Abstract
Background:
Analgesic poisoning is common in Hong Kong. Analgesics containing salicylate are easily available. Salicylate poisoning may be encountered in the daily practice of the accident and emergency department. The issue of routine salicylate screening was discussed in a number of articles, and the viewpoints of most authors were that routine salicylate screening was unnecessary.
Objectives:
The primary outcome is to explore the predictive factors of supratherapeutic peak serum salicylate level (>2.17 mmol/L) and delineate the role of routine screening serum salicylate level in poisoned patients attending the accident and emergency department.
Methods:
This is a retrospective cohort study. Poisoning cases presented to Hong Kong Poison Information Centre between 1 July 2008 and 31 October 2020 with serum salicylate level checked were included. The demographics, laboratory parameters and clinical outcomes were reviewed.
Results:
There were 8673 cases included with serum salicylate level checked, accounting for 17% of all poisoning cases. Sixty-six (0.76%) cases had supratherapeutic peak serum salicylate level and 11 (0.13%) of them were undeclared salicylate ingestion. Worse clinical outcomes were observed in cases with supratherapeutic peak serum salicylate level. Univariate analysis showed that tinnitus, hyperthermia, alkalaemia, metabolic acidosis, respiratory alkalosis and therapeutic use of aspirin were significantly associated with supratherapeutic peak serum salicylate level. Variables with p < 0.3 were entered into the backward stepwise logistic regression by likelihood ratio. A clinical prediction rule with three attributes (tinnitus, therapeutic use of aspirin and partial pressure of CO2 <5 kPa) was derived. It showed 100% sensitivity in internal validation.
Conclusion:
Serum salicylate level should be checked in poisoned patients with (1) tinnitus, (2) the therapeutic use of aspirin or (3) partial pressure of CO2 <5 kPa in arterial or venous blood gas. Routine salicylate screening remained a tool for clinicians not experienced in toxicology.
Introduction
Analgesic is one of the most commonly involved poisons in Hong Kong every year.1 –12 Since analgesics containing salicylates are readily available, salicylate poisoning may be encountered in the daily practice of the accident and emergency (A&E) department.
The typical symptoms of salicylate poisoning include tachypnoea, tinnitus, deafness, lethargy, seizures, confusion or even coma.13,14 When a history of salicylate overdose is available, the diagnosis of salicylate poisoning is usually not difficult for most emergency physicians. However, in situations when the patient deliberately hid the salicylate overdose history or the patient is unaware of an accidental salicylate overdose, the diagnosis of salicylate poisoning relies on the clinical features and serum salicylate level screening. In general, the possibility of salicylate poisoning needs to be considered in patients with unexplained tachypnoea, fever and metabolic acidosis. 15
Whether or not to perform screening of serum salicylate level routinely remained debatable. Some authors suggested that routine screening of salicylate was not necessary. A paper published in 1991 showed that a positive history of salicylate ingestion, tinnitus and hyperventilation were the best predictive indicators of salicylate ingestion. A thorough history and clinical assessment were better than routine analytical screening of salicylate. 16 Another 1996 paper also concluded that universal screening for serum salicylate level was neither cost-effective nor indicated, and salicylate poisoning should be suggested by high anion gap metabolic acidosis or altered mental state. 17 In a 2005 paper, negative history of salicylate ingestion had a high negative predictive value (NPV) of 98% for undetectable serum salicylate level. 18 The findings in the above studies concur with that in Hong Kong. A study in Hong Kong disclosed only negligibly above normal plasma salicylate concentrations in 1.1% of patients who were not suspected of ingestion of salicylate. 19 In another study, merely 1.7% of patients who were clinically salicylate-exposure negative were found to have either mildly elevated plasma salicylate levels or be mildly symptomatic. 20 Moreover, a local prospective observational study demonstrated that there was no undeclared salicylate ingestion if the patient was fully conscious. 21
However, it is arguable that the clinician should have a low threshold to obtain a serum salicylate level as the clinical effects of salicylate poisoning are non-specific; history may be generally unreliable in a suicidal patient or an unresponsive patient, and a lack of metabolic acidosis does not rule out the possibility of salicylate poisoning. 22 Together with routine serum paracetamol screening, serum salicylate screening for intentional poisoning patients is still a common practice by emergency physicians in Hong Kong.
This study aimed to explore the predictive factors of supratherapeutic peak serum salicylate level indicating possible salicylate poisoning and develop a clinical prediction rule in cases of undeclared ingestion and delineate the role of routine screening serum salicylate level in patients with suspected poisoning attending the A&E department.
Methodology
Study setting
This is a retrospective cohort study. All consultations and reportings to the Hong Kong Poison Information Centre (HKPIC) from 1 July 2008 to 31 October 2020 were enrolled on this study. The HKPIC provides 24-h territory-wide telephone consultation to all health care professionals in Hong Kong on poison information and management advice on clinical toxicology. It also received reporting cases from the Hong Kong Hospital Authority (HA), mainly the A&E department. In 2019, the HKPIC received 2100 consultations and 1916 reporting cases in total. 12
All data were extracted from the Poison Information and Clinical Management System, the database of the HKPIC, and Clinical Data Analysis and Reporting System (CDARS), the database of the HA. Patient demographics (gender and age), the intention of poisoning, symptomology (tinnitus, hyperventilation or dyspnoea), initial consciousness level, suspected poison involved, any therapeutic use of aspirin, laboratory parameters (initial and peak serum salicylate level, and first blood gas parameters) and clinical outcomes (length of stay, admission to intensity care unit (ICU)/high dependency unit (HDU), requirement of endotracheal intubation, requirement of extracorporeal treatment and mortality) were also collected.
The poisons involved were evaluated for any salicylate in the ingredients by looking up in the database of the Hong Kong Drug Office 23 for registered pharmaceuticals and the database of the Chinese Medicine Council of Hong Kong for over-the-counter proprietary Chinese medicines. 24
Patient inclusion
Patients with serum salicylate level measured, either taken in the A&E department or the inpatient ward, were included. Exclusion criteria included non-hospital authority patients, clinical admissions without A&E department attendance and inpatient poisoning. The reason for such exclusion criteria was that the details of the poisoning were usually not traceable, including the timing, symptoms, initial vitals and subsequent blood results.
Definitions
Patients were divided into groups by peak serum salicylate level. There were changes in reporting of the lowest serum salicylate level in Hong Kong throughout the study period. The values below reporting limit were shown as 0 in the database. A pharmacokinetic study showed that a serum salicylate level between 1.08 mmol/L and 2.17 mmol/L should be regarded as a therapeutic concentration. 25 We adapted half of the lower limit, which is 0.5 mmol/L, as a cut-off of a detectable level. Supratherapeutic peak serum salicylate level was defined as higher than 2.17 mmol/L, based on the same pharmacokinetic study.
Acidaemia and alkalaemia were defined by the presenting pH being <7.35 and >7.45, respectively. The presenting partial pressure of CO2 (pCO2) was defined as low if it was lower than 4.6 kilo-Pascal (kPa) and high if it was >6 kPa. The presenting base excess (BE) was defined as low if it was <−2 mmol and high if it was >+2 mmol.
An altered mental state was defined as Glasgow Coma Scale (GCS) <15 or not alert on the AVPU (alert, verbal, pain, unresponsive) scale upon presentation. Clinical hyperventilation was defined as a respiratory rate of >20/min or any respiratory distress reported by the referring clinician. Length of stay was rounded up to the nearest integer and discharge in A&E department is defined as 0 day.
Declaration of intake of salicylate-containing poison was evaluated by individual record review in the group with detectable peak serum salicylate level. Declared salicylate poisoning was defined as documented exposure to salicylate-containing poison in the A&E department record.
The outcomes of the cases were classified by senior doctors in HKPIC into five grades: no effect, mild effect, moderate effect, major effect or death according to the American Association of Poison Control Centers’ National Poison Data System. The poisoning–outcome relationship was graded as definite, probable, possible, not related or undetermined/not applicable according to the available information. The cases were further reviewed by a second senior doctor if they were classified into major effect or death. 12
Statistical analysis
Data analysis was performed by using the software SPSS v26. Categorical data were reported as frequency and percentage and continuous data were presented as median and interquartile ranges. In the univariate analysis, the chi-square test (Fisher’s exact test when appropriate) and the Mann–Whitney U test were employed for the analysis of categorical and continuous variables, respectively. A p value < 0.05 was considered significant.
The whole data set was divided into a derivation set and a validation set in a 2:1 ratio, stratified by with or without supratherapeutic peak serum salicylate levels. A multivariate logistic regression analysis was applied for significant variables. The resulting model in the derivation set was assessed by the Hosmer–Lemeshow test and area under the receiver-operating characteristic curve (AUROC).
A clinical prediction rule was then derived and assessed in the validation set. The sensitivity, specificity, positive predictive value (PPV), NPV, positive likelihood ratio (PLR) and negative likelihood ratio (NLR) were reported.
Results
Throughout the study period, there were in total 10,764 records with serum salicylate level measured (Figure 1). Of those, 8673 records remained after exclusion. Most of them were female (66.9%). The median age was 36 years old. The majority (75%) had suicidal or self-harm intentions. Salicylate-containing poisons were identified in 5.2% of the case population. Table 1 showed the summaries of different groups.
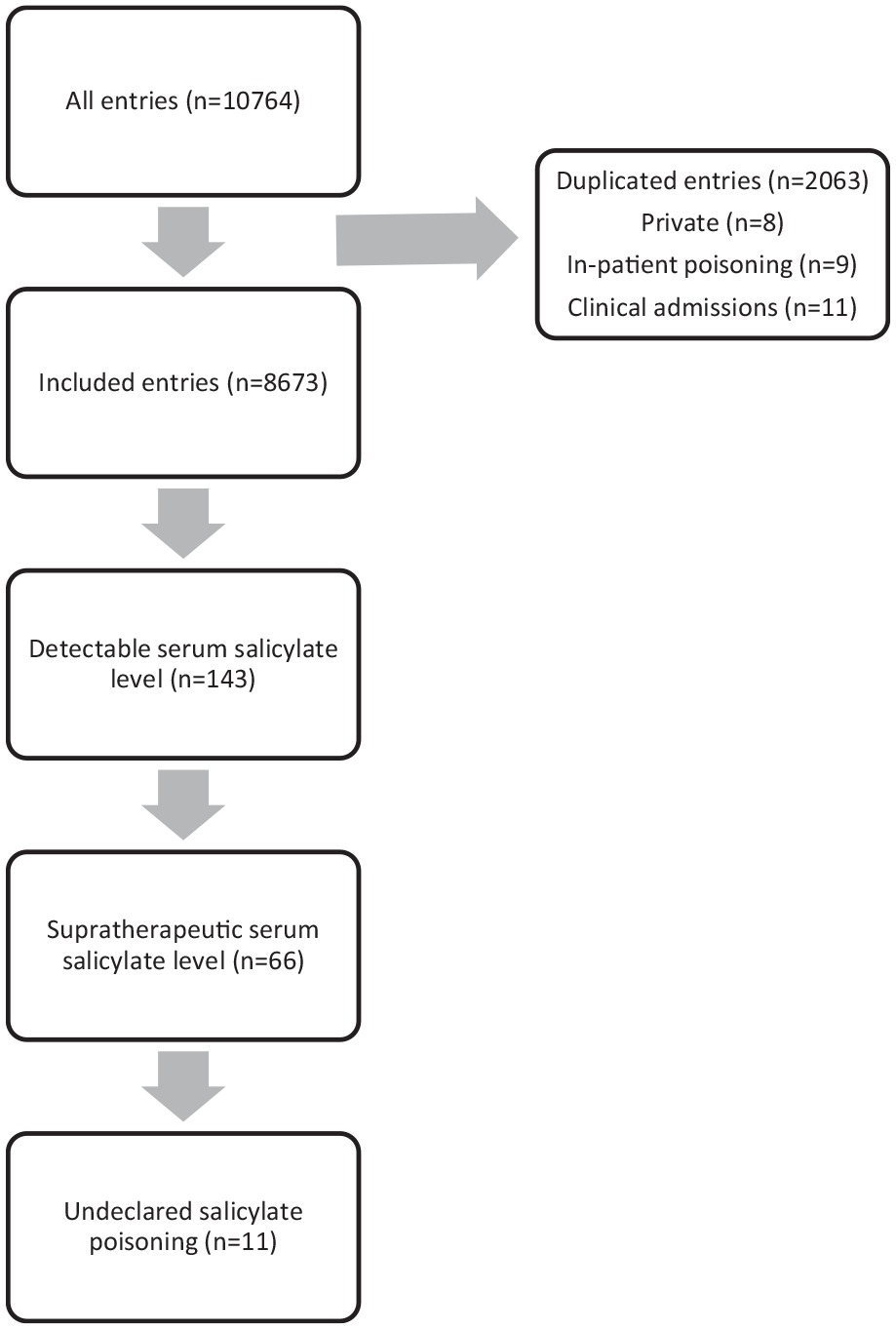
Flowchart of recruitment.
Clinical characteristics and outcome of the cohort of poisoning patients.
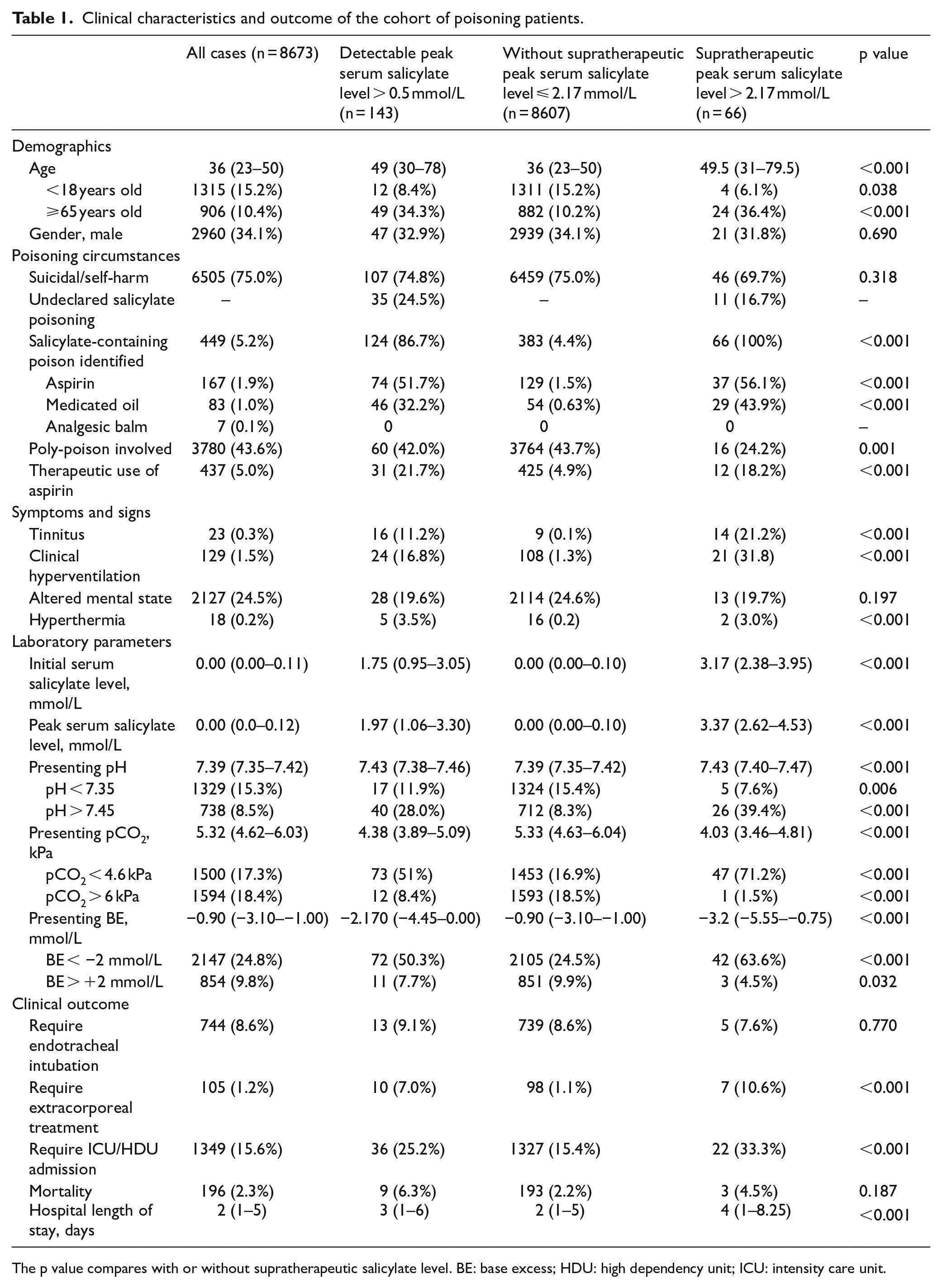
The p value compares with or without supratherapeutic salicylate level. BE: base excess; HDU: high dependency unit; ICU: intensity care unit.
Supratherapeutic peak serum salicylate level
Sixty-six cases (0.76%) were found to have supratherapeutic peak serum salicylate level. There was no significant difference in gender and intention of poisoning to self-harm in cases with or without supratherapeutic peak serum salicylate level. Elderly patient (⩾65 years old) was found to be more likely to have supratherapeutic peak serum salicylate level (p < 0.001). There was also a significantly higher ratio of the therapeutic use of aspirin in those with supratherapeutic peak serum salicylate level.
For presenting symptoms, tinnitus, clinical hyperventilation and hyperthermia, but not altered mental state, were associated with supratherapeutic peak serum salicylate level (both p < 0.001). Alkalaemia (p < 0.001), low BE (p < 0.001) and respiratory alkalosis (p < 0.001) were observed more frequently in this group of patients as well.
Patients with supratherapeutic peak serum salicylate level were more likely to require ICU/HDU admission and extracorporeal treatment (p < 0.001). They were more likely to have a longer length of stay (p < 0.001). The mortality was higher in the group with supratherapeutic peak serum salicylate level (4.5% vs 2.2%) but was not statistically significant (p = 0.187).
For salicylate-containing poison involved in this group of patients, 37 cases (56.1%) involved aspirin and 29 cases (43.9%) involved medicated oil containing methylsalicylate.
Undeclared salicylate exposure
Thirty-five patients did not declare exposure to salicylate poison in A&E department but were found to have detectable peak serum salicylate level. Of those, 11 patients had supratherapeutic peak serum salicylate level, which made up 0.13% of all cases in this study.
There was no significant difference between declared and undeclared salicylate exposure in the group with supratherapeutic peak serum salicylate level, except for lower pCO2 and longer length of stay in the hospital (Table 2).
Comparison of undeclared and declared salicylate exposure in patients with supratherapeutic peak serum salicylate level.
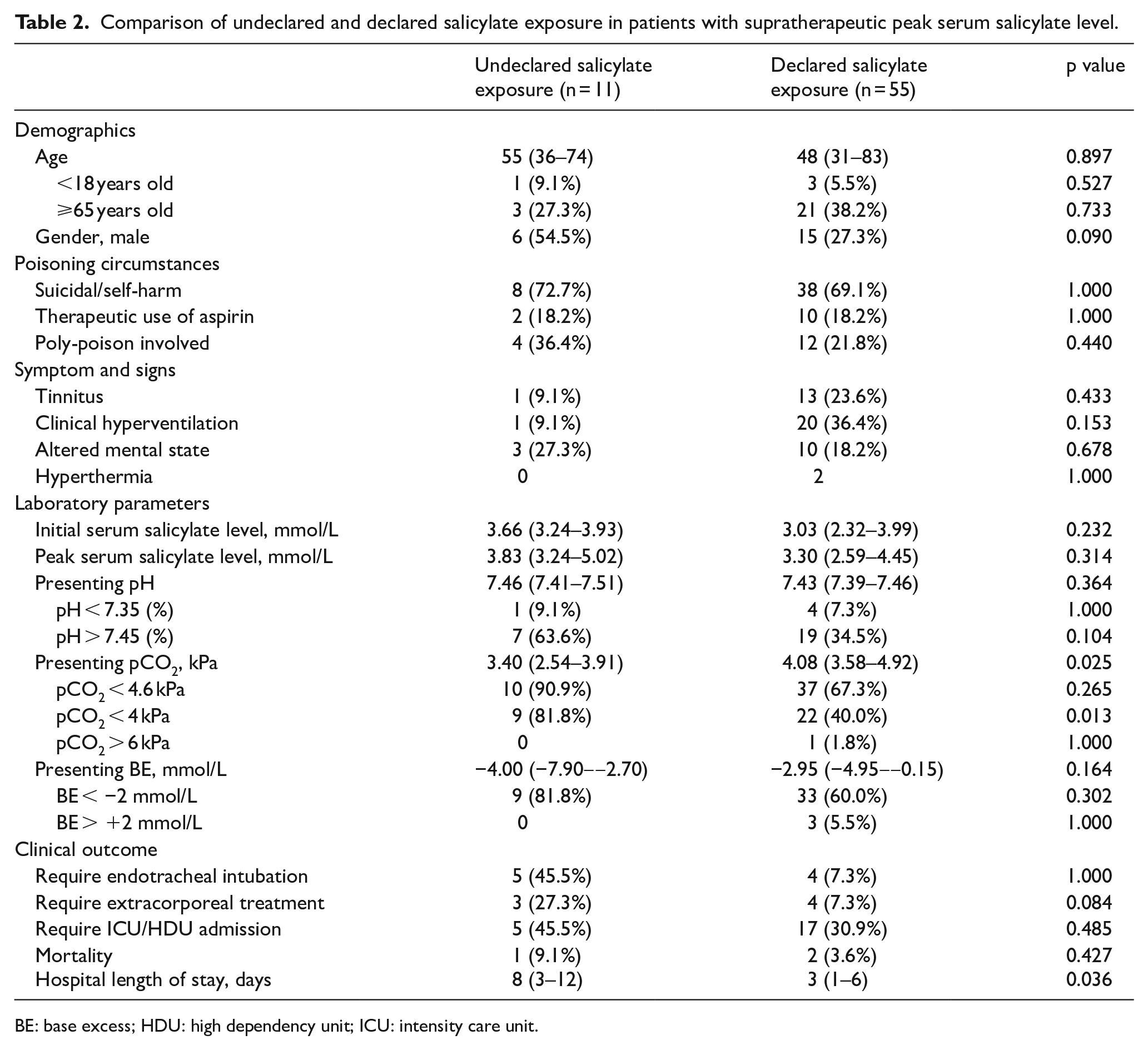
BE: base excess; HDU: high dependency unit; ICU: intensity care unit.
Fatal cases with supratherapeutic peak serum salicylate level
There were three fatal cases with supratherapeutic peak serum salicylate level (Table 3). They were all elderly patients. All of them were reported to be isolated salicylate poisoning without other poison involved. The salicylate poisons involved were all medicated oil containing methylsalicylate.
Clinical details of the fatal cases with supratherapeutic peak serum salicylate levels.
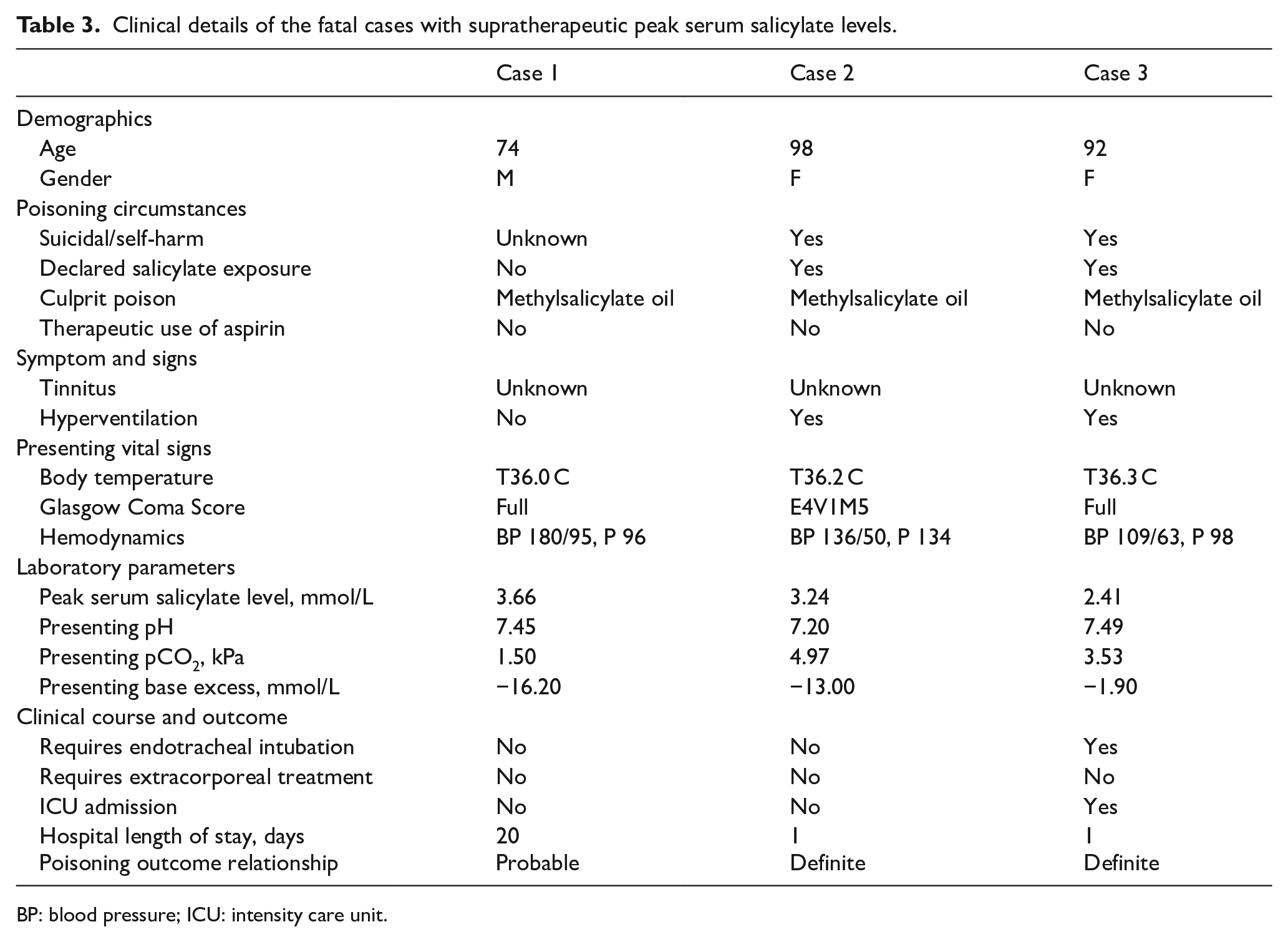
BP: blood pressure; ICU: intensity care unit.
Case 1 attended the A&E department for severe abdominal pain and requested discharge with acknowledgement of medical advice. He was found unconscious on the same day and was subsequently admitted. Serum salicylate level was found to be high. It was suspected that the patient applied a large amount of methylsalicylate oil for pain control. The patient was later diagnosed with terminal malignancy with hepatic encephalopathy and passed away. The poisoning-outcome relationship was probable. Cases 2 and 3 were elderlies who ingested medicated oil with rapid downhill despite treatments.
Clinical prediction rule modelling
The whole data set was divided into a derivation set and a validation set in a 2:1 ratio, stratified by serum salicylate level. A backward stepwise logistic regression by likelihood ratio (LR) was performed in the derivation set with variables with p < 0.3 recruited to identify predictors of a supratherapeutic peak serum salicylate level. There were six variables included as follows: therapeutic use of aspirin (p < 0.001), tinnitus (p < 0.001), hyperthermia (p < 0.001), altered mental state (p = 0.197), presenting pCO2 (p < 0.001) and presenting BE (p < 0.001). Missing data were managed by simple deletion, and 3070 cases were excluded in this part.
Age was not put into the model as it confounded with the therapeutic use of aspirin while 33% of cases elder than 65 years old had aspirin on hand and none of the aged <18 years old had aspirin on hand. Moreover, the age distribution in the group with supratherapeutic peak serum salicylate level was not highly correlated (Figure 2).
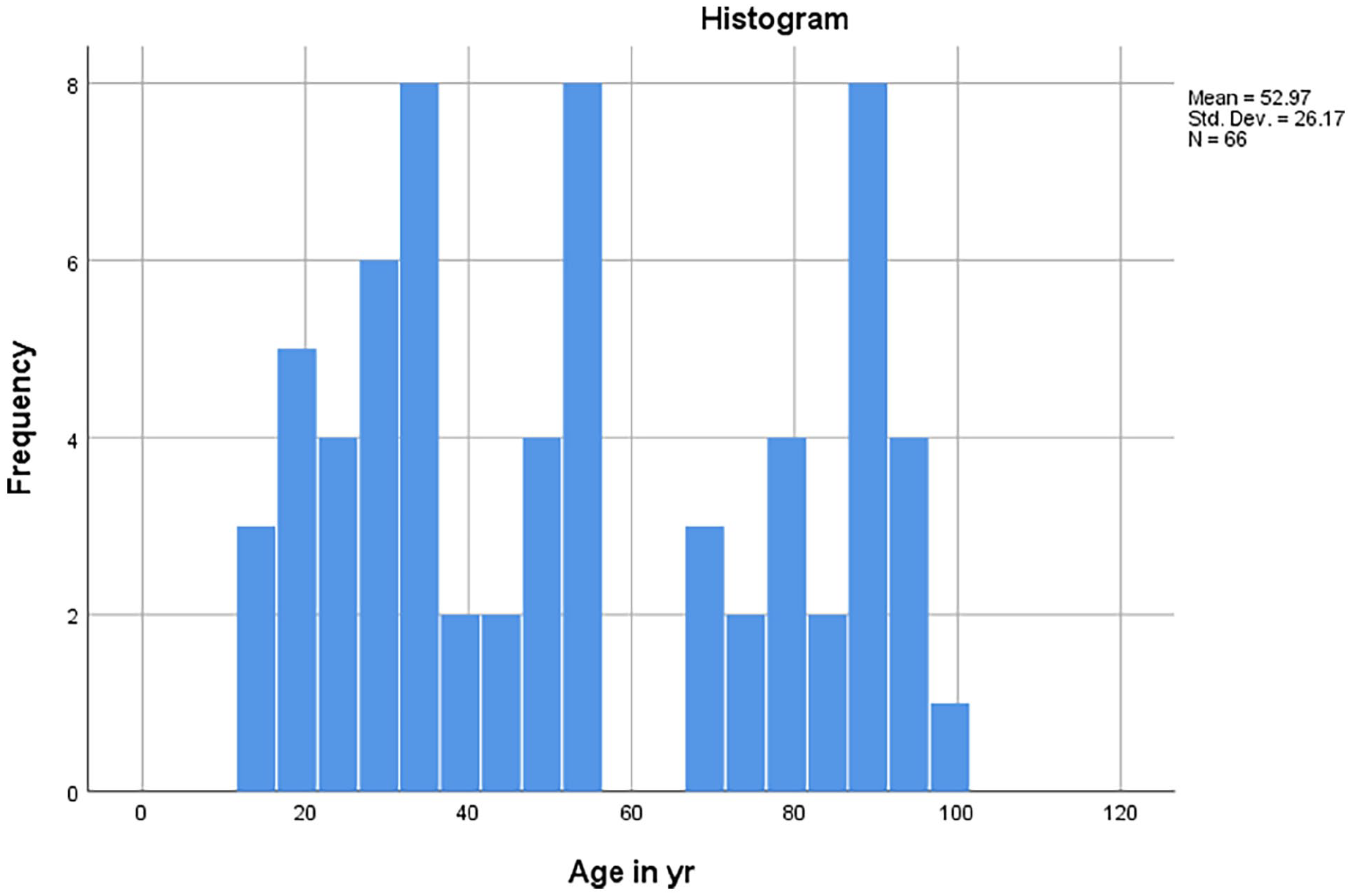
Age distribution of patients with supratherapeutic peak serum salicylate level.
Clinical hyperventilation was highly reflected by pCO2 and presenting pH was likely a combined effect of pCO2 and BE, and thus not recruited as well.
At each step, a variable was eliminated based on the lowest probability of the LR until all variables in the model had a p value < 0.1. Three variables remained in the final model: tinnitus, therapeutic use of aspirin and pCO2. The AUROC of the derived model was 0.845 (Figure 3). The Hosmer–Lemeshow test was insignificant (p = 0.160) (Table 4). The optimal pCO2 cut-off was 5 kPa with the best sensitivity and comparable specificity. A clinical prediction rule was derived accordingly (Figure 4). If any one of the factors was present, that is (1) tinnitus, (2) the therapeutic use of aspirin or (3) partial pressure of CO2 <5 kPa in blood gas, then there is a risk of having supratherapeutic peak serum salicylate level.
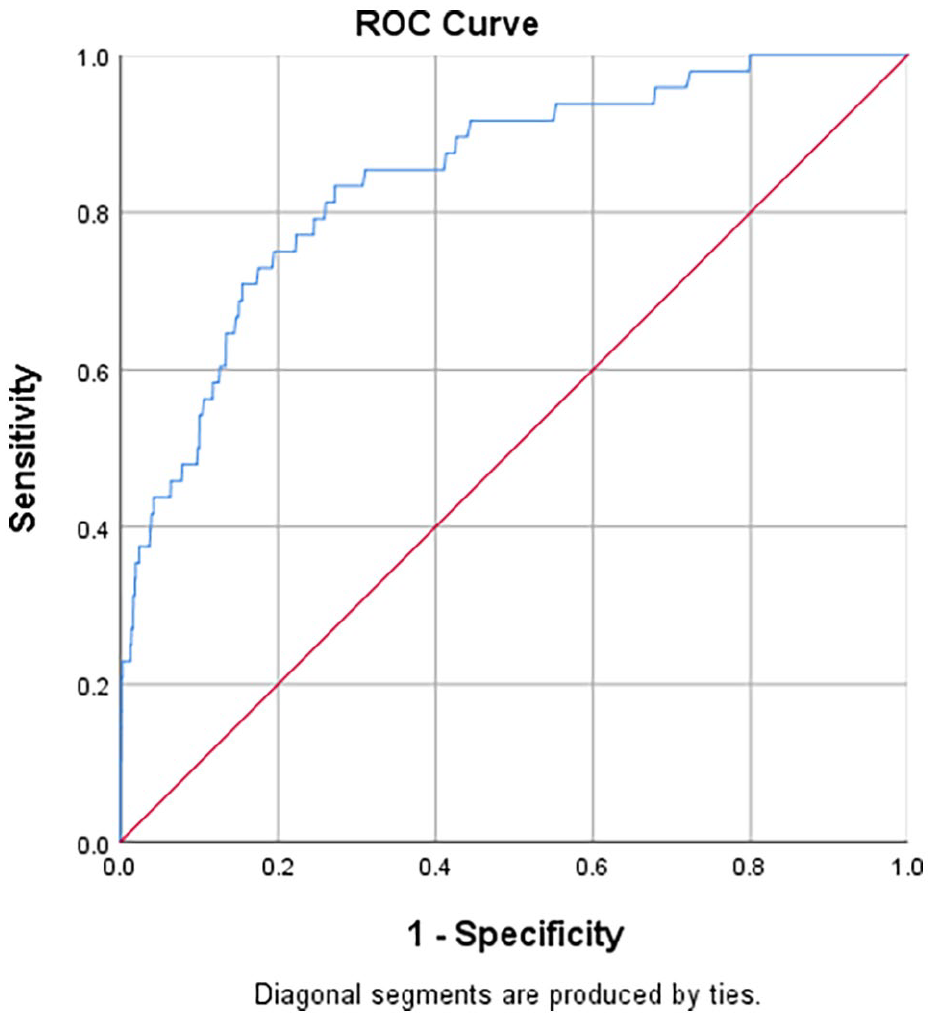
Receiver operating characteristic curve of the logistic regression model.
Multivariate logistic regression model to predict patients with supratherapeutic peak serum salicylate level among poisoned patients.

Hosmer–Lemeshow test p = 0.160.

Clinical prediction rule to predict supratherapeutic peak serum salicylate level.
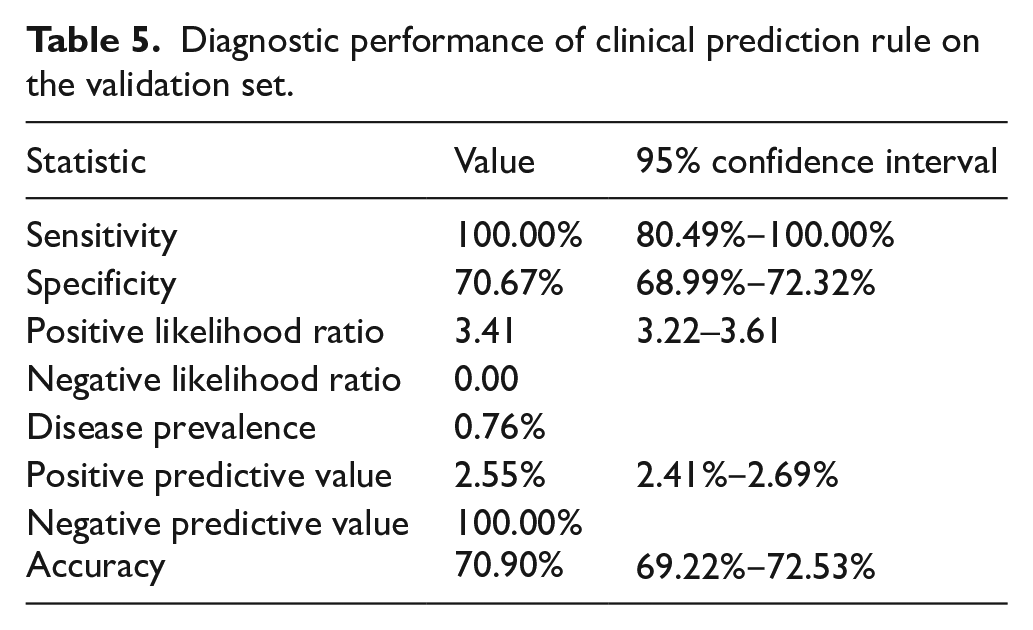
Internal validation on the validation set (Table 5) showed that the derived clinical prediction rule had 100% sensitivity (95% confidence interval (CI) 80.49–100) with 70.6% specificity (95% CI 68.99%‒72.32%). The NLR was 0.00 and the NPV was 100% while the PLR was 3.41 (95% CI 3.22–3.61) and PPV was 2.55% (95% CI 2.41%‒2.69%).
Discussion
In this retrospective study reviewing data of 13 years, there were 8673 cases in total with serum salicylate level checked. In our observation, routine serum salicylate screening was still a common local practice where it was done in 17% of all poisoning cases recorded by HKPIC during the study period.
Among them, there were 143 cases (1.65%) with detectable peak serum salicylate level and 66 cases (0.76%) with supratherapeutic peak serum salicylate level. This was compatible with the finding of a paper in 1984 suggesting that <5% had positive serum salicylate level in screening. 26 The mortality in the group with supratherapeutic peak serum salicylate level (4.5%) was 2 times that of the group without supratherapeutic peak serum salicylate level (2.2%), though not statistically significant (p = 0.187). This clinically significant difference may be underpowered due to the low incidence of supratherapeutic peak serum salicylate level. There was also a longer length of hospitalisation and a higher rate of HDU/ICU admission in the group with supratherapeutic peak serum salicylate level, which also signified that this group of patients tended to be more ill.
Diagnostic performance of clinical prediction rule on the validation set.
There were 35 (0.40%) undeclared salicylate poisoning with detectable peak serum salicylate level in this study and 11 of them (0.13%) had a supratherapeutic peak serum salicylate level. This was also compatible with the finding of a 1996 US study which found that only 0.16% had a toxic serum salicylate level not suggested by history. 17 That paper also suggested that such undeclared intoxication should be suggested by high anion gap metabolic acidosis and altered mental state. However, in this study, there were only 19.7% of cases with supratherapeutic peak serum salicylate level presented with alteration in mental state, which was lower than the control group (24.6%), though some of them became unconscious when the condition progressed. Among them, 16 cases in total (30.2%) presented with normal base excess. There might be a chance of delaying the diagnosis of salicylate poisoning if solely relying on the change in consciousness and high anion gap metabolic acidosis. In contrast to a paper in 1991 27 which found tinnitus as a non-sensitive or non-specific indicator of serum salicylate concentration, this study found that tinnitus was significantly higher in the group with supratherapeutic peak serum salicylate level. Also, respiratory alkalosis and metabolic acidosis, the classical blood gas pattern, were individually associated with supratherapeutic peak serum salicylate level. However, there were 12 cases (18.1%) with supratherapeutic peak serum salicylate level having normal blood gas patterns, meaning the lack of the above findings could not rule out the possibility of having supratherapeutic peak serum salicylate level.
Based on our study result, the derived clinical prediction rule had excellent sensitivity in predicting supratherapeutic peak serum salicylate level in poisoned patients. One of its strong points was that the presenting blood gas in this study included both arterial and venous blood gas. It is known that pCO2 does not correlate well in arterial and venous blood gas and cut-off is different. In day-to-day practice, venous blood gas is usually the initial investigation done in poisoned patients. Allowing venous blood gas for analysis made the clinical prediction rule easier to apply. Theoretically, routine salicylate screening is not necessary for clinicians with good awareness of undeclared salicylate poisoning and the ability to perform this clinical prediction rule.
Nonetheless, in real-life situations, not all poisoning cases were evaluated by clinicians with toxicological experience in diagnosing undeclared salicylate. Performing routine serum salicylate screening has a detection rate of about 1 in 800 in this study. Screening serum salicylate level remains an important tool to rule out acute salicylism in cases with unexplained high anion gap metabolic acidosis with or without tachypnoea.
Moreover, salicylate poisoning may still be possible despite an initially undetectable salicylate level. 28 It was suggested that 3.5% of salicylate-poisoned patients had initially undetectable serum salicylate level in a study in 2019. 29 There were cases reported on salicylism with initially undetectable serum salicylate level with possible mortality.30,31 Our study also found that there were rising serum salicylate levels upon recheck in 107 cases and 8 of them rose from normal level to supratherapeutic. Clinicians should be reminded to repeat serum salicylate level whenever in suspicion.
It was not surprising to find that medicated oil containing methylsalicylate was involved in all fatal cases with supratherapeutic peak serum salicylate level. The potential toxicological concern of products containing methylsalicylate was well described by different papers.32,33 Methylsalicylate is widely available in many over-the-counter liniments, ointments, lotions or medicated oils for the relief of musculoskeletal pains. 34 There were over hundreds of medicated oil or liniments containing methylsalicylate according to the database of the Chinese Medicine Council of Hong Kong. A local study found that the medicated oils contained 15%‒40% methylsalicylate and up to 67% in one 60-mL product, which was 55.2 g aspirin equivalent dose for ingesting one bottle. 35 Owing to its concentrated liquid state with high lipid solubility, ingestion of such medicated oil may cause rapid-onset, severe salicylism.32,33 Nonetheless, topical application of salicylate products can result in severe poisoning. A local volunteer study 36 confirmed that massive dermal application of methylsalicylate can produce unpredictable salicylate poisoning. It was also reviewed in a dermatologist paper. 37 Clinicians should be well aware of the potential danger in cases of exposure to medicated oil of unknown nature.
Limitation
There were limitations owing to the retrospective nature of this study.
The largest weak point of this study was that a supratherapeutic peak serum salicylate level may not necessarily be salicylism and vice versa. There was concern about the poor correlation of circulating serum salicylate level with the severity of intoxication.14,38 However, serum salicylate level still imparts diagnostic and prognostic information and defines therapy in overdose. 39 In this study, worse clinical outcomes were demonstrated in the group with supratherapeutic peak serum salicylate level. Yet, the cases without supratherapeutic serum salicylate level, with the possibilities of having salicylism, were not studied.
Also, this study did not differentiate between acute or chronic salicylate poisoning. Chronic salicylism can display clinical manifestations at concentrations much lower than acute toxicity. 14 Symptomatic patients without supratherapeutic peak serum salicylate level should still ring the alarm of clinicians about the possibility of chronic salicylate poisoning.
Another limitation was the possibility of reporting bias. Tinnitus may be over-reported when patients with elevated serum salicylate level and under-reported in patients without. A prospective study may be useful to delineate its significance in salicylate poisoning.
Nonetheless, there was also a lot of missing data in our study. There were 3070 (35.4%) cases with missing data, including 490 cases without consciousness level and 2580 cases without complete blood gas results. They were excluded in the multivariate analysis and the remaining 5603 cases were used to construct the clinical prediction rule. Moreover, the timing of blood gas, whether before or after treatment, cannot be assessed. The therapeutic use of aspirin was based on CDARS which did not include private drugs on hands. The route of entry was not examined in this study as well.
A further prospective study is needed to establish the clinical benefit of this clinical prediction rule and explore factors predicting severe outcomes in salicylate poisoning.
Conclusion
Serum salicylate level should be checked if a poisoned patient has aspirin on hand, tinnitus or blood gas showing pCO2 <5 kPa as it may be undeclared salicylate poisoning. Initial serum salicylate level can be undetectable and clinicians should repeat serum salicylate level whenever in suspicion. Routine serum salicylate level may detect 0.13% of undeclared salicylate poisoning with supratherapeutic peak serum salicylate level. Clinicians should be aware that poisoned patients with exposure to medicated oil containing salicylate can have serious outcomes. Routine serum salicylate screening detected supratherapeutic peak serum salicylate level at a rate of 1 in 800. It remained a tool for clinicians not experienced in toxicology.
Footnotes
Acknowledgements
None.
Author contributions
Chan Suen Wah wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Availability of data and materials
The data of the study were retrieved from the Hong Kong Poison Information Centre database (Poison Information and Clinical Management System) and the Clinical Data Analysis and Reporting System of Hospital Authority.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Patient consent was waived by the Research Ethics Committee.
Ethical approval
The study was approved by the Research Ethics Committee of Kowloon East Cluster of Hospital Authority of Hong Kong with the reference number KC/KE-21-0015/ER-2.
