Abstract
Objective
Anticholinergic burden refers to the cumulative effects of taking multiple medications with anticholinergic effects. This study was carried out in a public hospital in Singapore, aimed to improve and achieve a 100% comprehensive identification and review of measured, anticholinergic burden in a geriatric psychiatry liaison service to geriatric wards. We evaluated changes in pre-to post-assessment anticholinergic burden scores and trainee feedback.
Method
Plan Do Study Act methodology was employed, and Anticholinergic Effect on Cognition scale (AEC) was implemented as the study intervention. A survey instrument evaluated trainee feedback.
Results
There was no measured anticholinergic burden in a baseline of 170 assessments. 75 liaison psychiatry assessments were conducted between June and November 2021 in two cycles. 94.7% of pre-assessments (at the time of assessment) and 71.1% of post-assessments (following assessment) had a record of AEC scores in clinical documentation in cycle one, improving in the second cycle to 100%, 94.6%, respectively. A high post-assessment AEC score of 3 and over reduced from 15.8% in cycle one to 5.4% in cycle two. The trainee feedback suggested an enriching educational experience.
Conclusions
Using the AEC scale, the findings support the feasibility of comprehensive identification and review of measured anticholinergic burden in older people with neurocognitive disorders.
Keywords
Anticholinergic burden refers to the cumulative effects of taking multiple medications with desired or undesired anticholinergic effects. 1 Anticholinergic drugs are associated with delirium, 2 cognitive decline, 3 falls risk 4 and mortality 5 including increased all-cause mortality risk specifically in persons with dementia. 5
Leveraging advances in research in medication safety is critical to enhancing care quality in older people with delirium and dementia. We describe a healthcare improvement study harnessing an anticholinergic burden scale in a geriatric psychiatry liaison service to geriatric wards in a public hospital in Singapore with 1000 beds. The study’s theoretical rationale was that introducing a validated anticholinergic burden scale to quantify anticholinergic burden would improve prescribing practice in a liaison psychiatry service context.
The study’s primary aim was to improve and achieve a comprehensive identification and review of measured anticholinergic burden in the geriatric psychiatry liaison service to geriatric wards.
We also evaluated changes in pre-to post-assessment anticholinergic burden scores and trainee feedback.
Methods
Context
Changi General Hospital is a public hospital covering the eastern part of Singapore with four geriatric wards housing subsidised patients and one private and subsidised mixed geriatric and rehabilitation ward.
The Geriatric Psychiatry Liaison Programme is a liaison service initiative that has been operational in geriatric wards since 2013. 6 The service operates twice weekly, covering four geriatric wards with 116 beds.
The Geriatric team makes referrals for new assessments and reviews through an electronic system that defines referral criteria. The core older person’s liaison team comprises a Geriatric Psychiatrist, Psychologist and doctors in psychiatry training.
The study’s interdisciplinary composition comprised a Geriatric Psychiatrist, Pharmacist, Geriatric Advanced Practice Nurse from the Geriatric inpatient team, Consultants from Geriatric Medicine, Internal Medicine, and Gastroenterology. Two International mentors were also part of the study.
Study Baseline
The data management team in Changi General Hospital extracted 170 liaison psychiatry assessments between 1 November 2020 and 30 April 2021 for a 6-month baseline of referrals. The data were retrospectively extracted between April and early June 2021 at the study outset. The study had a baseline of 0% with no prior measured anticholinergic burden score using an anticholinergic burden scale (0/170 assessments).
Plan Do Study Act (PDSA) methodology and intervention
PDSA cycles,
7
and the timeline is illustrated (Figure 1). PDSA timeline chart.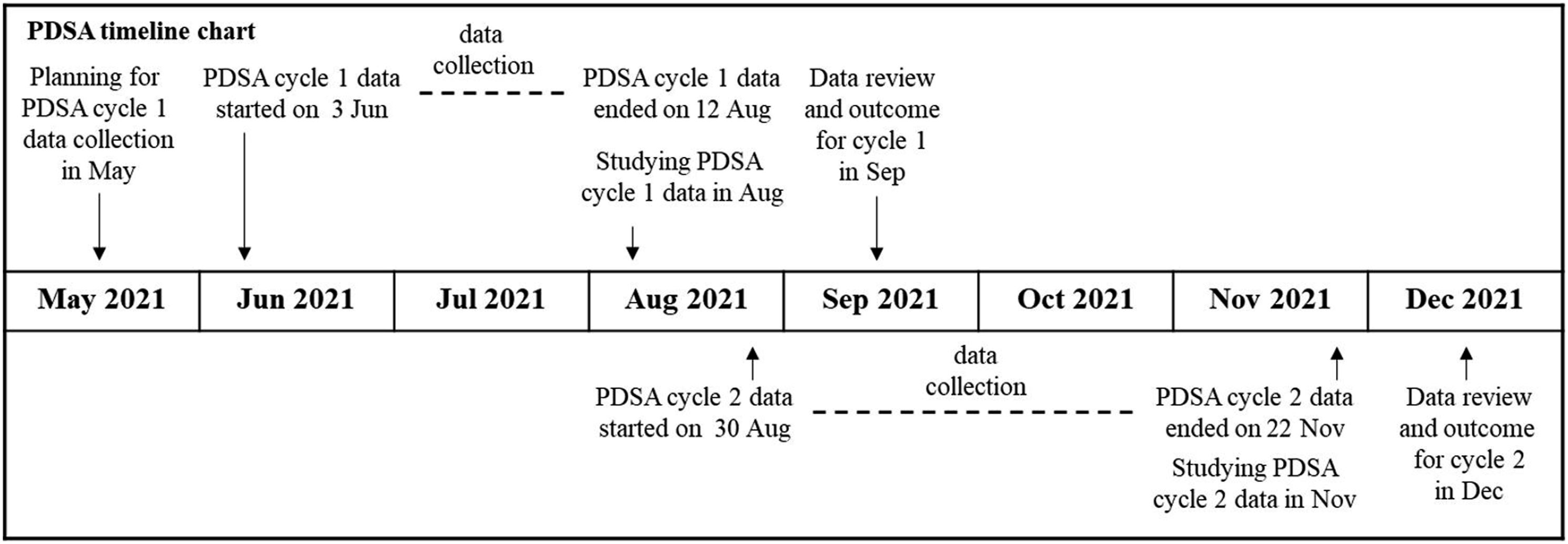
Several scales quantify the anticholinergic burden, including the Anticholinergic Risk Scale, 1 the Anticholinergic Drug Scale, 1 Anticholinergic Cognitive Burden Scale 1 and the Anticholinergic Effect on Cognition scale, AEC. 8 The study team discussed the introduction of the AEC, an innovative scale that addresses the muscarinic binding affinity of a given drug, the blood–brain barrier permeability and cognitive adverse effects.5,8 The scale addresses central anticholinergic activity and differs from the other currently available scales.5,8 AEC provides a traffic light system of drug classification with scores ranging from 0, 1, 2 and 3. A score of 0 indicates no central anticholinergic activity, and three indicates high central anticholinergic activity. 8
We defined and measured comprehensive identification as the total anticholinergic burden score at the time of liaison psychiatry assessment (pre-assessment total AEC score) evidenced in clinical documentation. The comprehensive review was defined and measured as the total anticholinergic burden score following assessment (post-assessment total AEC score). The liaison psychiatry team calculated the anticholinergic burden using the Medichec online resource (www.medichec.com). 9 The calculation of the total AEC score is illustrated (supplementary data).
Medication review strategies included substituting with a safer alternative (low AEC score medication), deprescribing and dose reduction of psychotropic medications.
The primary aim was achieved using the data of measured anticholinergic burden, with the record of pre-and post-assessment total AEC scores in clinical documentation at each liaison psychiatry assessment as the numerator and the total number of assessments the denominator.
We measured the changes in pre- to post-assessment anticholinergic burden scores as increased, decreased or unchanged. The standard for improvement was set at 100% within 12 months from April 2021, set as the study start date.
We commenced study cycle one and introduced the AEC scale between 3 June 2021 and 12 August 2021. The study PDSA data, demographic variables and diagnoses were prospectively collected on a Microsoft Excel spreadsheet by the study psychiatrist, examining the electronic records of referrals and assessments. All consecutive referrals were included in the study cycles, with one exclusion applied to requests for mental capacity assessments.
We studied the results of cycle one and implemented the second cycle between 30 August 2021 and 22 November 2021 for further improvement.
Driver diagram
7
illustrates the interventions, contextual elements and change drivers used to achieve the study aim (Figure 2). Driver Diagram.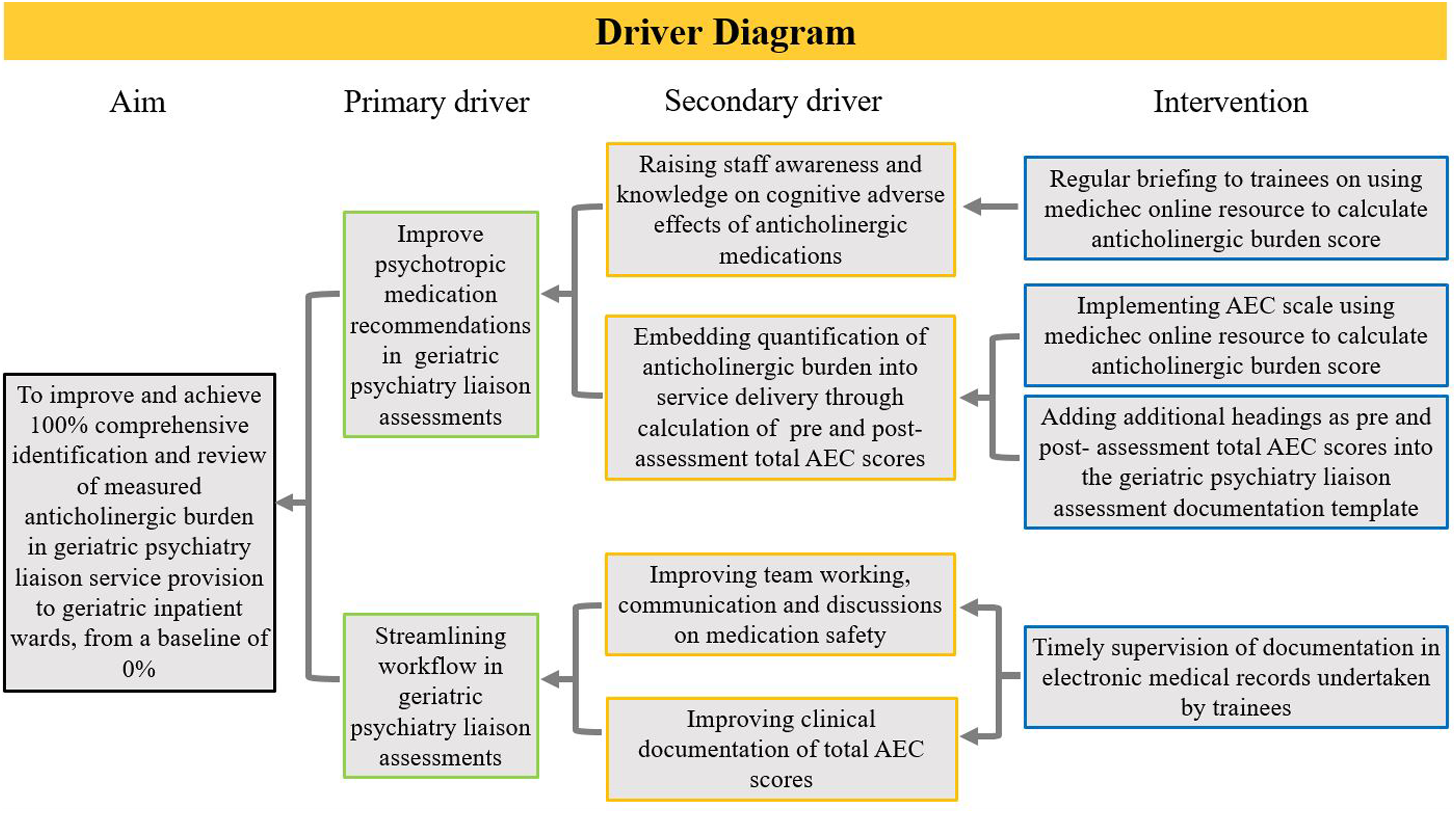
Data verification and accuracy of calculated scores for all assessments were undertaken.
Survey on trainee feedback
A government survey builder 10 was used to devise the survey questions, comprising a Likert scale, semi-structured questions and free text comments on feedback and improvement areas.
The study psychiatrist emailed the online anonymised survey in November 2021 to all past and current trainees. One trainee who assisted the study psychiatrist with devising the survey instrument was exempted from survey participation.
Analysis
Descriptive statistics were generated for all quantitative data using Excel. All free text comments in the survey results were included verbatim.
Results of PDSA cycles
Demographics and diagnoses
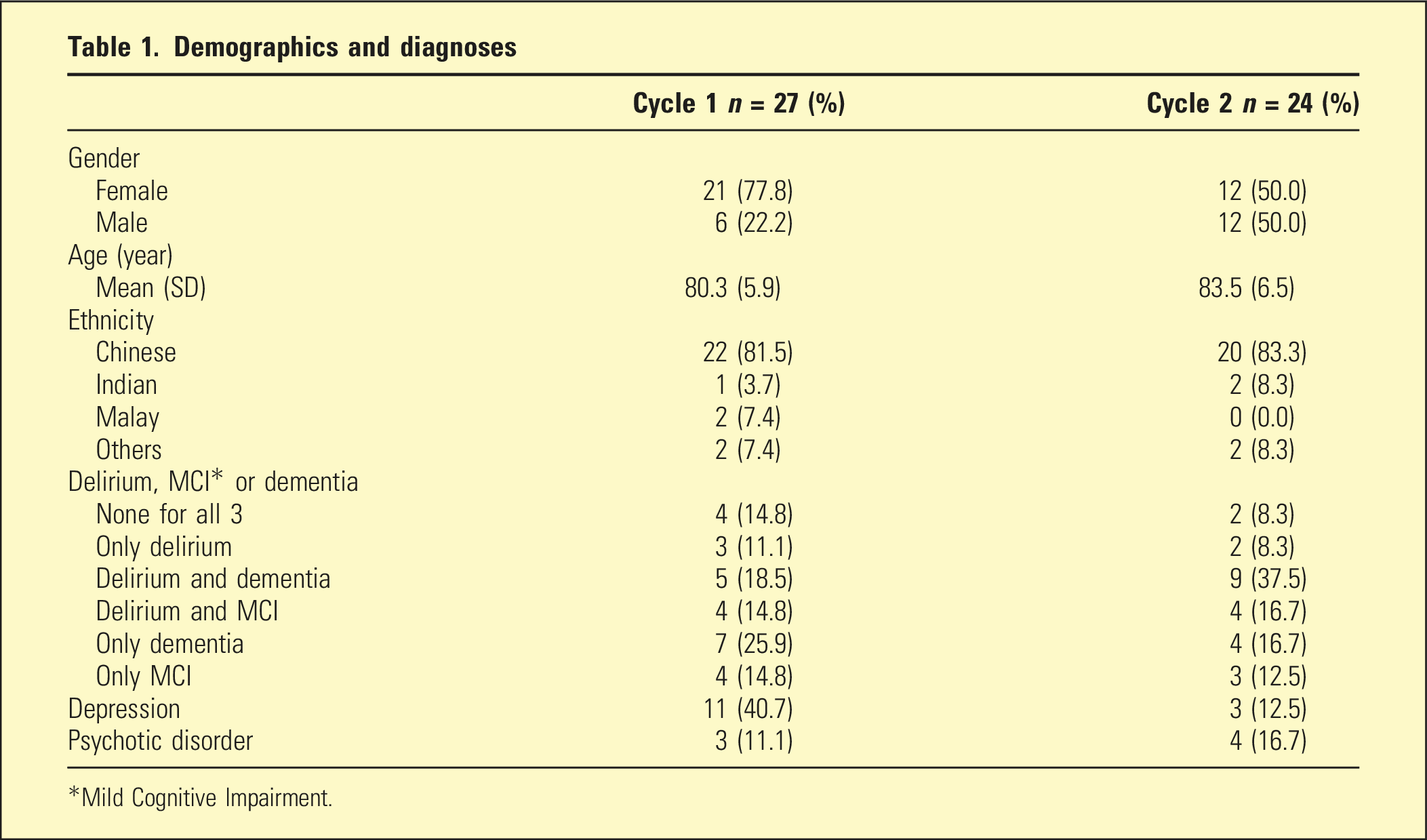
*Mild Cognitive Impairment.
Total AEC scores
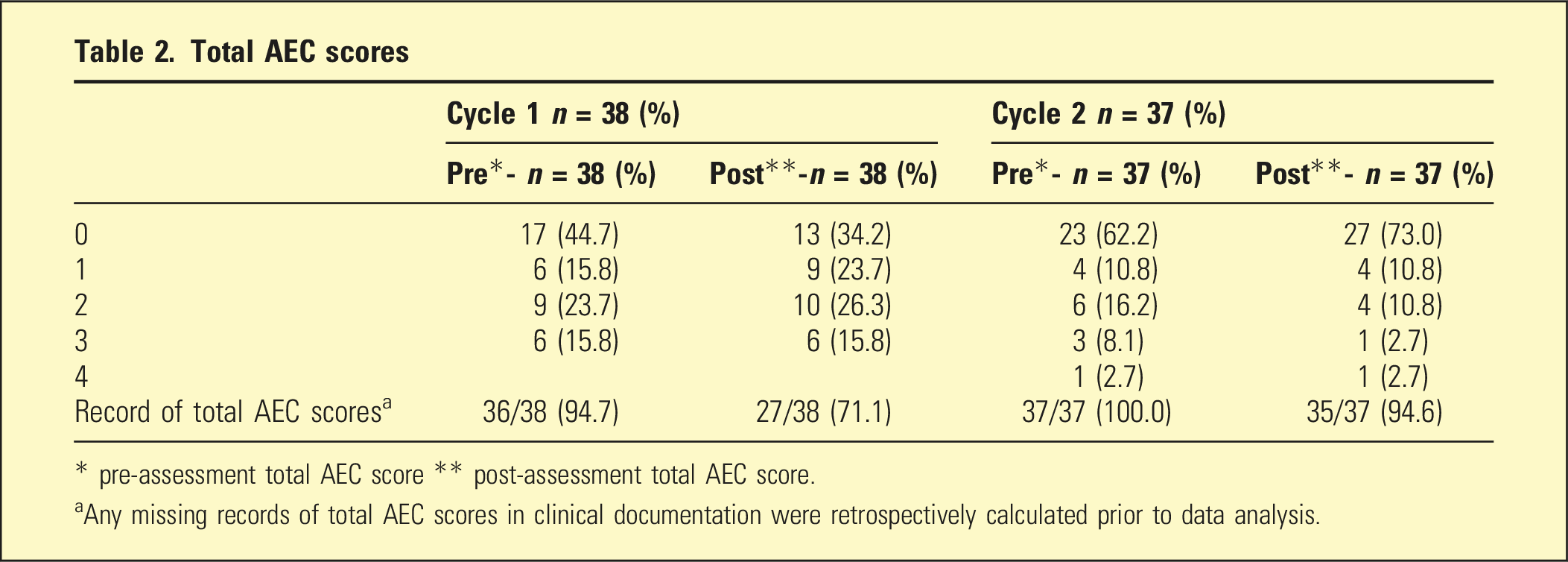
* pre-assessment total AEC score ** post-assessment total AEC score.
aAny missing records of total AEC scores in clinical documentation were retrospectively calculated prior to data analysis.
Four patients in the first cycle and five patients in the second cycle were assessed more than once.
Pre- and post-assessment total AEC scores of 1 and over were entirely attributable to antipsychotics and antidepressants in cycle one, single medication or a combination. Two other medications (domperidone and prednisolone) were accounted for in cycle two.
The main findings were on improvement in documented total AEC scores at pre-assessment and post-assessment (Table 2).
The results on secondary aim compared changes in pre-to post-assessment total AEC scores (Figure 3). Change in total AEC score from pre-assessment.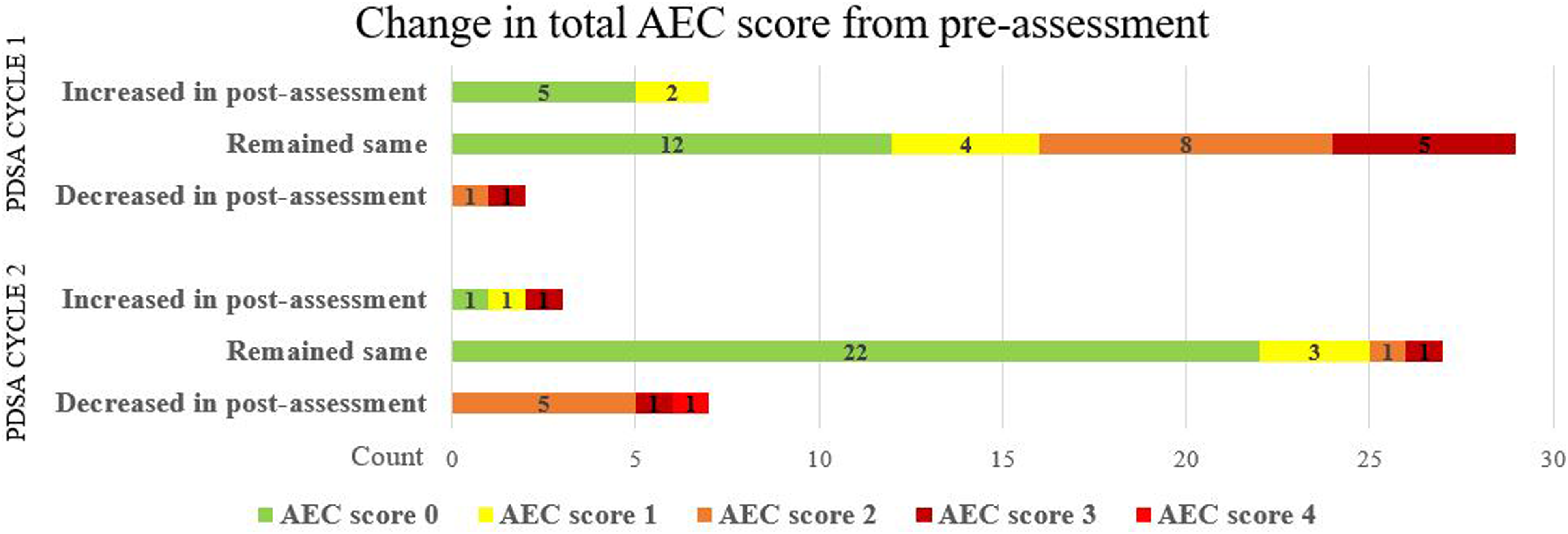
Results of Trainee feedback
There were 11 out of 14 survey respondents (78.6%).
The survey trainee results suggested an enriching educational experience (supplementary data).
Discussion
The main study results showed that implementing the AEC scale led to a 94.7% electronic documentation record of identification (pre-assessment total AEC score), leading to a record of review (post-assessment total AEC score) in 71.1% of assessments in cycle one and a further 100% documentation record of identification and 94.6% review in cycle two.
The review article by López-Álvarez et al. 11 notes that prescribing medications with anticholinergic effects may be inevitable or appropriate in conditions like depression and primary psychotic disorders, as evidenced in our study results. The post-assessment total AEC score of 2 and over paradoxically increased in cycle one (42.1%). However, we found that 40.7% had a diagnosis of depression, and 11.1% had a psychotic disorder in cycle one. In the second cycle, 12.5% had a diagnosis of depression, 16.7% had a diagnosis of a psychotic disorder and the post-assessment total AEC score of 2 and over showed a reduction to 16.2% from 42.1% in cycle one. A high post-assessment total AEC score of 3 and over reduced from 15.8% in cycle one to 5.4% in cycle two, demonstrating improvement in the second cycle.
A study by Hanlon et al. 12 concluded that anticholinergic burden was associated with adverse outcomes in middle-aged and older adults, with the AEC scale most strongly associated with dementia and delirium. Dementia risk has been found to be increased in association with exposure to several types of stronger anticholinergic drugs. 13 Therefore, medication review strategies to minimise anticholinergic burden are needed, balancing risks and benefits, medical comorbidities and other prescribed medications 11 when making prescribing decisions.
The study’s strengths involve a healthcare improvement study that attempts to bridge the gap between the clinical application of anticholinergic drug burden and advances in research and innovation. Other strengths were harnessing a digital aid using Medichec to quantify the central anticholinergic burden and the study team’s adaptability to ongoing challenges during the COVID-19 pandemic.
Limitations of this study include its relatively small sample in a geriatric psychiatry liaison setting led by the study psychiatrist and the possibility of improvement through chance in the second cycle. However, the sample size is consistent with health care improvement studies where improvement initiatives are evaluated on a small scale prior to upscaling. While cognitive testing using validated screening tools is embedded into the comprehensive geriatric inpatient assessment, we did not capture the data for this study.
An organisational approach to medication safety and progressive implementation into other specialist service provisions is underway.
Conclusions and Recommendations
Using the AEC scale, the study findings support the feasibility of comprehensive identification and review of measured anticholinergic burden in older people with neurocognitive disorders as part of routine care and assessments.
This study appears to be the first to exemplify the usage of an anticholinergic burden scale at assessment in a pragmatic geriatric psychiatry liaison clinical setting. We trust that this innovative study will spur the clinical application of the measured anticholinergic burden to refine clinical practice in neurocognitive disorders.
Supplemental Material
Supplemental Material - Improving quantification of anticholinergic burden using the anticholinergic effect on cognition scale – a healthcare improvement study in a geriatric ward setting
Supplemental Material for Improving quantification of anticholinergic burden using the anticholinergic effect on cognition scale – a healthcare improvement study in a geriatric ward setting by Bharathi Balasundaram, Wendy Swee Tee Ang, Robert Stewart, Delia Bishara, Chun How Ooi, Fuyin Li, Farooq Akram, and Andrew Boon Eu Kwek in Australasian Psychiatry
Footnotes
Author's Note
SQUIRE 2.0 Guidelines 14 have been used to devise the writing of this manuscript.
Acknowledgements
All trainees who participated in the Geriatric Psychiatry Liaison Service provision. Hong Hao Tan, trainee who assisted with devising the trainee survey questions. Tan Pei Ting former Analyst Health Services Research, Garry Semeniano and Johnathan Siew Jun Howe, Office of Improvement Science, Sandhiya Ramanathan, Date Management and Informatics, Changi General Hospital.
Author contributions
BB wrote the first draft of the article mentored by RS and DB. All authors were involved in the study design, planning, interpretation of data and critical revisions. All authors have consented to the publication in Australasian Psychiatry.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: RS declares research support in the past 3 years from Janssen, Glaxo Smith Kline, and Takeda Pharmaceuticals.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the RS is part-funded by: i) the National Institute for Health Research (NIHR) Biomedical Research Centre at the South London and Maudsley NHS Foundation Trust and King’s College London; ii) an NIHR Senior Investigator Award; iii) the National Institute for Health Research (NIHR) Applied Research Collaboration South London (NIHR ARC South London) at King’s College Hospital NHS Foundation Trust; iv) the DATAMIND HDR UK Mental Health Data Hub (MRC grant MR/W014386).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
