Abstract
Introduction:
Development of coagulopathy after anticoagulant rodenticide ingestion varies among patients. This study aimed to identify factors that were associated with coagulopathy after anticoagulant rodenticide ingestion.
Methods:
This was a retrospective cohort study, conducted in the Hong Kong Poison Information Centre. All patients who reported rodenticide exposure and presented to the Accident and Emergency Department from 1 January 2010 to 31 December 2019 were recruited. Coagulopathy was defined as International Normalized Ratio of 1.3 or above.
Results:
One hundred sixty-nine patients were included in the final analysis. The median age was 44 years old. Forty-nine patients developed coagulopathy (International Normalized Ratio ⩾1.3). Univariate analysis (at p < 0.05) showed that age (p = 0.003), ingestion of first-generation anticoagulant rodenticide (p = 0.017), ingestion of more than one pack (p < 0.001), intentional ingestion (p = 0.002), hypoalbuminemia (p < 0.001), elevated alanine aminotransferase level (p = 0.041) and abnormal estimated glomerular filtration rate (p = 0.005) on presentation, and co-ingestion with paracetamol (p = 0.018) were associated with coagulopathy after anticoagulant rodenticide ingestion. Among these, ingestion of more than one pack (p < 0.001; odds ratio = 19.8; 95% confidence interval = 6.78–65.7), ingestion of first-generation anticoagulant rodenticide (p = 0.006; odds ratio = 5.2; 95% confidence interval = 1.96–15.2), hypoalbuminemia (p < 0.001; odds ratio = 22.4; 95% confidence interval = 6.17–99.0) and elevated alanine aminotransferase level on presentation (p = 0.039; odds ratio = 7.11; 95% confidence interval = 1.58–33.1) were statistically significant in the multivariate analysis.
Conclusion:
Ingestion of more than one pack and ingestion of first-generation anticoagulant rodenticides were significantly associated with the development of coagulopathy after anticoagulant rodenticide ingestion. Patients who developed hypoalbuminemia or elevated alanine aminotransferase level as a result of anticoagulant rodenticide ingestion were also significantly associated with the development of coagulopathy.
Introduction
Rodenticides are products that are designed to kill rodents, mice, and other small animals and are used for control of rodent population. Rodenticides are composed of different agents, from highly toxic agents such as sodium monofluoroacetate (SMFA), tetramine, to less toxic agents such as anticoagulants.
According to the Registered Pesticides List by the Hong Kong Agriculture, Fisheries and Conservation Department (AFCD), rodenticides that are registered in Hong Kong for household use include anticoagulants, cholecalciferol, and cellulose. The most encountered rodenticide poisoning in Hong Kong is anticoagulant rodenticide poisoning. 1 In resource rich countries such as the United Kingdom and the United States, anticoagulants are also the most common cause in rodenticide poisoning.2,3
Anticoagulant rodenticide works by inhibiting vitamin K-1,25 epoxide reductase, an enzyme required for the regeneration of vitamin K. It interferes with the activation of vitamin K-dependent clotting factors II, VII, IX, and X, and proteins C, S and Z. The development of anticoagulation is delayed until body reserve of vitamin K is depleted and the pre-existing active coagulation factors are consumed. 4 A new group of anticoagulant rodenticide known as “superwarfarin” or second-generation anticoagulants had been developed in view of warfarin-resistant strain of rats. This includes the second-generation 4-hydroxycoumarins: brodifacoum, bromadiolone, difenacoum, flocoumafen and the indanedione derivatives chlorophacinone and diphacinone. They have a higher potency and longer duration of action due to higher affinity for vitamin K1-epoxide reductase, the ability to disrupt the vitamin K1-epoxide cycle at more than one point, liver accumulation and high lipid solubility and enterohepatic circulation.5,6
The clinical outcome of anticoagulant rodenticides poisoning varies among patients. Some develop coagulopathy with active bleeding, some have asymptomatic coagulopathy, while some do not develop coagulopathy.
A local epidemiological study in 2014 1 on household rodenticide poisoning with 87 patients from July 2008 to February 2012 showed that development of coagulopathy after rodenticide ingestion was significantly related to ingestion of warfarin group rodenticide and a higher ingestion dose. A study in Korea 7 on 31 patients with vitamin K-dependent coagulopathy presumed to be caused by brodifacoum showed that coagulopathy was significantly related to lower albumin level and simultaneous ingestion of rodenticide and alcohol.
Currently, there are limited local studies on factors associated with coagulopathy in anticoagulant rodenticide poisoning. This study aimed to investigate risk factors that would predispose patients to coagulopathy in anticoagulant rodenticide poisoning.
Method
Study design
This was a retrospective cohort study based on the territory-wide database of the Hong Kong Poison Information Centre (HKPIC). The database included consultation related to poisoning cases encountered by all healthcare workers in Hong Kong, and surveillance data from voluntary reporting of poisoning cases by all Accident and Emergency Departments (AED) under the Hospital Authority.
Patients who presented to the AED and reported rodenticide exposure from 1 January 2010 to 31 December 2019 were included in the study. The patients were recruited from the database of HKPIC. Exclusion criteria included the following: (1) patients with route of exposure other than oral ingestion, (2) patients who reported exposure to non-anticoagulant type of rodenticide, (3) patients who reported exposure to unknown type of rodenticide, (4) patients with incomplete records, (5) patients with unidentified context of exposure (e.g. cases of occult superwarfarin overdose in which the circumstances of exposure is unknown), and (6) patients with unknown dosage of rodenticide ingested. Formula of rodenticide ingested was based on the clinical history from patients or informants, and/or laboratory tests on urine, serum or rodenticide samples. Cases notes were reviewed by two authors to arrive at the decision of case exclusion.
Study variables included demographic characteristics, clinical presentation, clinical outcome, cause of ingestion, type and dosage of each poison ingested, medical and drug history, laboratory investigations and results, toxicology findings, and treatment. Data were extracted from the Accident and Emergency records and Electronic Patient Record System (ePR) of the Hospital Authority.
Coagulopathy was defined as the International Normalized Ratio (INR) of 1.3 or above. The dosage of rodenticide ingested was documented as packs in this study. According to a previous local study, 1 the mean package weight for one packet of household rodenticide across 15 different brands sold in Hong Kong was weighed to be 60.7 g (range = 42.5–100 g). Dosage documented in grams in the hospital records would be converted to packs based on the mean package size of rodenticide in Hong Kong. Co-ingestion was based on clinical history and/or laboratory findings.
Results from the first liver and renal function test taken after rodenticide ingestion were used for analysis in this study. Hypoalbuminemia was defined by a value smaller than the lower limit of the reference range of the respective laboratory. Elevated alanine aminotransferase (ALT) level, alkaline phosphatase (ALP) level, and bilirubin level were defined as a value higher than the upper limit of the reference range of the respective laboratory.
Estimated glomerular filtration rate (eGFR) was calculated using the four-variable Modification of Diet in Renal Disease (MDRD) Study equation: (eGFR = 175 × standardized serum creatinine − 1.154 × age-0.203 × 1.212 (if black) × 0.742 (if female)). 8 GFR less than 90 mL/min/1.73 m2 was classified as renal impairment according to the Kidney Disease Improving Global Outcomes (KDIGO) classification. 9
The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement Guideline was implemented in this article. This study was approved by the Hong Kong Hospital Authority Kowloon West Cluster Research Ethics Committee (reference no. KC/KE-20-01300/ER-3).
Statistical analysis
Statistical analysis was performed by R (statistical language program: https://www.r-project.org/about.html). Age and INR level were analyzed by Wilcoxon’s rank sum test to compare the difference in median age and INR level. Categorical factors were analyzed with the chi-square test or with Fisher’s exact test when at least one of the cell values was less than five. A multivariate logistic regression was performed on the significant factors from the univariate analysis. A p-value at <0.05 was regarded as statistically significant.
Results
Two hundred ninety-six patients were recruited into the study. One hundred twenty-seven patients were excluded from the study according to the exclusion criteria (Figure 1). One hundred sixty-nine patients were included in the final analysis.
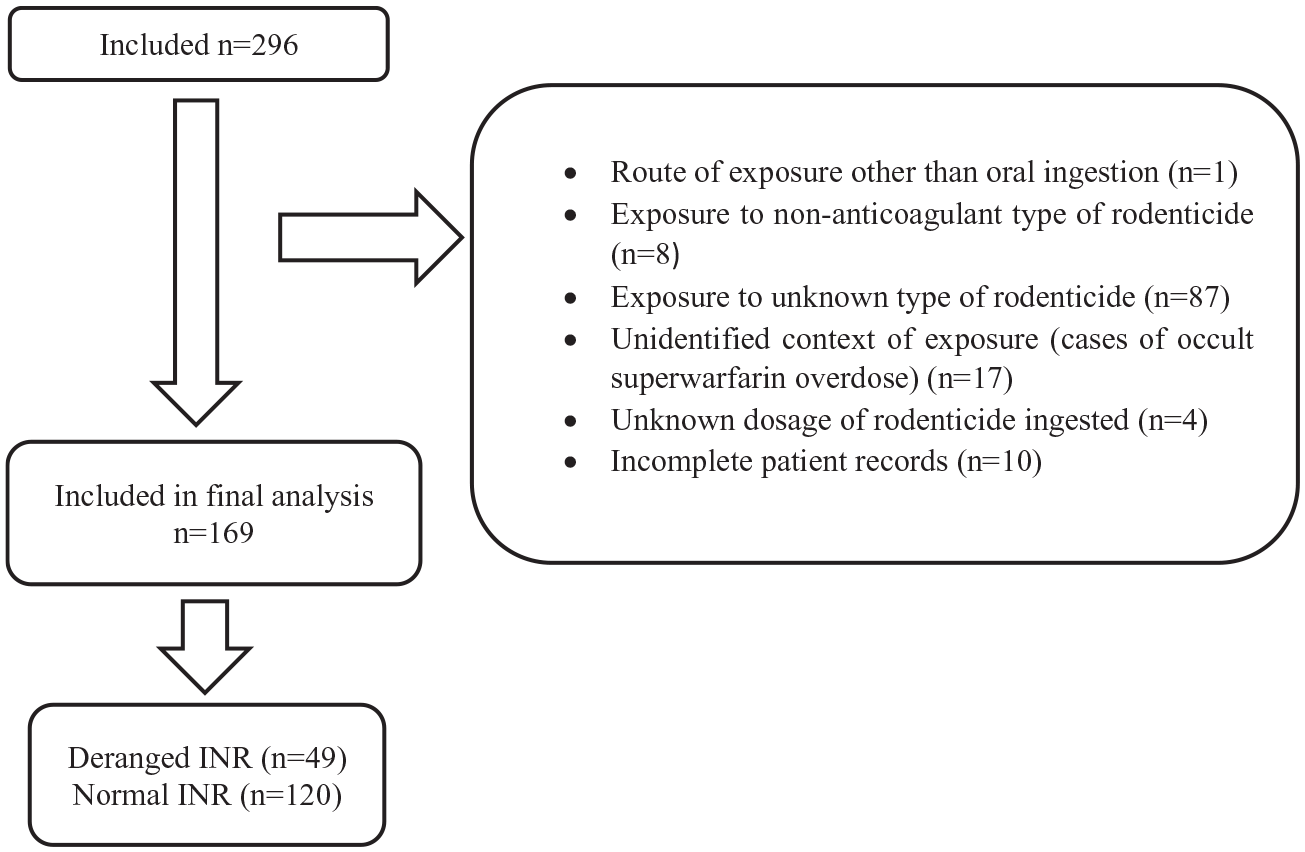
Flowchart of exclusion.
The median age was 44 years old (range from 11 months to 101 years old). Ninety patients (53.3%) were male and 79 patients (46.7%) were female. Forty-nine patients (29.0%) developed coagulopathy and 120 patients (71.0%) had normal clotting profiles after ingestion of anticoagulant rodenticide.
The documented rodenticide ingested included first-generation anticoagulant rodenticide: warfarin (n = 22), coumatetralyl (n = 13), bishydroxycoumarin (n = 1); second-generation anticoagulant rodenticide: bromadiolone (n = 98), brodifacoum (n = 23), mixed bromadiolone and brodifacoum (n = 5), unknown superwarfarin (n = 4), and flocoumafen (n = 2). One patient ingested mixed bromadiolone and warfarin.
One hundred forty patients (82.8%) ingested one pack of rodenticide or less, while 29 patients (17.2%) ingested more than one pack of rodenticide. One hundred forty-nine cases (88.1%) were intentional ingestion, and 20 cases were unintentional ingestion (11.8%). All the cases with unintentional ingestion had ingested less than one pack of rodenticide.
Ninety cases (41.4%) had co-ingestion, including alcohol (n = 25), sleeping pills (n = 21), paracetamol (n = 9), psychiatric medication (n = 9), household detergent (n = 8), and carbon monoxide poisoning (n = 5). Other co-ingestions included anti-hypertensive medications, oral hypoglycemic agents, insulin, histamine H2 antagonist, and traditional Chinese medicine.
Seven patients had history of liver disease, including hepatitis B carrier (n = 3), hepatitis C carrier (n = 1), fatty liver (n = 2), and liver cirrhosis (n = 1). Five patients had history of renal diseases.
On presentation, 18 patients (10.7%) had hypoalbuminemia, 72 patients (42.6%) had abnormal eGFR, and 12 patients (7.1%) had elevated ALT level. Forty-nine patients developed coagulopathy after rodenticide ingestion. The median peak INR levels was 2.2 (range = 1.3–8.6). Four patients (8.1%) suffered from bleeding symptoms (hemothorax, gum bleeding, hematuria, eye bruising, hemoptysis, and gastrointestinal bleeding); 45 patients (91.8%) were asymptomatic. Forty-one patients (83.7%) received vitamin K1 treatment. Two patients (4.1%) received fresh frozen plasma (both with INR >6.0).
Statistical analysis: deranged INR group versus normal INR group
Univariate analysis
Clinical characteristics were compared between the group that developed coagulopathy and the group that had normal clotting profile after ingestion. Age (p = 0.003), ingestion of first-generation anticoagulant rodenticide (p = 0.017), ingestion of more than one pack (p < 0.001), intentional ingestion (p = 0.002), hypoalbuminemia (p < 0.001), elevated ALT level (p = 0.041) and abnormal eGFR (p = 0.005) on presentation and co-ingestion with paracetamol (p = 0.018) had a statistically significant impact on development of coagulopathy after anticoagulant rodenticide ingestion (Table 1).
Comparison of clinical characteristics between the group with outcome of deranged INR and the other group.
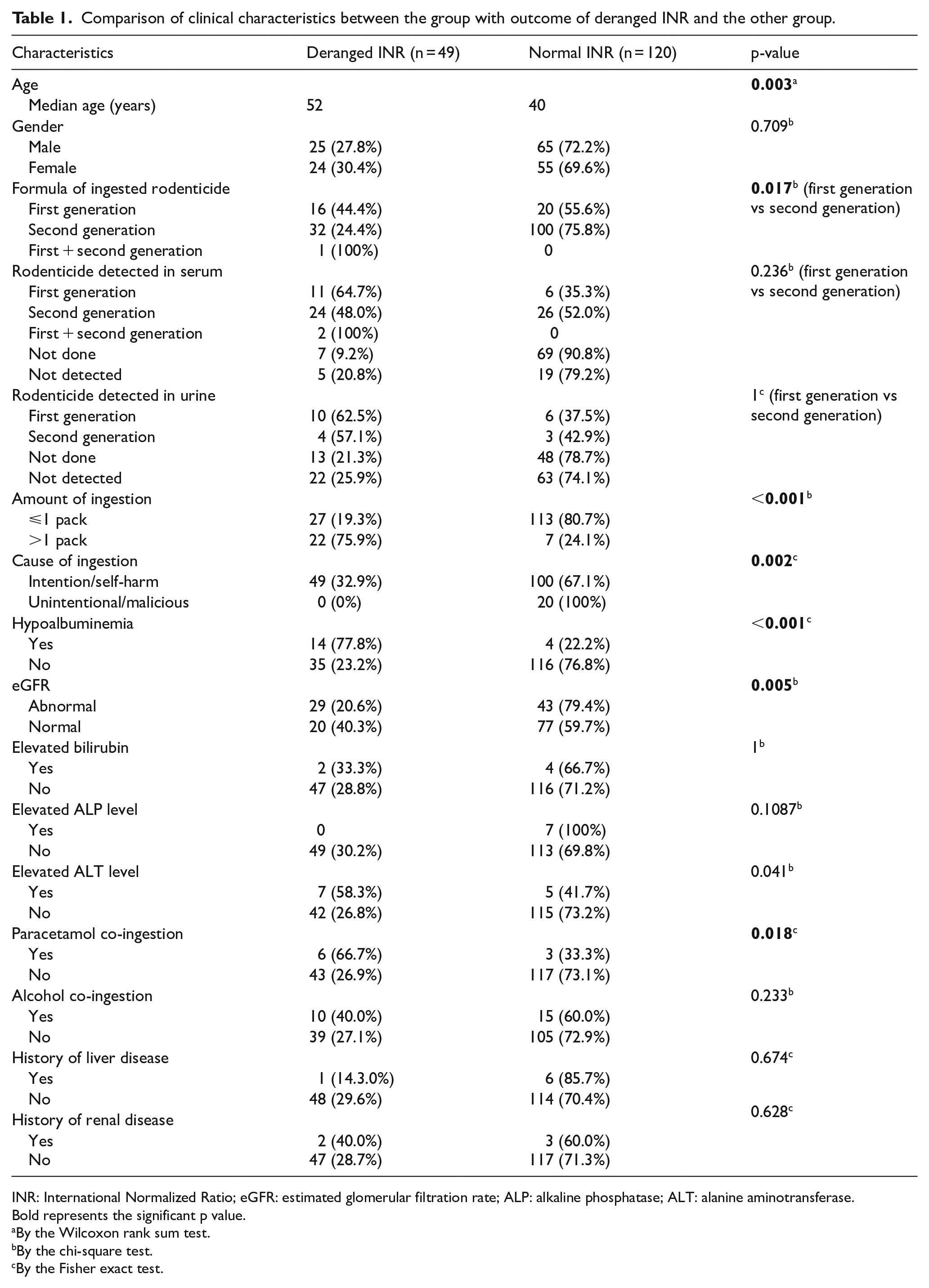
INR: International Normalized Ratio; eGFR: estimated glomerular filtration rate; ALP: alkaline phosphatase; ALT: alanine aminotransferase.
Bold represents the significant p value.
By the Wilcoxon rank sum test.
By the chi-square test.
By the Fisher exact test.
Multivariate logistic regression
Out of the eight factors that were significantly associated with coagulopathy after anticogulant rodenticide ingestion in the univariate study, the following four factors had reached a statistical significance at 95% confidence in the multivariate logistic regression model: ingestion of more than one pack (p < 0.001; odd ratio (OR) = 19.8; 95% confidence interval (CI) = 6.78–65.7), ingestion of first-generation anticoagulant rodenticide (p = 0.006; OR = 5.2; 95% CI = 1.96–15.2), hypoalbuminemia (p < 0.001; OR = 22.4; 95% CI = 6.17–99.0) and elevated ALT level on presentation (p = 0.039; OR = 7.11; 95% CI = 1.58–33.1). The R square value of the final logistic regression model was 0.4 (Table 2). Paracetamol co-ingestion had a near significance (p = 0.067) in the multivariate study.
Multivariate logistic regression model—p-value, odd ratios and 95% CI.

CI: confidence interval; ALT: alanine aminotransferase.
Discussion
This is the largest local retrospective study in Hong Kong on the development of coagulopathy among patients who ingested rodenticide. Around 29% of patients (n = 49) developed coagulopathy. In this study, the most common anticoagulant rodenticide agents ingested were bromadiolone (n = 98), followed by brodifacoum (n = 23) and warfarin (n = 22). This was different from the previous local study, 1 in which warfarin was the most common agent. This could represent an increase in prevalence of second-generation anticoagulant rodenticide in the market.
Factors related to development of coagulopathy (INR ⩾1.3)
Although bleeding was uncommon after rodenticide ingestion, the development of coagulopathy (29%) was not. Around 25% of our patients eventually required vitamin K1 treatment. We had identified factors that were associated with coagulopathy after rodenticide ingestion, including ingestion of more than one pack, ingestion of first-generation anticoagulant, hypoalbuminemia, and elevated ALT level on presentation. This would help early identification of high-risk group that might need close monitoring of INR level and guide the usage of vitamin K1 treatment. This was the first local study that had identified hypoalbuminemia and elevated ALT level being significantly associated with coagulopathy after anticoagulant rodenticide ingestion.
Amount of ingestion
Ingestion of larger amount of anticoagulant leads to coagulopathy. In this study, ingestion of more than one pack was significantly associated with coagulopathy, which was consistent with the previous local study. 1
Formula of anticoagulant rodenticide
Ingestion of first-generation anticoagulant was significantly associated with coagulopathy, compared to second-generation anticoagulant. In humans, the lowest published toxic dose of brodifacoum was 0.12–0.172 mg/kg. 10 One textbook listed the toxic dose of second-generation anticoagulant active ingredient as greater than 0.1 mg/kg. 11 In acute overdose of warfarin, the toxic dose was thought to be over 0.5 mg/kg. 12
The concentration of second-generation anticoagulant in the rodenticide in this study was around 0.005%, whereas the concentration of warfarin in the rodenticide in the study was around 0.1%, which was 20 times higher. For an average sized adult weighted 70 kg, the toxic dose of brodifacoum would be around 8.4 mg, and 35 mg for warfarin. Based on the mean package weight of rodenticide found in Hong Kong (63.7 g, range = 42.5–100 g), 1 one pack of rodenticide with warfarin 0.1% contained around 63.7 mg of warfarin (range = 42.5–100 mg), whereas one pack of rodenticide with bromadiolone 0.005% or bromadiolone 0.005% only contained around 3.19 mg of active ingredients (range = 2.13–5 mg).
Ingestion of one pack of first-generation anticoagulant could reach the toxic dose while one pack of second-generation anticoagulant might not. The much higher concentration in first-generation anticoagulant rodenticide might explain its significant association with coagulopathy after ingestion.
Albumin level
Our data suggested that hypoalbuminemia on presentation was associated with coagulopathy after anticoagulant rodenticide ingestion. Anticoagulants in rodenticides are vitamin K antagonists, which are more than 90% albumin bound in circulation. Only free, unbound vitamin K antagonist is biologically active. 13 Hypoalbuminemia increases the free fraction of vitamin K antagonist in plasma, 14 and thus enhances its anticoagulation effect. There had been studies which showed significant association between hypoalbuminemia and coagulopathy after anticoagulant usage. In a Korean study 7 on coagulopathy after brodifacoum exposure, lower albumin level was significantly associated with coagulopathy. An observational study of 402 patients in the United Kingdom 15 had shown that for patients with non-valvular atrial fibrillation receiving anticoagulation therapy with warfarin, hypoalbuminemia was a significant predictor of all bleeding in patients aged <75 years. A prospective cohort study on 755 patients in Japan 16 had shown that hypoalbuminemia increases the likelihood of supratherapeutic INR control and risk of major bleeding events in patients on warfarin for atrial fibrillation.
Alanine aminotransaminase
Alanine aminotransaminase (ALT) is a transaminase enzyme most commonly found in the liver, and can also be found in various body tissues such as kidney, heart and muscles. In this study, elevated ALT level on presentation was significantly associated with coagulopathy after anticoagulant rodenticide ingestion. Liver plays an important role in drug metabolism, protein and clotting factors synthesis, 17 elevated ALT can be related to hepatocyte damages. Both first- and second-generation anticoagulants undergo hepatic p450 metabolism,5,18 –21 and thus, the rodenticide metabolism might be affected in hepatocyte damages. A study on Asian patients on warfarin initiation therapy had shown that a high ALT level was significantly associated with a high INR level (INR ⩾4). 22
We took the results from the first liver and renal function test taken after rodenticide ingestion for analysis, as the liver and renal function status prior to anticoagulant rodenticide ingestion were not available in most patients due to the retrospective nature of this study. Among the poison co-ingested by patients, paracetamol and alcohol were the ones that might cause hepatocyte injuries. In our patients with elevated ALT level (n = 12), only two had paracetamol co-ingestion and none had alcohol co-ingestion, and in our patients with hypoalbuminemia (n = 18), none had paracetamol co-ingestion and only two had alcohol co-ingestion. Although whether the elevated ALT level and hypoalbuminemia were caused by anticoagulant rodenticide ingestion could not be statistically determined in this study, it is a logical deduction that they were likely due to anticoagulant rodenticide ingestion, and that these factors were associated with the development of coagulopathy based on our statistical analysis and the literature reviewed.
Co-ingestion
One hundred four patients had co-ingestion. The most common co-ingestion was alcohol, sleeping pills, and paracetamol. Patients who co-ingested paracetamol were significantly associated with coagulopathy after rodenticide ingestion in the univariate study (p = 0.018), and were close to significance in the multivariate regression model (p = 0.067).
There had been evidence proposed on interaction between warfarin and paracetamol. Five randomized control trials were performed,23 –27 with four showing a significant increase in INR in patients with concomitant use of warfarin and paracetamol. This could be explained by the effect of paracetamol on the level of functional factor VII. Two of the randomized control trials25,27 also found a significant reduction in functional clotting factors in patients with concomitant use of warfarin and paracetamol. A cohort study 28 had also shown that in patients with paracetamol poisoning, functional factor VII and factor IX were lowered.
A previous study 29 had found that the toxic metabolite from paracetamol overdose N-acetyl-para-benzoquinone imine (NAPQI) interferes with vitamin K-dependent gamma-carboxylase (VKD-carb) and vitamin K epoxide reductase (VKOR) activities. These two enzymes are involved in the vitamin K cycle and their inhibition affects the regeneration of vitamin K and subsequently interferes with the activation of vitamin K-dependent clotting factors. 30 The near significance of paracetamol co-ingestion in our multivariate regression study could be due to small sample size of patients.
Implication in clinical practice
Based on the findings in this study, we propose an updated management flowchart for patients with anticoagulant rodenticide ingestion (Figure 2). This helps to identify patients at higher risk of coagulopathy for closer monitoring, and reduce unnecessary hospital admissions and investigations in low risk cases.
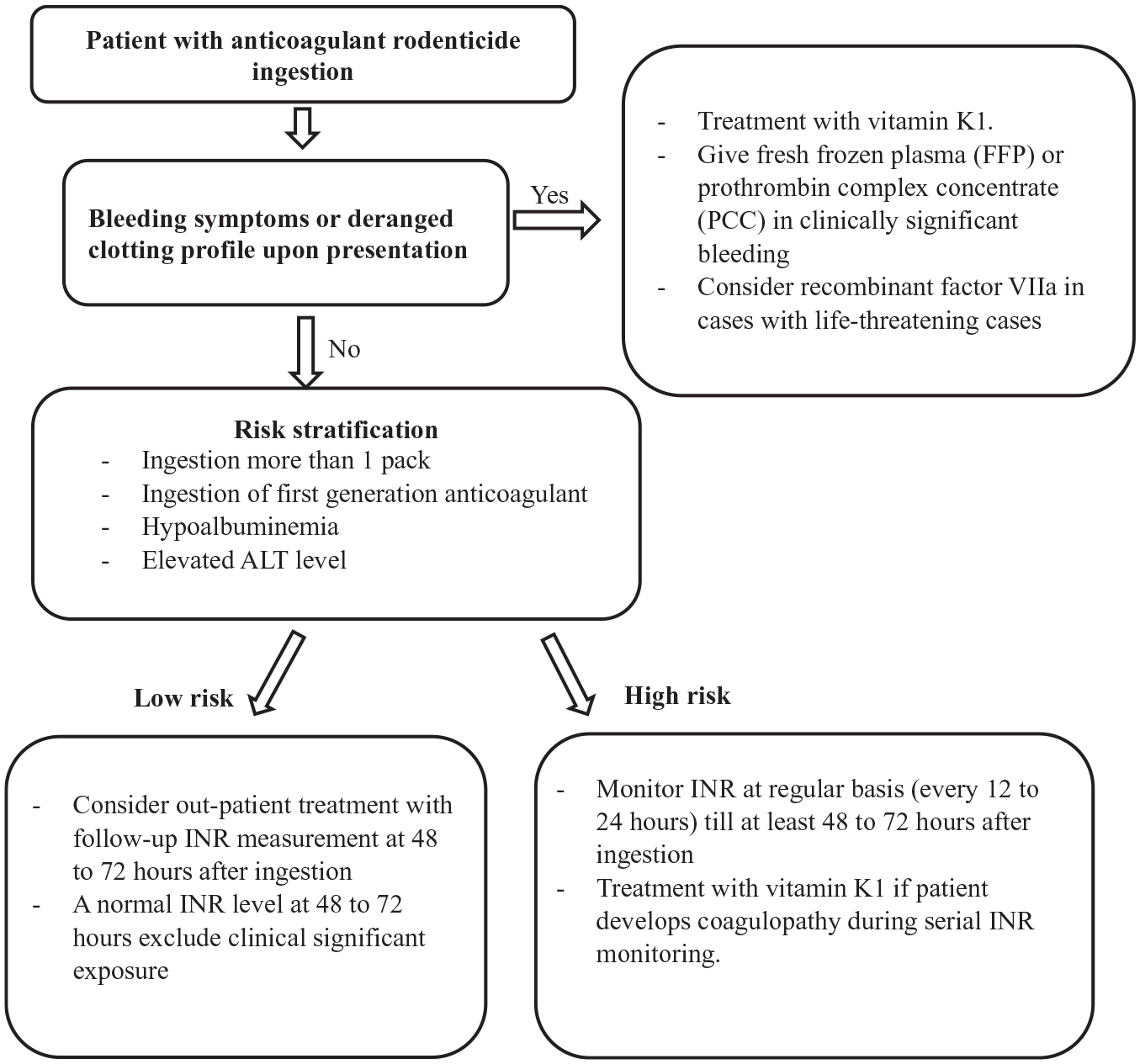
Management flowchart for patients with anticoagulant rodenticide ingestion.
Patients with coagulopathy after rodenticide ingestion
Forty-nine patients in this study developed coagulopathy after ingestion of rodenticide. Four patients developed bleeding symptoms, while 45 patients with deranged INR were asymptomatic. Comparing these two groups, patients who had bleeding symptoms had a significantly higher median first deranged INR level (3.95 vs 1.53, p = 0.024). The median peak INR level were higher in the bleeding group, but the difference was not significant (4 vs 2.1, p = 0.257).
Mortality was rare in this study where only two deaths were reported (1.67%). One was an 85-year-old female who ingested one pack of bromadiolone intentionally, and developed coagulopathy with a peak INR level of 2.1. She suffered from ST-elevation myocardial infarction and died 2 days after ingestion. Another death case was a 79-year-old male with terminal lung cancer who ingested one pack of bishydroxycoumarin intentionally. He developed coagulopathy with an INR level of 1.8 and complicated with development of hemothorax. He was given vitamin K1 treatment and INR level normalized afterwards. He died 4 days after ingestion, with his cause of death documented as terminal lung cancer.
Out of 49 patients with coagulopathy, 41 patients (83.7%) received vitamin K1 treatment and 8 patients (16.3%) did not. Further studies can be performed on topics related to vitamin K1 treatment, especially the relationship between albumin level, ALT level, and/or paracetamol co-ingestion and vitamin K1 dosage and duration.
Limitations
The unit of rodenticide ingested was not unanimous among the hospital records. Most were documented in terms of packs. Some were documented in grams, and were converted to number of packs in this study based on the mean package size of rodenticides available in Hong Kong. 1 This might cause discrepancy from the true ingested dose as the package weight might vary between different brands. In addition, there are currently no quantitative tests available for measurement of rodenticide level in urine or serum samples, causing difficulty in ascertaining the exact amount of rodenticide ingested.
Twelve patients in this study were given prophylactic vitamin K1 before the first INR result was available. This might lead to a false normal INR level. It is not advised to give vitamin K1 prophylactically after rodenticide ingestion until confirmation of coagulopathy, 31 as it may affect the subsequent monitoring of INR trend and give a false reassurance if there is a “normal” clotting profile.
In 58 cases, the toxicology tests (urine, serum or both) were negative. This could be due to the fact that tests for rodenticide identification in serum or urine samples are only available in the Toxicology Reference Laboratory (TRL) in Princess Margaret Hospital. In this study, not all samples were tested in the TRL. Laboratory toxicological confirmation tests (urine or serum) were not performed in 35 cases (20.7%). The diagnosis of anticoagulant rodenticide poisoning of these patients was solely based on the clinical history by the patients or informants.
The retrospective nature of the study and relatively small sample size may limit the power of multivariate analysis. Further studies can be performed on the association of age and underlying medical disease with development of coagulopathy after anticoagulant rodenticide ingestion.
Conclusion
Ingestion of more than one pack and ingestion of first-generation anticoagulant rodenticides were significantly associated with development of coagulopathy after anticoagulant rodenticide ingestion. Patients who developed hypoalbuminemia or elevated ALT level as a result of anticoagulant rodenticide ingestion were also significantly associated with the development of coagulopathy.
Footnotes
Acknowledgements
The authors thank Dr Tse Man Li of the Hong Kong Poison and Information Centre for his intellectual support to this study.
Author contributions
Dr K.W.T. (MBChB) designed and conceptualized the study, collected and analyzed the data, and drafted and revised the manuscript for intellectual content. Dr C.K.C. (MBBS, FHKCEM, FHKAM (Emergency medicine)) designed and conceptualized the study, interpreted the data, and revised the manuscript for intellectual content. Dr S.L. (MBBS, FHKCEM, FHKAM (Emergency medicine)) designed and conceptualized the study, interpreted the data, and revised the manuscript for intellectual content. All the authors approved the article to be published. All the authors participated sufficiently in the work to take public responsibility for appropriate portions of the content. The authors alone are responsible for its contents and writing of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
The data were retrieved from the HKPIC and Clinical Management System Database of HA.
Informed consent
Written informed consent was not required by research ethics committee as this is a retrospective study without patient intervention and no identifiable patient data have been included in the manuscript.
Ethical approval
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by The Kowloon Central Cluster Ethics Committee of the Hospital Authority (reference no. KC/KE-20-01300/ER-3).
Human rights
There were no human right conflicts to declare.
