, 35 and the presence of toxins, such as organophosphates 7 and/or heavy metals. 29 Spinal deformities can become a significant production issue in aquaculture, often resulting in increased morbidity and reductions in growth rate with the potential for severe economic losses. 17
Infections with Myxobolus spp. have been reported in association with spinal deformities of fish.12,24,26–28,30,38 Myxobolus spp. belong to the phylum Cnidaria (Myxosporea, Myxobolidae), which includes numerous pathogenic and nonpathogenic species infecting both freshwater and marine finfish. Fish are the intermediate hosts in the lifecycle of myxozoan parasites; annelids (oligochaetes in freshwater environments, and polychaetes in marine environments) are the definitive hosts. 25 M. cerebralis, the causative agent of salmonid whirling disease (or black tail), is perhaps one of the most well-known causes of spinal deformity, which has had significant impact on the salmonid aquaculture industry as well as wild populations in both Europe and North America.17,24 Clinically affected animals have reduced growth rate, and the spinal deformity can alter swimming performance. 13
M. acanthogobii is another myxozoan parasite associated with spinal deformities in fish. M. acanthogobii was first reported in yellowfin goby (Acanthogobius flavimanus) 31 with parasitic cysts present in the orbit of the eye and in the CNS; however, no spinal deformities were noted. Similarly, M. acanthogobii spores were found within the CNS of a forksnout searobin (Lepidotrigla alata) and a scribbled toby (syn. brown-lined puffer; Canthigaster rivulata), although again, no spinal deformity was observed. 30 In contrast, several studies have identified M. acanthogobii cysts within the CNS with evidence of scoliosis, kyphosis, or lordosis in Japanese amberjack, 12 Japanese bluefish, 28 and Japanese mackerel. 38
Flatheads of genus Platycephalus are finfish endemic to Australia and the Indo-West Pacific region. 2 They are of importance for both recreational fishing and commercial fisheries and are a popular table fish among consumers in New South Wales (NSW). 19 An estimated 100–200 tonnes of blue-spotted flathead (P. caeruleopunctatus) were caught in NSW estuaries and ocean waters across both recreational and commercial sectors between 2019 and 2020. 21 Dusky flathead (P. fuscus) has also been identified as a recreationally significant species for stock enhancement and has been the subject of hatchery production for release of juveniles.5,33 Gross deformity in members of this genus has potential implications for NSW wild-capture fisheries and hatchery production of these species.
Between 2015 and 2022, several reports from members of the public were made to the NSW Department of Primary Industries concerning wild-caught flathead with spinal deformities observed at the time of capture. Suitable samples of whole fish from 4 of these reports were submitted to the Elizabeth Macarthur Agricultural Institute (EMAI; Menangle, NSW, Australia) laboratory for diagnostic investigation, the results of which we report here.
Materials and methods
All gross postmortem examinations were performed at the EMAI. Brain and cyst material were retained for cytologic and histologic examination, differential interference microscopy, and molecular testing. Other tissues including spinal cord, gill, liver, heart, spleen, and gastrointestinal tract were retained in case 1. All retained fresh tissues were stored at −20°C, or at 4°C if collected into PBS.
Cytologic smears prepared from the cyst material from cases 1 and 4 were stained with Giemsa or Diff-Quik (Australian Biostain). For histology, tissues were fixed in 10% neutral-buffered formalin, paraffin-embedded, sectioned at 4–6-µm thickness, and stained with H&E (Australian Biostain). Select histologic sections were stained with Gram, Giemsa, or Ziehl–Neelsen (ZN; Australian Biostain).
Wet preparations of cyst material were prepared from cases 2 and 3 by rupturing the cyst wall, resuspending the contents, and pipetting onto glass slides for differential interference microscopy. Cysts from case 3 had been stored for 3 mo at 4°C in PBS before making the wet preparations. To induce polar filament extrusion, 2% potassium hydroxide was added to the wet preparation. Digital images were taken using a 100× objective and spore dimensions recorded.
For identification of parasites, DNA was extracted (DNeasy blood and tissue kit; Qiagen) from brain tissue and cystic material collected at postmortem examination. Extraction was according to the manufacturer’s instructions for the processing of tissue samples, with the exception that a micropestle was used to grind the tissue in the ATL buffer and proteinase K mixture before incubation. Amplification of the 18S rRNA gene was attempted using 3 published primer sets 6 : MX5-MX3, 1 ERIB1-ACT1R,4,22 and MYXGEN4f-ERIB10.4,11 PCR reactions contained 5 pmol of each primer, 1× BioTaq buffer, 37.5 nmol MgCl2, 5 nmol dNTPs, 2.5 U of BioTaq (Bioline), and 2 µL of DNA template in a total volume of 25 µL. Thermal cycling was carried out on a Mastercycler (Eppendorf). For the MX5-MX3 primer set, the cycling protocol involved an initial denaturation step at 95°C for 5 min, followed by 40 cycles of denaturation at 95°C for 30 s, annealing at 60°C for 30 s, extension at 72°C for 90 s, and 1 cycle at 72°C for 10 min as a final extension step. For the ERIB1-ACT1R and MYXGEN4f-ERIB10 primer sets, thermal cycling involved 1 cycle of 5 min at 95°C, followed by 35 cycles of denaturation at 95°C for 60 s, annealing at 56°C for 60 s, extension at 72°C for 90 s, followed by 1 cycle at 72°C for 5 min as a final extension step.
The same extraction method used for parasites was also used to obtain DNA for the barcoding of fish (case 4); however, in this case, a fin clip was used as the target tissue. Barcoding of flathead (from case 4) was achieved by amplifying a portion of the mitochondrial cytochrome oxidase I (COI) gene with published primers. 15 The PCR reaction mix was as described above but with 10 pmol LCO1490 and HCO2189 primers. Cycling conditions were 95°C for 10 min; followed by 5 cycles at 95°C for 30 s, 45°C for 90 s, 72°C for 60 s; 35 cycles at 95°C for 30 s, 51°C for 90 s, 72°C for 60 s; and 1 cycle at 72°C for 5 min.
All PCR products were analyzed on 1.5× Tris-borate EDTA (TBE) gels and electrophoresed at 100 V for 60 min. Amplicons were purified (QIAquick spin columns; Qiagen) and sequenced using Sanger dideoxy sequencing at the Australian Genomic Research Facility (AGRF; Westmead, NSW, Australia). Parasite 18S sequencing primers were MYXGEN4f-ERIB10, and fish barcoding primers were LCO1490 and HCO2189, as described above.
Consensus 18S sequences from cyst material were generated, aligned with reference myxozoan sequences from GenBank, and trimmed using Geneious v.R7.1 (Dotmatics). Phylogeny was inferred using the maximum-likelihood method with the Kimura 2-parameter model within MEGA X (https://www.megasoftware.net/), and phylogenetic trees were generated using the Neighbour-Join and BioNJ algorithms. Barcoding sequences derived from the flathead from case 4 were compared to the nucleic acid database within GenBank using BLASTN (https://blast.ncbi.nlm.nih.gov/Blast.cgi).
Results
Members of the public reported 11 cases of spinal deformity to the NSW Department of Primary Industries between 2015 and 2022 from various locations around NSW (Fig. 1). Four cases were submitted to the laboratory between August 2015 and April 2021, including 1 marbled flathead (P. marmoratus, case 1), 2 blue-spotted flathead (P. caeruleopunctatus, cases 2, 3), and 1 flathead that could not be identified based on morphology (case 4). DNA barcoding performed indicated that the flathead from case 4 was also P. caeruleopunctatus (Table 1). No reports of spinal deformity were made in other aquatic species during this time period. Several other reports of flathead with grossly similar skeletal abnormalities were made by members of the public during this time; however, suitable samples were not submitted or available for further investigation (Table 2).
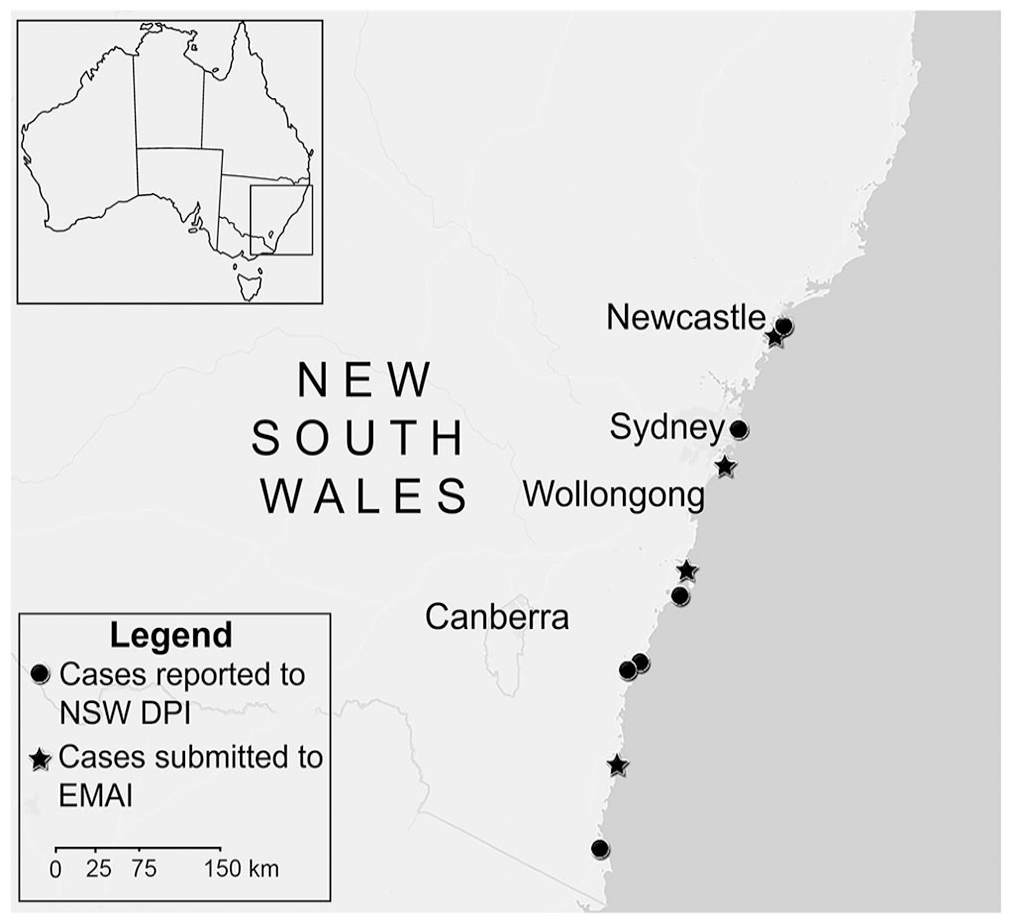
Distribution of cases of spinal deformity submitted to the Elizabeth Macarthur Agricultural Institute (EMAI) or reported to the New South Wales Department of Primary Industries (NSW DPI) between 2015 and 2022 in New South Wales, Australia.
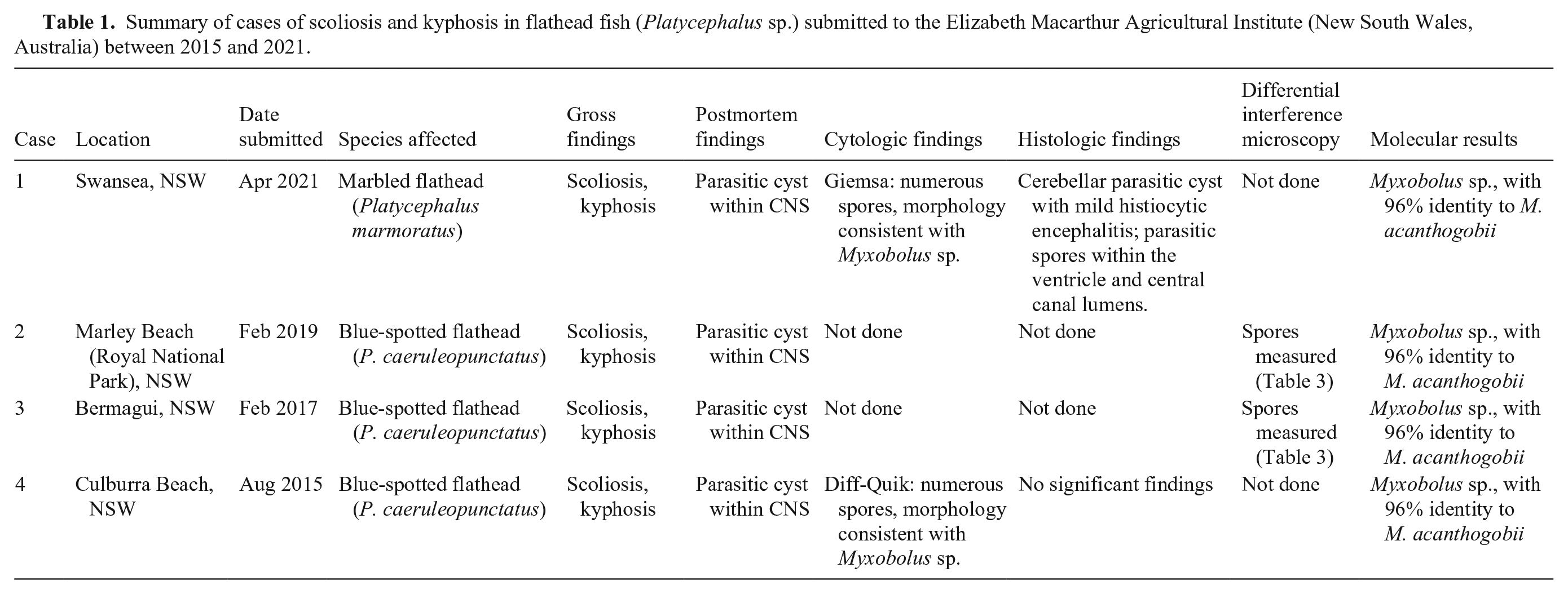
Summary of cases of scoliosis and kyphosis in flathead fish (Platycephalus sp.) submitted to the Elizabeth Macarthur Agricultural Institute (New South Wales, Australia) between 2015 and 2021.
Summary of reports made to New South Wales Department of Primary Industries by members of the public between 2015 and 2022 of flathead fish (Platycephalus sp.) with spinal deformities in which no suitable samples were submitted to the laboratory.
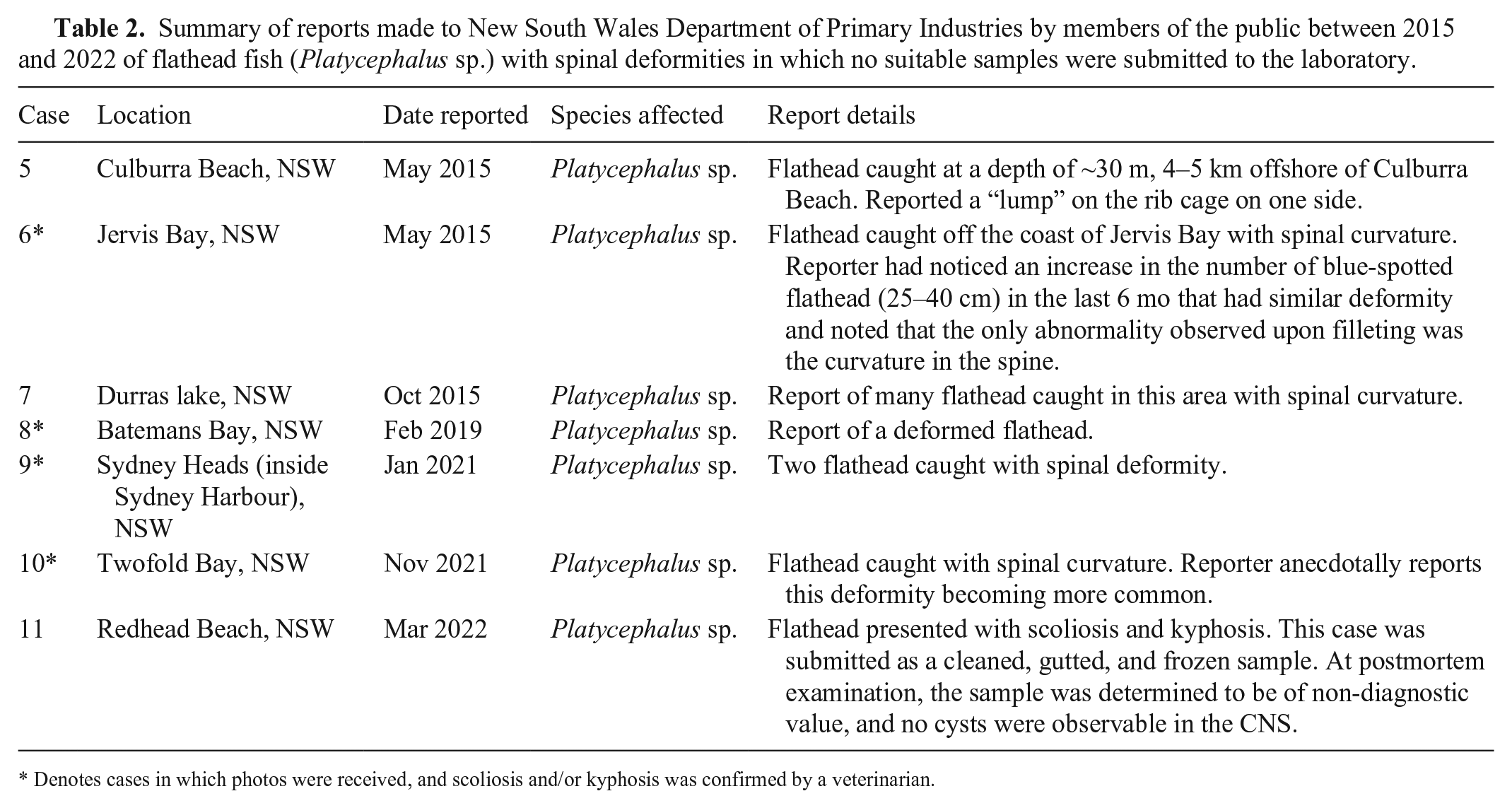
Denotes cases in which photos were received, and scoliosis and/or kyphosis was confirmed by a veterinarian.
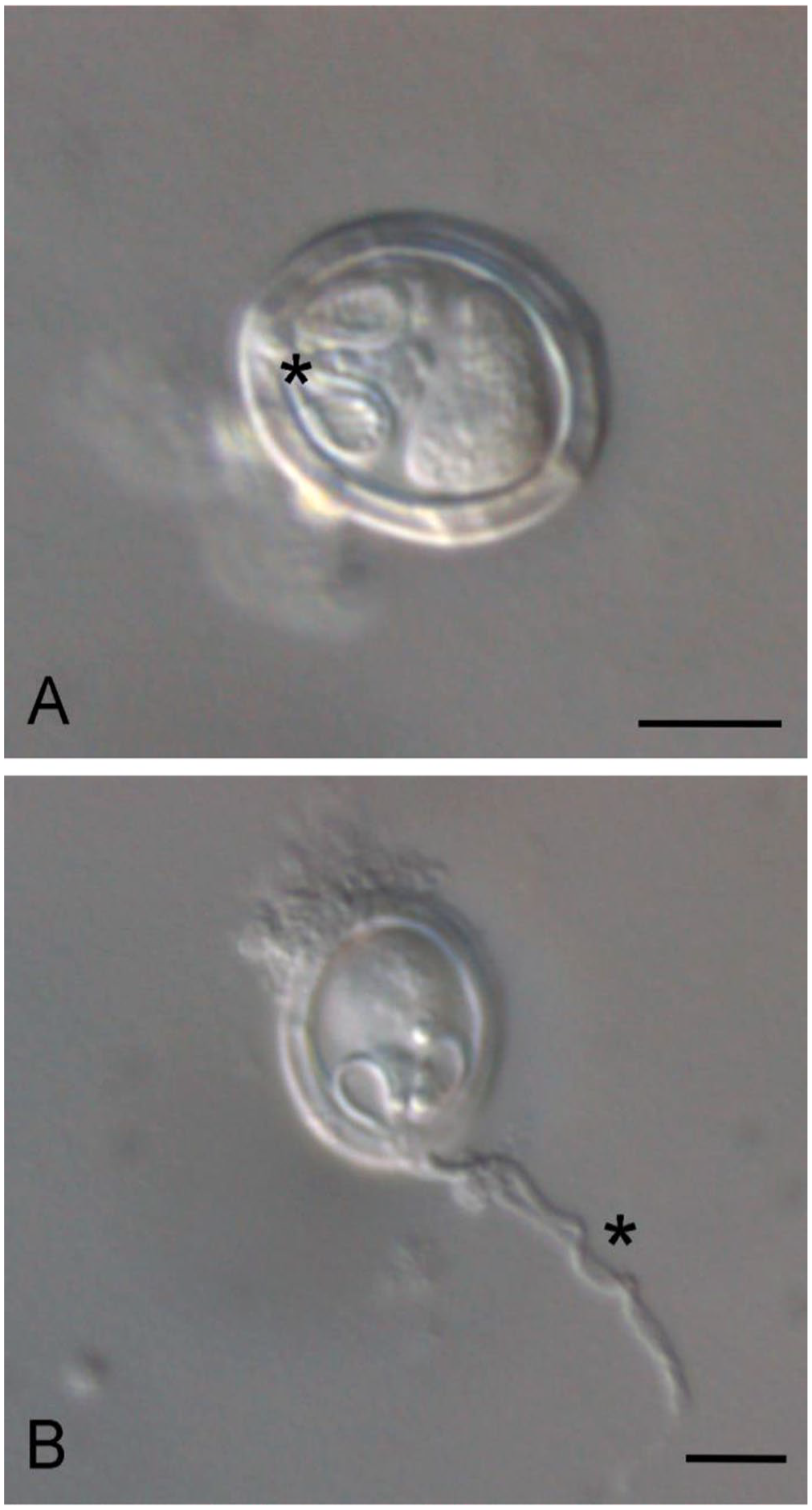
Grossly, all cases had scoliosis and kyphosis (Fig. 2A, 2B) with 1–2-mm, white to pale-yellow spherical parasitic cysts on the dorsal surface of the optic lobe or cerebellum. No other significant gross findings were identified.

In cases 1 and 4, a squash smear was prepared from cyst material. Numerous 10–12-µm, round-to-oval spores were present. Two pyriform polar capsules were occasionally observed, although they were difficult to visualize with both Diff-Quik and Giemsa stains. The polar capsules and sporoplasm were better visualized in the wet preparation for differential interference microscopy from cases 2 and 3. Cytologic smears were not prepared from cases 2 and 3.
Histologic examination was performed in cases 1 and 4. In case 1, a round, 2.1-mm cyst disrupted and expanded the center of the cerebellum and was surrounded by mild histiocytic encephalitis (Fig. 3A). The cyst was covered by a single layer of squamous epithelium overlying 100–200 µm of eosinophilic acellular material. The center of the cyst contained abundant oval spores. Small numbers of spores were also present within the lumen of the fourth ventricle and the central canal of the spinal cord (Fig. 3B). Spore dimensions (range [x̄ ± SD]) based on 20 spores were: 10.1–11.9 µm (11.2 ± 0.6 µm) long, 6.6–9.2 µm (8.0 ± 0.8 µm) wide. Some spores had visible symmetric pyriform polar capsules, although most were poorly stained by H&E. Gram and ZN stains of histologic sections stained spores adequately; however, spores and internal polar capsules were best visualized histologically by Giemsa stain (Fig. 3C). In case 4, no significant histologic findings were observed. No fixed samples from cases 2 and 3 were retained for histologic examination.
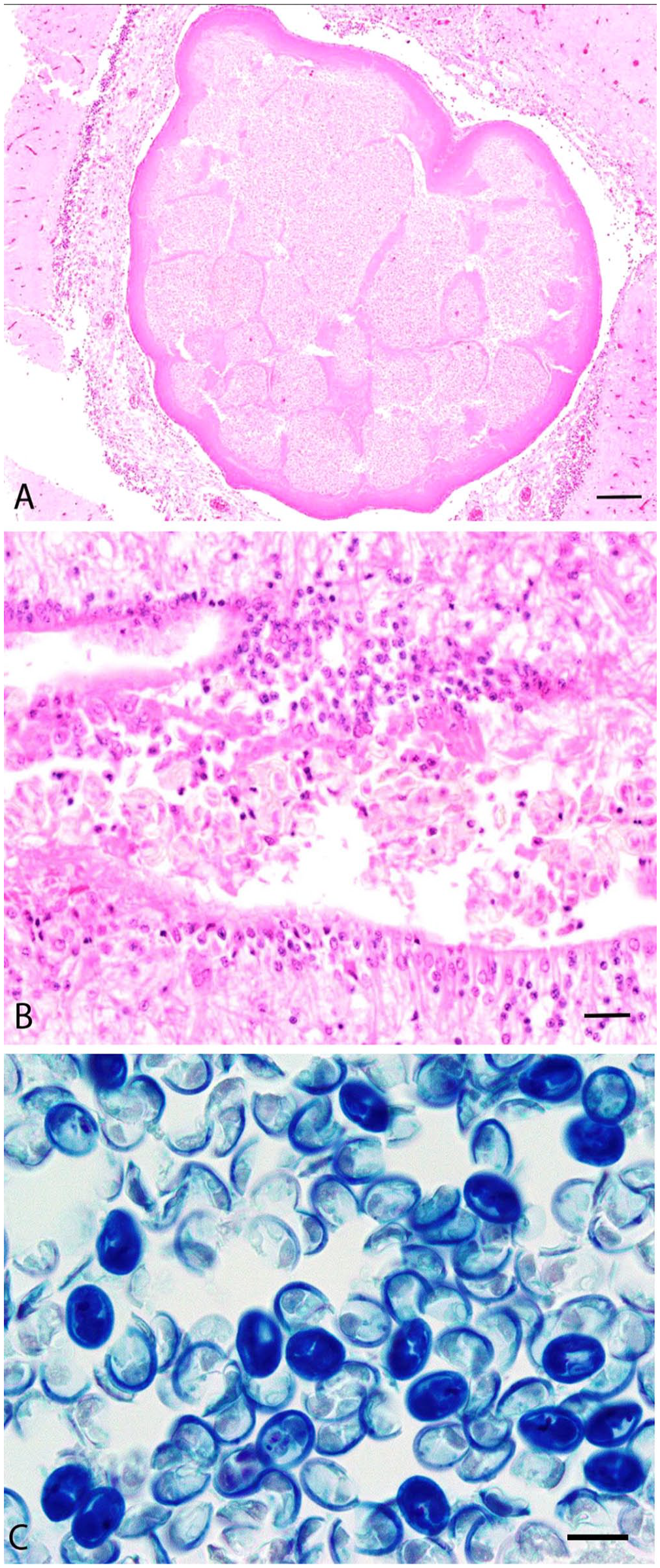
Wet preparations for differential interference microscopy were prepared from cyst material from cases 2 and 3. In frontal view, spores were oval with 2 equally sized pyriform polar capsules and homogeneous sporoplasm (Fig. 4A). Within the polar capsules, the coiled polar filament had 3 turns. An extruded polar filament was occasionally observed, measuring 19.5 µm (Table 3; Fig. 4B). Spore dimensions (x̄ ± SD) were: 14 ± 0.8 µm long, 11.5 ± 0.7 µm wide (Table 3).
Average spore dimensions of a Myxobolus acanthogobii–like organism and comparison to several other Myxobolus sp. that are known to cause skeletal deformity, including M. acanthogobii.
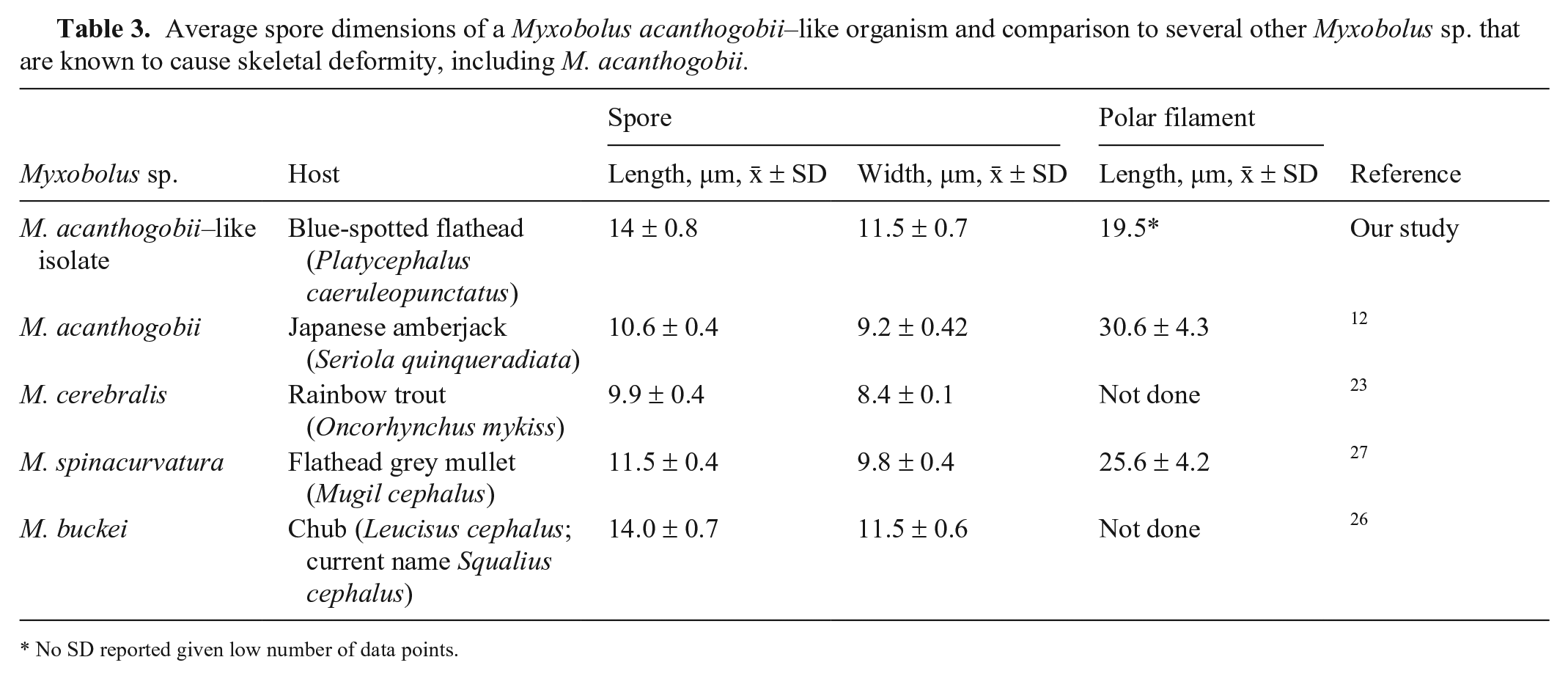
No SD reported given low number of data points.
Molecular testing and sequencing of retained cyst material collected at postmortem generated ~1,000 bp of sequence from the 18S rRNA gene using the MYXGEN4f-ERIB10 primer set (GenBank OR364499) and revealed that all cysts tested from cases 1–4 contained Myxobolus spp. with 100% identity to each other. Phylogenetic analysis performed on an alignment of ~1,000 bp of the 18S rRNA sequence from reference myxosporean species demonstrated that the flathead isolate clustered with M. acanthogobii (Fig. 5A) and that these strains shared 96.5% sequence identity. A shorter alignment of ~650 bp was also subjected to phylogenetic analysis given the limited sequence information in the database for some Myxobolus spp. This tree also indicated that M. acanthogobii is the closest named relative of the flathead myxosporean; however, 2 Myxobolus isolates from Malaysia infecting the fork-tailed threadfin bream (Nemipterus furcosus) and the bigeye scad (Selar crumenophthalmus)—GenBank MH425557 and MH425556, respectively—also clustered with M. acanthogobii and the flathead Myxobolus sp. (Fig. 5B). Based on the 650-bp alignment, the flathead Myxobolus sp. displayed 96.8% sequence identity with M. acanthogobii, but was also closely related (96.4% sequence identity) to Myxobolus sp. MH425557 isolated from N. furcosus. Consistent with the phylogenetic tree, the flathead Myxobolus sp. was more genetically distant from MH425556 from S. crumenophthalmus, with only 92.7% sequence identity between these 2 strains. Of the 4 strains from the M. acanthogobii clade, M. acanthogobii and MH425557 were each other’s closest relatives, sharing 98.7% identity. Interestingly, pairwise identity between the Myxobolus spp. in the M. acanthogobii cluster was similar to that seen between M. arcticus and M. neurobius (94.5%) and M. axelrodi and M. stellatus (92.6%) over the same region of 18S rRNA sequence, and these pairs of strains are deemed taxonomically to be separate species.
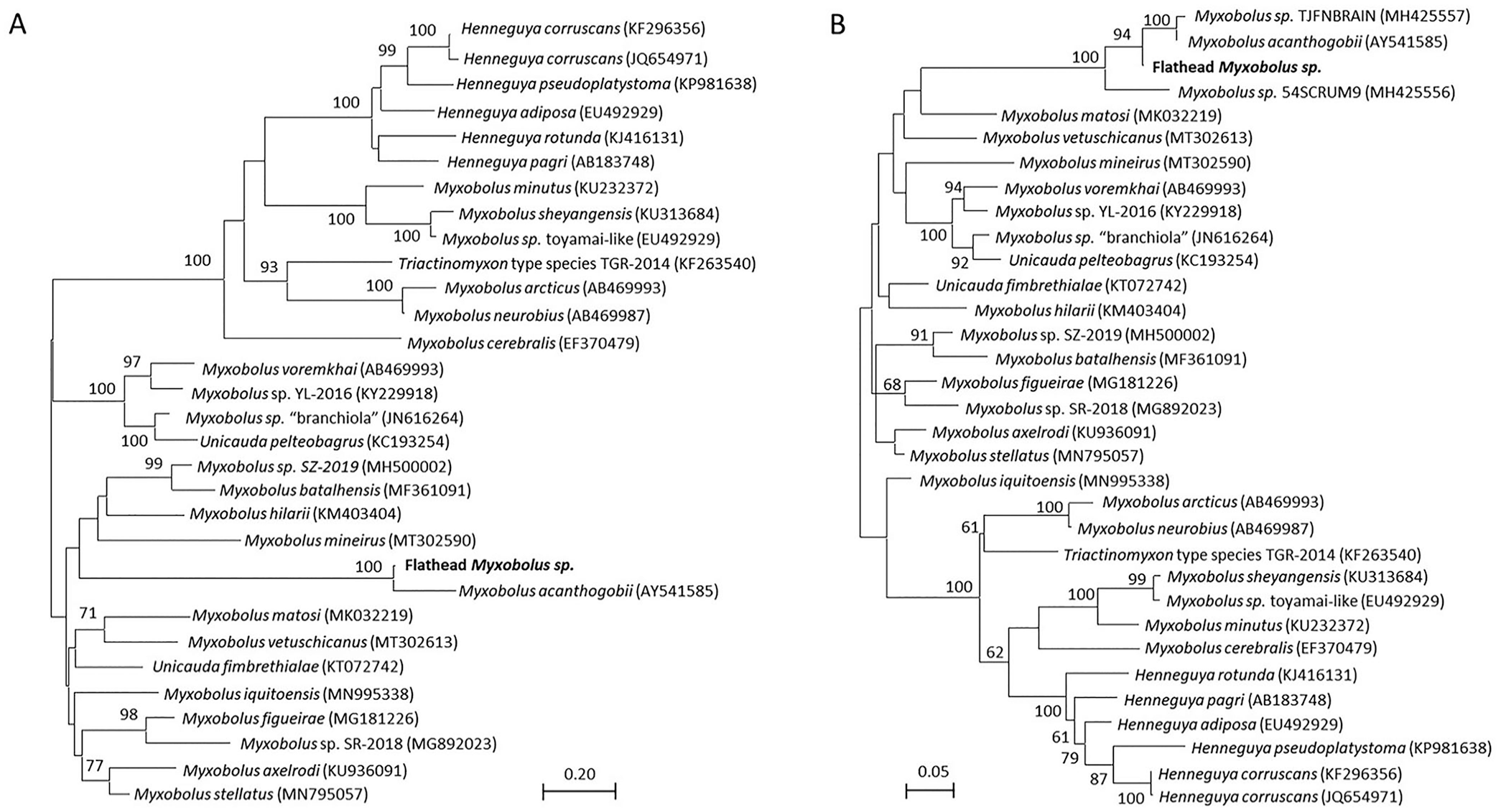
Maximum-likelihood trees generated based on (
Discussion
Our cases share remarkable similarity to gross pathology and spore morphology of M. acanthogobii reported from Japan.12,38 Scoliosis, lordosis, and kyphosis associated with CNS cysts were identified in Japanese amberjack (Seriola quinqueradiata) and Japanese mackerel (syn. chub mackerel; Scomber japonicus), with molecular testing confirming M. acanthogobii as the etiologic agent.12,38 However, molecular phylogenetic analysis demonstrated that, although the Myxobolus sp. in our cases involving flathead had a high degree of 18S rRNA sequence identity with M. acanthogobii, there were some differences, suggesting that our isolates could form a distinct species of Myxobolus. Indeed, in studies of microscopic eukaryotes, taxonomic classifications at the species level based on the use of the 18S rRNA gene have underestimated true species diversity. 36 Furthermore, 100% sequence similarity in the 18S rRNA gene has been identified as the lowest threshold for reliable speciation, using marine copepods as a case study. 37 Moreover, in lower eukaryotes, morphologic homogeneity can often disguise true genetic diversity. 36 Therefore, even though the flathead Myxobolus sp. is morphologically similar to M. acanthogobii, the fact that there was <97% sequence identity between these strains suggests that they are different species. Nonetheless, full sequencing of the 18S rRNA gene as well as other phylogenetic markers may be warranted given that we analyzed only ~1,000 bp of the 18S rRNA gene.
Studies of 3 Myxobolus sp. of cyprinids (M. cyprini, M. musculi, M. pseudodispar) similarly identified slight differences in spore dimensions, as well as morphology between the 3 species, in conjunction with sufficient differences in the DNA sequences of spores from different fish species to identify M. cyprinid, M. musculi, and M. pseudodispar as separate species, despite their similar host range and tissue predilection. 32 Similarly, the slight differences in spore dimensions between the flathead Myxobolus sp. and previously reported M. acanthogobii could suggest that they are different species. The small sample size limits our study, and further investigation of spore morphology and dimensions is warranted.
Similar gross and histologic lesions were identified in a previous case 34 in a sand flathead (P. bassensis), although no molecular testing was performed at the time and fresh material was not available to enable further identification of the Myxobolus sp. involved. Given the similarities to our findings, it is possible that the M. acanthogobii–like species that we report here was involved in the previous case. 34 The location of the cysts within the brain is consistent across M. acanthogobii cases with spinal deformity identified in Japan,12,38 and in the above-mentioned case. 34 The pathogenesis of the spinal deformity in these cases is unknown. It appears that M. acanthogobii has tropism for the CNS, with cysts present in similar anatomic locations across many cases. The location of the cyst could contribute to pathogenesis of the spinal deformity. Cyst location was suggested to be associated with axial muscle lesions or changes in muscle tone that could result in spinal deformity,34,38 especially if the animal was infected as a juvenile. It must be noted however that spinal deformity was not observed in all cases in which cysts or spores were observed within the CNS, such as in the yellowfin goby, 31 forksnout searobin, and scribbled toby. 30 Further histologic investigation of skeletal muscles, vertebrae, and nerves, particularly those that surround and support the spinal column, is warranted, to further understand the relationship between cyst location and spinal deformity.
Although other fish species have been reported to be affected by M. acanthogobii, only Platycephalus spp. were affected in our cases. The bottom dwelling nature of flatheads and presence of annelids (the definitive host for Myxobolus spp.) in the waterbed may be a key host–pathogen–environment interaction. The robustness of the spores in the environment may also be a key factor to infection. In case 3, cyst material retained for 3 mo had viable spores with active extrusion of the polar filaments when examined under differential interference microscopy. The polar filament attaches the spore to the host and is essential for host entry. 25 Although we are reporting only flathead (Platycephalus spp.) with skeletal abnormalities here, further investigation into whether other common bottom-dwelling fish can be affected, with or without skeletal abnormalities, would further clarify the host range. It is important to note that no freshwater species or cultured species were reported during this period, hence the risk to other species and the aquacultural industry in NSW is unknown. Investigation of all future reported cases of spinal deformity in any aquatic species is warranted to investigate this host range and potential risk, while concurrently giving proof of freedom from M. cerebralis, which is exotic to Australia.
The origin of the Myxobolus sp. identified here is uncertain. Yellowfin goby were introduced into NSW waterbodies in the early 1970s. 9 M. acanthogobii was first reported in 1940 31 in this species, although infection in the yellowfin goby is not associated with skeletal deformity. It is unlikely that the M. acanthogobii–like organism reported here diverged from the introduction of M. acanthogobii–infected yellowfin goby given the genetic distance between strains; however, limited numbers of reported M. acanthogobii cases performed gene sequencing, hence genetic diversity between those cases is also unknown. It is possible that the flathead Myxobolus sp. reported here is genetically identical or similar to a previous case of M. acanthogobii from Japan in which gene sequencing was not performed. Gross and histologic examinations in these cases were often limited by less-than-optimal sample preservation, and case presentation was opportunistic given the reliance on reports from members of the public. If further optimal samples become available, gross and histologic examination of other tissues, including thorough examination of the spinal column and supporting musculature, could help elucidate and expand knowledge on the pathogenesis of spinal deformity resulting from parasitic infection.
Footnotes
Acknowledgements
We thank the technical staff in the veterinary pathology, histology, necropsy, and molecular laboratories at the Elizabeth Macarthur Agricultural Institute.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
