Abstract
Aim of this review was to identify and synthesize the results of primary studies on the relationship between oxytocin and music. This review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) criteria. A systematic literature search was conducted via PubMed/Medline, Google Scholar and PsychArticles. Our inclusion criteria were as follows: (1) music intervention, including either active music-making or passive music listening, and (2) pre- and post-measurement of oxytocin as a blood or saliva test. Only primary studies were selected for review. Article search, data extraction and methodological study were performed by two reviewers independently in October 2023. Of the 839 initially identified articles, 15 articles met the inclusion criteria. Two additional studies were added via literature cross-references resulting in a total number of 760 participants. Of the 17 included studies, four studies showed an increase in oxytocin levels, three studies showed a decrease in oxytocin levels and five studies showed unchanged or non-significant changes in oxytocin levels. Another five studies showed opposing changes in oxytocin levels depending on the type of music intervention or characteristics of the participants, such as the extent of empathy. The methodological quality of studies varied, with six studies classified as low risk studies, seven studies at moderate risk of bias and four studies as high studies. Due to contradictory results of previous studies, more scientific research is needed to further explore and better understand the evidence obtained to date. Individual differences and contextual factors will have a key role in better understanding the relationship between the oxytocinergic system and music.
Making or listening to music is one of the human cultural universals (D. Brown, 1991). The positive effects of music are widely known from personal experience, anecdotal stories and scientific practice. Time and again, the question arises as to the significance of music for the human species in an evolutionary context and why it has remained with us through all the changes right up to the modern world (Harvey, 2017; Mithen, 2006; Nikolsky & Perlovsky, 2020).
Numerous studies investigated the effect of music on subjective well-being (Daykin et al., 2020; Weinberg & Joseph, 2016), prosocial behaviour (Buren et al., 2021; Schellenberg et al., 2015), feelings of connectedness and social cohesion (Bullack et al., 2018; Koelsch, 2013), cognitive abilities (Corrigall et al., 2013; Schellenberg, 2005), memory (Ferreri & Verga, 2016), but also on neuroplasticity (Chorna et al., 2019; Merrett et al., 2013; Münte et al., 2002), physiology (Kulinski et al., 2022; McCrary & Altenmüller, 2021) as well as immunological (Kuhn, 2002; Wachi et al., 2007), endocrinological responses (Bittman et al., 2001; Fancourt et al., 2014) and stress response (Bradt et al., 2013; Fu et al., 2019). Music has long been used in a therapeutic context to modulate pain perception (Golino et al., 2019; Kühlmann et al., 2018), in neurorehabilitation for dementia (van der Steen et al., 2018), multiple sclerosis (Ostermann & Schmid, 2006) and stroke patients (Q. Liu et al., 2022; Sihvonen et al., 2017), anxiety disorders (Lu et al., 2021) and depression (Aalbers et al., 2017), children with developmental delay (Groß et al., 2010; Kennelly, 2000) and many more. Despite the often limited data available and the need for further research, the overall results show a great therapeutic potential of music.
Principles of action and the signalling cascade of music in the human organism, however, remain poorly understood. How are the effects that are so clearly demonstrated mediated? Supported by as yet weak evidence it is hypothesized that the effects of music on health are mediated by neurochemical changes, that is via neuromodulators (Chanda & Levitin, 2013). In various studies, parameters like cortisol (CORT), adrenocorticotropic hormone (ACTH), corticotropin-releasing hormone (CRH), dehydroepiandrosterone (DHEA), prolactin (PRL), growth hormone (GH), oxytocin (OT), testosterone (Test) and chromogranin A (CgA) were measured before and after a music intervention. In a review by Fancourt et al. (2014), 32 studies with the above-mentioned endocrinological parameters were included and analysed. The results show a clear sensitivity of the hormonal biomarkers to musical stimulation. Nevertheless, the review concludes that further research is needed to identify underlying factors as well as musical characteristics responsible for the alterations (Fancourt et al., 2014). This review is intended to make a contribution by focusing on the relationship between oxytocin and music.
Theoretical background
Oxytocin is a peptide hormone mainly synthesized in the supraoptic (SON) and paraventricular (PVN) nuclei of the hypothalamus and is released both centrally and peripherally (Harvey, 2020; Iovino et al., 2021). The central projections of the hypothalamic oxytocin neurons extend to all main regions of the forebrain as well as to the mesencephalon, brainstem and limbic system (Grinevich et al., 2016; Jirikowski, 2019). Peripheral release occurs via axonal transport to the posterior pituitary gland, where oxytocin is stored and released into the bloodstream upon adequate stimulation (Mitre et al., 2018; Uvnäs-Moberg et al., 2005). Alongside brain regions, oxytocin receptors are also found in various other tissues, including the heart, kidneys, thymus, uterus, ovaries and gastrointestinal tract (Gimpl & Fahrenholz, 2001; Szczepanska-Sadowska et al., 2021). Typical release stimuli are sensory stimulation such as skin-to-skin contact, warm temperature and sexual activity as well as in response to food intake, labour and breastfeeding (Uvnäs-Moberg et al., 2015).
Oxytocin is a simple peptide with a multitude of complex systematic and neurobiological effects. The functions of oxytocin have primarily been associated with uterine contractions, mother–child bonding and breastfeeding (Ross & Young, 2009). However, increasing interest in the hormone oxytocin is being driven by its diverse effects on prosociality, cooperation, empathy, trust and social cognition in humans (Bartz et al., 2011; Erdozain & Peñagarikano, 2020; Geng et al., 2018; Kosfeld et al., 2005). Similar to the effect of music, it has also been shown that oxytocin has an influence on the immune system (Li et al., 2016; Wang et al., 2022), the cardiovascular system (Jankowski et al., 2020; Reiss et al., 2019; Szczepanska-Sadowska et al., 2021), stress response (Matsushita et al., 2019; Winter & Jurek, 2019), appetite regulation (Blevins et al., 2003; C. M. Liu et al., 2021; Velmurugan et al., 2013) and nociception (Arnold et al., 2024; Boll et al., 2018; Xin et al., 2017).
Oxytocin is most commonly used as a therapeutic agent in gynaecology for the treatment of complications in obstetrics (Osilla & Sharma, 2023). The first experimental therapy trials have been conducted in connection with nasally applied oxytocin in psychiatric disorders, including autism spectrum disorder, post-traumatic stress disorder, affective disorder, schizophrenia and borderline personality disorder (Giovanna et al., 2020; Peled-Avron et al., 2020), as well as to reduce pain perception (Arnold et al., 2024; Lopes & Osório, 2023) and to modulate the stress response (Romney et al., 2019; Wirth et al., 2015). The results are ostensibly promising, however not yet sufficiently conclusive, so that a clear evidence-based benefit of oxytocin treatment remains to be established (Quintana et al., 2021; Winterton et al., 2021). Abnormalities in the oxytocinergic system have also been identified in various other neuropsychiatric disorders such as depression, eating disorders, attention deficit disorder and addiction. Due to the multitude of its effects, oxytocin is, therefore, increasingly gaining attention as a therapeutic agent in these areas of medicine (Cid-Jofré et al., 2021; Romano et al., 2015). A therapeutic approach for these disorders could, therefore, involve either a stimulation of the endogenous release or an exogenous supply of oxytocin (Crockford et al., 2014). Yet a deeper understanding of the mechanisms of central oxytocin release and the oxytocinergic system in general is a prerequisite for a mechanism-based and safe therapy (Grinevich et al., 2016). Therefore, further investigation is also required into the interactions with other neuroregulatory signalling pathways and medication such as SSRIs and antipsychotics (Peled-Avron et al., 2020).
To a considerable extent, the effects of oxytocin overlap with the effects that can be observed when making or listening to music. It is, therefore, possible that the biology of oxytocin mirrors at least parts of the positive and diverse effects of music (Harvey, 2020; Keeler et al., 2015).
Previous studies on music and oxytocin, however, show contradictory results with either increasing, decreasing or unchanged oxytocin levels when measured before and after a music intervention (Bowling et al., 2022; Schladt et al., 2017). Overall, the neuroendocrine response to music interventions and how oxytocin can promote bonding in a musical setting is poorly understood.
A growing body of literature, nevertheless, explores an evidence-based approach to music interventions using reproducible scientific experiments (Chanda & Levitin, 2013). To date, neuroscientific research in the field of music has primarily relied on neural imaging. The effect of music on neuroendocrinology has only been increasingly investigated in recent years (Keeler et al., 2015).
The aim of this review is, therefore, to systematically record, analyse and summarize the results of the primary studies conducted to date to identify further steps for future research. The additional parameters collected in the primary studies will be considered in the analysis and correlations with oxytocin may be identified. Furthermore, the underlying mechanisms linking music-making to physical and mental health will be investigated.
Methods
The main objective of this article was to synthesize scientific evidence regarding music and oxytocin. This review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) criteria to improve transparency in the documentation of review methods. These guidelines, therefore, consist of a set of items designed to ensure a minimum standard for the reporting of systematic reviews and meta-analyses (Shamseer et al., 2015).
A focused systematic literature search strategy was designed using the databases Google Scholar, PubMed, APA PsycArticles, PBSC and MEDLINE (the last three via EBSCO). The search was intended to initially cover all available literature on oxytocin and music, so that PubMed and EBSCO Host were searched exclusively for ‘oxytocin’ AND ‘music’. Due to the high number of results and to ensure that the search was as comprehensible as possible, Google Scholar was searched more specifically for synonyms of changes in oxytocin levels: ‘oxytocin level’ music, ‘differences in oxytocin’ music, ‘change in oxytocin’ music and more holistically with ‘allintitle: oxytocin music’ to capture the entire existing body of literature in the end. The literature search was performed in September and October 2023.
Two reviewers independently extracted data on intervention and biomarker characteristics in several steps according to title, abstract and full text. The findings were compared afterwards, and differences were resolved by consensus.
The inclusion criteria consisted firstly of a music intervention, which could consist of both active music-making and passive music listening. The second inclusion criterion consisted of a pre- and post-measurement of the parameter oxytocin. This could be performed both as a saliva test or as a blood test. Finally, animal studies were excluded. Only primary studies were included in this review, and solely German- or English-language literature was assessed.
The selection of studies was not limited by time of publication, subject demographics or study design. No database filters were used to minimize the risk of loss. Mixed interventions studies were only included if effects on oxytocin changes could be traced back to the music intervention. Studies which used music as a confounding, distracting or supportive factor were also excluded.
Next, the most relevant data from the primary studies were extracted, including the name of the authors, year and country of publication, study design, sample size, objectives, intervention and, if applicable, control group data, outcomes and main results. Further endocrinological parameters, physiological parameters or questionnaires, were also recorded to take them into account in the subsequent analysis and to identify any correlations.
To assess methodological quality of the studies, the risk of bias for randomized controlled trials was assessed by the Cochrane Risk of Bias Tool 2 (RoB 2) (Sterne et al., 2019). This tool evaluates bias across five domains: randomization process, deviations from intended interventions, missing outcome data, measurement of outcomes and selection of the reported result. Each domain is rated as having ‘low risk’, ‘some concerns’ or ‘high risk’ of bias. For non-randomized studies (observational studies and pre–post studies), the Risk Of Bias In Non-randomized Studies–of Interventions (ROBINS-I) tool was used (Sterne et al., 2016), which assesses bias across seven domains, including confounding, selection of participants, classification of interventions, deviations from intended interventions, missing data, measurement of outcomes and selection of the reported result. Each domain in ROBINS-I is rated as ‘low’, ‘moderate’, ‘serious’ or ‘critical’ risk of bias.
Results
As shown in Figure 1, 839 publications were initially identified during the primary search. After screening for eligibility criteria, 15 studies met the requirements for this review. Two further studies were added via literature cross-references so that a total of 17 studies were ultimately included in the qualitative analysis. Table 1 depicts the data extracted from the primary studies in terms of authorship, publication, study design, objectives, sample, intervention, outcomes evaluated and main results.
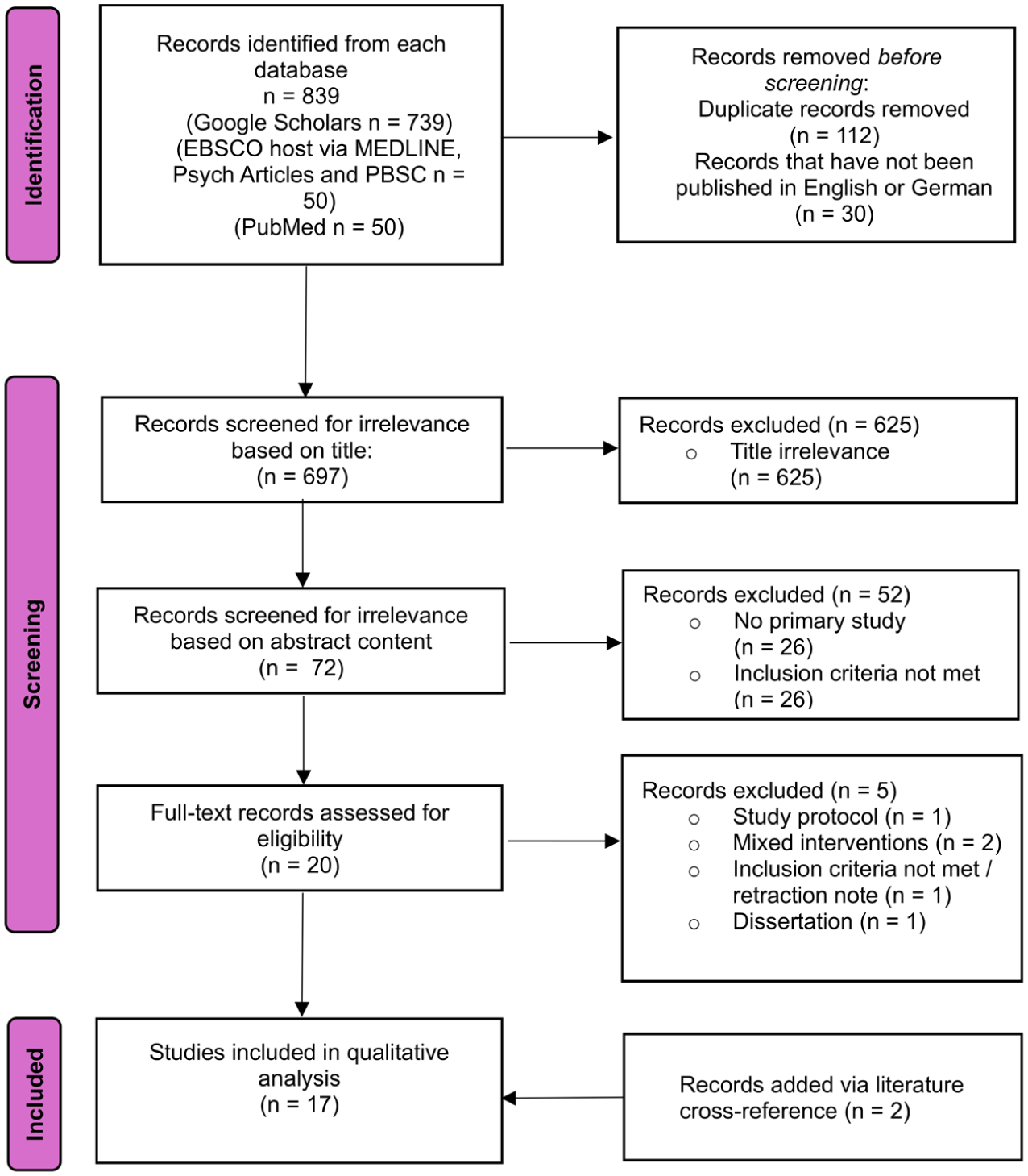
Prisma flow chart – identification of studies via databases.
Characteristics of the included studies.
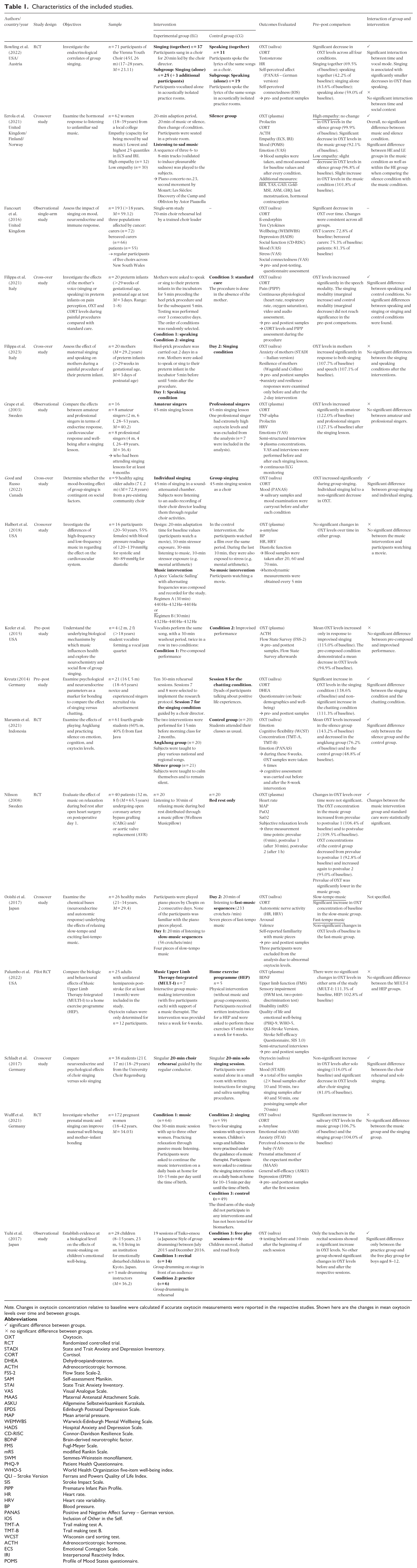
Note. Changes in oxytocin concentration relative to baseline were calculated if accurate oxytocin measurements were reported in the respective studies. Shown here are the changes in mean oxytocin levels over time and between groups.
✓ significant difference between groups.
× no significant difference between groups.
OXT Oxytocin.
RCT Randomized controlled trial.
STADI State and Trait Anxiety and Depression Inventory.
CORT Cortisol.
DHEA Dehydroepiandrosteron.
ACTH Adrenocorticotropic hormone.
FSS-2 Flow State Scale-2.
SAM Self-assessment Manikin.
STAI State Trait Anxiety Inventory.
VAS Visual Analogue Scale.
MAAS Maternal Antenatal Attachment Scale.
ASKU Allgemeine Selbstwirksamkeit Kurzskala.
EPDS Edinburgh Postnatal Depression Scale.
MAP Mean arterial pressure.
WEMWBS Warwick-Edinburgh Mental Wellbeing Scale.
HADS Hospital Anxiety and Depression Scale.
CD-RISC Connor-Davidson Resilience Scale.
BDNF Brain-derived neurotrophic factor.
FMS Fugl-Meyer Scale.
mRS modified Rankin Scale.
SWM Semmes-Weinstein monofilament.
PHQ-9 Patient Health Questionnaire.
WHO-5 World Health Organization five-item well-being index.
QLI – Stroke Version Ferrans and Powers Quality of Life Index.
SIS Stroke Impact Scale.
PIPP Premature Infant Pain Profile.
HR Heart rate.
HRV Heart rate variability.
BP Blood pressure.
PANAS Positive and Negative Affect Survey – German version.
IOS Inclusion of Other in the Self.
TMT-A Trail making test A.
TMT-B Trail making test B.
WCST Wisconsin card sorting test.
ACTH Adrenocorticotropic hormone.
ECS Emotional Contagion Scale.
IRI Interpersonal Reactivity Index.
POMS Profile of Mood States questionnaire.
Of the 20 full texts assessed, a total of five articles were excluded from the review. The reasons for this were that only a study protocol was available (Beck et al., 2018), the inclusion criteria were not met (Minutillo et al., 2021) a study was conducted as part of a dissertation (Wulff, 2020) and was later published and already included in the review (Wulff et al., 2021) as well as the use of mixed interventions (Jezova et al., 2013; Riedl et al., 2017). In the study by Riedl et al. (2017), for instance, a trust game was conducted in which music was played in the background in the intervention group and no music was played in the control group, so that changes in oxytocin levels cannot be clearly attributed to the music intervention. Similarly, in the study by Jezova et al., (2013), a combined intervention with both mental and physical stress procedures was performed, with additional music being played forwards (pleasant) or backwards (unpleasant), so that here, too, a change in oxytocin levels cannot be reliably traced back to the music intervention.
Overall study characteristics
The studies included were published in the period 2003–2023 and were all available in English. The studies were conducted in the following eight countries: USA (n = 3, 17.6%), Germany (n = 3, 17.6%), Japan (n = 2, 11.8%), Sweden (n = 2, 11.8%), Italy (n = 2, 11.8%), Austria (n = 1, 5.9%), Finland (n = 1, 5.9%), the United Kingdom (n = 1, 5.9%), Canada (n = 1, 5.9%) and Indonesia (n = 1, 5.9%).
Study designs and samples
Among the 17 studies, there were seven crossover trials, five randomized controlled trials, three observational studies and two pre–post studies. The total number of subjects included in the review was n = 760. The number of participants included in each study ranged from 4 to 193 (M: 45 participants, median: 23 participants). Note that the number of participants given here applies to those who actually underwent an oxytocin test and whose results were ultimately included in the analysis.
Due to incomplete data, only limited statements can be made about the average age and gender distribution in the total sample. However, 10 studies provided information on the average age of the study participants, resulting in an average age of 37.6 years (median: 34.03 years). In addition, the proportion of female study participants was 58.8%, although one study did not provide any information on this and, therefore, only 16 studies could be included in this calculation.
The subject characteristics were very diverse. Healthy subjects and patients who had undergone cardiac surgery, stroke patients and cancer patients were included. In addition, the overall sample covered an age range from preterm infants to pensioners, including traumatized children, students, pregnant women and mothers. Both musicians and non-musicians were part of the sample.
Objectives
The objective of most of the studies was to investigate the neuroendocrinological correlates of music, in particular singing, but also other music interventions, in more detail (Bowling et al., 2022; Eerola et al., 2021; Fancourt et al., 2016; Grape et al., 2002; Keeler et al., 2015; Kreutz, 2014; Maramis et al., 2021; Ooishi et al., 2017; Schladt et al., 2017; Yuhi et al., 2017). In addition, some studies attempted to address a wide range of additional questions. For example, Good & Russo (2022) aimed to determine the extent to which the mood-boosting effects of group singing are dependent on social factors. In a further study, the effect of the mother’s singing and speaking voice during painful procedures on her premature babies was investigated on the pain perception of these newborns in a first study (Filippa et al., 2021) and on the mothers’ well-being in a second study (Filippa et al., 2023). Wulff et al. (2021), on the contrary, investigated the effect of music on the well-being of pregnant women and on mother–child bonding. Other studies also looked at the effect of music on relaxation after cardiac surgery (Nilsson, 2009), the effects of high- and low-frequency music on the cardiovascular system (Halbert et al., 2018) and a special form of music therapy (MULTI-I) for stroke patients (Palumbo et al., 2022).
Music interventions
The inclusion criteria have allowed any form of music intervention to be included. The variety of music interventions performed is correspondingly diverse. A total of five studies included a passive music intervention, that is listening to music only. Participants were played sad music (Eerola et al., 2021), fast and slow music (Ooishi et al., 2017), music with changing frequency (Halbert et al., 2018) and relaxing music (Nilsson, 2009; Wulff et al., 2021).
A wide variety of scenarios were also investigated in the active music interventions. The effect of choral singing was frequently investigated in a total of five studies (Bowling et al., 2022; Fancourt et al., 2016; Good & Russo, 2022; Kreutz, 2014; Schladt et al., 2017). Solo singing (Bowling et al., 2022; Filippa et al., 2021, 2023; Good & Russo, 2022; Schladt et al., 2017) was examined in further studies or as a control group for the above-mentioned studies. In several studies, singing interventions were also compared with speech interventions (Bowling et al., 2022; Filippa et al., 2021, 2023; Kreutz, 2014). A comparison was also made between professional singers and amateur singers (Grape et al., 2002) and between a composed and an improvised performance (Keeler et al., 2015).
As a solely instrumental music intervention, two studies used an instrument that is traditional in each of these countries. The first was an angklung group, a traditional Indonesian instrument (Maramis et al., 2021), and the second was a Taiko-Ensou drumming session, a traditional Japanese style of group drumming (Yuhi et al., 2017) There was only one music therapy intervention in which music was made in a small group under the guidance of a music therapist (Palumbo et al., 2022).
In both active music interventions, such as singing in a choir, and passive music interventions, such as listening to music, the music was usually provided by the study authors. For example, participants were either given choral pieces (Bowling et al., 2022; Fancourt et al., 2016; Good & Russo, 2022; Kreutz, 2014; Schladt et al., 2017) or participants were played a specific pre-selected piece of music (Eerola et al., 2021; Halbert et al., 2018; Nilsson, 2009; Ooishi et al., 2017).
An exception to the predetermined choice of music includes, therefore, studies in which either musical improvisation (Keeler et al., 2015), music therapy (Palumbo et al., 2022), singing lessons (Grape et al., 2002) or free singing to contact one’s own newborn (Filippa et al., 2021, 2023) constituted as the music intervention. Thus, there was no study in which the music preferences of the study participants were systematically recorded and considered in the implementation of the music intervention.
In addition, previous musical knowledge or training was only partially recorded in the included studies. In a total of two studies, professional musicians (Grape et al., 2002) or music students (Keeler et al., 2015) were selected as the study population. More frequently, however, amateur musicians with previous musical experience, for example in choral singing (Fancourt et al., 2016; Good & Russo, 2022; Schladt et al., 2017) or participants without any previous musical experience were included in the studies (Nilsson, 2009; Palumbo et al., 2022; Wulff et al., 2021).
In most studies, a different music intervention was used as a control group. Beyond this, there were control groups in which participants were in silence groups (Eerola et al., 2021; Maramis et al., 2021), underwent standard medical care (Filippa et al., 2021; Nilsson, 2009), watched a film (Halbert et al., 2018) or carried out an exercise programme at home (Palumbo et al., 2022).
Outcome measures
The main outcome was the oxytocin concentration measured before and after the music intervention. In 11 studies, the oxytocin concentration was measured in saliva, and in the other six studies, a blood sample was used. In the majority of studies, other endocrinological parameters were also examined. Cortisol was measured most frequently in 10 studies. Other parameters collected were ACTH, prolactin, testosterone, ß-endorphin, cytokines, TNF-alpha, a-amylase, DHEA and BDNF. In a number of studies, physiological parameters such as heart rate, blood pressure and heart rate variability were also recorded. Moreover, various questionnaires were collected on mood, empathy, well-being, social function, stress, social connectedness, anxiety, pain and many more (cf. tab. 1).
Oxytocin measurement
Various radio- and enzyme-based immunoassays were used in the studies to determine oxytocin levels in saliva or plasma (see Table 2). However, a detailed discussion of this important topic is beyond the scope of this review, so reference is made here to the relevant literature (Gan et al., 2023; Harvey, 2020; Jurek & Neumann, 2018; Tabak et al., 2023), which describes the difficulties in measuring oxytocin levels with regard to the context of sample collection and technical implementation.
Listed are the enzyme-based and radioimmunoassays used in the studies to determine oxytocin levels.
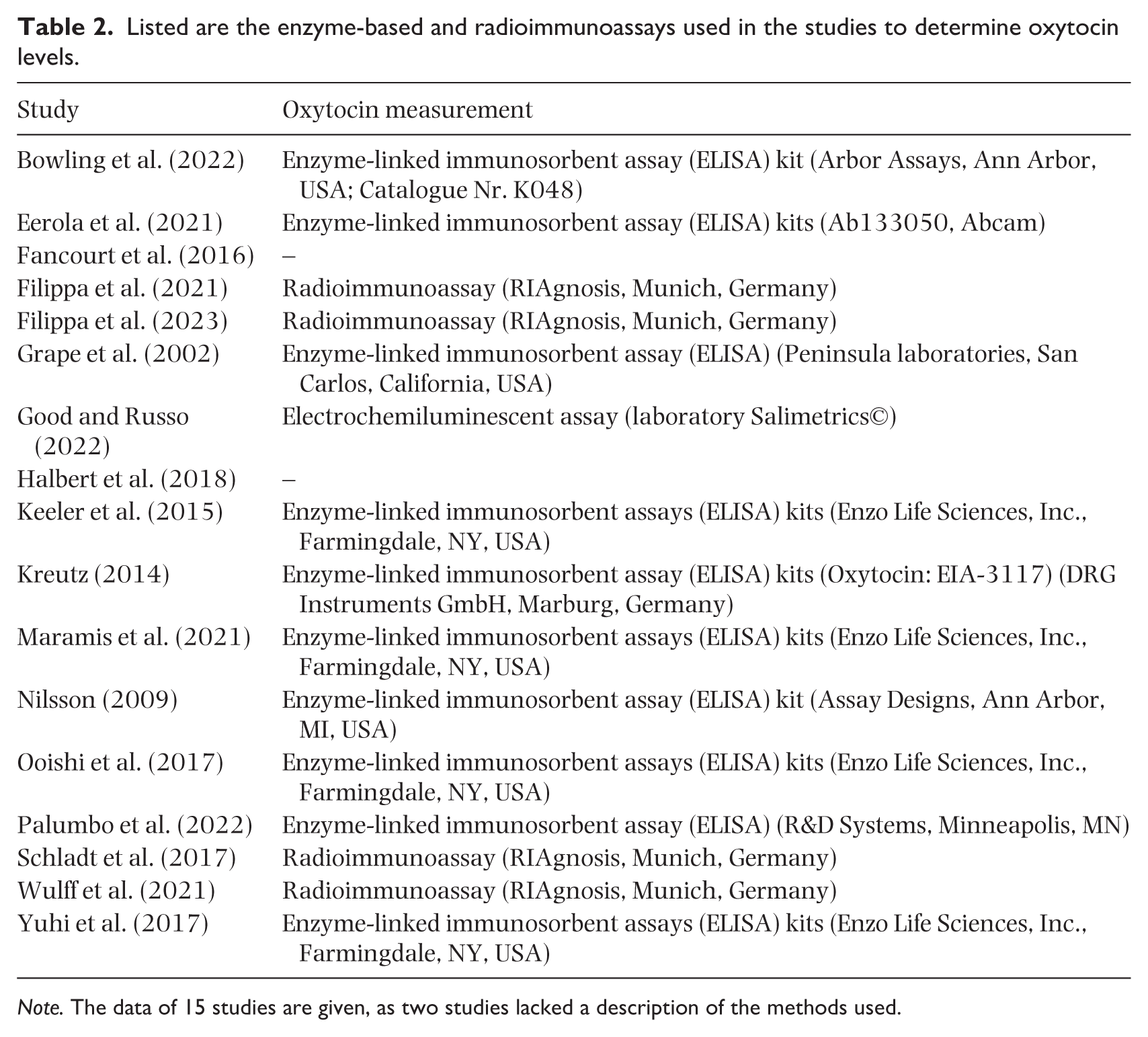
Note. The data of 15 studies are given, as two studies lacked a description of the methods used.
Main effects of intervention
Overall, it is difficult to summarize the results in a meaningful and comprehensible way due to the heterogeneity of the studies. The results of the studies can be roughly divided into four groups: Oxytocin increase (four studies), oxytocin decrease (three studies), unchanged oxytocin levels or non-significant results (five studies), and opposing changes in oxytocin levels depending on the type of music intervention in the respective study arms over time (five studies). It should be noted that the evaluation of the study results refers exclusively to the results of the music intervention.
Without differentiating between the type of music intervention or saliva and blood tests, four studies (Filippa et al., 2023; Grape et al., 2002; Kreutz, 2014; Wulff et al., 2021) found an increase in oxytocin levels and three studies (Bowling et al., 2022; Fancourt et al., 2016; Maramis et al., 2021) found a decrease in oxytocin levels at pre–post measurement.
A further five studies (Filippa et al., 2021; Halbert et al., 2018; Nilsson, 2009; Palumbo et al., 2022; Yuhi et al., 2017) showed unchanged or non-significant changes in oxytocin levels in a before-and-after comparison. However, in the study by Yuhi et al. (2017) the subgroup of teachers in the recital session showed a significant increase in oxytocin.
The last group described opposing changes in oxytocin levels depending on the type of music intervention (Good & Russo, 2022; Keeler et al., 2015; Ooishi et al., 2017; Schladt et al., 2017) or on characteristics of the participants such as the extent of empathy (Eerola et al., 2021). For instance, Keeler et al. (2015) investigated the effect of a composed performance compared with an improvised performance. They found a decrease in oxytocin concentration in the pre-composed performance and an increase in oxytocin concentration in the improvised performance. Furthermore, Ooishi et al. (2017) had participants listen to fast and slow music. The slow music led to an increase in oxytocin, while oxytocin levels remained unchanged in the fast-music condition. Finally, Eerola et al. (2021) not only looked at musical parameters but also included individual characteristics of the participants in the analysis. When listening to sad music, participants with a high empathy rating based on a questionnaire showed a drop in oxytocin levels, while participants with a low empathy rating showed a non-significant increase in oxytocin concentration.
In particular, studies comparing choral singing with solo singing show contradictory results. Bowling et al. (2022), Fancourt et al. (2016) and Schladt et al. (2017) reported a significant drop in oxytocin levels when singing together. However, Bowling et al. (2022) also showed a decrease in oxytocin when singing alone, while Schladt et al. (2017) showed a non-significant increase in oxytocin. In contrast, Good and Russo (2022) and Kreutz (2014) found an increase in oxytocin levels during choral singing. When singing solo, however, Good and Russo (2022), like Bowling et al. (2022), showed a decrease in oxytocin levels. The study by Kreutz (2014) and Fancourt et al. (2016) did not include an intervention group for solo singing, so no comparable data is available.
A total of four studies compared singing and speech interventions, with interventions differing greatly and the results remaining inconclusive. In two studies Filippa et al. (2021, 2023), research was first conducted into the effect of the mother’s singing and speaking voice on the premature infant during a painful procedure. In a second step, the effect of the mother’s singing and speaking voice on the mother herself was examined in reverse. The preterm infant showed a significant increase in oxytocin while the mother spoke to the infant, but not when the mother sang to the infant. Conversely, both maternal talking and singing led to an increase in the mother’s oxytocin concentration.
Bowling et al. (2022), on the contrary, investigated four conditions: singing alone, singing together, speaking alone and speaking together. All four conditions showed a drop in oxytocin. In another study, Kreutz (2014) compared singing together with participants talking in dyads about positive life experiences. This revealed a significant increase in oxytocin levels when singing together and a non-significant increase in oxytocin levels when chatting with each other.
Exploratory analyses on the influence of factors such as age, gender, sample size or measurement instruments on the direction of the pre–post results were not conducted in the included studies. Nevertheless, several studies have investigated the relationship between psychological variables and biomarkers (Fancourt et al., 2016; Filippa et al., 2023; Good & Russo, 2022; Kreutz, 2014), but again, no correlations between basal scores and the degree of response to the musical intervention in terms of a mood change and biomarker response were explored.
Interaction effects of intervention and group
When comparing between the groups, five studies (Good & Russo, 2022; Kreutz, 2014; Nilsson, 2009; Schladt et al., 2017) showed a significant difference between the music intervention group and the control group or between two music interventions. A further four studies (Eerola et al., 2021; Filippa et al., 2021; Maramis et al., 2021) only showed significant results when comparing subgroups or individual study arms. Additional six studies showed no significant differences when comparing the intervention and control groups (Filippa et al., 2023; Grape et al., 2002; Halbert et al., 2018; Keeler et al., 2015; Palumbo et al., 2022; Wulff et al., 2021). In the study by Ooishi et al. (2017), no group comparisons were made and in the study by Fancourt et al. (2016) only one study arm was conducted.
Risk of bias
Of the 17 studies, 9 studies were rated with a low risk of bias, 3 studies with a moderate risk of bias and 4 studies with a high risk of bias. The results of this qualitative analysis are shown in Figure 2. The risk-of-bias assessment for the respective studies relates exclusively to the survey of the parameter oxytocin.
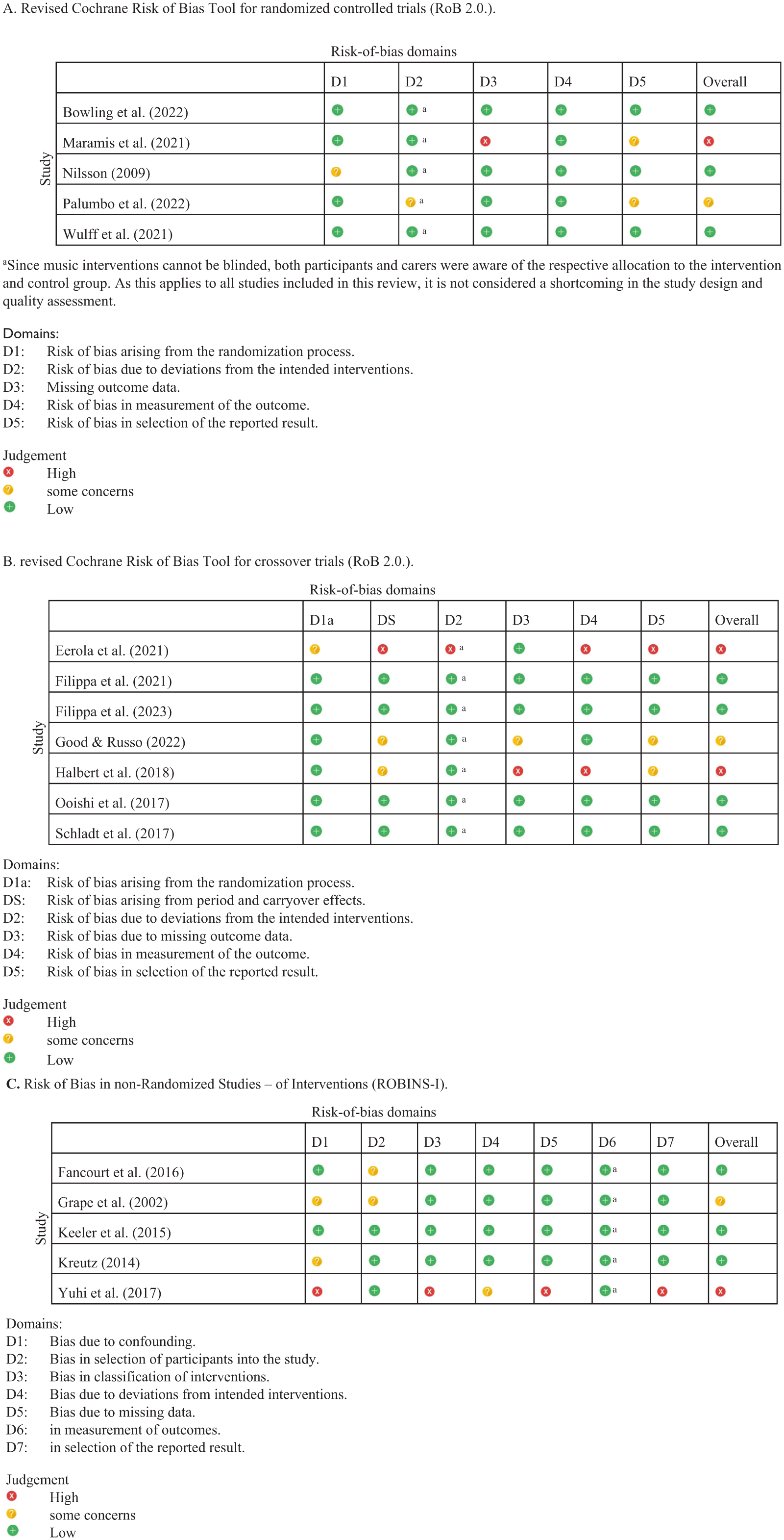
Risk-of-bias assessment of included randomized controlled trials (A) and of crossover trials (B) using the Revised Cochrane Risk of Bias Tool (RoB 2.0). Observational studies and pre–post studies (C) were assessed using the risk of bias in non-randomized studies of interventions assessment tool (ROBINS-I).
Looking exclusively at the seven studies that were rated with a low risk of bias (i.e. green) (Bowling et al., 2022; Filippa et al., 2021, 2023; Keeler et al., 2015; Ooishi et al., 2017; Schladt et al., 2017; Wulff et al., 2021), there were also contrasting results with four increases, three non-significant changes and three decreases in oxytocin levels. Similarly, the two included randomized controlled trials, which were rated at low risk of bias (Bowling et al., 2022; Wulff et al., 2021), showed one increase and one decrease in oxytocin levels. Thus, even stricter inclusion criteria do not appear to provide a clearer picture.
Discussion
The purpose of this review was to provide a systematic qualitative analysis of the current literature surrounding the mediating role of oxytocin in music. To conclude, it can be stated that the studies found are difficult to compare in terms of study design and music interventions, and the results are still equivocal or even contradictory in some cases.
The selected inclusion criteria allowed the data to be analysed without constraint in terms of demographic characteristics, geographic location, time of publication, number of participants, type of study or form of music intervention. Therefore, the studies were highly heterogeneous in terms of study design, participant characteristics, type and method of music interventions and sample material for oxytocin measurement. The studies included also contained some pilot and feasibility studies with very small case numbers, so that the results have no or only limited statistical significance, and the conclusions should be regarded tentatively.
The authors of the various studies repeatedly point to difficulties in the depiction of a naturalistic environment, as the design of the studies is inherently highly susceptible to interference (Bowling et al., 2022; Kreutz, 2014). First, because music interventions cannot be blinded, and second, because there is always the possibility of further non-musical interactions during group interventions, which could potentially confound the results. To prevent this, participants were asked, for example, to avoid physical contact with their fellow musicians while singing together (Keeler et al., 2015).
The design of the music intervention itself must also be carefully considered, as the individual musical parameters such as rhythm, melody and harmony may determine the effect of the music on the participants (Treider et al., 2023). In addition to the style of music, however, the question of the participants’ music preferences and the significance of self-selected versus prescribed music choices also arises time and again (Nilsson, 2009; Ooishi et al., 2017). The different susceptibility to cultural connotations of music (Bowling et al., 2022) as well as the musical experience and background of the participants may also play a role that should not be underestimated (Fancourt et al., 2016). To obtain reproducible and comparable results, the effect of the duration of the music intervention and the effect of repeated interventions will also need to be investigated in more detail (Nilsson, 2009).
Various challenges exist not only regarding the study design but also with regard to the parameter oxytocin. To date, there are no standardized values for oxytocin in the scientific literature. It is known, however, that there is a high intra-individual variability in the baseline values of oxytocin (Martins et al., 2020). This variation in the release and effect of oxytocin between individuals is dependent on age and gender, health status, early attachment and previous experiences (Carter et al., 2020; Crockford et al., 2014; Marazziti et al., 2023). The significance of the variability in baseline levels, as yet, is not understood. One hypothesis is that oxytocin could act as a biomarker that increases individual sensitivity to social stimuli (Bartz et al., 2011).
Another important factor is the high degree of context sensitivity that has been described for the hormone oxytocin (Bartz et al., 2011). The release and the effect of oxytocin are assumed to be dependent on the intimacy of the social interaction, the extent of collaborative action and the circumstances, such as a stressful or supportive environment. The relationship between subjects may, therefore, be critical to the potential for social bonding and thus changes in oxytocin levels (Bowling et al., 2022; Good & Russo, 2022). This purposeful joint interaction is a characteristic of both singing lessons (Grape et al., 2002) and musical improvisation (Keeler et al., 2015), in which an increase in oxytocin has been shown.
Furthermore, there are still some inconsistencies and unanswered questions regarding the measurement of oxytocin. A variety of measuring methods are used to determine peripheral and central oxytocin, including different biochemical assays and assay techniques as well as different approaches to sample collection, sample preparation and sample storage (MacLean et al., 2019; Tabak et al., 2023). However, it is still unclear whether or how peripherally measured oxytocin concentrations are related to centrally measured oxytocin concentrations (Crockford et al., 2014; Lefevre et al., 2017; Martin et al., 2018; Valstad et al., 2017). It remains unknown, at present, how exactly the hydrophilic oxytocin reaches saliva and whether an altered salivary flow during speaking or singing could possibly distort the measured oxytocin levels (Tabak et al., 2023). One approach could, therefore, be to calculate the flow rate of saliva based on the volume collected (Schladt et al., 2017).
Furthermore, a comprehensive understanding of the complexity of the interaction between oxytocin and other neuroendocrine and somatosensory signalling pathways is only gradually emerging (Grinevich & Stoop, 2018; Krause et al., 2021).
In the context of the contradictory changes in oxytocin concentrations before and after a music intervention, especially when singing together, a theory is gaining ground and is increasingly being supported by data: On one hand, music is associated with an increase in bonding response, which may be reflected by an increase in oxytocin levels (Kreutz, 2014). In addition, however, a drop in oxytocin concentrations in the pre–post measurement may be caused by a downregulation of the stress response, which could mask the opposite process of bond formation (Fancourt et al., 2016). As a result, oxytocin levels may be more dependent on HPA-axis activity than on social experience (Schladt et al., 2017). In a meta-analysis, C. A. Brown et al. (2016) also showed a correspondingly positive correlation between cortisol and oxytocin, which exists across individual differences but is strongly context dependent. Some of the studies included in this review also indicate that the oxytocin and cortisol response to group singing resembles each other (Bowling et al., 2022; Fancourt et al., 2016; Kreutz, 2014; Schladt et al., 2017). However, other studies showed less conclusive results regarding a positive correlation between oxytocin and cortisol (Good & Russo, 2022; Grape et al., 2002; Ooishi et al., 2017). Nevertheless, it can be assumed that changes in peripherally measured oxytocin depend on various social contexts and affective states, notably both social anxiety and social connectedness (Tabak et al., 2023). In this respect, it is essential to ensure a control for the HPA-axis activity in further studies (Schladt et al., 2017).
Limitations of this review
The diverse range of study designs, participant characteristics and music interventions included in the analysis contributes to significant heterogeneity across studies. As a result, comparing findings and drawing definitive conclusions becomes challenging, potentially limiting the robustness of the synthesized evidence.
The presence of pilot and feasibility studies with small sample sizes introduces limitations in terms of statistical power and generalizability. Consequently, the conclusions drawn from such studies may lack sufficient evidence to support their validity, necessitating caution in extrapolating findings to broader populations.
The inherent inability to blind participants to music interventions poses a methodological limitation, as it introduces the potential for bias and confounding variables. Furthermore, the possibility of non-musical interactions during group interventions complicates the interpretation of results and calls for rigorous control measures in future research.
Variations in musical parameters, participant music preferences and cultural connotations of music introduce complexity into the study of oxytocin’s mediating role. Failure to account for these factors adequately may obscure the true mechanisms underlying oxytocin modulation in response to music, highlighting the need for more nuanced approaches in future investigations.
The predominance of cross-sectional or acute intervention studies limits our understanding of the long-term effects of music on oxytocin modulation. Longitudinal studies tracking changes in oxytocin levels over extended periods are needed to elucidate the sustained impact of music interventions on social bonding and stress regulation.
Implications of the results for clinic, future research
The heterogeneous nature of the included studies presents challenges in comparing and synthesizing findings. Future research should strive for more standardized study designs and interventions to facilitate meaningful comparisons. Specifically, attention should be paid to controlling for confounding variables and ensuring consistency in music interventions across studies.
The influence of individual characteristics such as music preferences, cultural backgrounds and musical experiences on oxytocin response remains poorly understood. Future studies will, therefore, need to explore how these factors interact with music interventions to better elucidate the mechanisms underlying oxytocin modulation.
Furthermore, the high intra-individual variability in oxytocin levels underscores the need for a deeper understanding of the oxytocinergic system and the contextual factors that influence oxytocin release and its subsequent effects. Beyond this, research will need to further investigate how social contexts, collaborative actions and environmental stressors modulate the oxytocin response during musical interventions.
Another area for further investigation is the relationship between oxytocin and cortisol levels in response to music interventions. Future studies should incorporate measures of HPA-axis activity as an essential tool to disentangle the interplay between stress regulation and social bonding processes mediated by oxytocin.
Moreover, inconsistencies in oxytocin measurement methods and discrepancies between peripheral and central oxytocin concentrations necessitate methodological refinement. Future research should aim for standardized approaches to sample collection, assay techniques and correlation between peripheral and central oxytocin levels to improve the reliability and validity of findings.
With respect to speech or singing interventions, the impact of altered salivary flow on measured oxytocin levels requires further investigation. Future studies should consider the potential distortion of oxytocin levels due to variations in salivary flow and explore methods to account for these effects in data analysis.
In terms of analysing the study results, a more detailed investigation into the influence of age, gender, sample size and measurement instrument on the direction of the pre–post outcomes may be of particular interest for subsequent studies. As most studies included in this review examined a relatively homogeneous group of participants, considering a more heterogeneous study population could be valuable for conducting further studies to gain a better understanding of the key factors influencing oxytocin levels and relating these to clinical parameters.
Conclusion
The data from this review provides an insight into the complexity of oxytocin in the musical domain. To better understand the response of oxytocin to music interventions, studies with larger numbers of cases, controlling for contextual factors, individual differences and HPA-axis activity will need to be conducted. Only a better understanding of the physiological functioning of the oxytocinergic system will allow oxytocin to be used as a therapeutical agent.
Footnotes
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
