Abstract
Objective
The objective of this article is to present a decision aid in the form of a flowchart, designed to assist psychiatrists in navigating the challenges of off-label medication use. This tool is believed to be the only one currently available that combines guidance from medical indemnity insurers, RANZCP guidelines, CATG frameworks, and relevant legislation to provide a practical guide for everyday psychiatric practice.
Conclusions
There are clinical, legal, ethical, and financial considerations to take into account each time an off-label prescription is ordered. There is no legal barrier to prevent psychiatrists from prescribing off-label in Australia and New Zealand. The medication should be utilised if it is in the best interests of the patient. However, practitioners must be able to defend the rationale for their off-label medication use with evidence of sound quality. Off-label use medication use can lead to improved patient outcomes and quality of life. On the other hand, poor practice in off-label prescribing can result in adverse events, patient harm, legal implications, and damage to professional reputation. By evaluating available evidence, considering patient characteristics, obtaining informed consent, and documenting the rationale for off-label use, clinical decision-making can be enhanced whilst mitigating the risks associated with off-label prescribing.
In the realm of medicine, the landscape of treatment options is constantly evolving, and healthcare providers often find themselves faced with complex decisions regarding off-label medication use. While the concept of prescribing drugs for purposes beyond their approved indications is not uncommon, it poses unique challenges for doctors, especially as little guidance to prescribing off-label is provided. Balancing the potential benefits and risks requires a careful and well-informed approach. In this ever-evolving medical landscape, a decision tree for prescribing and risk assessment in off-label medication use emerges as a valuable tool. This flowchart not only offers guidance to psychiatrists, but also serves as a legally modelled framework, drawing from a diverse range of sources such as medical indemnity insurers, regulatory guidelines, and relevant legislation. By navigating this intricate decision-making process, practitioners can enhance patient care while minimising potential pitfalls. This article delves into the intricacies of off-label medication use, offering insights into the evaluation of evidence, patient considerations, obtaining informed consent, and the crucial documentation of the rationale behind such prescribing decisions.
When would a psychiatrist wish to prescribe an off-label medication?
A drug is considered to be ‘off-label’ when it used for an indication not currently approved by the Therapeutic Goods Administration (TGA) or the New Zealand Medicines and Medical Devices Safety Authority, 1 or is prescribed at a higher than usual dose, via an alternative route of administration, or in a patient group that is not included in the product information.2,3 Nevertheless, a medication without TGA approval does not mean it cannot be prescribed as it is ineffective, contraindicated or disapproved. It may mean that it is not economically viable for the sponsor (usually the drug manufacturer seeking access to the marketplace) to seek approval, even if there may be supporting evidence for its use for the indication in question. 4 In fact, particularly in specialised settings such as psychiatric practice, it can prove clinically valuable to contemplate off-label prescriptions. This becomes especially pertinent when confronted with a scarcity of alternative treatment approaches, or when emerging evidence sheds new light on a drug’s efficacy, or use of the drug is widely considered common practice amongst peers. 5
The TGA does not regulate the clinical decisions made by medical practitioners; instead it serves to perform as a regulatory body supervising the safety, quality, and efficacy of therapeutic goods. 6 The decision to prescribe off-label is the decision of the prescriber. However, the TGA does enforce the legislation and regulations that govern the overall supply, availability, and use of therapeutic goods in Australia. 6
Doctors may consider off-label medication use in certain scenarios where approved treatments have proven ineffective or inadequate for a particular patient. The benefits of off-label prescribing lie in the potential to expand treatment options, offering patients alternative therapies that may address their unique needs. It can also allow doctors to stay at the forefront of medical advancements by exploring emerging evidence and innovative approaches. However, there are inherent pitfalls and challenges associated with off-label medication use – and to be medicolegally defensible, must reflect current evidence-based treatment. Accordingly, one major challenge is the limited evidence base supporting the efficacy and safety of novel treatments, as they may not have undergone rigorous testing for specific off-label indications. This lack of data raises potential risks, including adverse reactions and unforeseen drug interactions. Medicine prescription accounts for the greatest proportion of preventable harm in all healthcare. 7 The Pharmaceutical Society of Australia has released data indicating that over 250,000 hospital admissions per annum are the result of medication-related problems, along with an additional 400,000 annual emergency department presentations, of which 50% are avoidable. 8
For obvious ethical reasons, the act of prescribing an off-label medication is not the opportunity to learn about the experience of a newly encountered medication on a chosen patient; it is a psychiatrist’s (or any medical practitioner’s) duty to ensure that they are both knowledgeable and capable in the area they chose to manage. This extends to being aware of gaps in their knowledge, such as with prescribing a new medication, and their experience with this medication to date. 9 This can become ethically challenging when a patient requests a specific medication, often due to media publicity, placing the practitioner in the position of needing to balance the needs of the individual patient with established standards of care, whilst being pressed by external influences. Being unaware of the mechanisms of a new drug being prescribed, its addictive properties, or other adverse effects due to unfamiliarity is not a defence.
Given the complexity, issues related to informed consent and patient understanding of the off-label nature of the prescribed medication can arise. Cost considerations also require input, as off-label use may not have insurance coverage or shall likely result in higher out-of-pocket expenses for patients. Medications with punitive pricing or which are difficult to source are more likely to lead to non-compliance than those which are less expensive or subsidised by the PBS. 10 These complexities highlight the need for a comprehensive decision tree and risk assessment framework to guide practitioners through the process of off-label prescribing, mitigating potential problems, while maximising the benefits for patient care.
Prescribing drugs of dependence involves their own unique risk assessment process which is not thoroughly explored in this article. Practitioners should be aware of drug-seeking behaviour protocols.
Considerations when prescribing off-label
In the realm of off-label medication use, doctors face complex decisions that require careful consideration of various factors. Psychiatrists should be aware of all of the treatment options available for a condition they are currently treating, as well as the implications of employing drugs outside of their usual scope of practice. Accountable prescribing requires a commitment to evidence-based practice, the use of medicines with proven effectiveness and the avoidance of medicines when they do not help or cause harm. 11
Practitioners must evaluate the available evidence, assess patient characteristics, obtain informed consent, and document the rationale behind their off-label prescribing decisions. 12 To provide a structured approach, a decision tree serves as a valuable tool, guiding practitioners through these considerations and assisting to facilitate safe and effective off-label prescribing. The flowchart takes into account the Royal Australian and New Zealand College of Psychiatrists (RANZCP) clinical guideline on off-label prescribing, 13 the Guiding Principles Framework from the Council of Therapeutic Advisory Groups (CATAG) 4 and the consensus amongst medical professional indemnity insurers operating in Australia and New Zealand.11,14–16
The CATAG published seven guiding principles to follow,
4
including: 1. Consider the off-label use of a medicine only when all other options, including medicines approved by the TGA, are unavailable, exhausted, not tolerated or unsuitable 2. Use high-quality evidence to determine appropriateness of off-label medicine use 3. Involve the patient/carer in shared decision-making when recommending an off-label medicine 4. Consult the Drug and Therapeutics Committee when prescribing an off-label medicine, except when the use of a medicine off-label is considered routine 5. Ensure appropriate information is available at all steps of the medicines management pathway 6. Monitor outcomes, effectiveness, and adverse events 7. Consider liability and accountability when using medicines off-label (Figure 1). A guided framework for assessing off-label medication use in Psychiatry.
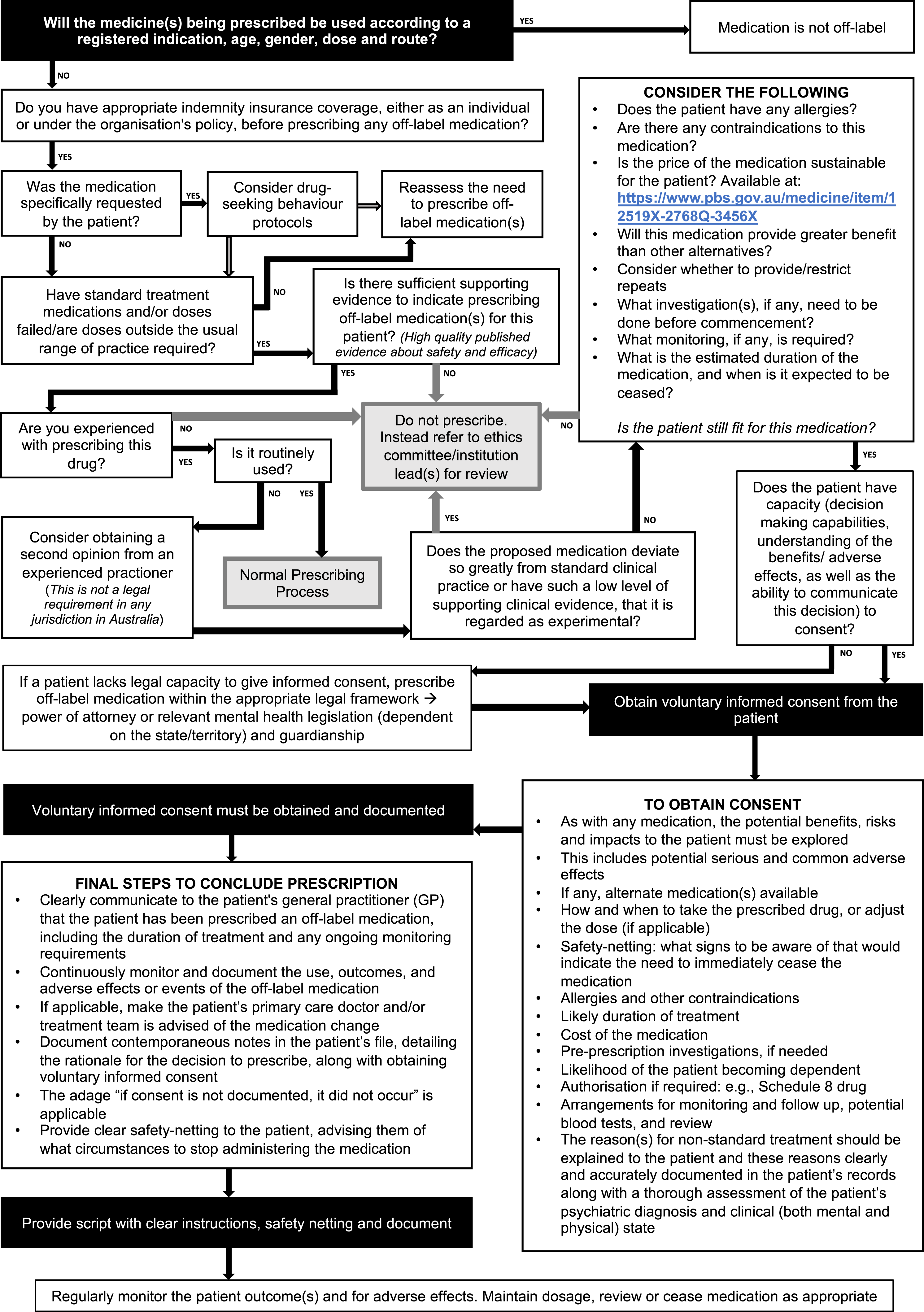
The practitioner is obliged to keep clear and contemporaneous records regarding their assessment of the patient, which includes: • Management plan • Documented informed consent, including risks and benefits explained to the patient • Notes and outcomes on any ongoing assessment and consultation of the patient • The patient notes must be legible (if not typed) • Satisfactory information to identify the patient, their diagnosis and detail supporting information justifying the diagnosis and treatment.
The notes should comprise requisite detail to allow another practitioner to assume continuity of the patient’s care if required. 17
In regards to finding evidence to support a prescription, sources may include (not an exhaustive list): • Decisions of competent regulatory bodies in other countries • Secondary or summarised sources of high-quality research evidence • Evidence-based therapeutic guidelines • Primary sources of high-quality research evidence, published in peer-reviewed literature, such as speciality-specific journals.
4
The decision to prescribe must be well-informed.
Irrespective of this, the commonly held perception within the medical profession is that if prescribing off-label, the practioner and organisation they work within is exposed to heightened medico-legal risk. The elements for a claim of negligence include owing a duty of care (which practitioners and medical organisations have with their patients), a breach of that duty (whether the practitioner failed to act with reasonable care and skill in their treatment), which results in damage (i.e. harm to the patient) and that the failure to exercise reasonable care caused the patient’s injury. 18 So, the question a doctor may ask is: ‘when could prescribing an off-label medication constitute a breach of my duty of care?’
If the prescribed medication is accepted by practitioner’s peers as competent practice in that particular clinical scenario, the use of the medication is supported by quality evidence, and the patient has provided voluntary informed consent which has been documented, then prescribing off-label medications should not infer negligence. 4
Limitations
The guided framework presented in this paper is based on current Australian and New Zealand regulations and consensus. It shall require updates as best practices and legislation evolve. It does not incorporate the individual policies of public and private institutions which vary between health services. The decision aid does not cover all patient prescribing scenarios and requires clinical judgement in conjunction with its use. While this paper provides a series of considerations and recommendations for psychiatrists to utilise when prescribing off-label, it does not account for the intricacies of prescribing drugs of dependence, obtaining requisite authority that may be required, forensic outcomes or the unique challenges faced in other medical specialities.
Conclusion
It should be stated that there is no legal barrier to prevent psychiatrists from prescribing off-label in Australia and New Zealand. The medication should be utilised if it is in the best interests of the patient. However, to avoid an actionable scenario, practitioners must be able to defend the rationale for their off-label medication use with evidence of sound quality. 19
Overall, it is essential to know and understand the rationale for prescribing off-label, to accurately document this, along with the patient’s consent and to inform the patient and their primary care doctor of the intended duration of treatment and any required monitoring. To be clear, it is best if your patient knows and understands that their drug is off-label and why an off-label selection has been made.19,20 There are clinical, legal, ethical, and financial considerations to take into account each time an off-label prescription is ordered.
Furthermore, it is imperative to balance the abundant benefits with the potentially unintended or harmful consequences associated with off-label prescribing. The potential benefits of off-label use include expanded treatment options, personalised patient care, and the ability to address unmet medical needs. It can lead to improved patient outcomes and quality of life. On the other hand, poor practice in off-label prescribing can result in adverse events, patient harm, legal implications, and damage to professional reputation. It underscores the critical importance of responsible and evidence-based decision-making in off-label prescribing to ensure patient safety and to mitigate potential risks.
Not all indemnity insurers provide the same coverage for off-label prescribing to their policyholders. Practitioners should contact their insurer and review their PDS prior to prescribing off-label drugs, particularly if they have not done so before. Individual state-run health services will have their own policies and procedures in place to govern the process. This article and flowchart do not constitute or replace legal advice or clinical judgment. If in doubt, please contact your medical indemnity insurer or legal representative.
Footnotes
Disclosure
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Authors’ Note
We both approve of submission of this manuscript (author’s approval) in conjunction with the terms and conditions of Australasian Psychiatry. We confirm that the manuscript content (in part or in full) has not been concurrently submitted or previously published elsewhere.
