Abstract
Background:
Patients with severe euvolemic hyponatremia can be challenging to treat, and the COVID-19 pandemic has added to the complexity of the situation.
Methods:
Data were collected from all patients treated in a local intensive care unit (ICU) with the protocolized proactive desmopressin plus 3% saline (PD-3 S) approach in March 2022.
Results:
Six patients were treated with the protocol, whose initial serum sodium level (SNa) ranged from 98 to 108 mmol/L. SNa of one patient dropped further due to delayed initiation of protocolized PD-3 S because of access block. But otherwise, the protocol had been effective in achieving a controlled rise of SNa in all patients. No major complications have been observed, and the feedback from both medical and nursing staff was overall positive.
Conclusion:
This case series shows that the protocolized PD-3 S is safe and effective. This protocol is currently being used in two ICUs in our cluster. And our unit is currently working with the emergency department to initiate the protocol earlier, before ICU admission.
Keywords
Introduction
Severe hyponatremia is defined as serum sodium level (SNa) below 120 mmol/L. If left untreated, it can lead to cerebral edema, seizures, or even brainstem herniation and death, whereas overcorrection of SNa more than the recommended range may result in osmotic demyelination syndrome, another devastating complication. 1 Current expert guidelines recommend limiting SNa rise to no more than 10 mmol/L in the first 24 h and no more than 8 mmol/L every 24 h thereafter.2,3 As such, patients with severe hyponatremia frequently warrant admission to the intensive care unit (ICU) for close monitoring and treatment.
Recently, we have observed a surge in the number of ICU admissions for patients with severe hyponatremia in our hospital. Many of these patients had concurrent or recent COVID-19 with or without clinical features of pneumonia. This observation is in line with published findings that COVID-19 is associated with hyponatremia. 4 In the context of the COVID-19 pandemic, there are competing priorities for ICU admission, which could contribute to patient boarding in the emergency department (ED) 5 , that is, access block situation. Given that SNa could change dramatically—sometimes even unpredictably—with treatment especially when frequent monitoring is not practically feasible due to resource limitation, exploring a way to treat these severely hyponatremic patients in a safe, efficient, and reliable manner is of utmost importance.
A proactive desmopressin plus 3% saline approach (PD-3 S) 6 has been described for treatment of severe euvolemic hyponatremia. The basic principles of PD-3 S involve
Regular desmopressin to limit urine free water loss.
Estimate the required sodium load to raise the SNa at a safe rate. Most equations (e.g. Adrogue-Madias formula) 7 are based on the Edelman equation. 8
Infuse 3% saline as the sodium source to replenish sodium deficit.
Close monitoring of SNa to adjust the rate of 3% saline infusion.
Limit the overcorrection of SNa rise with 5% dextrose water (D5 W) when indicated.
Simplistic as PD-3 S may seem, wide individual variations exist when adopting this approach, including the dose of desmopressin and the frequency of its administration, frequency of blood and urine sampling, preparation of 3% saline, the initial rate of 3% saline infusion, the route of administration of 3% saline, and the response to SNa deviations from the expected range. Furthermore, doctors newly rotating into our department may not be familiar with this approach. Therefore, we protocolized PD-3 S in order to create a framework that provides guidance to clinically safe and effective therapy, yet demanding minimal additional training or expertise to adopt, and is not overly rigid to impede the exercise of clinical judgment when indicated.
Methods
A protocol is created for PD-3 S, standardizing several important variables in practice. 9 The key steps are as detailed below:
Confirm inclusion and exclusion criteria Inclusion criteria: Severe hypo-osmolar euvolemic hyponatremia with SNa below 120 mmol/L Patient age at least 18 years
Exclusion criteria: Profound renal failure If control of body volume status is significantly affected by other clinical factors, for example, volume overload or when large volume fluid resuscitation may be needed Patients in whom bolus hypertonic therapy has recently been given
Regular desmopressin may still be started, and the clinician may consider initiating 3% saline infusion 24 h later if SNa rise is inadequate
Administer desmopressin 2 mg intravenously (IV) or subcutaneously immediately and then 1 mg IV every four hourly thereafter
Reliable IV access is established Peripheral cannulae are preferred Insertion of central venous catheter solely for 3% saline administration is deemed unnecessary
Start 3% saline infusion Dilution: 45 mL of 23.4% NaCl added into 455 mL of 0.9% NaCl Initial infusion rate according to patient’s body weight (BW) BW lower than 50 kg: start at 10 mL/h BW between 50 and 70 kg: start at 15 mL/h BW higher than 70 kg: start at 25 mL/h
Monitor paired serum and urine biochemistry every six hourly at a fixed schedule (or more frequently at the clinician’s discretion)
If SNa is out of target range, follow-up actions will be carried out: If rise of SNa is inadequate: 3% saline infusion rate will be increased by 5 mL/h If rise of SNa is excessive: D5 W bolus will be given, by default 250 mL over 30 min 3% saline will be withheld during D5 W infusion, and then be resumed at an infusion rate directed by the doctor
The SNa target range and 3% saline infusion rate will be reviewed at least once daily during morning round by the doctor.
Regular desmopressin and 3% saline infusion may be stopped once SNa reaches at least 125 mmol/L at the doctor’s discretion.
This protocolized PD-3 S has been regularly used since late February 2022 in our unit—a 18-bed ICU in a regional hospital with 697 inpatient beds and an ED with, on average, approximately 200 daily attendance. All junior ICU doctors were briefed about the protocol. An online platform was setup to provide them with easy access to information guiding the troubleshooting of some commonly encountered problems with the operation of the protocol. 9 When they are in any doubt, they are advised to consult the senior ICU specialist on duty for opinion.
After obtaining informed consent from the patients (written consent was preferable whenever feasible, but otherwise verbal consent was obtained in situations such as the patient being under isolation for active COVID-19 infection), we have collected the data from all patients with severe euvolemic hyponatremia treated in our ICU from 1 March 2022 to 31 March 2022 to observe the safety and efficiency of protocolized PD-3 S. Ethics approval was not required for the nature of this case series. After the aforementioned period, feedback on the protocol was collected from the staff involved, primarily junior doctors and senior nurses.
Results
In March 2022, six patients were admitted to the ICU for severe hyponatremia, of which five had concurrent or recent COVID-19. The patients’ baseline clinical characteristics and biochemical data are listed in Table 1. The trends of their SNa rise are plotted in Graph 1. In general, a controlled rise in SNa could be reliably achieved. The rate was in most instances within an acceptable range.
The baseline clinical characteristics and biochemical data of the hyponatremic patients.
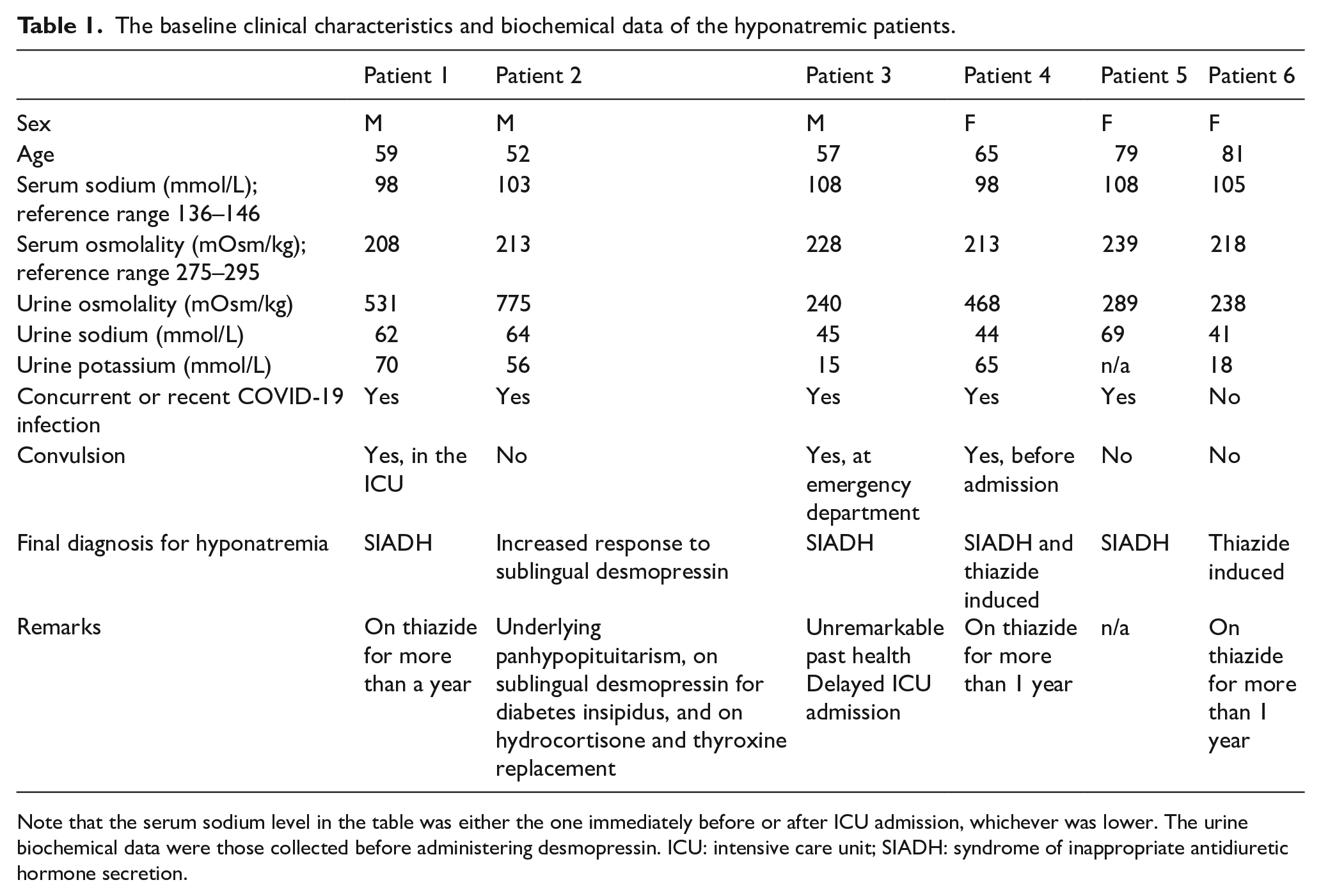
Note that the serum sodium level in the table was either the one immediately before or after ICU admission, whichever was lower. The urine biochemical data were those collected before administering desmopressin. ICU: intensive care unit; SIADH: syndrome of inappropriate antidiuretic hormone secretion.
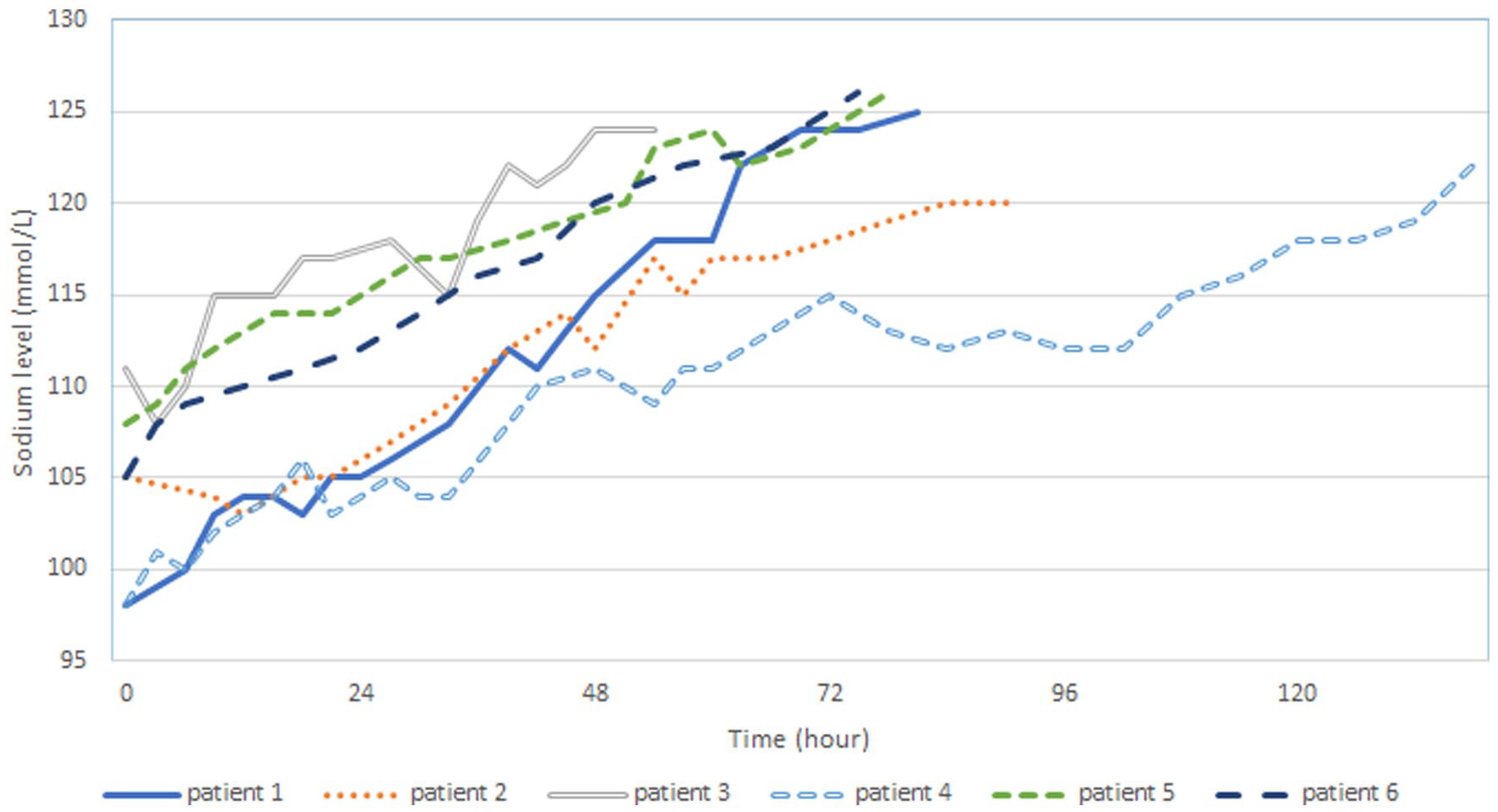
The trends of the patient’s sodium level. The sodium level of patients 2 and 3 showed an initial dip because they had waited several hours before ICU admission.
Patient 1 had a convulsion soon after admission to the ICU despite steady SNa rise with protocolized PD-3 S from 98 to 100 mmol/L. The seizure readily subsided with a 100-mL bolus of 3% hypertonic saline. Otherwise, no adverse reaction was observed with the protocolized PD-3 S approach in this patient.
Patient 3 had a convulsion in the ED and yet had been boarded at the resuscitation area for several hours as a vacant ICU bed was not immediately available. Because the protocolized PD-3 S was not started until his admission to the ICU, his SNa dropped initially despite already being given a bolus dose of hypertonic solution in the ED. Therefore, he was started on a higher initial rate of 3% saline infusion to keep his SNa on a rising trend. Nevertheless, his SNa rise in the initial 24 h was still within 10 mmol/L, and the infusion rate was subsequently reduced to avoid overly rapid correction.
Patient 4 had a seizure before attending the ED, and she subsequently developed some degree of renal salt wasting since day three of protocolized PD-3 S (corresponding to the SNa plateau close to the 72 h mark in Graph 1), for which 3% saline infusion rate had to be increased accordingly to compensate her renal sodium loss in addition to replacing sodium deficit.
Patient 2 had a relatively complex medical background. For his panhypopituitarism, he had been on sublingual desmopressin for central diabetes insipidus, as well as thyroxine and hydrocortisone replacement. While his biochemical and endocrine parameters had been stable for years, he presented with severe hyponatremia with concurrent COVID-19 infection. Despite the exact cause of his hyponatremia being uncertain when he first admitted to the ICU, protocolized PD-3 S was initiated (together with stress dose steroid) because the overriding priority then was to achieve tight control of his SNa as soon as possible. The risk of SNa further dropping—from escalating desmopressin dose—was considered minimal because his fluid and sodium input was strictly controlled by 3% saline infusion and kept nil by mouth. This approach has been proven effective and safe in this patient, and the endocrinologist could continue working up for the cause of his severe hyponatremia once PD-3 S was stopped after his SNa had reached a safer level.
All patients were discharged from ICU only when SNa was at least 120 mmol/L. The SNa rise in the first 24 h was within 8 mmol/L for all patients except patient 3 who received a bolus of hypertonic saline for his seizure in the ICU. D5 W was used to rectify SNa overshooting in five patients; three of whom required no more than 500 mL. The ICU length of stay for the majority, except patient 4 who had renal salt wasting, was approximately 3 days. Three-percent saline infusion via peripheral cannulae (20 gauge or 18 gauge cannulae over dorsum of hand or forearm) was well tolerated by all patients. No complication (e.g. pain, thrombophlebitis, or skin necrosis) was reported.
All the 16 senior nurses (advanced practice nurses, ward managers, and department operations manager) in our unit were interviewed, whose response was overall positive. They found protocolized PD-3 S to be less labor-intensive than non-protocolized approaches. As an example, predictability of the SNa rise allows blood-taking frequency to be safely reduced to every 6 h. Furthermore, instead of frequently changing the IV fluid between 0.9% saline, 3% saline, and D5 W, most of the time only the infusion rate of 3% saline had to be titrated to maintain a steady rise of SNa.
The experience of the junior ICU doctors was equally encouraging. All nine junior doctors in our unit found titrating 3% saline infusion simpler and easier to manage than frequent switching between various IV fluids. This also means they could be less distracted from other important clinical duties, particularly during a busy night shift.
Limitation
There are some limitations of this case series, including a small sample size, data collected over a short period of time, single-center setting, and primarily ICU-based experience. Therefore, the results may not be directly generalizable to non-ICU settings and thus required validation from further prospective studies before wider clinical adoption.
Discussion
Treatment of severe euvolemic hyponatremia remains a clinical challenge for physicians and intensivists because the renal response to the underlying etiology or to the therapy administered could be unpredictable and rapidly altering; in which case, SNa could change erratically no matter how careful the clinician calculates the sodium deficit or which equation he or she uses. PD-3 S makes use of desmopressin to limit one’s urine-free water loss, thereby reducing the degree of uncertainty; although in actual clinical practice, starting 3% saline with a weight-based infusion rate and titrating to response is likely more reliable than exclusively relying on any one equation. 10
Three-percent saline was once believed to be unsuitable for peripheral administration. However, its safety profile has been proven by many clinical studies. In fact, our experience is well aligned with that of other groups who found no significant complications associated with PD-3 S.6,10 Consequently, central venous catheter insertion solely for 3% saline infusion is likely not warranted.
We observed that patients with an initial clinical course similar to patient 3’s were not uncommon because the diagnosis of severe hyponatremia is usually not immediately evident when patients first present to the ED. And their initial SNa result may take several hours to become available, by then some IV fluid might have been administered already followed by a consequent renal response, at times leading to an unpredictable change in SNa. ED boarding due to COVID-19 could only complicate the matter even more.
Therefore, we have started working with our ED colleagues in the cluster hospitals, primarily aiming to initiate treatment at ED when ICU admission is expected to be delayed. With the advice from the intensivist, the emergency physician could help administer the first dose of desmopressin and stop all running IV fluid and insert a urinary catheter for the first set of urine biochemistry and to monitor urine output. To date, this approach has been working smoothly, and we have not yet encountered an abrupt, unexpected change of SNa in patients to whom desmopressin was started in the ED.
Because it is uncommon for these patients to experience ED boarding for more than 6 h in our hospital, we have not yet needed to start 3% saline infusion or administer a second dose of desmopressin at ED. While we prefer severe hyponatremia to be managed in the ICU setting whenever possible, we believe that our protocolized PD-3 S practice is safe to potentially be initiated in a non-ICU acute care setting, ED or acute medical wards, especially when a delay in ICU admission is anticipated or when competing priorities for admission exist.
Conclusion
Our case series have demonstrated that protocolized PD-3 S is a safe and highly effective approach for treating severe euvolemic hyponatremia and is showing promise in reducing manpower demand and clinicians’ cognitive load. Emergency physicians play an important role in the care of severely hyponatremic patients not only in the early diagnosis but also—especially in the contingency situation due to the current COVID-19 pandemic—in managing the severe symptoms from the low SNa and initiating therapy early on.
Further data collection is now underway to establish the efficiency of protocolized PD-3 S. This protocol has already been adopted in two ICUs in our cluster. Working closely with our nurses and our regional EDs, our group hopes to further simplify this protocol to ultimately enable its safe execution outside of higher dependency areas.
Footnotes
Acknowledgements
The authors would like to thank Dr Richard H. Sterns, MD, for his advice on the initial design of the treatment protocol.
Author contributions
Conceptualization: J.C.H.C., E.H.L.C., K.N.L. Data acquisition: J.C.H.C. Writing original draft: J.C.H.C. Review and editing: J.C.H.C., E.H.L.C., L.H., C.L.T., Y.Y.Y., K.N.L.
Availability of data and materials
The data sets analysed are available from the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was obtained from all the patients for their anonymized information to be published in this article.
Ethical approval
Ethics approval is not required for this case series.
