Abstract
A 3-day-old Thoroughbred colt was originally presented for treatment of neonatal isoerythrolysis, which was treated with a blood transfusion. However, persistent neutropenia was observed despite the absence of detectable infection. Subsequently, a granulocyte agglutination test was performed by incubating the colt’s neutrophils with the mare’s serum; results were positive, leading to a clinical diagnosis of alloimmune neonatal neutropenia. The diagnosis was further supported via flow cytometric analysis. The colt was hospitalized and treated prophylactically with antimicrobials and 4 separate doses of recombinant human granulocyte colony-stimulating factor (rhG-CSF; 1.4–3.5 µg/kg, subcutaneously) in attempts to maintain the neutrophil count within reference intervals over a 4-week period. The colt’s neutrophil count increased after administration of rhG-CSF and eventually stabilized within reference intervals by day 20. The colt maintained normal neutrophil counts after discharge and was reportedly healthy at 6 months of age. Alloimmune neonatal neutropenia should be considered in foals with persistent neutropenia in the absence of infection. Alloimmune neonatal neutropenia can be treated with prophylactic antimicrobials combined with rhG-CSF with a favorable outcome.
A 3-day-old, 59-kg Thoroughbred colt was presented to the Lloyd Veterinary Medical Center (Ames, Iowa) for suspected neonatal isoerythrolysis (NI). The colt’s dam was a 5-year-old primiparous mare without reported medical disorders during gestation or parturition. Upon initial examination, the colt was quiet and lethargic but still responsive, with a rectal temperature of 38.0°C, heart rate of 160 beats/min, and respiratory rate of 80 breaths/min. Marked jaundice of the mucous membranes and sclera was observed. Test results of a hematocrit of 11%, positive direct Coombs test and positive minor cross-match between the colt’s red cells and mare’s plasma provided a clinical diagnosis of NI. Two separate blood samples were collected under strict aseptic conditions for bacterial culture to assist in ruling out an infectious process. Other relevant clinicopathologic findings included leukopenia (3.51 × 103 cells/µl; reference [ref.] interval: 5.1–10.1 × 103 cells/µl) characterized by a marked neutropenia (0.95 × 103 cells/µl; ref. interval: 3.21–8.58 × 103 cells/µl) and hyperbilirubinemia (5.9 mg/dl; ref. interval: 0.5–3.9 mg/dl). Agglutination of red cells was not observed on the blood smear. After identification of an appropriate donor via major and minor cross-match, a blood transfusion consisting of 1.5 liters of packed red cells was performed over 6 hr; no adverse reactions were observed. Administration of amikacin (25 mg/kg, intravenously [IV], every 24 hr) and ceftiofur (5 mg/kg, IV, every 12 hr) also was initiated. The colt rapidly appeared much brighter and energetic with the hematocrit rising to 19% shortly after blood transfusion was complete. The hematocrit slowly increased during hospitalization and was 29% at the time of discharge on day 30 (Fig. 1). Blood typing of the mare identified the Aa, Ca, Qa, Qb, and Qc antigens on maternal red blood cells; Ca, Ka, Qa, Qb, and Qc antigens were identified on the colt’s red blood cells, suggesting the Ka erythrocyte antigen as the cause of NI in this colt.
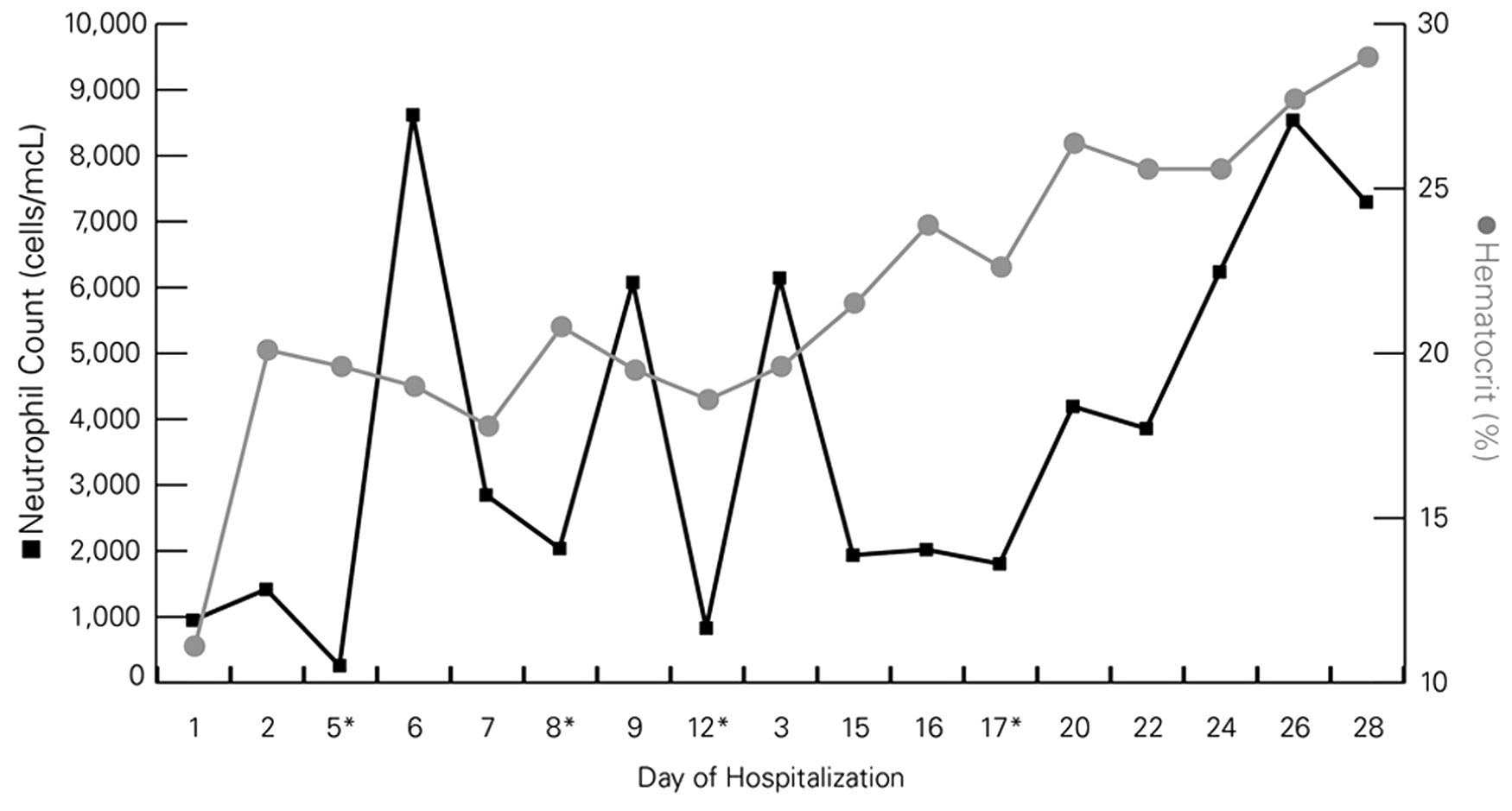
Serial measurements of neutrophil count and hematocrit in a foal with alloimmune neonatal neutropenia and neonatal isoerythrolysis over 30 days of hospitalization. Asterisk (*) indicates administration of recombinant human granulocyte colony-stimulating factor on days 5, 8, 12, and 17.
Despite the gradually increasing hematocrit, leukopenia characterized by marked neutropenia was identified on both automated a and manual evaluation of blood smears during the initial 5 days of hospitalization (Fig. 1). A thorough diagnostic investigation to identify a possible source of infection that could result in persistent neutropenia was conducted including repeated physical examinations and thoracic and abdominal ultrasonography. The colt was consistently bright and alert and maintained a normal body temperature; no abnormal findings were observed via ultrasonography, and blood cultures did not yield bacterial growth. Because an obvious source of infection could not be identified in conjunction with continued and profound neutropenia along with the absence of immature or toxic neutrophils, an immune-mediated neutropenia was considered. On day 5 of hospitalization, 180 ml of the mare’s and colt’s blood was collected separately, using acid–citrate–dextrose as an anticoagulant, to perform a granulocyte agglutination test (GAT). To perform this test, serum was harvested from the mare, colt, and a healthy adult control horse. A white cell suspension of polymorphonuclear cells (PMNs) was prepared from each of the 3 samples by diluting the individual blood samples with an equal volume of phosphate buffered saline (PBS) and centrifugation at 750 × g for 20 min as previously reported 16 ; plasma and buffy coats from each animal were aspirated and frozen. Subsequently, erythrocytes were lysed with 2 volumes of phosphate buffered distilled water for 1 min and then the PMN cell suspension was restored to isotonicity using phosphate buffered sodium chloride (2.7%). The PMNs were recovered from the lysed and restored erythrocyte fraction by centrifugation at 500 × g for 10 min followed by 3 washes with PBS. Cell pellets were resuspended in PBS to a final concentration of 10 × 106/ml. To document the kinetics of PMN agglutination, serial dilutions (1:2, 1:4, and 1:8) of the final foal PMN suspension (100 μl, final volume) and 100 μl of serum from the mare or control horse were made. Two additional sets of each dilution were made and incubated at 37°C and examined grossly and microscopically for neutrophil agglutination at 15 min and 30 min. In addition, 100 µl of undiluted foal PMNs were combined with 50 µl of serum from the mare or control horse in glass tubes and examined grossly and microscopically for neutrophil agglutination immediately following gentle mixing (Table 1). Agglutination was subjectively graded according to a previously published grading scale: no agglutination (0); microscopically positive; 1+ (small macroscopic agglutinates); 2+ (medium-sized macroscopic agglutinates); 3+ (several large macroscopic agglutinates); or 4+ (one solid macroscopic aggregate). 7 Auto control using the PMNs and serum from both the foal and the mare was also examined. Agglutination of the foal’s PMNs with the mare’s serum was graded as a 1–2+ after a 15-min incubation and 2+ agglutination after 30 min compared with microscopic agglutination when the foal’s PMNs were incubated with serum from the control horse. A complete blood cell count (CBC) was performed on the mare to eliminate the possibility of a maternal autoimmune neutropenia; the neutrophil count was within reference intervals. Based on these results, a clinical diagnosis of alloimmune neonatal neutropenia (ANN) was established.
Results of the granulocyte agglutination test in a foal with alloimmune neonatal neutropenia.*
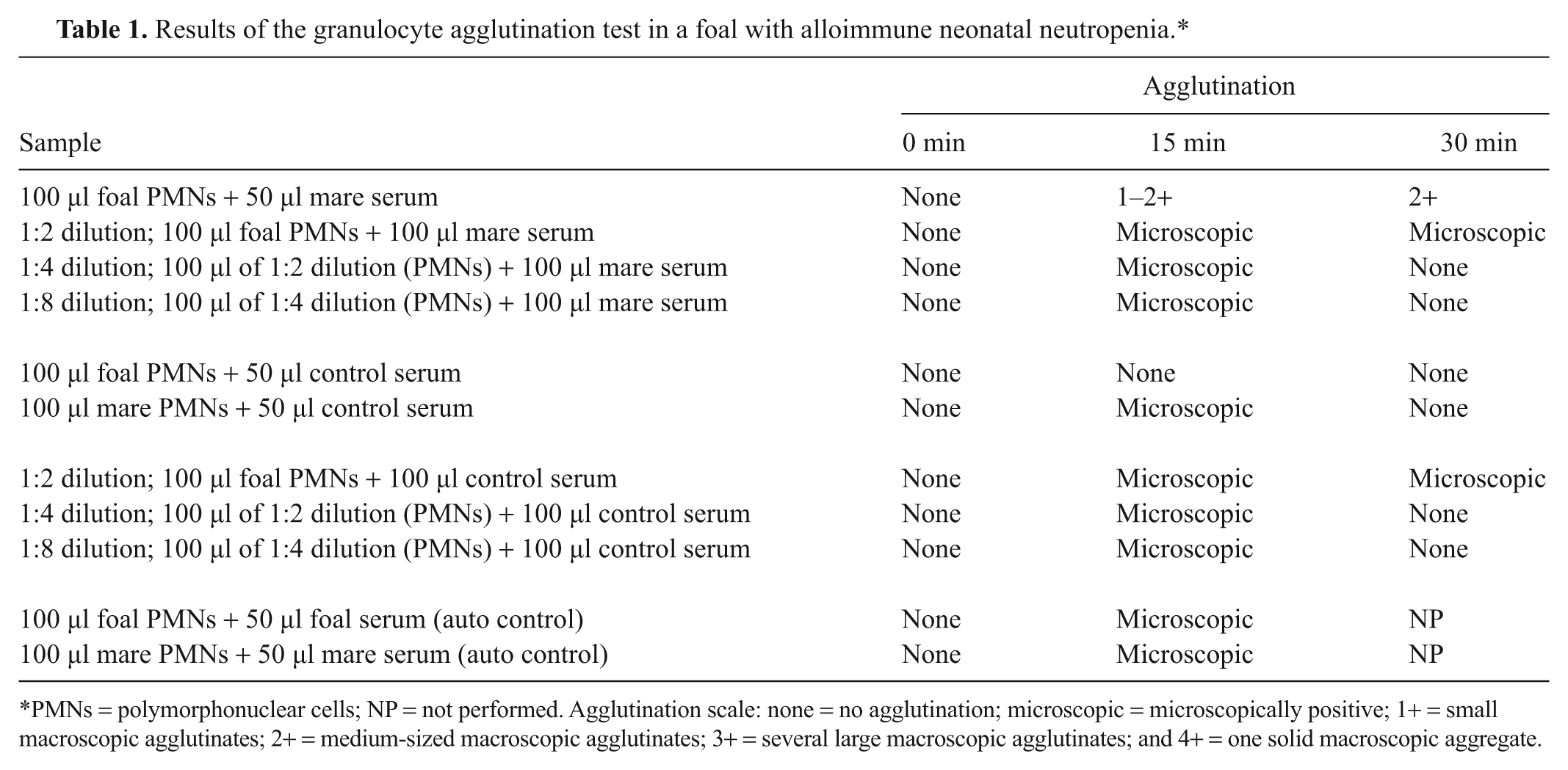
PMNs = polymorphonuclear cells; NP = not performed. Agglutination scale: none = no agglutination; microscopic = microscopically positive; 1+ = small macroscopic agglutinates; 2+ = medium-sized macroscopic agglutinates; 3+ = several large macroscopic agglutinates; and 4+ = one solid macroscopic aggregate.
In addition to the GAT, the PMNs, isolated as above from the mare and colt, were subjected to flow cytometric analysis. Aliquots of 100 µl/tube of isolated PMNs were incubated according to the following test conditions: each test sample contained 600 µl of PBS plus 300 µl (30% w/v) of mare or foal serum plus 100 µl (1 × 106) of either mare or foal PMNs. Samples were incubated for 30 min at 37°C and 5% CO2. After incubation, flow cytometry was performed using a 2-color flow method to verify the identity of mare and foal PMNs as granulocytes and to detect the presence of anti-neutrophil immunoglobulin (Ig)G. Cell handling and staining were performed as previously described in a 96-well, round-bottomed plate, with minor modification. 31 Antibodies utilized consisted of mouse monoclonal anti-cluster of differentiation (CD)172a, b which specifically stain equine monocytes and granulocytes, and cyanine (Cy)2-conjugated goat anti-horse IgG.c,34,36 R-phycoerythrin–labeled rabbit anti-mouse polyclonal antibodies d were used as the secondary detection reagent for CD172a staining. After final staining washes, cells were transferred to flow tubes and kept in the dark at 4°C until flow cytometric analysis. Samples were subsequently analyzed in a flow cytometer e using specific software f for data acquisition from 22,000 total events for each sample. Cell analysis software g was used to analyze flow data. Cells were displayed as forward-angle versus side-angle light scatter plots, and the live-cell neutrophil population was identified (Fig. 2). Among live cells, a decrease in foal granulocytes incubated with autologous (Fig. 2d) or mare (Fig. 2e) sera was apparent, suggesting maternally derived antibody-mediated lysis of foal PMNs. The mechanism resulting in the observed in vitro lysis of neutrophils was not examined further, but was suspected as being due to complement, a rare occurrence described in a 2009 report. 1 To determine whether maternal antibody could account for the clinically detectable neutropenia, foal PMNs were assayed for the presence of surface bound IgG (Fig. 3). As a control, mare PMNs were incubated with mare serum, and virtually no surface IgG was detected. Mare PMNs incubated with foal serum had little surface IgG, and foal PMNs incubated with autologous serum contained a slight increase in surface IgG. However, a log-fold increase in surface IgG of foal PMNs admixed with mare serum was apparent, consistent with maternally derived antibody as a cause of the foal’s neutropenia. Based on these findings, along with the positive GAT, a diagnosis of ANN was confirmed.
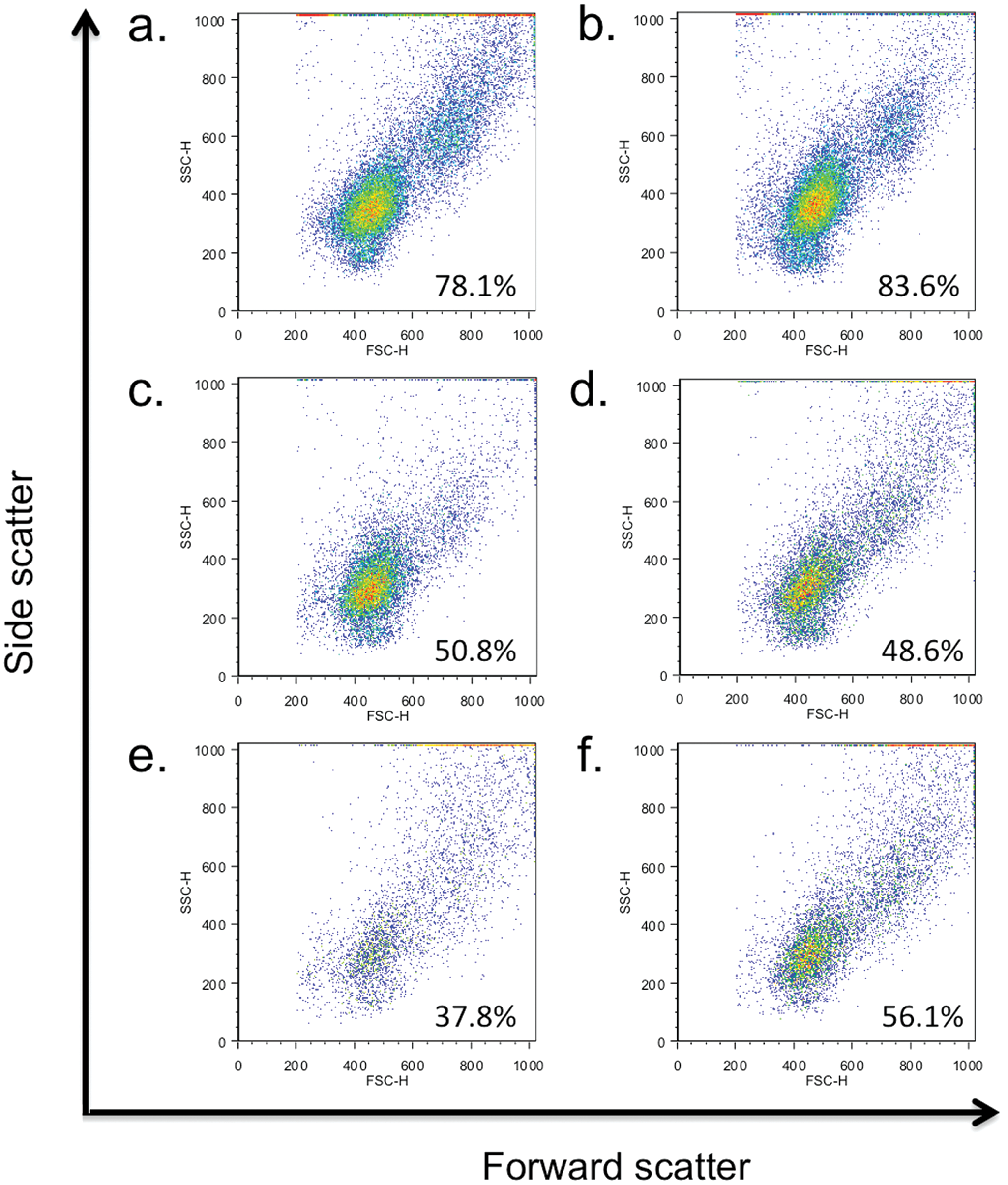
Forward-scatter versus side-scatter dot plots of live cells from mare and foal polymorphonuclear cells admixed with mare or foal serum, with percent live cells within gate: (
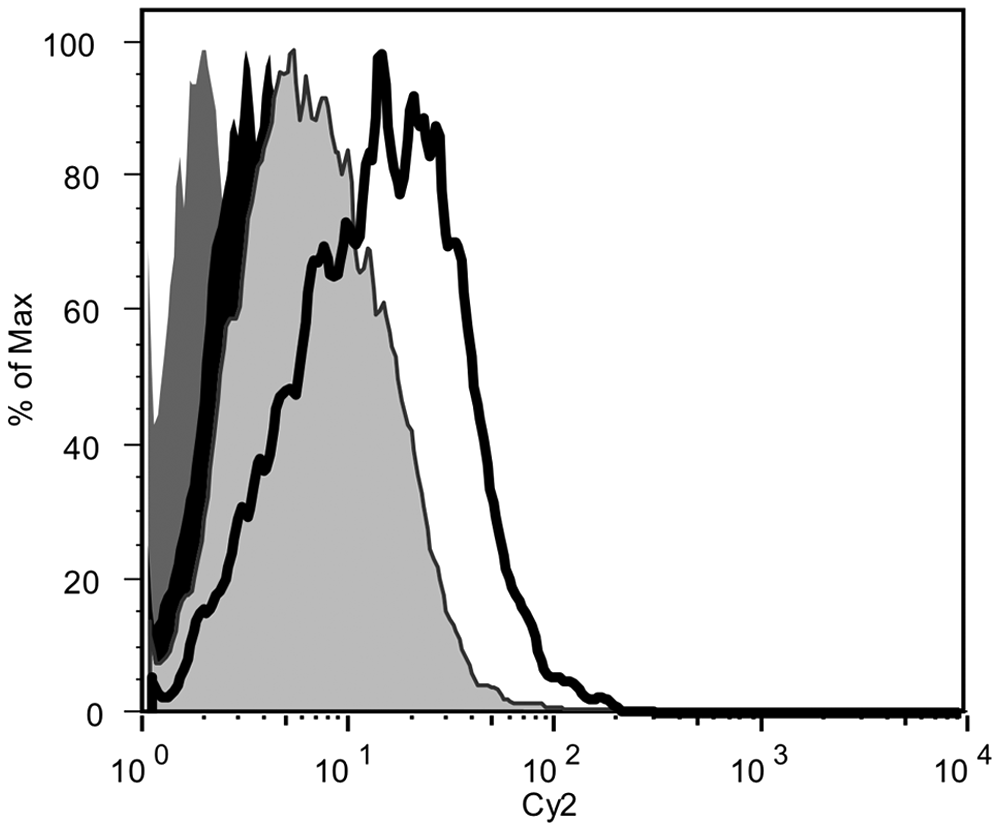
Histogram of neutrophils with surface-bound immunoglobulin (Ig)G as detected by mouse anti-equine IgG. Dam neutrophils admixed with dam serum (dark gray histogram); foal neutrophils alone (black histogram); foal neutrophils admixed with autologous serum (gray histogram); and foal neutrophils admixed with dam serum (open histogram).
Treatment at this point included continued administration of prophylactic antimicrobial therapy, as noted above, and the administration of recombinant human granulocyte colony-stimulating factor h (rhG-CSF; 3.5 µg/kg, subcutaneously [SC]) on day 5. An increase in total neutrophil count (8.62 × 103 cells/µl) was observed upon reevaluation 24 hr (day 6) after rhG-CSF administration (Fig. 1). However, gradually declining neutrophil counts were observed on days 7 and 8 on subsequent CBC evaluations. Therefore, rhG-CSF was again administered subcutaneously on days 8 (2.1 µg/kg), 12 (1.6 µg/kg), and 17 (1.4 µg/kg) to help maintain the neutrophil count within the reference interval. Repeated evaluations of CBC were used to determine when to administer rhG-CSF. The colt’s antimicrobial therapy was changed to trimethoprim–sulfamethoxazole (30 mg/kg, orally, every 12 hr) on day 10. After administration of rhG-CSF on day 17, the neutrophil count remained within the reference interval, suggesting that the immune-mediated destruction of neutrophils had diminished or was absent. During the hospitalization period, the colt was normothermic, gained weight daily, and remained alert and active. Antimicrobial therapy was discontinued on day 30, and the foal was discharged with all CBC parameters within reference intervals. The neutrophil count remained within reference interval on follow-up examinations 5, 10, and 22 days after discharge, and the colt was reportedly healthy at 6 months of age.
Neonatal isoerythrolysis is a common cause of anemia in neonatal foals and has been described thoroughly. 6 Interestingly, in the case presented herein, the mare was primiparous with no known history of previous administration of a blood transfusion. Thus, the inciting cause in which the mare developed anti-erythrocyte antibodies is unknown. Inferences have suggested that transplacental sensitization of the mare to fetal erythrocytes in early gestation is possible, but confirmation of this supposition remains difficult. In the colt in the present study, the Ka antigen was implicated as the causative erythrocyte antigen resulting in the development of NI which, to the authors’ knowledge, has not been previously reported in the foal. Although anti-erythrocyte antibodies against the Ka antigen have been documented in Standardbred mares, testing for antibodies against the Ka antigen in the mare in the current report was not performed. 3 Subsequent treatment and recovery from NI in this colt was uneventful, with a gradual increase in erythrocytes over the 30-day hospitalization. In contrast to the well-described syndrome of NI, the case presented herein is only the second reported description of ANN in a foal and provides further description of the disease process as well as additional diagnostic procedures that can be performed to establish the diagnosis of ANN. 15 While ANN is typically a self-limiting problem in infants and a presumably rare occurrence in foals, recognition of ANN in veterinary medicine is important to allow adequate prophylactic treatment to be implemented with the intent of avoiding infections.27,28 Infrequent reports have documented immune-mediated neutropenia in dogs and cats, whereas immune-mediated neutropenia is well described in human beings.8,9,27,28,30 In infants, 3 main disorders related to immune-mediated neonatal neutropenia have been documented and include ANN, neonatal autoimmune neutropenia, and autoimmune neutropenia of infancy. 27
The pathophysiology of ANN is analogous to NI but involves the foal’s neutrophils rather than erythrocytes. In such a clinical situation, the mare becomes immunized to a specific paternal granulocyte surface antigen that is absent on the mare’s neutrophils, but expressed on the foal’s neutrophils. Upon consumption of colostrum, IgG is absorbed and attaches to the foal’s neutrophils, which are subsequently destroyed by splenic macrophages.27,28 The severity of the neutropenia is partially governed by antibody titer along with the amount of IgG consumed. In infants, a wide variety of antigenic targets have been identified on neutrophils with approximately half of all cases of ANN in the United States being attributed to antibodies that bind to human neutrophil antigen (HNA)-1a, HNA-1b, or HNA-2a antigens 27 ; however, causative antigens remain unidentified in the other half of ANN cases. 18 Identification of equine neutrophil antigenic targets has not been adequately investigated although flow cytometry has been preliminarily explored for this purpose in horses.15,22 Other causes of immune-mediated neutropenia include neonatal autoimmune neutropenia whereby the mother has an autoimmune disease in which anti-neutrophil autoantibodies are produced, resulting in immune-mediated neutropenia in the mother; these autoantibodies are subsequently passively transferred to the fetus culminating in transient immune-mediated neutropenia in the neonate.20,27,28 In the case presented herein, the mare’s neutrophil count was within reference intervals making neonatal autoimmune neutropenia unlikely. In autoimmune neutropenia of infancy, the neonate produces cross-reacting antibodies that react with the neutrophils.27,28 The underlying mechanism remains unclear but has been associated with viral infections or administration of β-lactam antimicrobials.33,35 The β-lactam antibiotic ceftiofur was administered to the foal presented herein but autoimmune neutropenia of infancy seems implausible as the foal had neutropenia prior to initiation of ceftiofur administration, and a bacterial or viral infectious agent was not suspected. As with the NI in the current case, the means by which the primiparous mare was sensitized to exogenous neutrophil antigens remains unknown, and the authors can only speculate that either a previous undocumented blood transfusion was administered to the mare or transplacental contamination of fetal blood occurred during gestation, sensitizing the dam to paternally derived erythrocyte and neutrophil antigens.
In the only other case report of ANN in foals, severe leukopenia (white blood cell count: 1.2 × 103 cells/µl) characterized by neutropenia (0.62 × 103 cells/µl) was observed in an otherwise bright, alert, and responsive 12-hr old Arabian colt. 15 A fluorescent antibody test was used to document that the affected foal had 55% of the neutrophils coated with IgG as compared with <5% in an age-matched healthy foal. The procedure was repeated over time, and by 2 weeks of age, the affected foal had similar values as the control. Granulocyte colony-stimulating factor was also administered (6 µg/kg, SC, once) in that case, and the foal recovered without incident. 15 A clinical description of neonatal foals that develop ulcerative dermatitis, severe thrombocytopenia (0–30,000 platelets/µl), and mild neutropenia (500–1,800 neutrophils/µl) also has been reported. 29 Although the exact pathophysiologic mechanisms are unknown, there may be a causal relationship between colostral antibodies (or other factors within colostrum) and dermatitis and thrombocytopenia.
The incidence of ANN in infants is low (0.1–0.2% of pregnancies) but the diagnosis may be underdetected as many infants demonstrate no overt clinical signs, thus negating indications to evaluate the neutrophil count.12,38 In a human prospective study involving 247 cord blood samples, the frequency of ANN was higher (0.81% of pregnancies) suggesting that ANN occurs more frequently than clinically diagnosed. 38 However, the incidence is still relatively low and does not justify prospective screening in infants. Common clinical manifestations of ANN in symptomatic infants include skin infections, fever, omphalitis, otitis media, or pneumonia during the first few weeks of life.12,17,27,28 Sepsis is a less common but potential occurrence. 26 The overall mortality rate in ANN is approximately 5%. 27 The foal in the present case was examined for clinical manifestations of anemia rather than neutropenia, thus specific clinical signs associated with ANN in the foal are unknown. The authors suggest that foals with ANN may have minimal clinically recognizable manifestations. 15 Conversely, recurrent clinical signs of antimicrobial-responsive infections may be a possible indicator of ANN in foals. It would appear that the incidence of ANN in foals is relatively low, but ANN may not be identified in asymptomatic foals or foals with mild infections and therefore may be under-detected and -reported.
Diagnosis of ANN first depends on the presence of persistent neutropenia in the absence of evidence of increased consumption or decreased production of neutrophils. In the case presented herein, the foal was neutropenic for 5 days, and disease processes such as bacterial sepsis and equine herpesvirus infection were initially considered; however, the absence of documented infection based on normal physical examination findings, lack of fever or evidence of the systemic inflammatory response syndrome, negative blood culture results, and normal ultrasonographic findings within the thoracic and abdominal cavities made a serious infectious disease process less likely. Additional testing available to document ANN include the GAT and the granulocyte immunofluorescence test (GIFT). The GAT, used in the case presented herein, is performed by combining the mare’s serum (antibodies) with the foal’s neutrophils and the subsequent demonstration of neutrophil agglutination.11,12 In the current case, mild microscopic neutrophil agglutination with the control serum as well as the auto control samples suggested that minor neutrophil agglutination may transiently occur with sample handling or processing; however, the reaction between the foal’s neutrophils and dam’s serum was much more pronounced. In GIFT, a fluorescent dye–labeled anti-human antibody detects antibodies bound to granulocytes. 10 Unfortunately, neutrophil serological testing such as GIFT is extremely specialized and species-specific, and, to the authors’ knowledge, is not available for equine neutrophils. In the case presented herein, flow cytometry was performed to detect IgG attached to the foal’s neutrophils. Indeed, flow cytometry revealed that the serum from the foal contained IgG that specifically recognized and bound to the foal’s neutrophils. The authors speculate that the mare was the likely source of IgG, as an equivalent amount of the mare’s serum incubated with the foal’s neutrophils demonstrated even greater surface-bound IgG. Evaluation of bone marrow samples in infants with ANN are either normal or hypercellular with numerous metamyelocytes, band neutrophils, and mature segmented neutrophils.27,28 However, when evaluating infants that are suspected of having ANN, bone marrow biopsy is not indicated unless the neutropenia persists for more than 2–3 days, remains unresponsive to rhG-CSF therapy, is severe (<500 neutrophils/µl), or the etiology of neutropenia remains unclear. 27 Evaluation of bone marrow was not performed in the colt in the present study as bone marrow evaluation findings are not pathognomonic for ANN and the response to rhG-CSF was adequate in this case.
Alloimmune neonatal neutropenia is self-limiting in infants and typically resolves over several weeks to months as maternal antibodies diminish. Similarly, equine studies have demonstrated that the half-life of passively acquired maternal antibodies in foals is approximately 30–40 days.21,25 In the interim period, treatment of ANN primarily consists of administration of prophylactic antimicrobials and rhG-CSF. 15 Neonatal intensive care units commonly use a rhG-CSF dose of 5–10 µg/kg per day (IV or SC) for 3 consecutive days with additional doses being titrated to keep the neutrophil count over 1,000 cells/µl. 13 Experimental equine studies have evaluated canine recombinant G-CSF administered intramuscularly at 20 µg/kg on the day of birth followed by 10 µg/kg for 13 consecutive days to increase neutrophil counts significantly with the maximum mean neutrophil count of 49.71 × 103 cells/µl on day 10. 40 The initial dose of rhG-CSF administered in the present case was 3.5 µg/kg; subsequent doses were decreased (2.1, 1.6, 1.4 µg/kg). The lower doses were chosen based on the adequate response to the initial dose and the high cost of the medication (approximately $280 U.S. dollars per 300-µg vial). In addition, the frequency to which rhG-CSF had to be administered to the colt decreased as time elapsed, from every 3–4 days (doses 2 and 3) to 5 days (dose 4). This suggests that maternal anti-neutrophil antibodies began to diminish over the first 3 weeks of life. Administration of rhG-CSF has been reported to cause severe neutropenia in dogs after an initial neutrophilia during the first 20 days of treatment as a result of antibodies to rhG-CSF cross-reacting with endogenous G-CSF. 19 Neutropenia secondary to cross-reacting antibodies to endogenous G-CSF was considered in the foal presented here but was not observed. This response may have not occurred in the current case because only 4 doses were administered over a 17-day period; alternatively, rhG-CSF may not cross-react with equine G-CSF, as the horse’s immune response may not recognize rhG-CSF as foreign. In the foal in the current case, the neutrophil count was within reference intervals and considered stable when the foal was 24 days of age based on serial neutrophil counts.
Other therapeutic modalities that have been used in infants with ANN include administration of intravenous immunoglobulins (IVIG).12,39 The use of IVIG has been explored in dogs with immune-mediated disorders with variable results.4,5,32,37 The exact mechanism by which IVIG imparts beneficial effects is unknown but administration of IVIG may result in a nonspecific blockade of the reticuloendothelial system by the Fc portion of the infused IgG molecules, modulation of expression of Fc receptors, decrease in immunoglobulin production, or interference with activation of B and T cells.14,24 Reports in infants with ANN document a positive response rate to IVIG of approximately 50% but the use of this modality has not been reported in horses. 27 Recently, it has been demonstrated that the protective effect of IVIG in mice results from sialylation of the Fc fragment and subsequent binding to its cognate FcγRIIB receptor, Sign-R1 (DC-SIGN in human beings), which then leads to enhanced expression of this inhibitory receptor on effector macrophages resulting in decreased macrophage activation, and subsequently, decreased inflammation.2,23 IVIG was not administered to the foal in the current study based on the high cost of IVIG in this size foal (approximately $3,700 if given at a dose of 0.5 g/kg), the lack of knowledge in regard to the clinical utility in horses (i.e., whether sialylated human IgG binds to equine ortholog), and positive response to rhG-CSF. Corticosteroids are a final major treatment modality for immune-mediated neutropenia, primarily autoimmune neutropenia. Corticosteroid administration to infants with ANN has been reportedly less efficacious, is not currently recommended, and was not utilized in the case presented herein. 27
Alloimmune neonatal neutropenia is a rare disorder in foals and the current case represents only the second documented case of ANN in the foal and the first case in which ANN was observed in combination with NI. Also notable is the fact that the affected foal was from a primiparous mare. In retrospect, it would have been interesting to document if the same antigen were present on both red blood cells and neutrophils resulting in the anemia and neutropenia. Alloimmune neonatal neutropenia should be considered as a differential diagnosis in foals with persistent neutropenia that lack evidence of infection, in the presence or absence of NI.
Footnotes
a.
Advia 120 Hematology System, Siemens Healthcare Diagnostics Inc., Deerfield, IL.
b.
IgG1, VMRD Inc., Pullman, WA.
c.
Goat Anti-Horse IgG, Jackson ImmunoResearch, West Grove, PA.
d.
IgG, AbD Serotec, Raleigh, NC.
e.
FACScan, BD, San Jose, CA.
f.
CellQuest Software, BD, San Jose, CA.
g.
FlowJo software 9.0.1, TreeStar Inc., San Carlos, CA.
h.
Neupogen, Amgen Inc., Thousand Oaks, CA.
The authors declared that they had no conflicts of interests in their authorship and publication of this contribution.
The authors acknowledge partial support from NIH R15 HL103 488-01.
