Abstract
A 27-mo-old, spayed female mixed-breed dog was presented with left forelimb pain, which progressed to full thickness necrosis of the soft tissues of multiple limbs. Clinical imaging and postmortem examination suggested multiple large arterial thromboemboli. Histologic examination of vascular lesions revealed markedly thickened tunica intima with polypoid intraluminal projections, which partially to entirely occluded the arterial lumen. The expanded tunica intima was comprised of intimal accumulation of Alcian blue–positive matrix with scattered spindle-to-satellite cells. These cells were positive for von Willebrand factor and vimentin but negative for α–smooth muscle actin, suggesting endothelial origin. Deposition of the intimal mucoid matrix was observed in the elastic and muscular arteries associated with regional ischemic changes. Mucoid emboli, likely from fragmentation of proliferative intimal tissue, were identified in smaller vessels supplied by affected arteries. Based on these findings, we diagnosed systemic mucoid degeneration of the arterial tunica intima. Such systemic arterial degeneration characterized by deposition of mucoid matrix in the tunica intima has not been reported previously in dogs, to our knowledge, and should be distinguished from thromboembolism and other degenerative vascular diseases.
Degenerative arterial diseases, such as arteriosclerosis, atherosclerosis, and arteriolosclerosis, are observed occasionally in older dogs. These diseases cause thickening, loss of elasticity, and luminal narrowing in arteries or arterioles as a result of proliferative and degenerative changes in the tunica media and intima. 10 Aside from some extensive cases of atherosclerosis, clinical consequences of degenerative arterial diseases are rarely reported in dogs. Here, we report an unusual case of systemic arterial degeneration resembling multiple arterial thromboemboli in a dog, with severe clinical consequences.
A 27-mo-old, spayed female mixed-breed dog was presented because of left forelimb pain and possible ataxia, and was placed on doxycycline (5 mg/kg, q12h) and prednisone (0.5 mg/kg, q6h) for presumptive neurologic disease. The dog failed to respond to treatment after 6 wk and was referred to Hokkaido University Veterinary Teaching Hospital (HUVTH; Sapporo, Japan). At presentation at the HUVTH, the dog had deep ulcers of the skin on the left forelimb. Hematologic and biochemistry profiles were within RIs except for some elevated liver enzyme activities (alanine aminotransferase 218 IU/L, RI: 17–78 IU/L; aspartate aminotransferase 67 IUL, RI: 17–44 IU/L; alkaline phosphatase 1,165 IU/L, RI: 47–254 IU/L; gamma-glutamyl transferase 16 IU/L, RI: 5–14 IU/L), and the total T4 level was low (10.3 nmol/L, RI: 13–50 nmol/L). An MRI did not identify any lesions in the brain. The patient was continued on prednisone (0.5 mg/kg, q6h), and the antibiotic was switched to minocycline (10 mg/kg, q12h). The patient returned 2 mo later because of development of full-thickness soft tissue necrosis (gangrene) with bone exposure of the right forelimb (Fig. 1), development of additional foci of dermal necrosis on the hindlimbs and nasal planum, and minimal improvement of the initial left forelimb lesions. Sonographically, the abdominal aorta contained a mildly echogenic mass that extended into both external iliac arteries. Blood flow to the hindlimbs was minimal. Computed axial tomography confirmed the mass and also revealed an intraluminal mass occluding segments of the aorta, specifically the aortic arch proximal to and extending into the brachiocephalic artery. Echocardiography did not reveal any abnormalities. A presumptive diagnosis of arterial thromboembolism was made, and the dog was given anticoagulant therapy and underwent amputation of the right distal forelimb; however, 2 wk later, the dog was euthanized because of a poor prognosis.
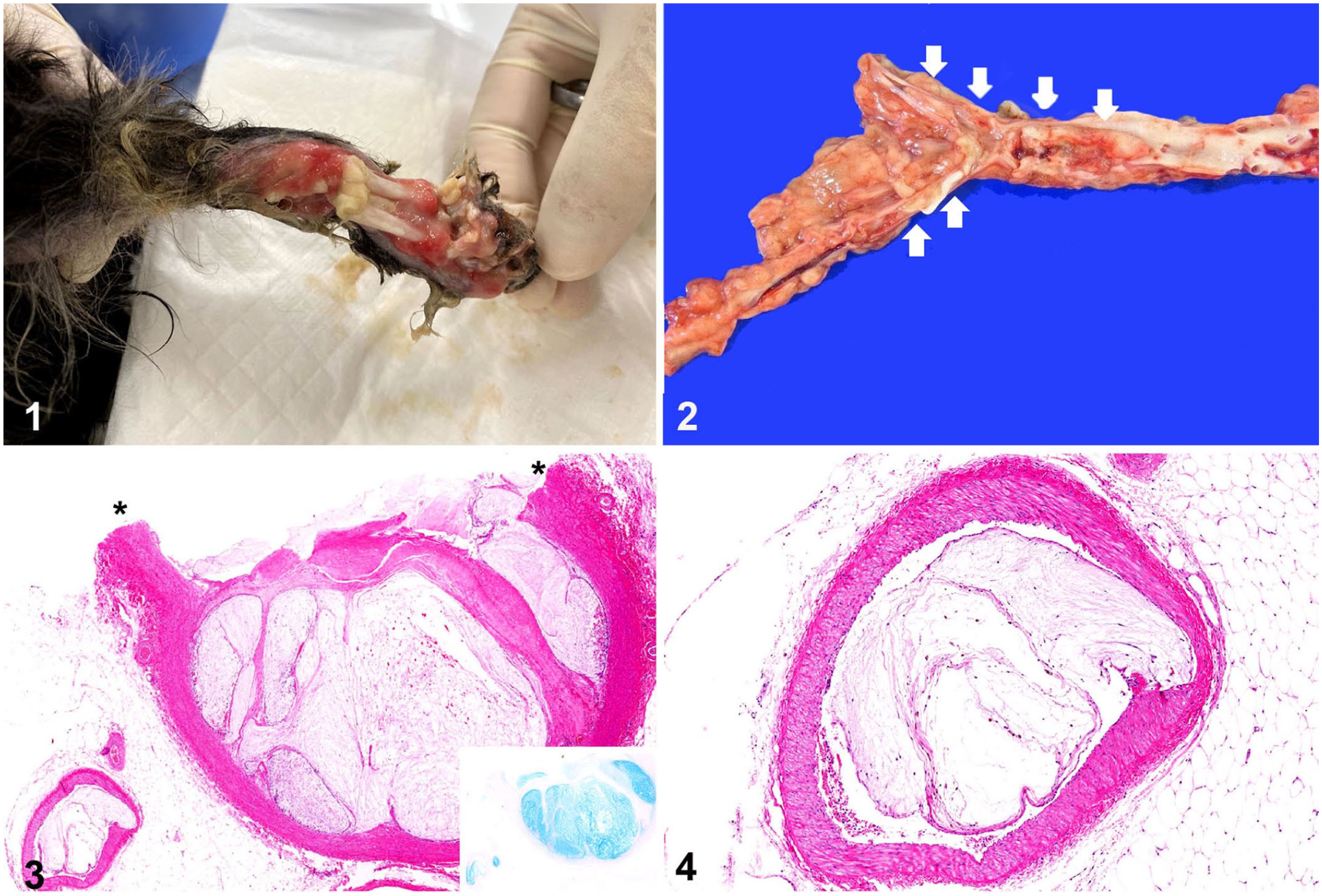
Systemic intimal mucoid degeneration of the arteries in a dog.
At postmortem examination, the distal left forelimb was detached at the carpus as a result of ischemic necrosis of all soft tissues (gangrene). The hindlimbs, tail, and nose had multifocal-to-coalescing well-demarcated foci of alopecia, which were often ulcerated and accompanied by underlying soft tissue necrosis and bone exposure. The exposed soft tissues were dry and discolored dark gray-tan. Segments (3–6 cm long) of the abdominal aorta, brachiocephalic artery, and mesenteric artery were occluded by ill-defined, pink-to-tan soft intraluminal masses (Fig. 2). The masses could not be detached from the arterial wall. Multiple wedged-shaped, dark-red foci were identified in the spleen, kidneys, and ileum.
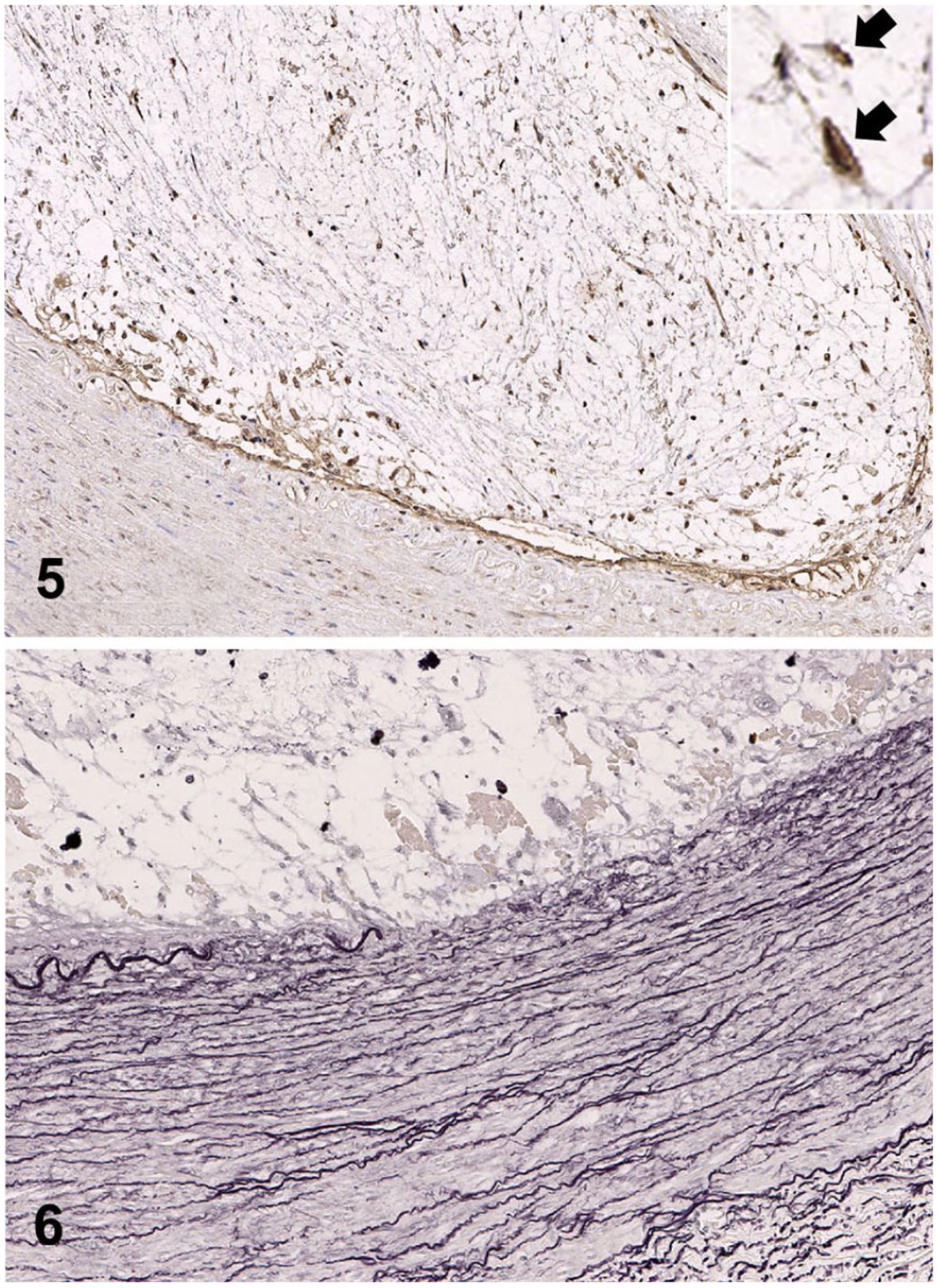
Samples of the arteries, amputated limb, and major organs were fixed in 10% neutral-buffered formalin, processed routinely, and sections stained with hematoxylin and eosin. Additional sections of the arteries, kidney, and spleen were stained with Alcian blue (pH 2.5) and Elastica van Gieson stains, and then subjected to immunohistochemistry using primary antibodies directed against von Willebrand factor (vWF; A00821, 1:500, Dako), α–smooth muscle actin (α-SMA; N1584, 1:2, Dako), and vimentin (IS630, 1:2; Dako). The intraluminal masses were demonstrated histologically to be markedly thickened tunica intima expanded by intimal accumulation of a hypocellular pale basophilic matrix (Figs. 3, 4) that stained deeply blue with Alcian blue stain (Fig. 3, inset). Within the mucoid matrix, spindle-to-satellite cells with elongate hyperchromatic nuclei and scant cytoplasm were sparsely distributed. These cells were positive for vWF and vimentin but negative for α-SMA (Fig. 5). The tunica intima was partially to circumferentially thickened, such that the lumen of the affected arteries was often obscured or markedly narrowed leaving slit-like spaces. The intimal mucoid matrix was sometimes separated from the overlying endothelium by a thin layer of collagenous connective tissue. Recanalization was occasionally observed within the connective tissues. Endothelium was morphologically normal. Focally, the intimal mucoid matrix compressed the outer vessel wall, with destruction of the internal elastic lamina and elastic fibers in the tunica media (Fig. 6). The amount of Alcian blue–positive substance in the tunica media of these arteries was similar to that of unaffected arteries. There were no apparent histologic changes in the tunica externa.
Systemic intimal mucoid degeneration of the arteries in a dog.
The same histologic changes with variable severity were observed in the following arteries: abdominal aorta, brachiocephalic artery, mesenteric artery; and medium and small muscular arteries in the nose, right forelimb, spleen, pancreas, duodenum, and kidneys. Infarcts were found in the spleen, kidneys, and ileum in fields supplied by the affected arteries. Some glomerular capillaries and splenic sheathed capillaries were occluded by pieces of Alcian blue–positive hypocellular matrix, which were morphologically similar to the proliferative intimal tissue of the large arteries but not connected to the capillary walls. Other histologic lesions in the arteries included vacuolation of the medial smooth muscle cells of the splenic trabecular arteries, and fibrinoid necrosis of the left renal interlobar and arcuate arteries. The latter lesions were accompanied by minimal infiltrates of neutrophils and macrophages. There were no apparent histologic abnormalities in the brain.
On the basis of these findings, we diagnosed systemic mucoid degeneration of the arterial tunica intima. Although arterial thromboembolism was suspected based on clinical imaging and at postmortem examination, the intraluminal masses were demonstrated histologically to be marked expansion of the tunica intima with accumulation of hypocellular mucoid matrix. This unusual intimal mucoid deposition likely caused luminal occlusion and subsequent ischemia, manifested as infarction and necrosis. In addition, emboli morphologically similar to the mucoid intimal tissue were observed in capillaries. Such disseminated mucoid emboli might be observed in cardiac myxoma 7 ; however, our case had no evidence of neoplasia. Other diseases that could predispose to emboli or thromboemboli, such as cardiac disease, pancreatitis, hyperadrenocorticism, necrotizing myelopathy, or massive trauma,2,6,10,14 were not evident in our case. Given these findings, the mucoid emboli are considered to be products of degeneration of the intimal mucoid matrix in the upstream arteries.
Although fibrinoid necrosis accompanied by inflammation was observed in the left renal arteries, it is unlikely that inflammatory processes were involved in the myxoid change seen in most arteries. Chronic vasculitis can cause deposition of mucoid matrix in the vascular wall 16 ; however, the majority of the affected arteries lacked inflammation. In contrast, the inflamed left renal arteries did not exhibit the intimal proliferation or mucoid matrix deposition noted elsewhere in this dog. Our findings suggest a distinct pathogenesis for each lesion. Hypertension is a known cause of fibrinoid necrosis with inflammation. 2 Although antemortem blood pressure was not obtained in our case, hypertension resulting from major arterial occlusion is a possible cause of the vascular lesion in the left renal arteries.
Our case has histopathologic features distinct from well-known canine degenerative cardiovascular diseases such as atherosclerosis or endocardiosis. In atherosclerosis and endocardiosis, the majority of intimal and subendocardial proliferating cells are smooth muscle cells and fibroblasts, respectively, 10 which contrasts with immunohistochemical findings that the proliferating cells were positive for the endothelial marker vWF in our case.
Several degenerative arterial diseases accompanied by mucoid matrix deposition are reported in human medicine, including intimomedial mucoid degeneration (IMD), Erdheim–Gsell cystic medial necrosis (EGCMN), and mucoid intimal edema (MIE). However, the histologic features of our case are not entirely consistent with any of these diseases. The mucoid changes in IMD and EGCMN are mainly present as localized deposition in the tunica media of large arteries,1,12,17 which is distinct from our case in which there was systemic mucoid deposition in the tunica intima. Some IMD patients also have mucoid matrix deposition in the tunica intima; this change is always accompanied by medial mucoid matrix deposition.1,3,5,12,15,17 Furthermore, the typical consequence of IMD and EGCMN is a focal aneurysm as a result of disruption of the inner elastic lamina, rather than luminal occlusion as observed in our case.1,3,15,17,18 In addition, our case had no histologic evidence suggestive of Marfan syndrome or Ehlers–Danlos syndrome, which could cause EGCMN.9,13 Our case had several similarities with MIE, which has intimal mucoid deposition in arterioles and luminal occlusion as a consequence. However, our case is characterized by proliferation of vWF-positive cells, instead of the concentric proliferation of α-SMA–positive myointimal cells associated with MIE.4,8
Given that the T4 level was below the RI, hypothyroidism cannot be excluded as a possible cause of mucinous matrix accumulation in the arteries. There is, however, no report describing mucinous accumulation of arteries in animals or humans with hypothyroidism. Furthermore, in our case, clinical imaging and postmortem examination did not reveal abnormalities in either thyroid gland or any clinical sign such as hyperkeratosis, myxedema, or hyperpigmentation that would support a diagnosis of hypothyroidism. 11
Although we did not identify an etiology for the vascular changes in our case, given the age of the dog, congenital factors should be considered. In cases of suspected thromboembolism, atherosclerosis and other degenerative arterial diseases should be considered, especially when patients do not have known conditions predisposing to thromboemboli.
Footnotes
Acknowledgements
We thank all the members of the Laboratory of Comparative Pathology, Hokkaido University for giving useful advice on writing this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
