Abstract
Ricin, a lectin from the castor bean plant (Ricinis communis), is considered one of the most potent plant toxins. Ingestion of masticated seeds results in high morbidity, with vomiting and watery to hemorrhagic diarrhea. The prognosis varies with the number of seeds ingested, the degree of mastication, individual susceptibility, and the delay in treatment. Low mortality restricts assessment of histologic lesions, and the literature on toxicologic analysis for ricin is limited. This report describes a fatal case of castor bean ingestion in a 12-week-old Mastiff puppy, with confirmation of ricin exposure through detection of the biomarker ricinine by liquid chromatography/mass spectrometry (LC/MS). Despite supportive therapy, the puppy died several hours after presentation for acute vomiting, diarrhea, and lethargy. At necropsy, a segment of jejunum and mesenteric lymph nodes were congested. When the owner reported the presence of castor beans in the dog's feces, selected formalin-fixed and unfixed tissues were submitted for diagnostic evaluation. Histopathologic findings included superficial necrotizing enteritis of the jejunum and occasional, random foci of coagulative necrosis in the liver. The alkaloid ricinine was detected in gastric content by using a newly developed LC/MS method. This confirmation of exposure is important in the diagnosis of ricin toxicosis, because ingestion of castor beans is not always fatal, histologic lesions are nonspecific, and the degree of mastication can influence the effective dose of ricin.
Ricin, from the castor bean plant (Ricinis communis), is considered one of the most potent plant toxins and is listed as a Category B bioterrorism agent by the Center for Disease Control and Prevention. 4 In humans, the estimated oral lethal dose of ricin may range from 1 mg/kg 11 up to 20 mg/kg body weight, or about 8 seeds. 2 Castor beans are cultivated to produce castor oil and are ubiquitous ornamental plants. The toxic lectin, released when the hard, tick-like seeds (Fig. 1) are cracked during mastication, inhibits protein synthesis, leading to cell death, and can affect a variety of domestic mammals, 1,3,9,12,14,18 poultry, 3,9,10 and humans. 2,3,15,17 Ricin poisoning may result in high morbidity with vomiting, watery-to-hemorrhagic diarrhea, and abdominal pain. 1,3,9,10,12,14,15 The severity of poisoning and the outcome depend on the number of seeds ingested, the degree of mastication, individual susceptibility, and delay in treatment. 1,2,9,11,14,18 Low mortality restricts assessment of histologic lesions, and toxicologic testing for ricin exposure has been limited, primarily based on the large size of the molecule. 11 Extraction of ricinine, an alkaloid with low molecular weight present in all parts of the castor bean plant, 5,6,13 provides a biomarker for exposure to castor bean extracts and, therefore, exposure to the toxic lectin, ricin. 5,11 This report describes the findings in a fatal case of castor bean ingestion in a puppy, with confirmation of ricin exposure through the detection of the biomarker ricinine by liquid chromatography/mass spectrometry (LC/MS).
A 12-week-old, 14.6 kg, female Mastiff puppy was presented with vomiting and diarrhea of less than 24 hours' duration. The puppy was lethargic, with pale mucous membranes and delayed capillary refill time. Fecal testing for parvovirus by using a commercial enzyme-linked immunosorbent assay (ELISA) kit a was negative, as was the direct fecal examination for parasites. Laboratory test results included a packed cell volume of 51% (reference range, 37%-55%), elevated alkaline phosphatase (424 units/ L; reference range, 46–337 units/L), increased blood urea nitrogen (54 mg/dl; reference range, 7–29 mg/dl), and hyperphosphatemia (13.6 mg/dl; reference range, 5.1-10.4 mg/dl). The puppy was hospitalized and treated with intravenous fluids, b oral activated charcoal gel, c intravenous dexamethasone sodium phosphate, d intramuscular enrofloxacin, e and subcutaneous vitamin K. f It remained depressed and lethargic, with hemorrhagic diarrhea, and died several hours after presentation. Subsequently, the owners reported finding castor beans, consumed from ornamental plants at the residence, in the dog's feces.
Postmortem examination revealed approximately 30–60 cm of dark red to purple jejunum, with swelling and congestion of corresponding mesenteric lymph nodes. Stomach, small intestine, large intestine, liver, spleen, kidney, and lymph node samples in 10% neutral buffered formalin and unfixed stomach and kidney samples were submitted to the Animal Disease Diagnostic Laboratory at Purdue University.

Seeds from castor bean plant, demonstrating mottled seed coat and “engorged tick” appearance. Bar = 1 cm.
Formalin-fixed tissues were paraffin embedded, sectioned at 4- to 5-μm thickness, and stained routinely with hematoxylin and eosin for microscopic examination. Jejunal mucosa was eroded with disruption of villous tip architecture, infiltration by degenerate leukocytes, and abundant nuclear debris (Fig. 2). Crypt epithelial cells were crowded with frequent mitotic figures. Increased numbers of eosinophils were in the deep lamina propria and submucosa. Smooth muscle cells of the tunica muscularis had frequent cytoplasmic vacuolization; aggregates of myocytes with pyknotic nuclei and hyalinized cytoplasm were also present. The mucosal and muscular layers had mild, multifocal hemorrhage. Occasional random foci of hepatic degeneration and necrosis ranged from hepatocytes with indistinct cytoplasmic vacuoles to those with hypereosinophilic cytoplasm and pyknotic nuclei surrounded by a few granulocytes (Fig. 3). Increased fibrous connective tissue surrounded some central veins; lymphatic vessels were dilated. Leukocytes, predominantly neutrophils, were distributed in increased numbers in portal tracts, around central veins, and within sinusoids. Splenic follicles had numerous lymphocytes with pyknotic or karyorrhectic nuclei and hypereosinophilic cytoplasm. Frequent apoptotic bodies were scattered through the mesenteric lymph node. Lymph-node follicles were hypocellular with pale germinal centers.
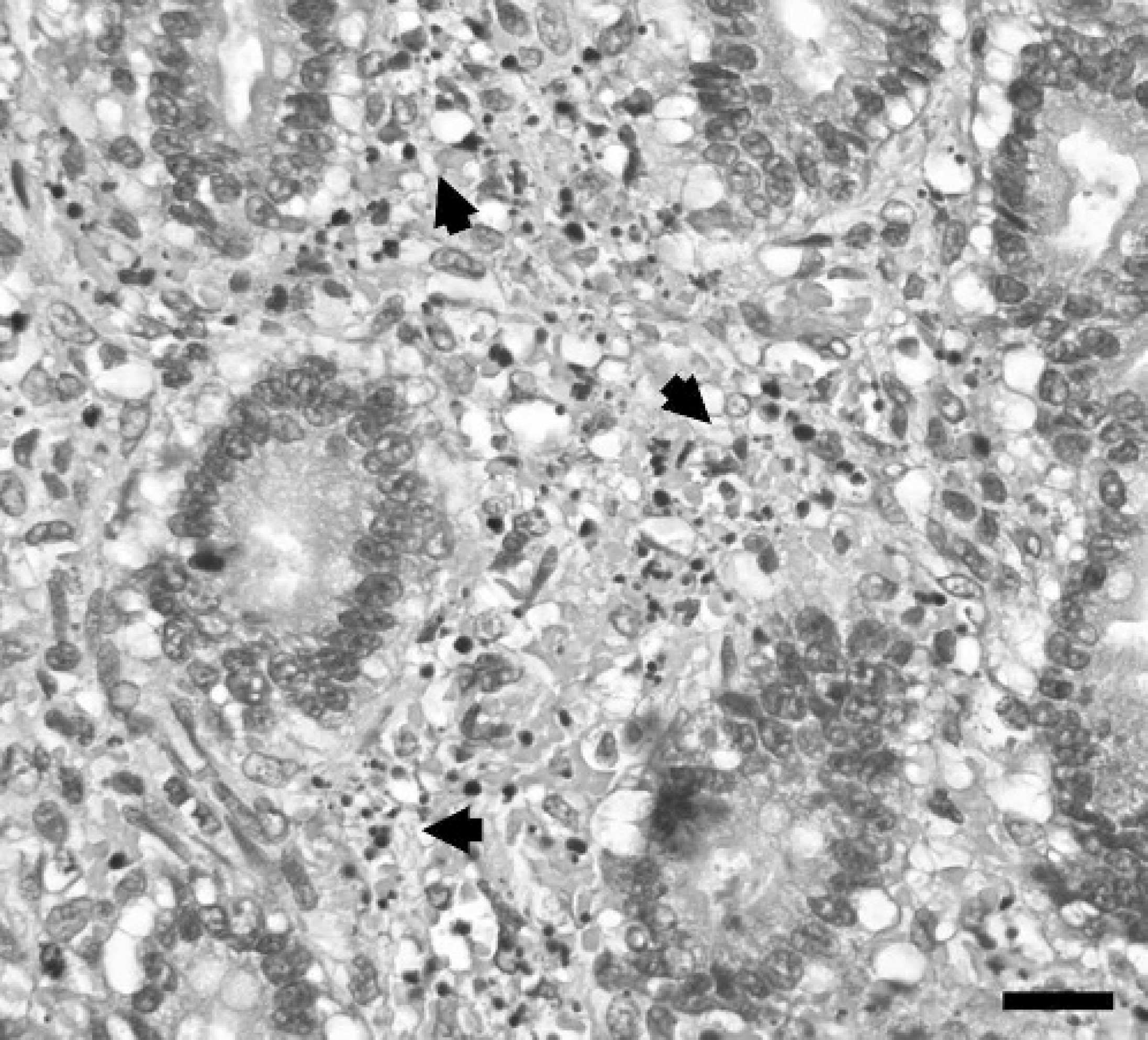
Photomicrograph of jejunal mucosa demonstrating numerous degenerated leukocytes and nuclear debris (arrowheads) in lamina propria. HE. Bar = 20 μm.
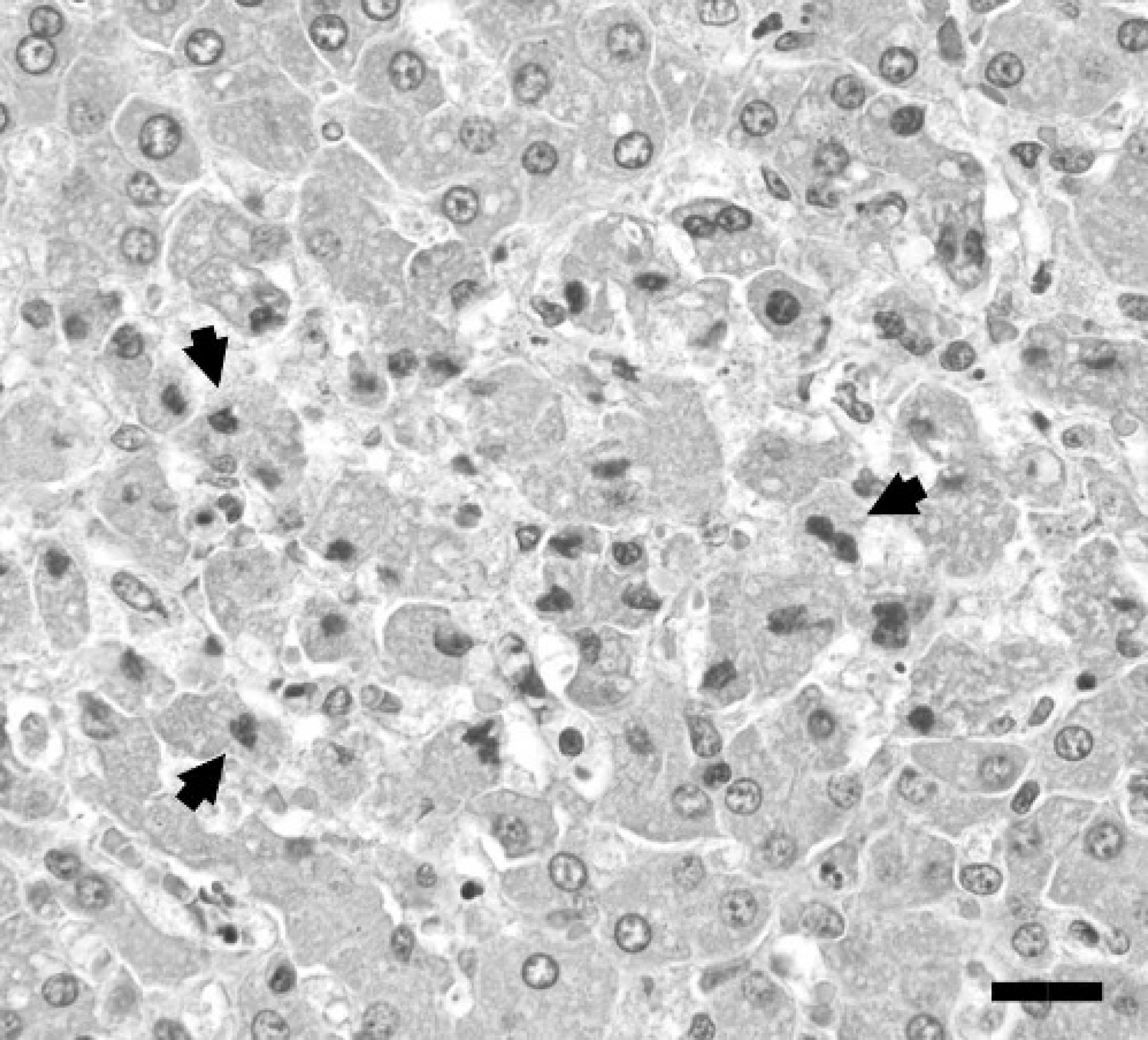
Photomicrograph of liver depicting a focus of hepatocellular necrosis with several pyknotic nuclei (arrowheads). HE. Bar = 20 μm.
Aerobic bacterial culture of lymph node and liver samples was negative, as was Salmonella culture on tetrathionate broth, when using mesenteric lymph node. A fluorescent antibody (FA) test, using polyclonal antibody, was negative for canine parvovirus on samples of intestine and spleen, and positive for canine coronavirus (intestinal samples only). No virus was isolated from intestinal or splenic samples. Immunohistochemistry (IHC) using anticoronaviral monoclonal antibody g was negative on jejunal sections.
To confirm castor bean toxicosis, a sample of stomach was submitted to the toxicology laboratory at the California Animal Health and Food Safety Laboratory System at the University of California, Davis, for evaluation of the presence of ricinine. Gastric contents were analyzed for ricinine by using LC/MS. In brief, the ricinine was extracted from gastric contents with methanol.
The resulting extract was purified by solid-phase extraction by using 3 ml Sep-pak VAC C18 cartridges. h , 13 > C6-Ricinine i was added to each extract for use as an internal standard. High-pressure liquid chromatography (HPLC) separation was performed by using an Agilent 1100 HPLC system equipped with a Synergi RP-Polar column. j The HPLC system was interfaced with an linear ion trap quadropole mass spectrometer k and ricinine was detected in full scan MS/MS mode. The method detection limit for ricinine in stomach content was estimated at 1 ng/g. Ricinine was positively identified in the gastric content, based on comparison of the retention time and mass spectrometry/ mass spectrometry (MS/MS) spectrum obtained from analysis of a purified ricinine standard (Fig. 4). l
On the basis of pathologic and toxicologic findings, a diagnosis of castor bean toxicosis was established. This case demonstrates intestinal and hepatic necrosis with lymphoid atrophy in a puppy that presented with fulminating gastrointestinal signs after castor bean consumption. The positive FA test for coronavirus in the face of negative IHC could be a false-positive test, reflecting the decreased specificity of the polyclonal FA versus the monoclonal antibody for IHC. However, if present, coronaviral damage to intestinal mucosa could have facilitated enteric absorption of ricin.
Ricin exposure was confirmed through LC/MS detection of ricinine in gastric content, because serum and urine samples were unavailable in this case. The two most common methods to detect ricin in blood or bodily fluids are radioimmunoassay (RIA) and ELISA. 5 These immunological assays are not suitable for testing gastrointestinal contents or other matrices for which the assays have not been validated. In addition, it is known that ricin is quickly metabolized before excretion and may be missed in samples such as blood or urine. 16 The development of a method to detect a biomarker of castor bean exposure, ricinine, allows for rapid, highly specific, and sensitive detection of this alkaloid. 5 Significant levels of ricinine have been detected via LC/MS in urine up to 48 hours after exposure. 11 Ricinine is a central nervous system stimulatory compound, most likely interacting at a specific gamma aminobutyric acid receptor (GABAA), that can lead to hyperactivity, seizures, and respiratory arrest at high doses. 7,8 The highest concentrations of ricinine are found in the leaves and the pericarp of the fruit, whereas ricin itself appears to be the primary compound in castor bean seeds. 6,13 The detection of ricinine in biological samples collected from suspected poisoning cases confirms exposure to any part of the castor bean plant. 5,11 In animals, a diagnosis of castor bean toxicosis is generally based on observing ingestion or emesis of seeds or plant components, 1,9,18 rather than by confirming exposure through toxicologic testing. Thus, many cases of castor bean toxicosis may be misdiagnosed or remain unidentified.
The findings in this case lead to the following conclusions. First, castor bean ingestion can cause histologically identifiable tissue necrosis not limited to the gastrointestinal tract. Second, significant illness and, in this case, death, may result from consumption of castor beans. Finally, LC/MS analysis of gastrointestinal content is an effective method to assay for ricinine. Reports of castor bean ingestion in humans and domestic animals vary in the description of systemic effects and prognosis. The question of whether ricin is poorly 1,12,15 or easily 18 absorbed from the gastrointestinal tract is unresolved, as is the pathogenesis and incidence of delayed hepatotoxicity and multiorgan failure. 1,2,12,15,17 A review of human cases of castor bean toxicosis listed a death rate of 1.9%, with most deaths documented in the late 19th or early 20th century. 17 That study challenged the seriousness of systemic effects after consumption of castor beans, indicating that hospitalization and prolonged monitoring were unnecessary. 17 In contrast, a child, hospitalized because of known ingestion of castor beans, demonstrated evidence of hepatotoxicity through alterations in clinicopathologic parameters at 72 hours after seed consumption. 15 A recent review of castor bean toxicosis in 98 dogs documented 76% morbidity and 9% mortality, the latter including euthanized animals. 1 Elevated hepatocellular enzymes in the serum were among the most common laboratory test abnormalities in clinically affected dogs. 1 A single case report of fatal ricin intoxication in a young adult Rottweiler dog after consumption of castor bean cake fertilizer described intestinal hemorrhage and hepatic degeneration, as well as cardiac hemorrhage and necrosis. 18 In contrast to the present case, the pattern of hepatic degeneration in the Rottweiler was described as centrilobular, 18 rather than random and multifocal, as illustrated here. Important differences between human and animal intoxication that may influence disease progression and prognosis include known exposure, early recognition of clinical signs, election to treat versus monitor, and aggressiveness of therapy. In addition, humans might be less inclined than animals to chew the hard seeds, failing to release significant amounts of ricin toxin. In the dog of this report, the time of castor bean consumption was unknown, as were the number of seeds ingested and the degree of the mastication. Because these factors are often undetermined in domestic animal castor bean toxicosis, monitoring of patients for systemic effects is warranted.
The diagnostic approach described here provided information crucial in the diagnosis of ricin poisoning in a dog. It is imperative to consider castor bean toxicosis when evaluating a dog that is presented with vomiting and diarrhea, even if there is no history of exposure to household plants or the outdoors. In suspected ricin poisoning cases, serum and urine samples should be collected at various time points, beginning as early after exposure as possible for use with ELISA or RIA for ricin (serum, urine) or with LC/MS for ricinine (urine). In postmortem presentations, gastrointestinal contents are suitable for ricinine testing to confirm exposure. The suspected plant or seed material or vomited gastrointestinal contents should also be saved for analysis. The procedure described here is especially suited to veterinary diagnostic laboratory situations for which rapid diagnosis of exposure to R. communis is necessary.
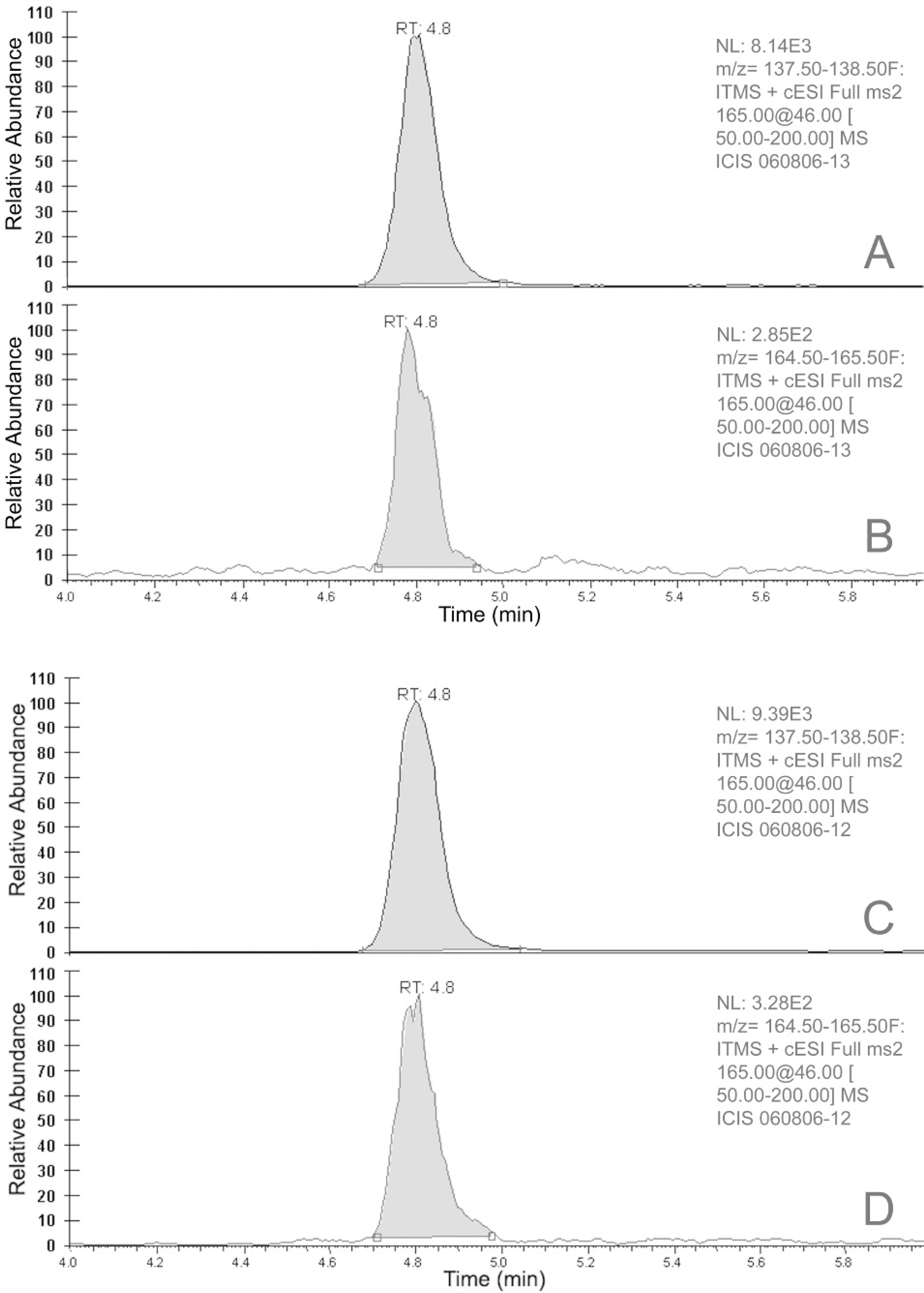
Selected product ion chromatograms from analysis of negative control matrix fortified with 1 ppb of ricinine (
Footnotes
a.
Idexx Laboratories, Westbrook, ME.
b.
Normosol R, Abbott Laboratories, Abbott Park, IL.
c.
UAA gel, Nich Marketers Inc, Columbus, OH.
d.
VedCo Inc, St. Joseph, MO.
e.
Baytril, Bayer Corporation, Shawnee, KS.
f.
Veda-K1, VedCo Inc, St. Joseph, MO.
g.
Custom Monoclonals International, West Sacramento, CA.
h.
Waters Corp, Milford, MA.
i.
Cambridge Isotope Labs, Andover, MA.
j.
Phenomenex, Torrance, CA.
k.
Thermo, San Jose, CA.
l.
Sigma, St. Louis, MO.
