Abstract
A 15-y-old Miniature horse mare had a 6-mo history of an ulcerated mass on the right lower eyelid. An incisional biopsy and a subsequent excisional biopsy were submitted to the Michigan State University Veterinary Diagnostic Laboratory for microscopic evaluation. Histologically, the incisional biopsy was composed of sheets of large neoplastic vacuolated polygonal cells. A few regions contained poorly differentiated neoplastic round-to-basaloid cells that rimmed the sheets of highly vacuolated polygonal cells. Both vacuolated and basaloid cells exhibited strong perimembranous and cytoplasmic immunoreactivity for E-cadherin and cytokeratin 5/6, respectively. Vacuolated polygonal cells were histochemically negative for periodic acid–Schiff, mucicarmine, and oil red O, consistent with a diagnosis of poorly differentiated carcinoma. Within the excisional biopsy specimen, there were anastomosing cords and nests of neoplastic squamous epithelial cells that merged with sheets of similar vacuolated polygonal cells. These findings are consistent with a squamous cell carcinoma with clear cell differentiation. In addition, in the adjacent dermis, there was solar elastosis suggestive of ultraviolet (UV) damage. A clear cell variant of squamous cell carcinoma is a rare entity in humans that previously has not been described in animals, to our knowledge, and is often associated with chronic UV exposure.
A 15-y-old Miniature horse mare was presented to the referring veterinarian because of a 6-mo history of blepharospasm of the right eye associated with a non-healing wound on the lower eyelid. Physical examination revealed a 3-cm, ovoid, ulcerated mass on the right lower eyelid that exuded yellow discharge. An incisional biopsy and a subsequent excisional biopsy 30 d later were submitted to the Michigan State University Veterinary Diagnostic Laboratory (East Lansing, MI) for microscopic evaluation.
Tissues from both the incisional and excisional biopsies were fixed in 10% neutral-buffered formalin, and processed routinely; sections were stained with hematoxylin and eosin. The initial incisional biopsy was difficult to interpret because the sample was composed primarily of sheets of large vacuolated polygonal cells (Fig. 1). These cells had large peripheralized nuclei with finely stippled chromatin. A few areas were characterized by rows and trabeculae of poorly differentiated neoplastic round-to-basaloid cells that surrounded the vacuolated polygonal cells. Anisokaryosis of these cells was moderate, and there were 0–2 mitoses per high power field (HPF). The proliferation of round-to-basaloid cells rimming the vacuolated cells was concerning for a neoplasm of epithelial origin; however, an inflammatory reaction with xanthomatous, lipid-laden macrophages, or an adipocytic neoplasm, was also considered.
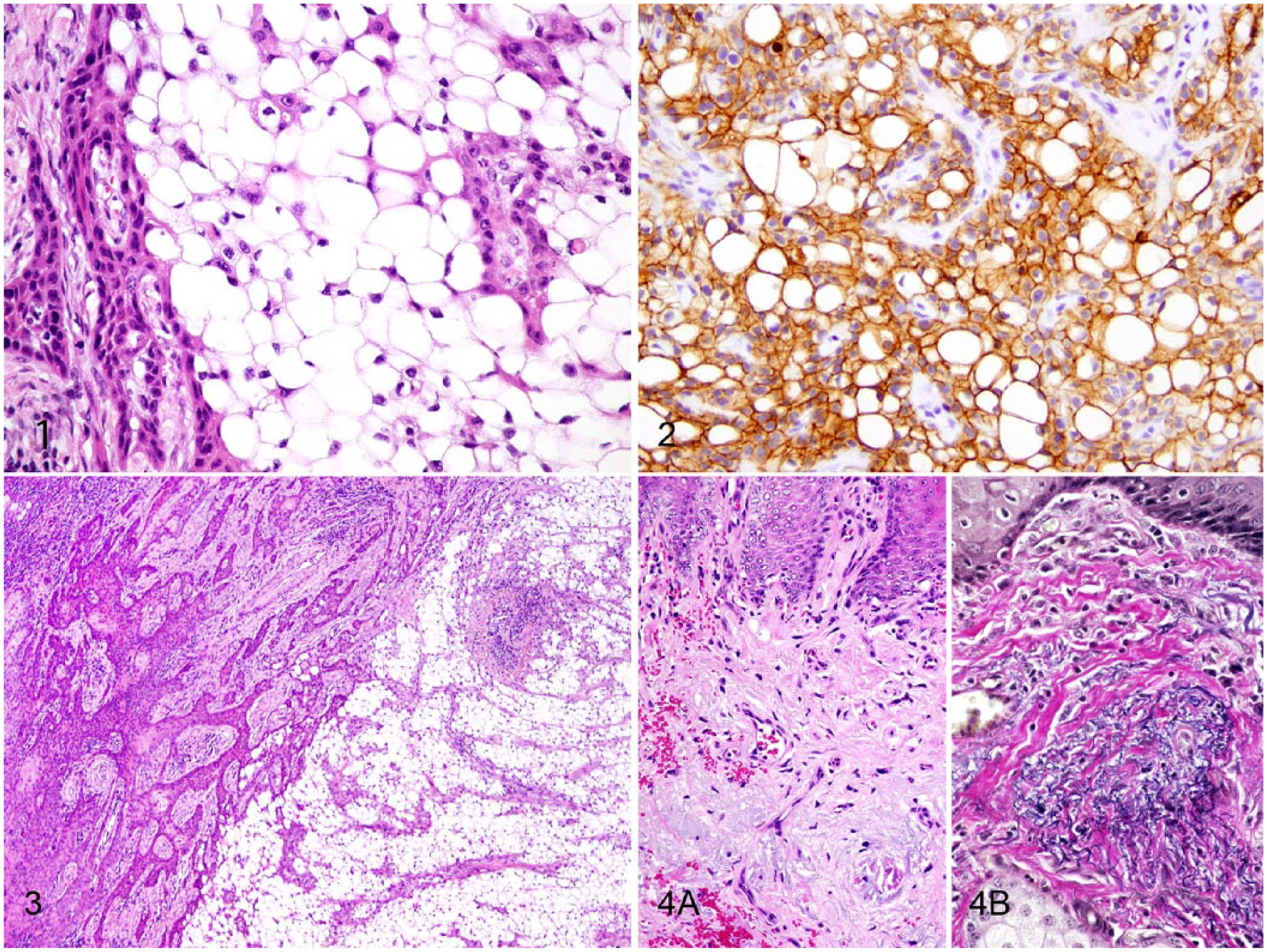
Squamous cell carcinoma with clear cell differentiation in the eyelid of a Miniature horse.
Immunohistochemistry for E-cadherin (mouse monoclonal, clone 36; BIOSCI 610181, BD Biosciences, Sparks, MD) was performed to confirm an epithelial origin. 11 The round-to-basaloid cells and vacuolated polygonal cells had strong perimembranous labeling for E-cadherin (Fig. 2). Immunohistochemical labeling for cytokeratin (CK) 5/6 (mouse monoclonal, clone D5/16 B4; DAKO M7237, Dako North America, Carpinteria, CA) followed to further prove epithelial origin, and the round-to-basaloid cells and vacuolated polygonal cells had strong diffuse cytoplasmic labeling for this marker. 14
Special staining with periodic acid–Schiff (PAS), for polysaccharides, glycoproteins, and glycolipids, and mucicarmine for mucin did not identify positive material within the cytoplasm of vacuolated polygonal cells. Additionally, special staining with oil red O for lipid was performed on frozen sections of formalin-fixed tissue to further classify the cytoplasmic content of the vacuolated polygonal cells. There was no evidence of oil red O staining within the cytoplasm of these cells. A preliminary diagnosis of a carcinoma with clear cell differentiation was made. A clear cell squamous cell carcinoma (SCC) was the primary differential, followed by a clear cell basal cell carcinoma. A sebaceous or Meibomian carcinoma was considered less likely given the lack of sebaceous production evidenced by lack of PAS-positive material within the cytoplasm of vacuolated cells. An excisional biopsy was recommended to completely excise the neoplasm and to confirm this diagnosis.
A complete excisional biopsy was submitted ~1 mo later. Histologically, the excisional biopsy was comprised of a fairly well-demarcated, ulcerated dermal mass composed of overtly neoplastic squamous epithelial cells arranged in anastomosing cords and nests that extended from the surface into the underlying dermis and merged with dense sheets of vacuolated polygonal cells similar to those described in the incisional biopsy (Fig. 3). Cords and nests of neoplastic cells near the surface and along the lateral margins of the mass exhibited central keratinization. Toward the center of the mass, these cords and nests often rimmed central sheets of vacuolated polygonal cells. Neoplastic squamous cells were round-to-basaloid to polygonal, contained a small-to-moderate amount of eosinophilic cytoplasm, and had round-to-ovoid, finely stippled nuclei with a single large central nucleolus. Anisokaryosis was moderate-to-marked, and there were occasional binucleate cells. There were 0–2 mitoses per HPF. Nests and cords of neoplastic squamous epithelial cells invaded the underlying dense collagenous connective tissue, focally extended into the underlying skeletal muscle, and often surrounded nerves. Neoplastic cells extended to the examined tissue margins; therefore complete excision could not be confirmed. In addition, perivascular and perineural inflammatory infiltrates were comprised of moderate-to-large numbers of lymphocytes and plasma cells. In some regions, there was sebaceous gland hyperplasia along the surface of the haired skin. Along the lateral margins of the neoplasm, the dermis also had evidence of solar elastosis (Fig. 4A). Within the superficial dermis, collagen bundles were separated by tangles of thickened, curled, lightly basophilic fibers that stained black with Verhoeff–van Gieson, consistent with elastin (Fig. 4B).
Although neoplastic cells were arranged in various patterns, including extensive sheets of vacuolated cells, there was overt squamous differentiation in several areas. These histologic findings, in conjunction with immunohistochemical and histochemical staining results, were consistent with a clear cell SCC. There was no true tubular or acinar formation, and vacuolated polygonal cells lacked cytoplasmic lipid, glycogen, and mucus, further excluding neoplasms of adnexal or adipocyte origin.5-7,9,15,16
In humans, clear cell SCC is a rare neoplastic entity in the skin, also referred to as hydropic SCC. The clear cell appearance is caused by hydropic degeneration of neoplastic squamous cells causing accumulation of intracellular fluid and not glycogen, lipid, or mucin.8,13 Human clear cell SCC is divided into 3 histologic types: keratinizing (type I), non-keratinizing (type II), and pleomorphic (type III). Type I is defined by sheets or islands of neoplastic clear cells with peripherally displaced nuclei that are often indistinguishable from adipocytes, as well as some cells that have more vacuolated cytoplasm and resemble sebaceous cells. 9 Additionally, there are foci of keratinization or keratin pearl formation. Type II is characterized by anastomosing cords of neoplastic clear cells with dense lymphoplasmacytic infiltrates and no keratinization. Type III demonstrates marked pleomorphism of neoplastic clear cells, vascular and perineural invasion, foci of squamous differentiation, and microcysts with acantholytic neoplastic cells. In all 3 types, there is no glycogen or mucin accumulation within clear cells; thus, clear cells are negative for PAS, mucicarmine, and alcian blue stains. Multi-cytokeratin AE1/AE3 and CK 7 are used to confirm epithelial origin of the neoplastic clear cells.8,10 Anti-epithelium 1 (AE1) is a monoclonal antibody that detects high molecular weight cytokeratins 10, 14, 15, 16 and low molecular weight CK 19; AE3, which is also a monoclonal antibody, detects high molecular weight CKs 1–6 and low molecular weight CKs 7 and 8. E-cadherin and CK 5/6 were used in our case because, in our experience, these markers are more widely conserved in carcinomas of veterinary species than the pancytokeratin cocktail used in our laboratory, which contains multi-cytokeratin AE1/AE3 and CK 8/18.The case presented herein is most consistent with a type I clear cell SCC.
To our knowledge, clear cell SCC has not been reported in animals. Variants of SCC with clear cells have been described in dogs, but have not been classified as clear cell SCC. A signet-ring SCC was described in the scrotum of one dog but, ultrastructurally, clear cells contained intracytoplasmic lipid vacuoles and scattered glycogen. 5 The uncommon canine clear cell adnexal carcinomas are also negative for oil red O but, in contrast to our case, they stain with PAS as a result of intracytoplasmic glycogen accumulation. 16 Other cutaneous neoplasms with clear cells that have been reported in dogs include clear cell basal cell carcinoma, clear cell hidradenocarcinoma, sebaceous carcinoma, balloon cell melanoma, liposarcoma, and metastatic renal cell carcinoma.6,7,16,18 None of these entities is characterized by squamous differentiation, and the clear cells in adnexal neoplasms contain intracytoplasmic glycogen. In horses, clear cell differentiation has only been reported in cutaneous basal cell tumors; however, those tumors lacked the squamous differentiation observed in our excisional biopsy. 15 Metastatic clear cell renal cell carcinoma has only been reported in the oral cavity of a horse, and was excluded as a differential based on squamous differentiation and clear association with the overlying epithelium. 12
SCCs are the most common neoplasm of the equine eye and adnexa. 4 SCC of the eyelid typically has an aggressive, locally invasive behavior, and carries a poor prognosis. 17 Metastasis is uncommon, but may occur late in the course of disease and most often to regional lymph nodes. We speculate that clear cell SCC will exhibit similar behavior, although this cannot be confirmed given loss of follow-up in our case. For this reason, accurate recognition of this rare entity is warranted in order that cases may be identified for subsequent study of their behavior. The prognosis of clear cell SCC in humans is also unclear given the scarcity of case reports. Of 7 cases of clear cell SCC reported in humans, all but 1 occurred on the head or neck of elderly white males who worked outdoors. One case was reported in a dark-skinned adult male who also worked outdoors. Of these reported cases, 1 patient died of metastatic disease, 1 died post-operatively, and 1 had recurrence after 3 mo.1,13
In both humans and animals, chronic ultraviolet (UV) light exposure plays an important etiologic role in the development of cutaneous SCC.2,3,9,10,13 Solar elastosis is a non-neoplastic UV-induced lesion commonly observed in poorly pigmented skin of horses and humans with or without SCC. It is defined as an accumulation of abnormal elastin in the dermis and is characterized histologically by aggregates of thick interwoven elastic fibers mixed with degenerate collagen within the superficial to mid-dermis, as observed in our case. The pathogenesis of elastin accumulation is unclear, but is often seen in poorly pigmented or non-pigmented skin with chronic UV exposure, or in conjunction with other solar-induced changes such as solar (actinic) keratosis, epidermal plaques, and SCC. In horses, solar-associated SCC has been reported in the conjunctiva, eyelid, and vulvar epithelium, and is morphologically similar to solar-induced SCC in humans, often exhibiting solar elastosis within or adjacent to the neoplasm. 2 In humans, chronic UV exposure is hypothesized to play a role in the development of clear cell SCC. All 7 case reports describe tumors developing on chronically sun-exposed areas (i.e., neck, scalp, face), although histologic evidence of solar elastosis has not been clearly described.1,8,13 The solar elastosis observed in our case parallels the observation in human and equine cutaneous SCC, and suggests that chronic UV exposure may play an etiologic role in the development of clear cell SCC in horses.
Footnotes
Acknowledgements
We thank the submitting veterinarian and the diagnostic receiving and histology staff from the Michigan State University Veterinary Diagnostic Laboratory for their assistance with this case.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
