Abstract
In the present study, the peripheral blood lymphocyte subset distribution was compared between healthy dogs and dogs with chronic gastrointestinal disease, dental and skin conditions, and cancer. The immunophenotype of the group with chronic gastrointestinal disease and the group with dental and skin conditions showed no statistically significant differences with other groups of healthy or diseased dogs. When compared with healthy dogs, animals with cancer showed significantly lower absolute values of T cytotoxic cells (CD3+, CD8+) and lymphocytes that express major histocompatibility complex (MHC) class II (MHC-II+) in peripheral blood. The results suggest that peripheral blood immunophenotype is mainly altered in dogs with cancer but not in other diseases. Further studies are required to evaluate the clinical relevance of these findings.
Keywords
Immunologic status has been investigated in dogs with various pathologic conditions. Specifically, some studies have focused their investigations on dogs with tumors.4–6,11,15,17 It has been described that tumor-bearing dogs have a higher number of leukocytes than normal dogs. 6 Absolute cell and relative percentage of cluster of differentiation (CD)3+ T cells, CD4+ T helper (Th) cells, CD8+ T cytotoxic (Tc) cells, and CD21+ B cells in dogs with tumors are notably decreased in comparison with healthy dogs. 17 In addition, tumor-bearing dogs show a significant increase of plasma interleukin 6 and transforming growth factor–beta activities. 6 Previous studies suggested that the Th type 1 lymphocyte–to–Th type 2 lymphocyte (Th1/Th2) balance is polarized to Th2 in dogs with malignant tumors.4,5 An increased population of regulatory T cells has also been reported in dogs with various cancers.5,11,15
Despite extensive research in human beings, few reports evaluating the role of different peripheral lymphocyte subsets in dogs with non-oncologic diseases are available. In the present study, the immunophenotype found in the peripheral blood lymphocytes of dogs with several different pathologic conditions is evaluated.
Fifty-two dogs, classified into 4 groups, were used in the current study. The groups included 1) dogs with chronic gastrointestinal disease (
Peripheral blood was obtained from all dogs by cephalic venipuncture and collected in tubes with ethylenediamine tetra-acetic acid for hematology and flow cytometry (FC). Whole blood hematologic parameters were analyzed employing a hematology analyzer, a following Principles of Good Laboratory Practice (http://www.oecd.org/chemicalsafety/testing/oecdseriesonprinciplesofgoodlaboratorypracticeglpandcompliancemonitoring.htm). Samples were processed for hematology and FC within 2 hr of blood drawn. All procedures were approved by the Animal Research Committee of the Complutense University of Madrid.
Multiparametric FC was performed to analyze different lymphocyte subsets (CD3+ T; CD3+, CD4+ Th; CD3+, CD8+ Tc; CD21+ B; those that express major histocompatibility complex [MHC] class II; and non-T, non-B lymphocytes) in each sample. Absolute values were calculated using white blood cell (WBC) analysis in combination with FC data. Monoclonal antibodies b to canine lymphocyte cell surface antigens included: anti-canine CD3 (clone CA17.2A12) conjugated with fluorescein isothiocyanate, anti-canine CD4 (clone YKIX302 9) conjugated with phycoerythrin, anti-canine CD8 (clone YCATE55.9) conjugated with AlexaFluor 647, and anti-canine CD21 (clone CA2.1D6) conjugated with PE.
The FC protocol employed was previously described 16 and standardized before the beginning of the study. The specificity of primary antibody binding was also evaluated using the isotype controls suggested by the manufacturer (data not shown). Following incubation with antibodies, erythrocytes were lysed with red blood cell lysing buffer. b Samples were analyzed using a flow cytometer c and commercial software. c Stained cells were analyzed with an electronic gate set by forward-angle versus side-angle light scatter properties to determine fluorescence in FL1, FL2, or FL4 on lymphocytes only. A total of 10,000 events were acquired for each preparation.
Data analysis was performed using analysis of variance and Bonferroni tests, considering a level of significance of
When analyzing sex and age, no significant differences were found between groups (
Differences in peripheral blood (cell counts of) leukocyte subsets between healthy dogs and dogs with chronic gastrointestinal disease, dental and skin conditions, and cancer (mean ± standard deviation).
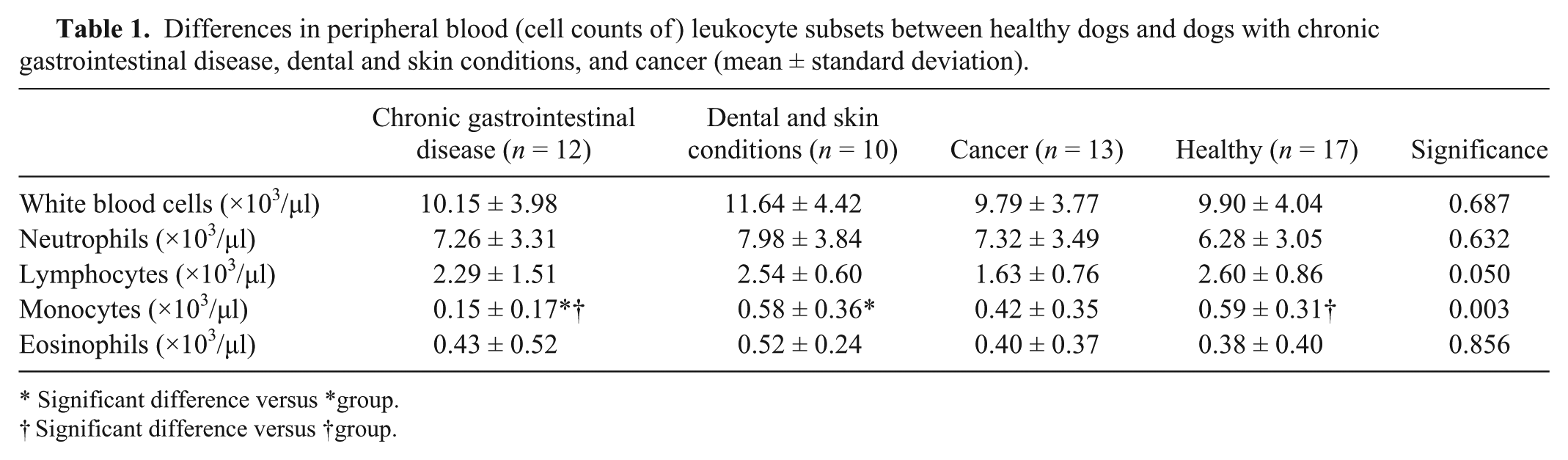
Significant difference versus *group.
Significant difference versus †group.
Results of lymphocyte subpopulations distribution in all groups are shown in Table 2. Significant differences between groups were observed in relation to absolute values of Tc cells (CD3+, CD8+;
Differences in peripheral blood lymphocyte subpopulations between healthy dogs and dogs with chronic gastrointestinal disease, dental and skin conditions, and cancer (mean ± standard deviation).*
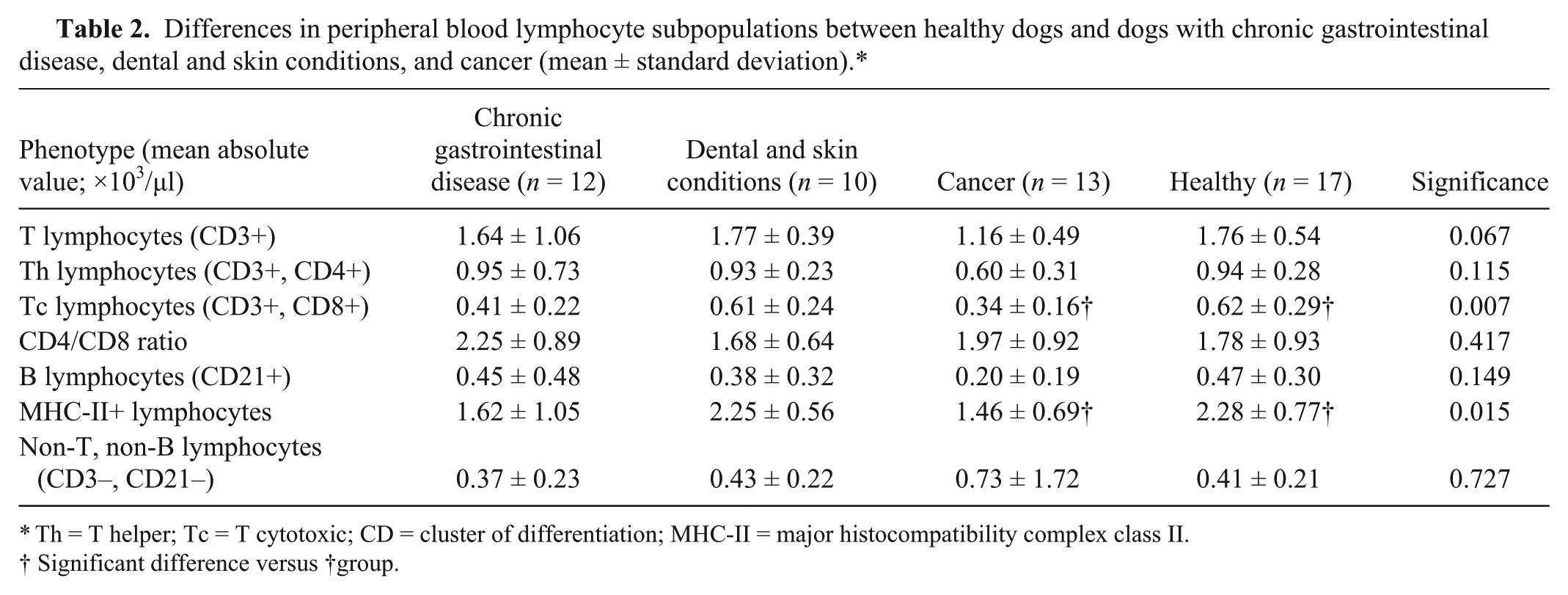
Th = T helper; Tc = T cytotoxic; CD = cluster of differentiation; MHC-II = major histocompatibility complex class II.
Significant difference versus †group.
Dental and skin conditions and chronic gastrointestinal groups showed no significant differences when compared with other groups of healthy or diseased dogs. When compared with healthy dogs, animals with cancer showed significantly lower absolute values of Tc cells and lymphocytes that express MHC-II in peripheral blood.
Despite extensive research in human beings, few studies evaluating the possible immunologic dysfunction presented in diseased dogs have been performed. Studies available describe immune status by means of peripheral lymphocyte subsets in dogs with cancer,6,11,14,17 hyperadrenocorticism, 10 leishmaniosis,1,9 or ehrlichiosis.8,16
The data presented in the current study showed that immunophenotype of peripheral blood lymphocytes in diseased dogs may be altered. Particularly, main differences occurred in dogs with cancer. Dogs with dental and skin conditions or gastrointestinal disease reported no differences in relation to the immunophenotype. This result should be cautiously interpreted taking into account the different diseases included in these groups together with the small number of evaluated animals.
White blood cell count was similar in all groups of dogs included in the study. The results contrast with the increase in WBC counts previously described in dogs with cancer.6,17 White blood cell counts in dogs with distant metastases are significantly higher than in dogs without metastases. 17 The increase in WBC count has been explained by a neutrophil leukocytosis that could be induced by infection, inflammation, and tissue necrosis associated with malignant neoplasms. 17 However, a decrease in neutrophil counts has also been described in some oncologic diseases. 14 In the present study, dogs with tumors had neutrophil counts within physiologic values, although a higher ratio of these cells was found in the tumor-bearing dogs when compared with healthy animals. This finding could suggest an activation of an early innate immune response or an inflammatory reaction at sites of tumor development and/or infection resulting from decreased immunity.
Absolute values of monocytes demonstrated a significant decrease in dogs with gastrointestinal disease compared to healthy dogs. However, it is worth keeping in mind that in all cases, mean values are within reference ranges. In dogs with IBD, nonspecific hematologic abnormalities are frequently described. 7 Specifically, monocytosis and neutrophilia reflecting chronic, active inflammation has been described in severe cases of IBD. 13
With regard to lymphocyte subsets, the absolute cell counts of Tc cells and lymphocytes that express MHC-II showed a significant decrease in dogs with tumors compared with healthy dogs. An age-related decline in absolute numbers of CD3+ T cells; CD3+, CD4+ Th cells; CD3+, CD8+ Tc cells; and CD21+ B cells has been reported.2,3,17 Therefore, it cannot be ruled out that some of these findings described in dogs with cancer could be associated with aging, although no significant differences were found when evaluating age differences between groups of dogs of the present study. However, it has been previously reported that all the lymphocyte subsets in dogs with tumors were notably decreased in comparison with age-matched controls. 17
The findings in the current study are consistent with earlier studies that have identified decreasing CD3+, CD4+, and CD8+ absolute values in dogs with cancer.6,11,17 The results suggest that oncologic dogs may be suffering from an immune-depressed state as evidenced by significant differences in lymphocyte subset populations. It was considered that the tumors in these dogs produced immunosuppressive substances and induced inflammatory reaction, thus suppressing cellular immunity (Th1) and activating humoral immunity (Th2). 6
The absolute number of lymphocytes that express MHC-II was significantly decreased in dogs with cancer compared with healthy dogs. Major histocompatibility complex class II expression on circulating lymphocytes has been less investigated than other lymphocyte surface molecules. An age-related decline of MHC-II expression by lymphocytes has been previously described. 12 It has been hypothesized that this phenomenon may extend to immunoregulatory or tolerance mechanisms acquired as a function of the continuous exposure to environmental stimuli. 12 In the current study, dogs with cancer were older than healthy dogs but not in a significant way, so it is unclear if this finding is related to aging or whether the diseases are implicated. In conclusion, peripheral leukocyte count and composition of lymphocyte phenotype were altered in dogs with cancer but not in other diseases. Further studies are required to evaluate the clinical relevance of these findings.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
a.
ADVIA 120, Siemens SA, Madrid, Spain.
b.
Monoclonal antibodies (see text), Erythrolyse red blood cell lysing buffer; AbD Serotec, Oxford, United Kingdom.
c.
FACSCalibur flow cytometer, Cell-Quest software; BD Biosciences, San Jose, CA.
d.
Statgraphics software (Centurion XVI version), StatPoint Technologies Inc., Warrenton, VA.
