Abstract
Liposarcomas are malignant tumors of adipocytes. The current report describes a liposarcoma in a 2.5-year-old, mixed-breed commercial sow that was detected during meat inspection. On gross examination, a firm, whitish, multinodular, 20 cm ×10 cm mass was observed in the perirenal area along with smaller nodules multifocally scattered within the renal parenchyma. Histological examination revealed an anaplastic sarcoma with clear intracytoplasmic lipidic vacuoles that were positive for Sudan black staining. Most of the cells were also positive for S100 and vimentin immunohistochemistry. Based on these results, a diagnosis of a perirenal liposarcoma was established. To the authors’ knowledge, no previous reports of liposarcomas in pigs have been published. This report also includes a review of the literature published on animal liposarcomas.
A 2.5-year-old commercial sow was slaughtered in an abattoir in Catalonia, Spain, and samples were submitted for examination. On questioning, the inspectors that submitted the sample reported no antemortem alterations. Regrettably, no further information could be obtained from the farm of origin. The macroscopic description referred from the veterinary inspector was that during the postmortem inspection, a multinodular, whitish mass, of firm consistency and measuring 20 cm × 10 cm, was observed in the right perirenal region. The cut section revealed different areas with grainy surface and small white nodules (Fig. 1A). Multiple nodules with the same macroscopic features were observed unilaterally scattered within the right kidney parenchyma (Fig. 1B).
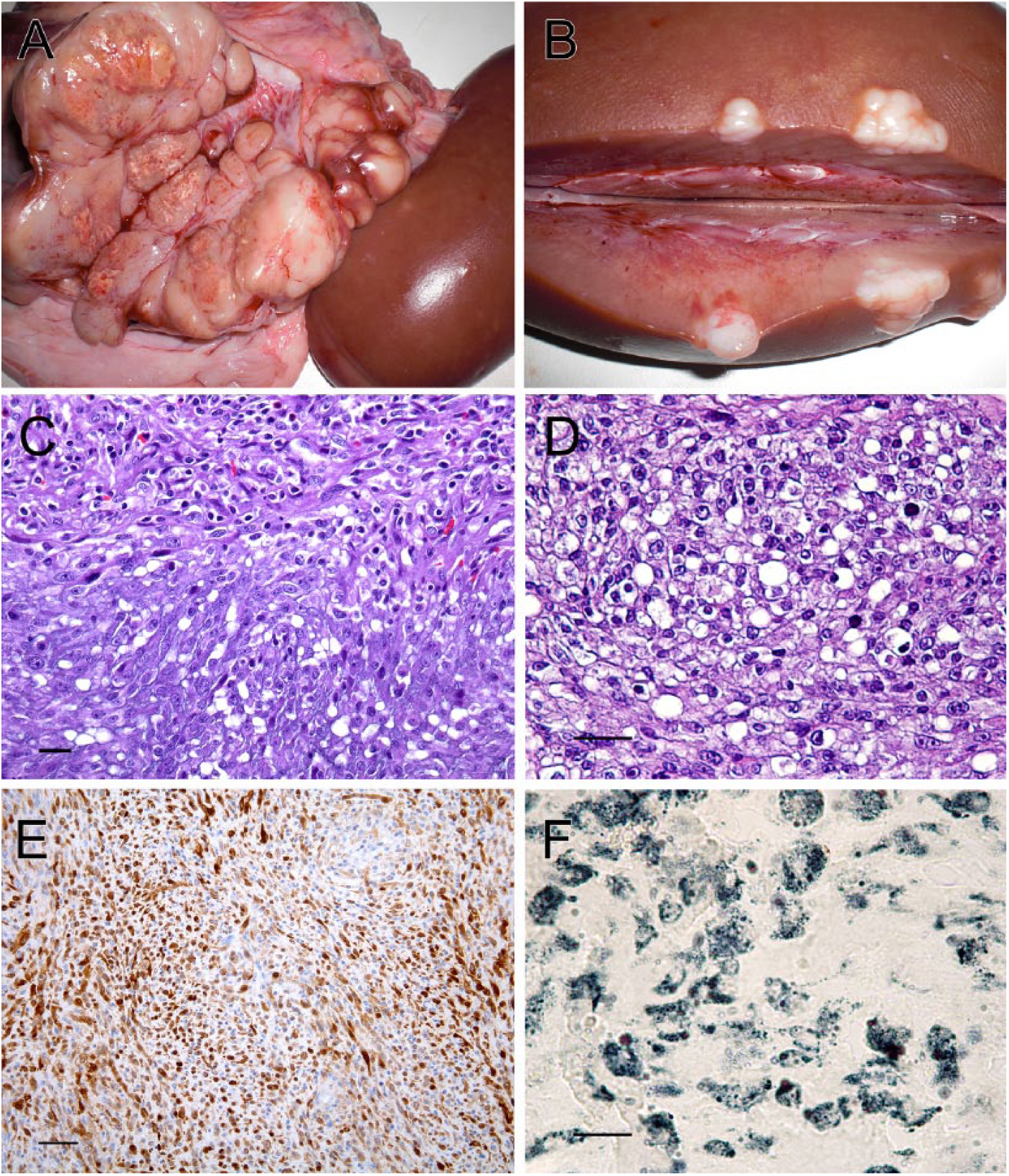
The kidney and different sections from the perirenal mass were collected, fixed in 10% formalin, and submitted to the Slaughterhouse Support Network (Servei de Suport a Escorxadors [SESC]) of the Center for Research in Animal Health (Centre de Recerca en Sanitat Animal [CReSA]; Barcelona, Spain) for diagnosis. The differential diagnosis by the meat inspectors included lymphosarcoma or an adrenal gland tumor. In the laboratory, samples were routinely processed for histology. Sections were processed for hematoxylin and eosin staining. Immunohistochemical staining (IHC) with antibodies against cluster of differentiation (CD)3 a and CD20 b were performed in order to rule out a possible lymphoid origin. Lysozyme c IHC was also performed to discard a macrophagic origin of the tumor and S100 d IHC to rule out a fibrosarcoma, which lacks immunoreactivity for this protein. 4 Finally, IHC against vimentin e was performed to confirm the mesenchymal origin of the neoplasm.
Briefly, heat-induced antigen retrieval was performed in an autostainer, f at 95°C with low pH target retrieval solution for 20 min before application of primary antibodies for CD3, CD20, S100, and vimentin. For the lysozyme IHC, slides were pretreated with proteinase K for 10 min at room temperature.
For IHC control purposes, porcine lymph node tissue for CD3 and CD20 was used. A cerebellum section was chosen as a control for S100 IHC, a tongue tissue section for vimentin IHC, and, for lysozyme IHC, skin with granulomatous dermatitis was used. Omission of the primary antibody in each slide served as nonspecific reaction control.
Sudan black staining g was performed on cryotome-obtained sections from formalin-fixed tissue, to identify the content of the cytoplasmic vacuoles. Regular adipose tissue was used as the technique control.
The mass was a densely cellular, infiltrative, and partially encapsulated neoplasm. Cells were arranged in compacted but disorganized bundles with abundant collagenous stroma, scant myxoid matrix, and a few blood vessels. Cells were medium sized and spindle shaped, with poorly defined borders. The cells had a moderate amount of fibrillar, pale, and eosinophilic cytoplasm. In some areas, cells had medium to large, well-defined, lipid intracytoplasmic vacuoles (Fig. 1C). Nuclei were paracentral, oval, and large with finely stippled chromatin and 1 or 2 eosinophilic nucleoli. Anisocytosis and anisokaryosis were evident, and some karyomegalic cells could be observed. Mitotic index was low (0–1 mitotic figures in 10 random fields at 400×). Mature eosinophils with some lymphocytes were scattered among the neoplastic cells. There were also multifocal large areas of necrosis and mineralization. In contrast, in the kidney, most neoplastic cells were polygonal and just a few had spindle shapes and intracytoplasmic lipid vacuoles. Foci of these tumor cells multifocally present within the renal cortex. Pleomorphism and mitotic index were higher (2–3 mitotic figures in 10 random fields at 400×). Based on the histological features of the perirenal mass, and a positive result against vimentin IHC on over 90% of the tumor cells, a diagnosis of anaplastic malignant mesenchymal cell tumor was established.
Differential diagnosis included histiocytic sarcoma, fibrosarcoma, and liposarcoma. Eighty percent of the cells, particularly those with spindle shapes, were intensely positive for S100 IHC within the cytoplasm and nuclei (Fig. 1E). The Sudan black–positive result confirmed the lipid-nature of the cytoplasmic vacuoles of the neoplastic cells (Fig. 1F). Hence, macroscopic and histological features of the perirenal mass, along with Sudan black and IHC, led to the diagnosis of a malignant liposarcoma. Although the cells of the kidney mass were initially suggestive of a round cell tumor, IHC of CD3, b CD20, c and lysozyme d were negative for the 3 markers in the kidney and perirenal tumors. These results ruled out the possibility of a round cell tumor and supported the hypothesis of a common mesenchymatous origin of both neoplastic lesions. The renal lesion was considered to be a metastasis from the primary liposarcoma. Based on the cellular morphology, the remitted masses were classified as a pleomorphic liposarcoma.
The most commonly reported swine tumors are those affecting young pigs, such as lymphosarcoma, embryonal nephroma, and melanoma. 15 This is because of the fact that a majority of pigs are slaughtered before reaching adulthood, when tumors often develop. 15 Based on abattoir surveys, lymphoma is the most frequent tumor of swine, commonly affecting 1-year-old or younger pigs.22,40 In a retrospective study in 63 pot-bellied pigs, the tumor incidence was summarized and a variety of neoplasms in pigs was described without any evidence of liposarcomas. The mean age of the tumor development in that study was 11.3 years. 39 There are very few reported cases of lipomas in pigs. 62
To the best of the authors’ knowledge, liposarcomas have not been reported in pigs. In the current report, we describe a liposarcoma that developed presumably from the perirenal fat, with metastasis to the renal parenchyma.
In most cases of liposarcomas reviewed in this article, IHC and histological (lipid) stainings have been used to reach the final diagnosis because histological examination revealed a poorly defined neoplastic proliferation.42,59 Such methods have also been used to confirm the microscopic diagnosis in some well-differentiated liposarcomas. 42 Frequently used markers are lipid stainings (such as oil red O) and vimentin and S100 IHC. All of these markers yield positive results, regardless of the subtype of liposarcoma and species in which they have been used.1,16,42,59,65 Although rare, this diagnosis should be included in the differential diagnosis of swine neoplasms.
Liposarcoma is a malignant tumor of adipocytes, first described by Rudolf Virchow in 1857. 48 Traditionally, great emphasis has been placed on the identification of lipoblasts for diagnosing liposarcomas, but their importance in some situations has been overemphasized as lipoblast-like cells may be seen in a variety of conditions. 67
Liposarcoma is among the most commonly described soft tissue sarcomas in adult human beings.10,11 The most recent World Health Organization (WHO) classification of soft tissue tumors recognizes 5 categories of liposarcomas: well-differentiated (which includes the adipocytic, sclerosing, and inflammatory subtypes), dedifferentiated liposarcoma, myxoid liposarcoma, round cell liposarcoma, and pleomorphic liposarcoma. 38 Although liposarcomas are divided into these subtypes, the categories most frequently used are well-differentiated liposarcoma, myxoid liposarcoma, and pleomorphic liposarcoma. 67 Well-differentiated liposarcomas are the most common form of reported malignant adipocytic neoplasms, accounting for approximately 40–45% of all liposarcomas followed by myxoid liposarcoma (30–35%).11,67 Pleomorphic liposarcoma is the least common malignant adipocytic neoplasm, accounting for approximately 5% of all cases. 11 The limited presence of cytoplasmatic lipid vacuoles may have led to misdiagnosis of liposarcomas as undifferentiated pleomorphic sarcoma. 41
In veterinary medicine, there is no accepted classification of liposarcomas, but based on cellular morphology and following the WHO classification, liposarcomas in animals are divided into 3 categories: well-differentiated, pleomorphic, and myxoid liposarcomas.3,22,38 In animals, liposarcomas are rare but can occur in many domestic and nondomestic species1,12,16,29,36,43,45,46,61 (Table 1). Liposarcoma has been described most commonly in dogs and, in this species, its incidence increases with age.3,13,22,31,66 The average age of affected dogs is 9–10 years.3,13,32,66 The etiology of canine liposarcomas is largely unknown although it has been associated with the presence of foreign bodies.34,63 In an 11-year-old dog, a liposarcoma in the forelimb was associated with a glass foreign body. 34 A low-grade liposarcoma was diagnosed in another 11-year-old dog with a subcutaneous mass located in the lateral region of the neck, at the site of a microchip implantation. 63 In cats, it has been associated with vaccination sites 21 and retrovirus infection even though it could not be proved that the tumor was caused by the virus. 57 The majority of liposarcomas are aggressive, locally invasive neoplasms. Location is unpredictable (Table 1). Recurrence is common but metastatic spread is rare except in cases with evident cellular anaplasia. In myxoid liposarcomas, metastatic rate can reach 60% and, in pleomorphic liposarcomas, between 30% and 50%. 11 In dogs, metastases have been reported mainly in lung, spleen, liver, kidney, and lymph nodes.3,17,38,65
Liposarcoma reports in the veterinary literature.*
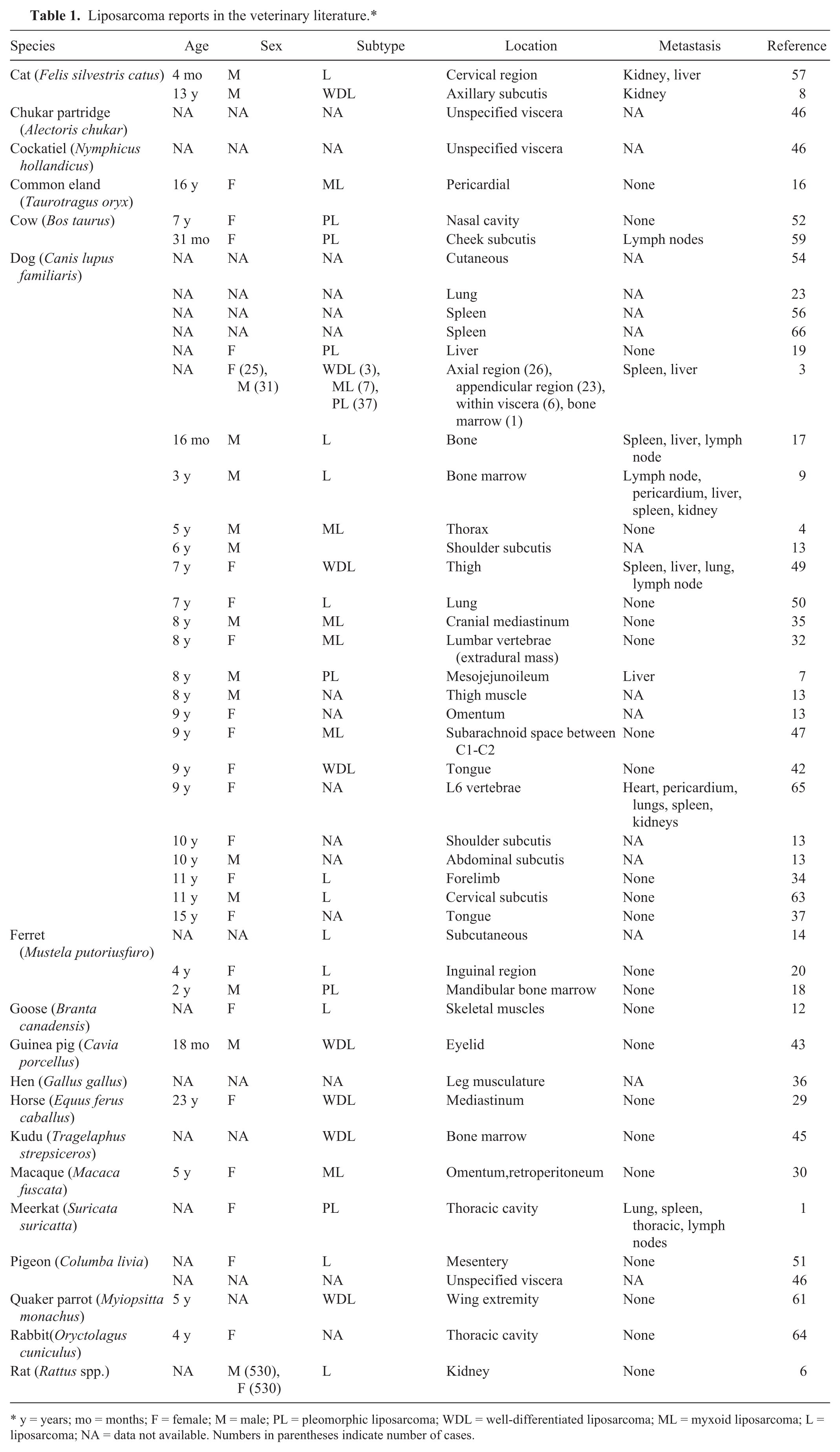
y = years; mo = months; F = female; M = male; PL = pleomorphic liposarcoma; WDL = well-differentiated liposarcoma; ML = myxoid liposarcoma; L = liposarcoma; NA = data not available. Numbers in parentheses indicate number of cases.
Well-differentiated variant
Considered the most common histologic subtype, 31 well-differentiated variant consists of multilobular, fairly well-circumscribed but unencapsulated neoplasms that arise from the subcutis.8,31 Cells can be round to polygonal and arranged in solid sheets. A moderate number of cells are well-differentiated adipocytes, with a single clear fat vacuole and peripheral nuclear displacement.22,49 Other cells have a central, round to oval nuclei and abundant cytoplasm that contains variably sized lipid droplets resembling large pleomorphic lipoblasts of varying maturity.22,45 Mitotic activity is low. The diagnosis in these cases is clear.8,22,31,42,45,49 This subtype must be differentiated from invasive lipomas (also named infiltrative lipomas), which are histologically similar but lack mitotic figures and show no evidence of anaplasia.24,28,54
Myxoid variant
The myxoid variant is an uncommon variant of liposarcoma, and is described as multilobular and nonencapsulated with poorly defined mass margins.4,30,31,35 This subtype is usually composed of a mixture of lipocytes, with a single large and clear cytoplasmic lipid vacuole compressing the nuclei, lipoblasts, and scattered spindle and stellate cells interspersed in a myxoid background with small number of collagen fibrils.4,35,47 A few multinucleated tumor cells can also be present.4,21,30,47 Anastomosing capillary vasculature is likewise present.31,35 Resembling myxosarcoma, this tumor is differentiated by the presence of cytoplasmic lipid-filled vacuoles.4,21,30,31,35,42,47
Pleomorphic liposarcoma
This variant has pleomorphic cells of variable size and shape, 52 and large bizarre multinucleated cells might be observed.7,31 Cells have abundant eosinophilic cytoplasm that may appear glassy or foamy. A few cells have intracytoplasmic distinct fat vacuoles.31,52 Nuclei also exhibit variability in size, shape, and chromatin pattern. Nuclear hyperchromatism is common, and single or multiple conspicuous nucleoli can be observed.7,52 Mitoses are frequent.7,22,52 This tumor mimics other pleomorphic mesenchymal malignancies, especially when they have scant lipoblasts. 67 Liposarcomas resemble a pleomorphic variant of histiocytic sarcoma but, in the case of liposarcomas, they lack significant collagenous stroma and spindle-cell population.1,7,22,31,42,52,59 Unlike histiocytic sarcoma, pleomorphic liposarcomas demonstrate focal labeling for S100 protein in their lipoblastic elements. 5 Even though there are many unclassified liposarcomas in the veterinary medicine literature, in a study including 56 liposarcoma reports in dogs, this was the most prevalent subtype. 3 In Table 1, a summary of the liposarcomas described in veterinary medicine literature is presented, including information on the animal species, age, sex, tumor site, and the occurrence of metastasis.
Regarding diagnosis, soft tissue sarcomas with a pleomorphic histological appearance can manifest similar immunoreactivity patterns. 16 Nevertheless, immunohistochemical characterization of the tumor is useful for definitive diagnosis. 42 The presence of vacuolated cells with positive vimentin and S100 immunostaining is suggestive of a liposarcoma.1,16,42,59,65 All pleomorphic dedifferentiated liposarcomas are intensely vimentin positive,1,59,65 approximately 70% are S100 positive, and just 30% of them are collagen type IV positive. Diagnostic intracytoplasmic fat vacuoles are usually present, but only in a small percentage of cells.22,59 The lack of cytokeratin labeling is useful in differentiating liposarcomas from other lipid-rich neoplasias, such as mesotheliomas.16,42
The histological staining reviewed in the literature of veterinary liposarcomas include oil red O,7,9,12,14,16,18,29,30,45,46,49,52,66 periodic acid–Schiff,1,34 osmium tetroxide,18,45,52 alcian blue, and toluidine.17,45 The immunohistochemical markers most frequently used were vimentin,1,14,16,42,43,47,59,52,65 S100,1,16,42,47,59,65 cytokeratin,1,16,42,43,47,50,52,59,65 CD3, 51 actin,14,51,65 and desmin.42,47,66
Liposarcomas can be diagnosed cytologically, by fine-needle aspiration, where dense aggregates of mesenchymal cells containing variable amount of lipid vacuoles can be recognized.4,20,35 Multinucleated cells may be present.35,44 In contrast, other authors consider cytological examination not suitable to reach a definitive diagnosis of liposarcoma, and limit this technique to support the choice between incisional or excisional biopsy. 58 Some studies suggest that oil red O staining may be an easy, inexpensive, and useful diagnostic tool for cytological diagnosis of canine liposarcoma and the differentiation from other mesenchymal neoplasms, 33 such as fibrosarcoma, undifferentiated sarcoma, and anaplastic carcinoma. 44
The first choice of treatment for human beings and animals is a wide surgical excision, the current established treatment for soft tissue sarcoma. 26 Although used in combination in some contexts to control recurrence, 7 the efficacy of radiation therapy and chemotherapy remains uncertain at this time. 26
Footnotes
Acknowledgements
The authors thank Agència de Salut Pública de Catalunya (ASPC), slaughterhouse veterinary meat inspectors for macroscopic pictures and case description, and the technical assistance of Blanca Pérez and Aida Neira of the Veterinary Pathology Diagnostic Service (SDPV-UAB). The authors thank also the Slaughterhouse Support Network (Servei de Suport a Escorxadors, SESC-CReSA), which is funded by ASPC of the Departament de Salut, Generalitat de Catalunya.
a.
CD3 (ref. A-0452), Dako Denmark A/S, Glostrup, Denmark.
b.
CD20 (ref. PAS-35313), Dako Denmark A/S, Glostrup, Denmark.
c.
Lysozyme (ref. A-0099), Dako Denmark A/S, Glostrup, Denmark.
d.
S100 (ref. Z0311), Dako Denmark A/S, Glostrup, Denmark.
e.
Vimentin (ref. M0725), Dako Denmark A/S, Glostrup, Denmark.
f.
PTLink autostainer, EnVision FLEX; Dako Denmark A/S, Glostrup, Denmark.
g.
Sudan black, Merck KGaA, Darmstadt, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
