Abstract
Objective:
To externally validate the use of the Oakland score in identification of low-risk lower gastrointestinal bleeding patients who can be safely managed without hospitalisation in an accident and emergency department in Hong Kong.
Methods:
This single-centre retrospective cohort study included adult patients who attended the accident and emergency department for lower gastrointestinal bleeding and were subsequently admitted to hospital from 1 January 2020 to 31 December 2020. Safe discharge was defined as absence of all of the following adverse outcomes after hospital presentation: blood transfusion; therapeutic colonoscopy, mesenteric embolisation, or laparotomy for bleeding; in-hospital death (all causes); and readmission with subsequent lower gastrointestinal bleeding within 28 days. The sensitivities, specificities and area under the receiver-operating characteristic curve of the Oakland score were calculated.
Results:
Among 376 patients who were included in this study, 114 (30.3%) of them experienced one or more adverse outcomes. The area under the receiver-operating characteristic curve for safe discharge was 0.88 (95% confidence interval, 0.84–0.91). Using an Oakland score threshold of ⩽8 points as in the original derivation study can achieve 100% sensitivity (95% confidence interval, 96.8%–100%) and 7.3% specificity (95% confidence interval, 4.4%–11.1%) for safe discharge. When the Oakland score threshold was extended to ⩽11 points, a much greater proportion of low-risk patients could be identified, with 97.4% sensitivity (95% confidence interval, 92.5%–99.5%) and 32.8% specificity (95% confidence interval, 27.2%–38.9%) for safe discharge.
Conclusion:
The Oakland score performed well in identification of lower gastrointestinal bleeding patients who were at low risk of experiencing adverse outcomes and could thus be safely managed without hospitalisation. Extension of the Oakland score threshold to ⩽11 points can allow identification of a greater proportion of low-risk patients while sensitivity is reasonably maintained.
Keywords
Introduction
Lower gastrointestinal bleeding (LGIB) is a common presentation in the emergency department and a common cause of hospital admission. In Hong Kong, LGIB had actually surpassed upper gastrointestinal bleeding (UGIB) as the leading source of bleeding among patients who are hospitalised for gastrointestinal bleeding, and it was predicted that there will be a further increase in the incidence of LGIB in the future. 1 In the United States, LGIB is the second most common reason for emergency department visit due to gastrointestinal disease, with more than 40% of the cases requiring hospital admission. 2 However, not all patients with LGIB require immediate interventions such as blood transfusion, therapeutic endoscopy, mesenteric embolisation or surgery. In fact, LGIB usually follows an indolent course. Bleeding stops spontaneously in 80% to 85% of the cases, 3 and the mortality rate is between 2% and 4%.3,4
The European Society of Gastrointestinal Endoscopy (ESGE) and the British Society of Gastroenterology recommended that in patients presenting with a self-limited bleed and no adverse clinical features, an Oakland score of ⩽8 points can be used to guide the clinician decision to discharge the patient for outpatient investigation.5,6 The Oakland score was developed in 2017 within a nationally representative sample of patients in the United Kingdom to identify patients who were at low risk of experiencing adverse outcomes and could thus be safely discharged, avoiding hospital admission. 7 This scoring system makes use of clinical and laboratory parameters for risk stratification. It contains seven variables (age, sex, previous hospital admission with LGIB, digital rectal examination results, heart rate, systolic blood pressure and haemoglobin concentration) and ranges from 0 to 35 points (Table 1). A higher score indicates a higher risk of experiencing an adverse outcome. An adverse outcome is defined as occurrence of any one of the following after hospital presentation: blood transfusion; therapeutic colonoscopy, mesenteric embolisation or laparotomy for bleeding; in-hospital death (all causes); and readmission with subsequent LGIB within 28 days. The original derivation and validation study stated that a score of ⩽8 predicted a 95% probability of safe discharge with the absence of any adverse outcome. In a subsequent external validation study, it was found that the Oakland score threshold to identify low-risk patients could be extended from ⩽8 points to ⩽10 points to allow identification of a greater proportion of low-risk patients. 8 In that external validation study, a score threshold of ⩽8 points identified 8.7% of patients as low risk with a sensitivity for safe discharge of 98.4%, while extension of the score threshold to ⩽10 points identified 17.8% of patients as low risk with a sensitivity for safe discharge of 96%. The aim of this study is to externally validate the Oakland score in Hong Kong, assess its performance and obtain an optimal score threshold to identify low-risk LGIB patients in Hong Kong who can be safely managed without hospitalisation.
Variables of the Oakland score.

LGIB: lower gastrointestinal bleeding.
Methods
Study design and setting
This was a retrospective cohort study conducted in the accident and emergency department of a regional hospital (Kwong Wah Hospital) in Hong Kong. Kwong Wah Hospital is a 24-h regional acute hospital with an annual accident and emergency department attendance of around 100,000. This study was approved by the Ethics Committee of the Kowloon Central/Kowloon East Cluster of the Hospital Authority. Written informed consent was not necessary because no identifiable patient data was included in the study.
Study population
Patients aged 18 or above who attended the emergency department of Kwong Wah Hospital for LGIB subsequently admitted to hospital from 1 January 2020 to 31 December 2020 were included. Patients with LGIB presented with bright, dark red blood or clots per rectum, blood mixed in with stool, or melaena without haematemesis. Exclusion criteria were the following: haematemesis or coffee ground vomiting on presentation; stomal bleeding; presence of another acute illness in addition to LGIB which warranted hospital admission; proven findings of UGIB on oesophagoduodenogastroscopy (OGD); development of LGIB after hospital admission; or patients who were transferred between hospitals or other treatment facilities.
Data collection
Patient data was retrieved through the Clinical Data Analysis and Reporting System (CDARS) of the Hospital Authority. The International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) code was used for case identification. The diagnoses of gastrointestinal haemorrhage (ICD-9-CM 578), diverticulosis of small intestine with haemorrhage (ICD-9-CM 562.02), diverticulitis of small intestine with haemorrhage (ICD-9-CM 562.03), diverticulosis of colon with haemorrhage (ICD-9-CM 562.12), diverticulitis of colon with haemorrhage (ICD-9-CM 562.13), haemorrhage of rectum and anus (ICD-9-CM 569.3), angiodysplasia of intestine with haemorrhage (ICD-9-CM 569.85), haemorrhoids (ICD-9-CM 455), malignant neoplasm of small intestine including duodenum (ICD-9-CM 152), malignant neoplasm of colon (ICD-9-CM 153), and malignant neoplasm of rectum, rectosigmoid junction and anus (ICD-9-CM 154) were used to retrieve patient records. Data including all the variables needed for calculation of the Oakland score and patient outcome were collected from the Clinical Management System. The Oakland score was calculated based on the criteria reported in the original study (Table 1). 7 Data collection and review of patient records were carried out by the author of this study.
Outcome measure
The primary outcome measure of this study was the area under the receiver-operating characteristic curve (AUC), which was used to assess the discriminatory performance of the Oakland score. Safe discharge was defined as the absence of all of the following after hospital presentation: blood transfusion; therapeutic colonoscopy, mesenteric embolisation or laparotomy for bleeding; in-hospital death (all causes); and readmission with subsequent LGIB within 28 days. These composite outcomes were used in previous Oakland score validation studies.
Statistical analysis
Estimates of the sensitivity and specificity with 95% confidence interval (CI) were calculated at each potential decision threshold for the Oakland score. The performance of the Oakland score was assessed by the AUC. The optimal cutoff score for safe discharge of low-risk LGIB patients with at least 95% sensitivity and at the same time the highest specificity was identified. Statistical analysis was performed with the software MedCalc version 20.215.
Sample size calculation
With AUC being the primary outcome measure, the sample size required to obtain a 95% CI with width ⩽0.1 is at least 265. This is based on the numbers in the validation study by Oakland et al. 8 (AUC = 0.87, proportion of subjects with safe discharge = 0.479), and the formulas by Zhou et al. 9
Results
A total of 792 patients were identified through the CDARS initially. Of those, 416 patients were excluded, including 157 patients who did not suffer from gastrointestinal bleeding, 113 patients with haematemesis or coffee ground vomiting, 5 patients with stoma bleeding, 64 patients with the presence of another acute illness in addition to LGIB which warranted hospital admission, 10 patients with findings of UGIB on OGD, 31 patients with development of LGIB after hospital admission, 35 patients who were transferred between hospitals and 1 patient with missing laboratory data.
As a result, a total of 376 patients were included in the study, of which 195 (51.9%) were men. The median age of the study population was 75 years (interquartile range (IQR), 62–85). There were 35 (9.3%) patients receiving oral anticoagulant medications at the time of hospital admission (Table 2).
Patient demographic characteristics.

IQR: interquartile range; LGIB: lower gastrointestinal bleeding.
Overall, 114 (30.3%) patients experienced one or more adverse outcomes, while 262 (69.7%) patients did not experience any adverse outcome and therefore met the criteria for safe discharge. In-patient colonoscopy was performed in 30 patients (8%). Among those who experienced adverse outcomes, 81 patients received blood transfusion, 7 patients received therapeutic colonoscopy for haemostasis, 2 patients received laparotomy for refractory bleeding, 11 patients died during hospitalisation and 38 patients were readmitted subsequently with LGIB. No patient underwent mesenteric embolisation.
The Oakland score was calculated in all 376 patients. The median Oakland score was 16 points (IQR, 12–22 points). The sensitivity and specificity for safe discharge at different cutoff points are shown in Table 3. A sensitivity of 97.4% (95% CI, 92.5%–99.5%) for safe discharge can be achieved at an Oakland score of ⩽11 points, with a specificity of 32.8% (95% CI, 27.2%–38.9%). The AUC of the composite outcome of safe discharge was 0.88 (95% CI, 0.84–0.91) (Figure 1). For the three patients who had an Oakland score of ⩽11 and experienced adverse outcomes, one of them required blood transfusion, while two of them were readmitted with subsequent LGIB within 28 days.
Sensitivity and specificity for safe discharge at different cutoff points.
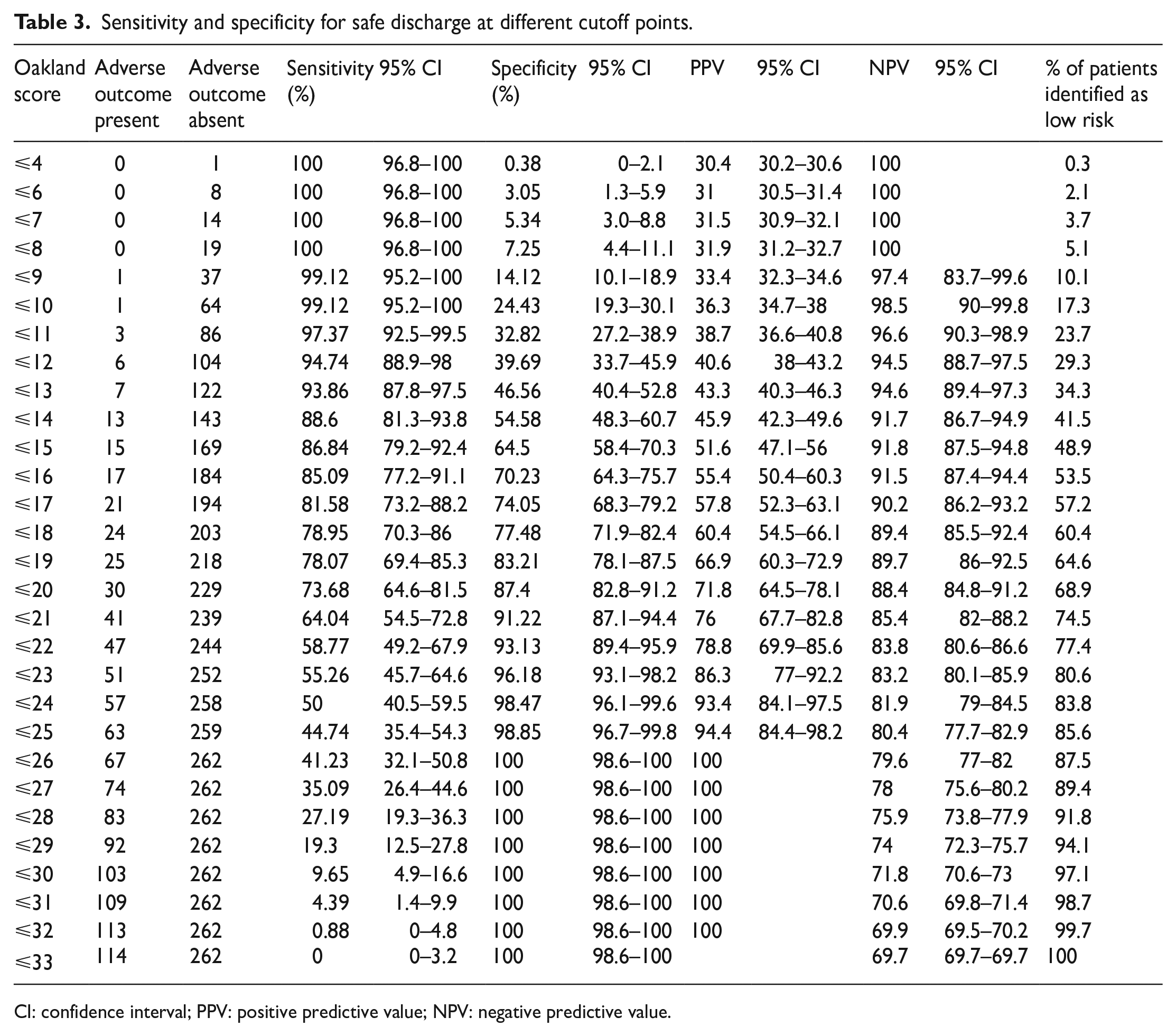
CI: confidence interval; PPV: positive predictive value; NPV: negative predictive value.
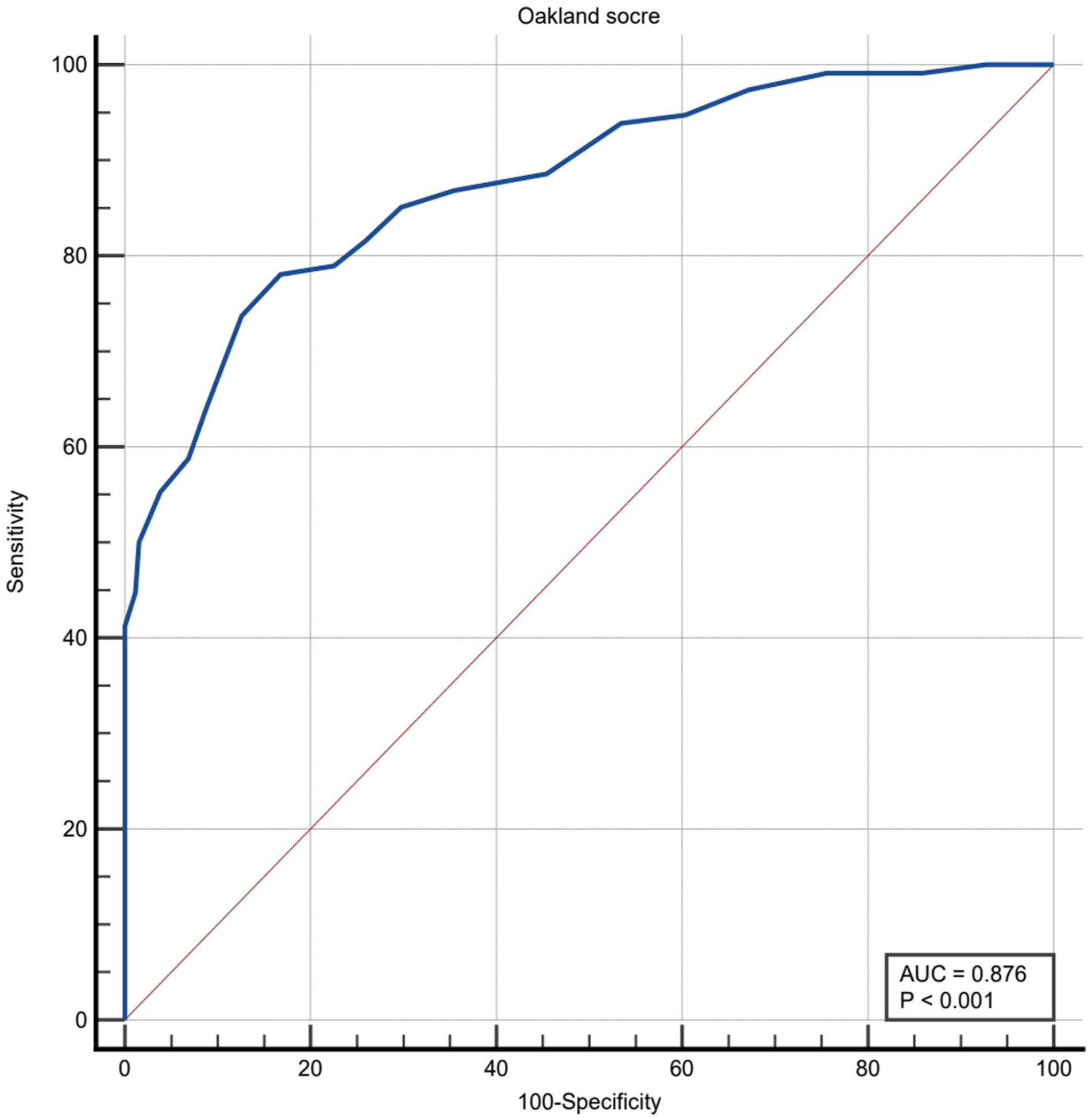
Receiver-operating characteristic (ROC) curve of the Oakland score for safe discharge.
Discussion
Over the years, numerous LGIB risk stratification scores have been developed. 10 Examples of such scores include the Oakland score, the BLEED (ongoing bleeding, low systolic blood pressure, elevated prothrombin time, erratic mental status and unstable comorbid disease) score, the NOBLADS (nonsteroidal anti-inflammatory drug use, no diarrhoea, no abdominal tenderness, blood pressure ⩽ 100 mmHg, antiplatelet drug use (nonaspirin), albumin < 3.0 g/ dL, disease score ⩾2 (according to the Charlson Comorbidity Index) and syncope) score, the Strate score and the Sengupta score. Some of the scores have been developed to identify low-risk patients, while others have been developed to predict severe bleeding, in-hospital complication or death. According to a recent systematic review and meta-analysis, the Oakland score was the most discriminative LGIB risk score for predicting safe discharge, major bleeding and need for transfusion. 11 The Oakland score was thus selected in the current study to be the risk score of choice to identify low-risk LGIB patients who can be safely managed without hospitalisation.
To the best of the author’s knowledge, this study is the first Oakland score validation study in a Chinese population. The Oakland score was previously validated in the United Kingdom, 7 the United States 8 and Vietnam, 12 and it was shown that the score can perform well in predicting safe discharge.
The AUC of 0.88 in the present study is in accordance with the AUC in previous external validation studies.8,12 This shows that the Oakland score can perform equally well in Hong Kong in identifying LGIB patients who are at low risk of adverse outcomes. Determining the cutoff score for safe discharge requires balancing the risk of patient misclassification with the need to include a greater proportion of low-risk patients for the score to be useful clinically. In the current study, using an Oakland score of ⩽8 points as a cutoff can achieve 100% sensitivity for safe discharge, but only 19 (5.1%) patients were identified as low risk. If a score threshold of ⩽11 points was used instead, 89 (23.7%) patients were identified as low risk, with a sensitivity of safe discharge more than 97%.
By identifying low-risk patients who can be safely discharged from the emergency department, unnecessary hospitalisations can be reduced. Longer hospital stays can be harmful, particularly for older patients, and avoidance of unnecessary hospital admissions can reduce the risk of inpatient complications, such as hospital-acquired infections and falls. 13 In addition, by reducing the number of hospitalisations, access block in the emergency department may be alleviated, while resources can be saved and re-invested into other areas. In 2020, the cost of general ward inpatient service in a public hospital in Hong Kong was $7240 HKD per patient day. 14 If the Oakland score threshold of ⩽11 points was used to identify low-risk LGIB patients who do not need hospitalisation, at least $644,360 HKD could be potentially saved within the study population alone.
One point to note is that the Oakland score may underestimate disease severity in patients who present early when haemoglobin drop has not yet occurred, or in patients who are resuscitated in the prehospital setting with improved haemodynamic status upon arrival to the emergency department. It is also noteworthy that the use of the Oakland score to discharge low-risk LGIB patients from the emergency department should be complemented with arrangement of appropriate outpatient follow-up. A stable patient with self-limited LGIB may not need inpatient management, but should be seen by a surgeon or gastroenterologist early, preferably within 28 days, so that appropriate gastrointestinal workup can be arranged. This is especially important if the patient is an elderly or there are risk factors for gastrointestinal malignancy. In view of the long waiting time for new case booking for specialist outpatient clinic in Hong Kong, if the Oakland score is to be widely adopted locally, the emergency department may need to collaborate with the surgical department to launch a fast track clinic for low-risk LGIB patients who are discharged from the emergency department.
There are several limitations to this study. First of all, this is a retrospective study which only included patients who were admitted to the hospital with LGIB, while those who were directly discharged from the emergency department were not included. In order to include all patients who attended the emergency department for LGIB, a prospective study is needed. Second, since only medical records from public hospitals can be retrieved through the CDARS, it is possible that discharged patients might have received medical care in private hospitals subsequently and that data were not included in the analysis. Third, whether to transfuse blood or perform therapeutic colonoscopy or surgery was subject to the decision of the attending surgeon or gastroenterologist. In addition, some of the admitted patients received oral or intravenous tranexamic acid, and this might also have an effect on the need for other interventions.
Conclusion
The Oakland score can identify LGIB patients who are at low risk of experiencing adverse outcomes and could thus be safely discharged from the emergency department. The original Oakland score threshold of ⩽8 points can be extended to ⩽11 points to allow identification of a greater proportion of low-risk patients while sensitivity is reasonably maintained.
Footnotes
Acknowledgements
The author would like to thank the Clinical Research Centre of the Kwong Wah Hospital for its assistance in this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Availability of data and materials
The datasets generated and/or analysed during the current study are available from the corresponding author.
Informed consent
Informed consent was waived by the Research Ethics Committee (Kowloon Central/Kowloon East).
Ethical approval
Ethical approval for this study was obtained from the Research Ethics Committee (Kowloon Central/ Kowloon East; Ref: KC/KE-22-0057/ER-4).
