Abstract
Bovine anaplasmosis is a worldwide infectious disease caused by the intraerythrocytic bacterium Anaplasma marginale, which is transmitted by ticks and fomites. A. centrale is a less virulent subspecies used as a live vaccine in cohorts of 8- to 10-mo-old calves that did not naturally reach enzootic stability. We developed 3 variants of a double-antigen sandwich ELISA (dasELISA) using a recombinant major surface protein 5 (MSP5) from A. marginale (dasELISAm) or from A. centrale (dasELISAc) or using MSP5 from both organisms (dasELISAmc). Each dasELISA was tested for the detection of antibodies against A. marginale and A. centrale. The tests were validated using serum samples from cattle not infected with Anaplasma spp. (n = 388), infected with A. marginale (n = 436), and vaccinated with A. centrale (n = 358), confirmed by nested PCR. A total of 462 samples were compared with a commercial competitive ELISA (cELISA). For dasELISAm, dasELISAc, and dasELISAmc, specificities were 98.7%, 98.7%, and 97.4%, and overall sensitivities were 92.6%, 85.7%, and 97.4%, respectively. For A. marginale–infected and A. centrale–vaccinated cattle, sensitivities were 97.7% and 86.3% for dasELISAm, and 77.7% and 95.5% for dasELISAc, respectively. Sensitivity of dasELISAmc was similar for both groups (>96%). The agreement rate between dasELISAmc and cELISA was 96.3% (κ = 0.92); the former test allowed earlier detection of seroconversion of vaccinated cattle than did cELISA. Based on these results, the test could be used to 1) determine the enzootic stability or instability of anaplasmosis in calves, 2) conduct epidemiologic studies, and 3) evaluate the immunogenicity of A. centrale live vaccine.
Keywords
Introduction
Bovine anaplasmosis is an infectious disease caused by the obligate intraerythrocytic bacterium Anaplasma marginale (order Rickettsiales, family Anaplasmataceae), which is endemic in tropical, subtropical, and some temperate areas of the world (Theiler A. Anaplasma marginale (gen. and spec. nov.): the marginal points in the blood of cattle suffering from a specific disease. Report of the Government Veterinary Bacteriologist, 1908–1909. Pretoria, South Africa: Government Printing and Stationery Office, 1910:7–64. Available at: http://hdl.handle.net/2263/10409).11,16 A. marginale is transmitted from infected to susceptible cattle by blood-sucking arthropods and blood-contaminated fomites. 16 In Argentina, anaplasmosis is endemic north of parallel 33°S, exceeding the distribution area of the cattle tick Rhipicephalus microplus, which is present north of parallel 30°S. 10 Cattle that become infected during the first months of life develop subclinical disease, whereas adult cattle suffer the severe form of anaplasmosis, characterized by anemia, abortion, weight loss, reduced milk production, and often death. Cattle that have recovered from the acute phase of infection acquire long-lasting immunity and remain infected carriers for life, serving as reservoirs for transmission to naive cattle.6,15 To prevent outbreaks of anaplasmosis in herds located in endemic areas, cattle should become infected naturally or by vaccination during the first year of life, when they are more resistant to the severe form of the disease.2,14
In Australia, Israel, and various countries of Africa and Latin America (including Argentina), A. centrale, a species of low pathogenicity, is used as a live vaccine, given that it produces cross-protection against A. marginale infection (Theiler A. Further investigations into anaplasmosis of South African cattle. First report of the Director of Veterinary Research. Pretoria, South Africa: Government Printer and Stationary Office, 1911:7–46. Available at: http://hdl.handle.net/2263/11310). Highly sensitive and specific serologic tests are needed to identify Anaplasma spp. carrier cattle, determine the epidemiologic status of anaplasmosis in calves from enzootic regions, and evaluate the immune response to the A. centrale vaccine. 11 Various tests have been developed to detect antibodies against Anaplasma spp., although ELISA is the most widely used worldwide.5,9,13,26 A competitive ELISA (cELISA) based on A. marginale recombinant major surface protein 5 (MSP5m) fused with E. coli maltose-binding protein (MBP-MSP5m) and the monoclonal antibody ANAF16C1 had a sensitivity of 96% and a specificity of 95% when evaluated in cattle of an endemic area of anaplasmosis.13,26 The MBP, which produces false-positive reactions, has been replaced with glutathione S-transferase (GST), and the resulting fusion protein (GST-MSP5m) was used to develop an indirect ELISA (iELISA) and a cELISA, both available commercially.4,19 The performance of these tests to evaluate the antibody response after vaccination with live A. centrale has not been evaluated. Soluble truncated MSP5 protein, without N-terminus transmembrane helix (tMSP5, residues 28–210) and without molecular chaperones, improved the specificity of the cELISA. 20 The fusion protein of MSP5 of A. centrale and A. marginale (MSP5c-MSP5m) increased the sensitivity of the cELISA when evaluated in a cattle population from endemic areas in which the live vaccine is used regularly. 20
In recent years, the double-antigen sandwich ELISA (dasELISA) has been used successfully to detect low levels of antibodies to several pathogens.8,12,17,21,24,25,27 In this test, specific antibodies present in serum samples bind to the immobilized antigen on the solid phase with one antigen-binding site. The remaining antigen-binding site on the antibody is free to bind with labeled soluble antigen, which serves as the detector reagent in the ELISA.
We evaluated the performance of 3 variants of dasELISA based on MSP5 of A. marginale (MSP5m) or MSP5 of A. centrale (MSP5c) or both together, without chaperone proteins, to identify cattle naturally infected with A. marginale and vaccinated with A. centrale.
Materials and methods
Cattle samples
To validate the serologic tests, sera and whole blood samples from cattle of different groups (G) with known status of anaplasmosis infection were obtained or selected from our own biological bank (Instituto Nacional de Tecnología Agropecuaria [INTA], Santa Fe, Argentina). A nested PCR (nPCR) was used as the gold standard; the current infection of cattle by Anaplasma ssp. was assessed from 2 samples obtained with an interval of 30 d. 23 The positive samples were analyzed using 2 specific nPCRs to differentiate A. marginale from A. centrale. 18 Blood samples were aseptically collected with and without 5% citrate as anticoagulant. Serum and whole blood samples were distributed in aliquots and kept at −20°C until use. Genomic DNA was obtained from blood samples using the phenol–chloroform method. 3
Serum samples from 436 cows naturally infected with A. marginale (G1) were obtained from 2 farms, located in Avia Terai (26°40’S 60°46’W) and Villa Angela (27°35’S 60°43’W), in Chaco province. Cattle in both locations have endemic infections of A. marginale and R. microplus. These samples were used to evaluate the sensitivity of the tests.
Serum samples from 759 cattle of different ages, raised in a dairy herd historically free of anaplasmosis located in Rafaela (31°11’S 61°30’W), a temperate area of Santa Fe province free of R. microplus, were selected. These cattle were used for the following experiments: 1) evaluation of the sensitivity of tests using 358 ten-mo-old calves, vaccinated with live A. centrale 4 mo previously (G2); 2) evaluation of the specificity of serologic tests, through the analysis of sera from 388 dairy cows free of anaplasmosis (G3); 3) duration of specific antibody in sera from 9-mo-old calves (n = 11) recently vaccinated with live A. centrale >18 mo after vaccination (G4); and 4) collection of 2 positive control sera, 60 d after the experimental inoculation of 2 steers with 107 A. marginale or A. centrale parasitized erythrocytes (G5).
The vaccine against A. centrale used in our study is approved by the National Service of Agri-Food Health and Quality (93159). Experimentally inoculated cattle were maintained in isolated pens with food and water ad libitum at the Rafaela Experimental Station of the INTA, and the clinical status was monitored daily over 45 d. All procedures were approved by the Animal Care Committee of the Faculty of Veterinary Sciences, National University of Litoral, Argentina (protocol 243/15).
Antigens
Cloning, expression, and purification of the recombinant proteins
Complementary DNA encoding residues 28–210 with a 6-histidine tag at the C-terminus of the A. marginale MSP5 (tMSP5m) or A. centrale MSP5 (tMSP5c) were amplified by PCR and cloned in a pET9b vector (Novagen, Madison, WI) to build ptMSP5m and ptMSP5c vectors. 20
tMSP5m and tMSP5c were expressed in E. coli BL21 RIL (DE3)pLysS cells (Novagen) and purified by pseudo-affinity with Ni2+-NTA agarose (Qiagen, Hilden, Germany). Briefly, E. coli BL21 RIL (DE3)pLysS competent cells transformed with ptMSP5m or ptMSP5c were cultured to an optical density at 600 nm (OD600nm) = 1 at 37°C in 500 mL of Luria–Bertani medium supplemented with 50 µg/mL of kanamycin and 34 µg/mL of chloramphenicol. Protein expression was induced with 1% lactose. After 3 h of incubation at 37°C, bacteria were harvested by centrifugation, suspended in 10 mL of lysis buffer (50 mM sodium phosphate, 300 mM NaCl, 10 mM imidazole, pH 8) containing 1:1,000 protease inhibitor cocktail set III (Calbiochem, La Jolla, CA), and lysed by 2 passes through a cell disruptor at 20,000 psi (Avestin, Ottawa, Ontario, Canada). After centrifugation (12,000 × g, 30 min, 4°C), the soluble fraction was separated and added to 2 mL of Ni2+-NTA agarose previously equilibrated with lysis buffer. After incubation at 4°C for 1 h, the suspension was poured into a 1.5 × 5.0 cm column and washed with 5 volumes of lysis buffer containing 30 mM imidazole. Bound tMSP5m or tMSP5c was eluted with 5 volumes of 100 mM imidazole lysis buffer. Finally, the buffer was exchanged into 50 mM sodium phosphate, 200 mM NaCl, pH 7.2, by overnight dialysis at 4°C. Molar concentration of the dialyzed proteins was calculated by absorbance at 280 nm using a molar extinction coefficient (ε280nm) equal to 8,940/M/cm or 10,430/M/cm for tMSP5m or tMSP5c, respectively.
Protein biotinylation
tMSP5m or tMSP5c (2 mg) was incubated with 0.8 mg of sulfo-NHS-biotin (N-hydroxysulfosuccinimidobiotin; Pierce Biotechnology, Thermo Fisher Scientific, Waltham, MA) in a final volume of 2 mL. The reactions were incubated on ice for 2 h. Free biotin was removed on a Zeba spin desalting column, 7K MWCO (Pierce). The number of biotin groups incorporated per molecule was determined using the 4’-hydroxyazobenzene-2-carboxylic acid (HABA) method (Pierce). The purity of tMSP5m-biotin and tMSP5c-biotin was evaluated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and western blot.
Western blot
SDS-PAGE was performed as described previously. 22 Proteins (0.2 μg) were transferred to nitrocellulose membranes. Free protein-binding sites were blocked by incubation with TBS (50 mM Tris-Cl, 150 mM NaCl, pH 7.6)–5% fat-free dried milk for 2 h. Peroxidase–streptavidin (Jackson ImmunoResearch, West Grove, PA) was added at 1:750 dilution in TBS–0.05% Tween 20–5% fat-free dried milk. After incubation at 25°C for 1 h, membranes were washed 5 times with TBS–0.05% Tween 20 and reacted with the colorimetric substrate 3,3’-diaminobenzidine tetrahydrochloride (DAB; MilliporeSigma, St. Louis, MO).
ELISA protocols
All samples and controls were assayed in duplicate. Incubations were performed at 25°C.
cELISA for detection of antibody against A. marginale
We used a commercial cELISA (VMRD, Pullman, WA), recommended by the World Organization for Animal Health (OIE), following the manufacturer’s instructions. Results were expressed as percentage of inhibition (%I) according to the following formula: %I = 100 [1 – (sample OD/negative control OD)]. The cutoff point recommended is ≥ 30%I. 26
dasELISA
The optimal concentrations of coating antigen and biotinylated antigen for the 3 dasELISA variants were determined by checkerboard serial-dilution analysis. The optimal working conditions are described below. Microplates were coated overnight at 4°C with 100 µL of 0.8 µg/mL of tMSP5m (dasELISAm) or tMSP5c (dasELISAc) or 50 µL of tMSP5m and 50 µL of tMSP5c (dasELISAmc), in buffer PBS (50 mM sodium phosphate, 150 mM NaCl, pH 7.2). After 2 washes with PBS, blocking buffer (10% fat-free dried milk in PBS, 300 μL/well) was added and the plates were incubated for 1 h. After washing 3 times with PBST (PBS containing 0.05% Tween 20), plates were incubated with the undiluted serum samples (100 μL/well) for 1 h. The plates were washed 5 times with PBST and then 100 μL of tMSP5m-biotin or tMSP5c-biotin (2 µg/mL) or 50 µL of both together, in PBST–10% fat-free dried milk , were added. After 1-h incubation, the plates were washed 5 times with PBST and bound protein-biotin was detected with peroxidase–streptavidin diluted 1:500 in PBST–10% fat-free dried milk, 100 μL/well, for 1 h. The final wash (5 times with PBST) was followed by the addition of the chromogenic substrate 1 mM 2,2’-Azinobis [3-ethylbenzothiazoline-6-sulfonic acid]-diammonium salt (ABTS; MilliporeSigma) in 0.05 M sodium citrate, pH 4.5, 0.0025% V/V H2O2 (100 μL/well). The plates were incubated in the dark for 16–20 h before the color reaction was measured at 405 nm with an ELISA plate reader. Results were expressed as percentage of positivity (%P) according to the following formula: %P = 100(sample OD/positive control OD).
Kinetics of the colorimetric reaction for dasELISA
The kinetics of the colorimetric reaction to reach OD405nm saturation for dasELISA was evaluated for 20 h in 50 cattle serum samples selected from: G1 cows infected with A. marginale with fast (n = 10) or slow (n = 10) colorimetric reaction; G2 calves vaccinated with A. centrale with fast (n = 10) or slow (n = 10) colorimetric reaction; and G3 cows free of Anaplasma spp. (n = 10). The OD405nm measurements were recorded every 5 min for 2 h, every 10 min during the next 2 h, every 30 min during the following hour, and then once at hours 6, 8, and 20 after the chromogenic substrate was added.
Comparison between dasELISAmc and cELISA
A total of 462 serum samples that included 145, 113, and 204 samples of the groups G1, G2, and G3, respectively, were analyzed by dasELISAmc and cELISA, and their results were compared.
Duration of antibody in serum against A. centrale after vaccination
Eleven 9-mo-old Holstein male calves free of detectable antibody against Anaplasma spp. were vaccinated with 107 erythrocytes parasitized with A. centrale by intramuscular injection (G4). After vaccination, serum and blood samples were collected weekly for 8 wk and then monthly for 18 mo.
Data analysis
The optimal cutoff point and the diagnostic sensitivity and specificity for the 3 variants of dasELISA were established using the positive serum samples specific for the antigen immobilized to the plate (G1 for dasELISAm, G2 for dasELISAc, and G1+G2 for dasELISAmc) and 388 negative sera (G3), via receiver operating characteristic (ROC) analysis. The cutoff point used for cELISA was obtained using 145 and 204 serum samples from G1 and G3, respectively, via ROC analysis, and compared with that recommended by the manufacturers. Sensitivity for each of the 3 dasELISA variants was determined using serum samples from 3 groups of cattle infected with Anaplasma spp. (G1+G2), A. marginale (G1), and A. centrale (G2), according to the following formula: Se = 100[n of dasELISA positives/(n of true positives + n of false negatives)]. The overall sensitivity for each test was defined using serum from Anaplasma spp. (G1+G2) infected cattle.
The inter-assay agreement between dasELISAmc and cELISA was determined using the kappa coefficient (κ). 1 MedCalc v.8.1.0.0 statistical software (MedCalc Software, Mariakerke, Belgium) was used. Differences among means of %P obtained with the 3 dasELISA protocols were evaluated using a Mann–Whitney test.
Results
Antigen production
Recombinant tMSP5m and tMSP5c (21.7 kDa) were purified from the soluble intracellular fraction of bacterial lysates by Ni2+-NTA agarose affinity chromatography. The purity of recovered recombinant proteins was >95%, and the yield was 40 mg/L and 60 mg/L of culture for each protein, respectively. The biotinylation reaction of tMSP5m and tMSP5c allowed us to incorporate 2 or 3 biotin groups per molecule, as determined by the HABA method. The molecular weight of biotin is 244 Da; therefore, no differences in molecular weight were observed between biotinylated and non-biotinylated proteins by SDS-PAGE (Fig. 1A). Western blotting with peroxidase–streptavidin showed an ∼ 22-kDa band, as expected for tMSP5m-biotin and tMSP5c-biotin (Fig. 1B).
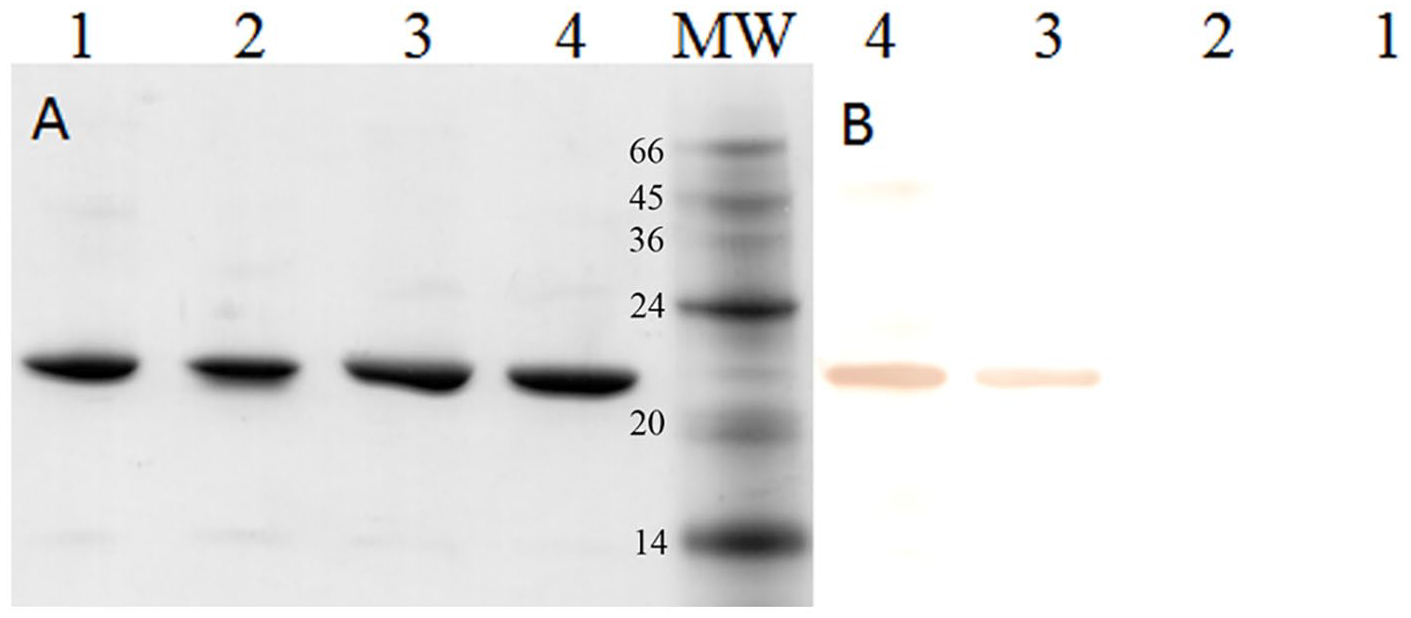
Purification and biotinylation of recombinant proteins. SDS-PAGE (
Antibody detection by ELISA
Sera with high concentrations of antibody against A. marginale or A. centrale showed a rapid colorimetric reaction that peaked (OD405nm ≈ 3) after 1 h of incubation, whereas sera with less antibody had slower colorimetric reactions that peaked at 8 h of incubation. The OD405nm values for sera negative for antibody against Anaplasma spp. were low (OD405nm < 0.2) after 8 h of incubation and remained constant through 20 h of incubation. These results allowed us to set the reading time of dasELISA at 16–20 h, when the difference between negative and positive control was maximum (Fig. 2).
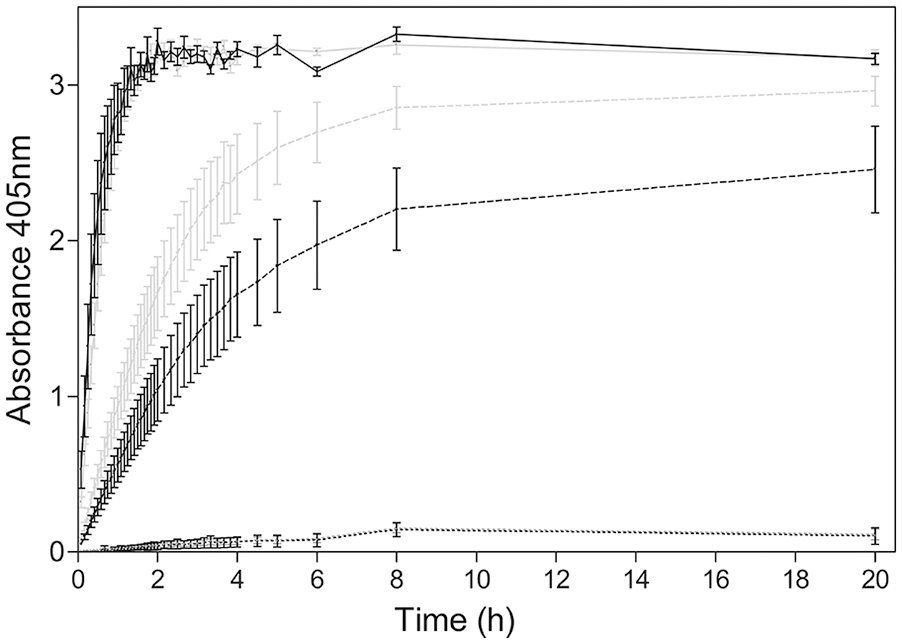
Kinetics of the colorimetric reaction for different cattle groups. The optical density (OD) mean of 10 strong-positive and 10 weak-positive sera for Anaplasma marginale (black) and for A. centrale (gray) are shown with a continuous line and a dashed line, respectively. The OD mean of 10 negative sera is shown with a black dotted line.
ROC analysis showed a cutoff point ≥ 25%P for the 3 variants of dasELISA when noninfected and nonvaccinated cattle (G3) were used as the negative group and A. marginale–infected (G1) and/or A. centrale–vaccinated cattle (G2) were used as the positive group. G1 was used for dasELISAm, G2 for dasELISAc, and both groups for dasELISAmc. The area under the ROC curve (AUC) was 0.998, 0.996, and 0.997 for dasELISAm, dasELISAc, and dasELISAmc, respectively. The cutoff point determined for cELISA was ≥30%I, which coincided with that recommended by the manufacturer.
Significant differences (p ≤ 0.05) in the mean of %P of G1 and G2 cattle were obtained when serum samples were analyzed by dasELISAm and dasELISAc (Fig. 3). Mean and SD of %P were 85 ± 19 for G1 and 68 ± 30 for G2 by dasELISAm, and 63 ± 34 for G1 and 83 ± 25 for G2 by dasELISAc (Fig. 3). No significant differences were observed between means of %P of G1 and G2 when the serum samples were analyzed by dasELISAmc (p > 0.05; Fig. 3).
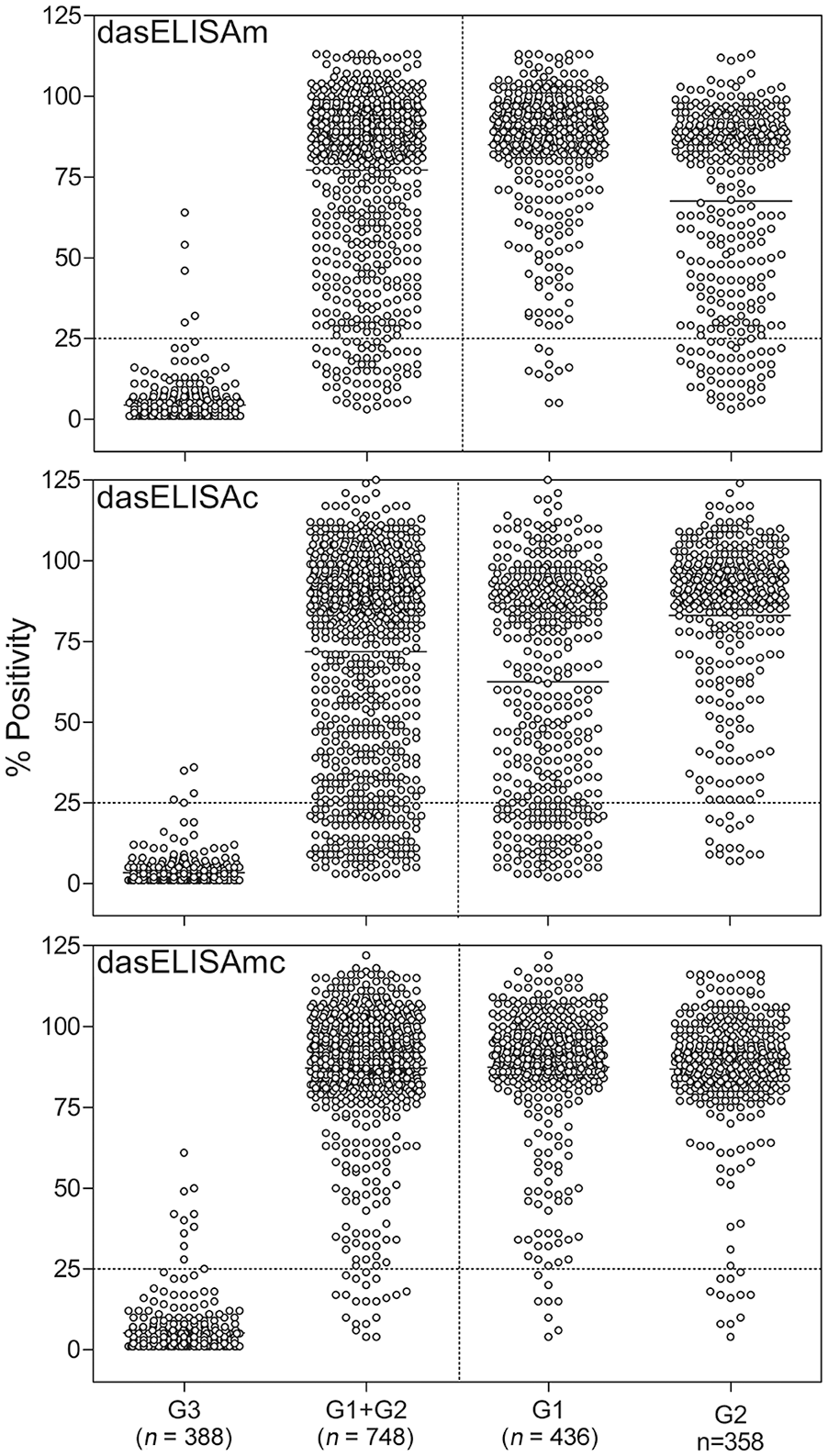
Results of dasELISAm, dasELISAc, and dasELISAmc for Anaplasma spp. antibody detection expressed as percentage of positivity (%P). Dot plot of %P for the negative sera from A. marginale noninfected and A. centrale nonvaccinated cattle (G3) and the overall positive samples from infected cattle of G1+G2 is shown on the left. Dot plot of G1 or G2 is shown in the separate dot plots on the right. The cutoff value for the 3 dasELISA variants was 25%P and is indicated with a dotted line.
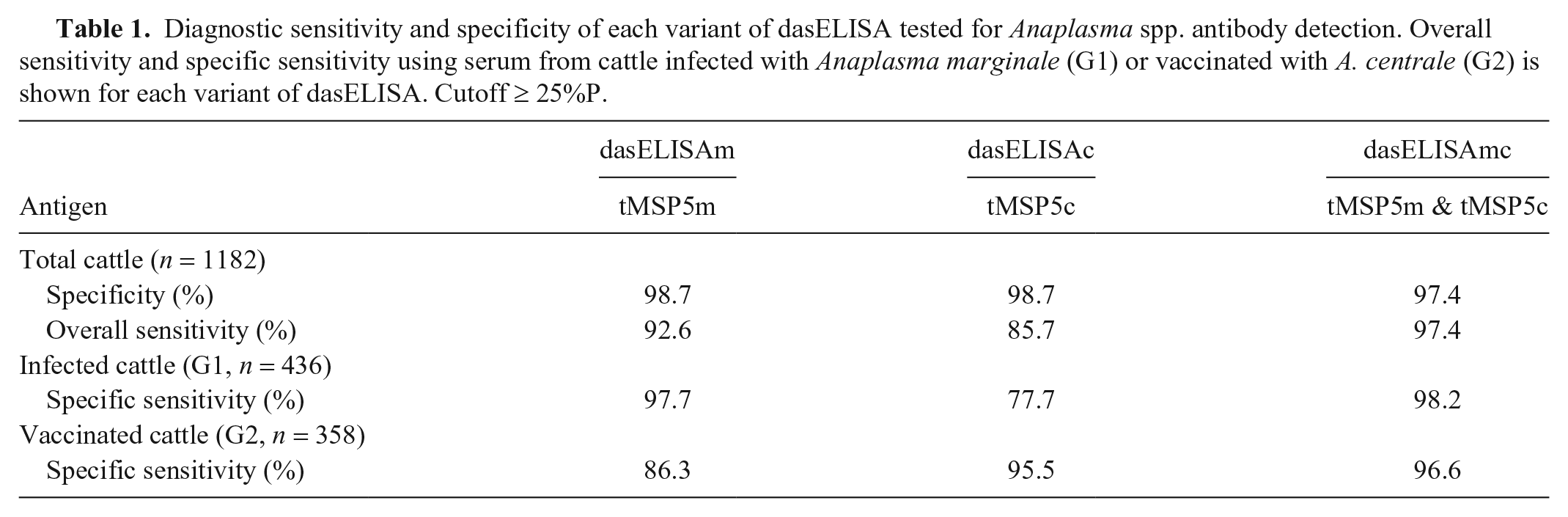
With a cutoff of ≥ 25%P, specificity was 98.7% for dasELISAm and dasELISAc, and 97.4% for dasELISAmc. Overall sensitivity was 92.6%, 85.7%, and 97.4% for dasELISAm, dasELISAc, and dasELISAmc, respectively. The lowest sensitivity was observed for dasELISAc (77.7%) when sera from A. marginale–infected cattle were analyzed, followed by dasELISAm (86.3%) when sera from A. centrale–immunized cattle were analyzed. For dasELISAmc, similar sensitivity values were obtained when A. marginale–infected (98.2%) or A. centrale–vaccinated cattle groups (96.6%) were analyzed (Table 1).
Diagnostic sensitivity and specificity of each variant of dasELISA tested for Anaplasma spp. antibody detection. Overall sensitivity and specific sensitivity using serum from cattle infected with Anaplasma marginale (G1) or vaccinated with A. centrale (G2) is shown for each variant of dasELISA. Cutoff ≥ 25%P.
dasELISAmc and cELISA showed an agreement rate of 96.3% (94.6%, 99.3%, and 95.6% for negative cows, cows infected with A. marginale, and cows vaccinated with A. centrale, respectively), with κ = 0.92.
After vaccination with A. centrale, the 11 male calves (100%) were positive to Anaplasma by nPCR, dasELISAmc, and cELISA at 38, 42, and 49 d, respectively (Table 2). Seroconversion was detected by dasELISAmc at least 4 d earlier than by cELISA. All cattle remained positive by both tests during 18 mo of follow-up.
Percentage of cattle positive for anaplasmosis detected by nPCR, dasELISAmc, and cELISA techniques in relation to time after vaccination.
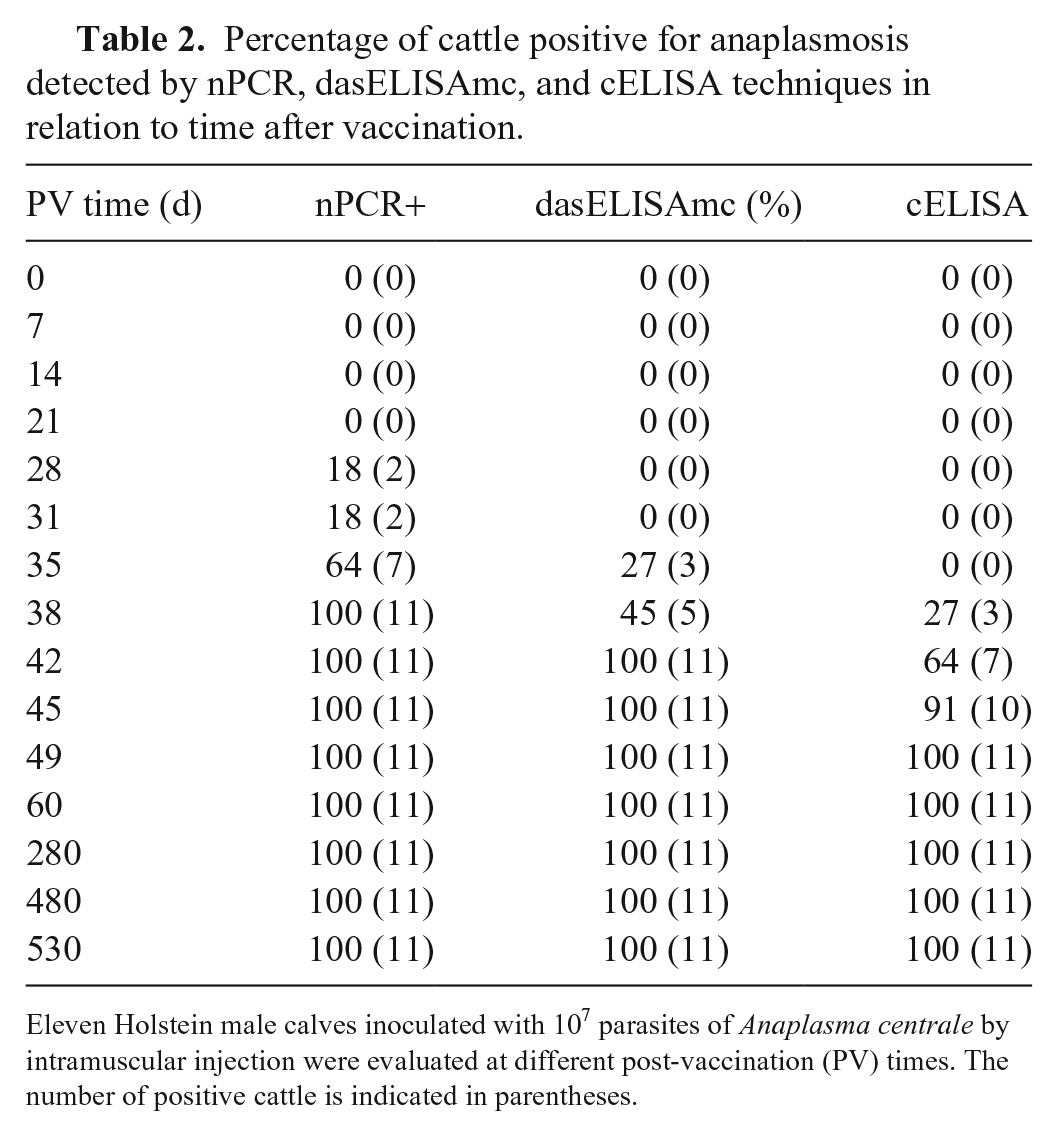
Eleven Holstein male calves inoculated with 107 parasites of Anaplasma centrale by intramuscular injection were evaluated at different post-vaccination (PV) times. The number of positive cattle is indicated in parentheses.
Discussion
Our dasELISAmc showed high sensitivity and specificity in the different epidemiologic contexts analyzed in our work. Although dasELISAm achieved 97.7% sensitivity when standardized using sera from A. marginale–infected cattle, this value decreased to 86.3% when the assay was evaluated with sera from A. centrale–vaccinated cattle. Similarly, when dasELISAc was used with sera from cattle vaccinated with A. centrale, sensitivity was 95.5% but decreased to 77.7% when the same antigen was evaluated with sera from A. marginale–infected cattle. In addition, the means of %P between A. marginale–infected (85% and 63%) and A. centrale–vaccinated (68% and 83%) cattle were significantly different (p < 0.05) when serum samples were analyzed by dasELISAm or dasELISAc, respectively. On the contrary, the mean of %P was 87%P for both cattle groups (G1 and G2) by dasELISAmc.
Although cELISA was developed to detect early infections and long-time carriers of A. marginale,13,23 it has also been widely used to detect antibodies against A. centrale. In our study, dasELISAmc was clearly more sensitive than cELISA in detecting early infections with A. centrale. Antibodies against A. centrale were detected by dasELISAmc 35 d after inoculation in 3 of the 11 vaccinated calves, 4 d earlier than by cELISA. In addition, 100% of the calves seroconverted 42 and 49 d after vaccination with A. centrale by dasELISAmc and cELISA, respectively. As expected, PCR detected the first A. centrale–positive samples 28 d after inoculation, with 100% detected 10 d later. Nested PCR, dasELISAmc, and cELISA remained positive until the end of our study, 530 d after vaccination. These results confirmed the infectivity of the vaccine (nPCR) and demonstrated that both serologic tests would be valuable tools for the detection of post-vaccinal antibodies. In Argentina, monitoring of the infectivity of A. centrale live vaccine is a routine practice and is carried out by serology 60 d after vaccination. Therefore, this post-vaccinal period would be adequate to perform the analysis through the 2 serologic tests, although the detection could be performed 15 d earlier using dasELISAmc.
Overall agreement between dasELISAmc and cELISA was substantial (96.3%); nevertheless, cELISA showed lower sensitivity to identify cattle vaccinated with A. centrale, and agreement was 95.6%, whereas agreement reached 99.3% for cattle naturally infected with A. marginale. This result can be attributed to the fact that both ELISAs contain the A. marginale MSP5 antigen, but only dasELISAmc has A. centrale MSP5 antigen. Although the inter-rater agreement between dasELISAmc and cELISA had “very good” valuation 1 (κ = 0.92), dasELISA had the advantage of maintaining the polarization between the %P of positive and negative sera, even with weak-positive samples. This advantage would be associated with the intrinsic characteristics of the dasELISA system, which allows weak-positive sera with slow colorimetric reaction (OD) to reach saturation 8–20 h after the addition of the chromogenic substrate. The dasELISA plates were read 16–20 h after adding the substrate, without affecting the specificity of the test, given that the %P of the negative sera remained at minimum levels (Fig. 3). Because dasELISA does not require secondary or monoclonal antibodies to reveal the antigen–antibody reaction, background reactivity is usually absent, which is a characteristic that cannot be avoided in the conventional iELISA or cELISA. In addition, unlike the iELISA and cELISA, whose antigens are based on the fusion proteins GST-MSP5 and GST/MBP-MSP5, dasELISA is based on the tMSP5 protein alone and avoids interference by MBP or GST.4,13,19 These fusion proteins could reduce both test specificity, as a result of antibodies against the chaperone protein, and test sensitivity, because the chaperone protein may hide important epitopes by steric hindrance.4,20
The performance of dasELISA was better than that reported for iELISA, 19 and similar to that reported for cELISA. 23 Similar to cELISA, dasELISA has the potential to be used for the determination of antibodies of different isotypes and with different animal species, given that secondary antibodies are not involved in the test. In addition, dasELISA does not require the use of monoclonal antibodies. Therefore, there is no need to use laboratory animals, which is a relevant aspect encouraged by ethics committees for animal welfare. 7
Footnotes
Acknowledgements
We gratefully acknowledge the technical assistance of Paola Amherdt for the processing of serum samples by cELISA and nPCR.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was supported by INTA, Asociación Cooperadora INTA Rafaela, and Agencia Nacional de Promoción Científica y Tecnológica (PICT0369).
