Abstract
Background:
Patients commonly visit the emergency department for pain after musculoskeletal injury, but the problem of oligoanalgesia is prevalent. Methoxyflurane (Penthrox®) is an inhalational analgesic for moderate to severe trauma-associated pain in stable and conscious patients. It is a fast-acting, effective analgesic that can be readily administered via a non-invasive route, making it an attractive agent for managing acute pain in the emergency departments.
Objectives:
The aim was to assess the analgesic efficacy of methoxyflurane in patients with acute traumatic pain by comparing it to ketorolac, a standard analgesic treatment for moderate pain in emergency departments in Hong Kong.
Methods:
This was a single-center, open-label, randomized controlled, parallel-group, non-inferiority pilot study that enrolled adult patients with moderate trauma-associated pain in an emergency department in Hong Kong. Patients were randomized 1:1 to the methoxyflurane group or the ketorolac group. The primary outcome was the change in pain intensity measured by visual analogue scale from baseline to 5, 15, 30, and 60 min after drug administration.
Results:
Twenty patients received methoxyflurane, and twenty patients received ketorolac. There were significant reductions in pain score over 60 min in both groups. The pain reduction at 5 min was significantly greater for the methoxyflurane group (−13.912 mm; 95% confidence interval = −20.008 to −7.817) than for the ketorolac group (−4.888 mm; 95% confidence interval = −10.983 to 1.208), with the treatment effect (−9.025 mm; 95% confidence interval = −17.656 to −0.393; p = 0.041) demonstrating superiority of methoxyflurane. The treatment effect at 15 and 30 min demonstrated non-inferiority of methoxyflurane versus ketorolac.
Conclusion:
Methoxyflurane provided non-inferior analgesia in the first 30 min with a faster onset of action when compared with ketorolac in moderate traumatic pain. It can be considered a non-invasive, rapid-acting, and effective first-line alternative to currently available analgesics for traumatic pain in emergency settings.
Background
Patients commonly visit the emergency department (ED) for pain after musculoskeletal injury and need early treatment with analgesics. Prompt and adequate pain relief can reduce suffering and promote early discharge and return to work. 1 However, the problem of oligoanalgesia is prevalent among many EDs. 2 Several barriers that hinder optimal pain management 3 include inadequate pain assessment, limited prescription of analgesics, long waiting time for analgesics, and patient reluctance due to fear of side effects, fear of masking symptoms, and so on. Current guidelines on pharmacological management of pain in EDs in Hong Kong follow the World Health Organization’s analgesic ladder principle: start with paracetamol for mild pain, step up to non-steroidal anti-inflammatory drugs (NSAIDs) or weak opioid for moderate pain, and strong opioid for severe pain. The oral route is the commonest and simplest means of drug administration, but the onset of action is relatively slow. Intravenous (IV) analgesics are preferred in emergency settings as they provide the fastest onset of action. Obtaining venous access, however, can be distressing and time-consuming with a 12%–26% failure rate for the first attempt in adults. 4 Intramuscular analgesics can be the alternative for patients with difficult venous access, but injections are painful and invasive that may result in refusal of treatment in needle-phobic patients. 5 Analgesic of other modalities such as Entonox (mixture of 50% nitrous oxide and 50% oxygen) has the benefit of being non-invasive with rapid onset of action, yet the bulky equipment delays its administration 6 and limits its practicality in prehospital settings. 7 Thus, there is an unmet need for a fast-acting, effective, and safe analgesic that can be readily administered via a non-invasive route.
Methoxyflurane, manufactured under the brand name Penthrox®, is an inhalational analgesic licensed to relieve moderate to severe pain associated with trauma or minor surgical procedures in stable and conscious patients.8,9 It is a volatile liquid that can be self-administered through a lightweight, portable, hand-held inhaler device. Methoxyflurane liquid is first added into the inhaler device, where it is absorbed by a polypropylene wick and vaporized. Pain relief should commence after 6–10 inhalations through the mouthpiece. 10 Each inhaler containing 3 mL of methoxyflurane can maintain an analgesic effect for 25–30 min with continuous inhalation, up to 60 min with intermittent inhalation. 10 The mechanism of the analgesic effect is unknown. Methoxyflurane belongs to the fluorinated hydrocarbon group of volatile anesthetics. It was first used for general anesthesia in the 1960s 11 but was later withdrawn for the risk of nephrotoxicity at high anesthetic doses (typically 40–60 mL). 12 However, low-dose methoxyflurane at subanesthetic concentration has been used extensively as an analgesic by paramedics and emergency physicians in Australia and New Zealand for the past four decades.8,9 The analgesic effect has been proven in a randomized placebo-controlled STOP! trial, which showed a significantly greater reduction in pain score for methoxyflurane. 10 Moreover, the low analgesic dose of methoxyflurane has a well-established safety profile. It is not associated with nephrotoxicity, and adverse events are usually transient and self-limiting.13,14
Methoxyflurane (Penthrox®) has been granted registration approval in Hong Kong since 2018. Despite having several advantages and fewer barriers to administration compared to other analgesics available, 6 it is still not widely used in this locality. In this study, we aimed to assess the efficacy of methoxyflurane for short-term pain relief in adult patients with acute traumatic pain by comparing it to intramuscular ketorolac, a standard analgesic treatment for moderate pain in EDs in Hong Kong. To our best knowledge, this is the first head-to-head randomized controlled trial to evaluate the analgesic efficacy of methoxyflurane against ketorolac.
Method
Study design and setting
This was a single-center, open-label, randomized controlled, parallel-group, non-inferiority pilot study comparing methoxyflurane with ketorolac. It was conducted in the Accident and Emergency Department of Ruttonjee Hospital, an acute district hospital in Hong Kong providing 24-h accident and emergency services with a daily attendance around 200 patients. Ethics approval was obtained from Hong Kong East Cluster Research Ethics Committee (reference no. HKECREC-2020-064). Written informed consent for participation was obtained from all patients before the study.
Patient selection
Patients of both sexes from 18 to 64 years of age presented to the Accident and Emergency Department in Ruttonjee Hospital for any musculoskeletal injury within 72 h of onset with moderate pain at screening (10-point Numeric Rating Scale (NRS) ⩾4 to ⩽7) were eligible for the study. Types of injury included contusion, sprain, crushing, burn, abrasion, laceration, fracture, and dislocation. Exclusion criteria included life-threatening condition or hemodynamic instability (systolic blood pressure <90 mm Hg, diastolic blood pressure <60 mm Hg, respiratory distress with respiratory rate >20 breath per minute, or oxygen saturation <95% on room air) requiring resuscitation; limb-threatening condition or any injury requiring immediate management; pregnant or breastfeeding woman; impaired consciousness from any cause such as head injury and acute intoxication; any physical, visual, or cognitive condition that may affect patient’s ability to use visual analogue scale (VAS) for self-assessment of pain intensity; concomitant use of other analgesic within 5 h (8 h for diclofenac sodium) prior to presentation to ED; other pre-existing chronic pain condition; inability or refusal to provide written informed consent; inability to understand and converse in the language spoken; and any contraindication to inhalational methoxyflurane or intramuscular ketorolac as per the Summary of Product Characteristics.8,15
Contraindications to inhalational methoxyflurane were known personal or family history of hypersensitivity to methoxyflurane or any fluorinated anesthetics; known pre-existing clinically significant renal or hepatic impairment; known personal or family history of malignant hyperthermia; concomitant use of nephrotoxic agents such as gentamicin, tetracycline, colistin, polymyxin B, and amphotericin B; and concomitant use of CYP 450 inducers such as alcohol, isoniazid, phenobarbital, and rifampicin. Contraindications to intramuscular ketorolac were known allergy to NSAIDs; known pre-existing clinically significant renal impairment or at risk for renal failure due to volume depletion; active major bleeding; suspected or confirmed cerebrovascular bleeding; history of peptic ulcer disease, gastrointestinal bleeding, or perforation; concomitant use of aspirin, other NSAIDs, anticoagulant, or novel anticoagulant agent, pentoxifylline and probenecid; known bleeding disorders such as hemophilia, thrombocytopenia; heart failure; perioperative period of coronary artery bypass graft (CABG) surgery; and concurrent asthmatic attack.
Randomization and intervention
Patients were enrolled on a convenience basis in the Accident and Emergency Department in Ruttonjee Hospital. After screening by investigators, patients who met all inclusion and non-exclusion criteria were randomly assigned to either the experimental group or control group in a 1:1 ratio by drawing from equal preset numbers of sealed and opaque envelopes. Randomization was in a block randomization scheme of 10 without stratification.
Patients were supplied with methoxyflurane or ketorolac as soon as possible after randomization. Patients in the experimental group were assisted to self-administer one Penthrox® inhaler under the supervision of trained personnel. After priming the inhaler with 3 mL of methoxyflurane by nursing staff, patients were instructed to inhale through the mouthpiece to obtain analgesia and then exhaled back into the mouthpiece so that any unmetabolized methoxyflurane could be adsorbed by the activated charcoal chamber. Patients were also instructed to control the level of analgesia by inhaling more frequently or covering the diluter hole with the index finger if stronger analgesia was required. Supplemental Appendix 1 showed the instruction for using the Penthrox® inhaler. 8 Patients in the control group received one dose of 30 mg intramuscular ketorolac injected at gluteal muscle by nursing staff as usual practice.
Patients and investigators were not blinded to the allocated treatment due to practical differences in the route of administration. Patients were advised that if pain relief were inadequate after study drug administration, rescue medication would be available immediately on request at any time before they were discharged from the ED.
Data collection and outcome measurements
Patient demographic data, types, and sites of injury were recorded on the study data form. After drug administration, patients were observed in the ED for at least 1 h before they were discharged home or admitted to the hospital.
The primary outcome was the change in pain intensity after drug administration. It was measured by VAS, an unmarked 100 mm horizontal straight line where the left end indicated “no pain at all” and the right end indicated “the worst possible pain I can imagine.” Patients were instructed to mark the pain intensity on the VAS before and at 5, 15, 30, and 60 min after drug administration. Measurement of pain intensity ceased if rescue medication was administered.
The secondary outcomes were the change in vital signs and level of sedation after drug administration, the incidence of treatment-related adverse events (TRAEs), the use of rescue medication, and the global level of satisfaction with pain control and overall acceptance of treatment. Vital sign parameters (including blood pressure, pulse rate, and oxygen saturation) and level of sedation in terms of the Ramsay Sedation Scale (RSS) were recorded before and at 5, 15, 30, and 60 min after drug administration. The nature and severity of any TRAEs observed by investigators or spontaneously reported by patients throughout the observation period were recorded. Any analgesics given in the ED after study drug administration were considered rescue medication. It was provided upon request or when pain improvement was insufficient based on the judgment of the attending physicians. The time of administration of rescue medication was recorded. At the time of discharge from the ED, patients were asked to rate their level of satisfaction with pain control and overall acceptance using a 5-point Likert-type scale.
Sample size calculation
Previous studies found the minimum clinically significant difference in VAS pain score was 13 mm,16,17 so the margin for this non-inferiority study was set to be 13 mm. A sample size of 34 patients (17 patients per treatment group) was required to provide 80% power at a 5% (one-sided) significance level to show non-inferiority, assuming no difference in mean VAS change from baseline between the two groups and the standard deviation for this parameter was 15 mm. Given the setting of the study, the drop-out rate was expected to be low, so 20 patients in the experimental group and 20 patients in the control group were planned to allow for a drop-out rate of 15%.
Statistical analysis
Data were analyzed on an intention-to-treat basis using IBM SPSS Statistics version 26.0 (IBM Corp, Armonk, NY, USA). No imputation of missing data was performed. Descriptive statistics were used as appropriate.
The primary outcome measure was the mean change in VAS pain score from baseline for each group at 5, 15, 30, and 60 min after drug administration. It was analyzed with linear mixed-effect model for repeated-measure analysis of variance, adjusted for baseline VAS pain score, and the interaction between time point and treatment. The treatment effect and the associated two-sided 95% confidence interval (95% CI) were estimated as the difference in the adjusted mean change of VAS score between the two treatment groups (methoxyflurane – ketorolac). Methoxyflurane was determined to be non-inferior to ketorolac if the upper bound of the 95% CI of the treatment effect was below 13 mm (non-inferior margin). Superiority of methoxyflurane was further concluded if the upper bound of the 95% CI of the treatment effect was below 0 mm.
The secondary outcome measure was the mean change in vital sign parameters from baseline for each group at 5, 15, 30, and 60 min after drug administration. It was analyzed with linear mixed-effect model for repeated-measure analysis of variance, adjusted for baseline vital sign parameters and the interaction between time point and treatment. For other secondary outcome measures, ordinal variables (level of sedation and global level of satisfaction with pain control and overall acceptance) were analyzed with Mann–Whitney U test, while dichotomous variables (the incidence of TRAEs and the use of rescue medications) were analyzed with chi-square test or Fisher’s exact test if any expected cell is less than 5.
Results
From October to November 2020, a total of 40 patients were enrolled and randomly assigned to two treatment groups (20 in the methoxyflurane group and 20 in the ketorolac group). All patients in each group received the treatment and completed the study. All of their data were included for analyses.
Demographic and baseline characteristics were similar in the two treatment groups (Table 1). The mean age was 39.4–39.8 years, and the majority of patients were male (50%–75%) and Chinese (75%–80%). Baseline injuries were also comparable between both groups. More than half of the patients had injuries in lower limbs (52.2%–55.6%). The dominant types of injury were fracture (39.1%) in the methoxyflurane group and contusion (33.3%) and sprain (33.3%) in the ketorolac group. A few patients had second (n = 4) and third (n = 3) injuries. Patients in the two treatment groups had similar baseline VAS scores (methoxyflurane: mean = 65.3 mm, standard deviation (SD) = 14.9 mm; ketorolac: mean = 63.4 mm, SD = 11.3 mm). Most patients were discharged home (65%–70%), while other patients were admitted to the emergency observation ward or orthopedics ward for further management. One patient in the ketorolac group with fracture right mid tibia and fibula was discharged against medical advice and referred to another hospital for geographical reasons. No patients were admitted due to TRAEs.
Demographic and baseline characteristics (intention-to-treat population).
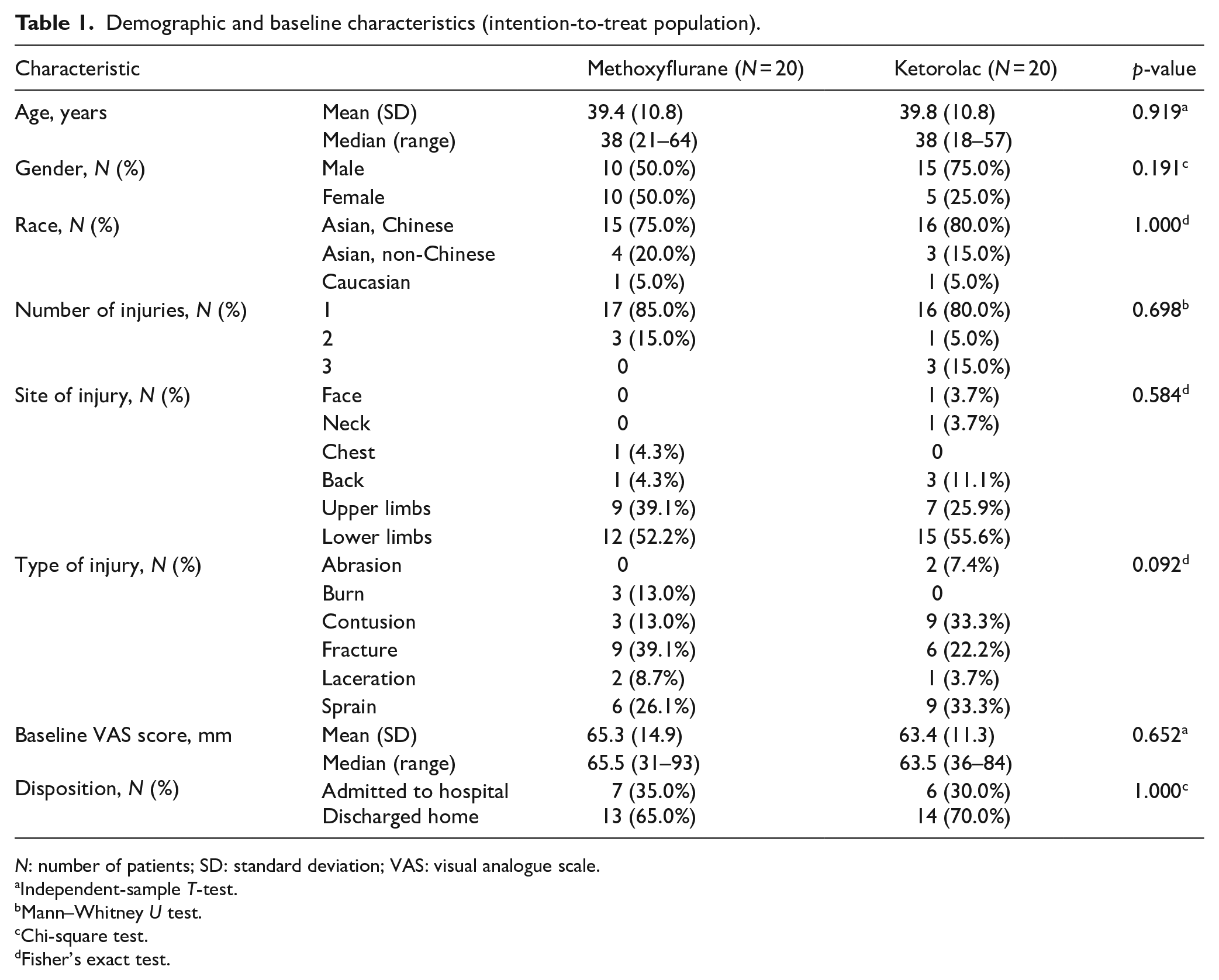
N: number of patients; SD: standard deviation; VAS: visual analogue scale.
Independent-sample T-test.
Mann–Whitney U test.
Chi-square test.
Fisher’s exact test.
The mean VAS pain scores at each time point are presented graphically in Figure 1. There were significant reductions in VAS pain score over 60 min in both treatment groups, from 65.30 to 34.20 mm for the methoxyflurane group (p < 0.001) and from 63.40 to 32.15 mm for the ketorolac group (p < 0.001). Table 2 shows the mean change in VAS pain score and the treatment effect (outcome difference between the two treatment groups) adjusted for baseline VAS pain score. There was no significant difference in the overall treatment effect considering all time points between the two treatment groups (−2.097 mm; 95% CI = −11.539 to 7.344; p = 0.655). However, pairwise comparison at each time point showed that the adjusted mean change in VAS pain score from baseline to 5 min was significantly greater for the methoxyflurane group (−13.912 mm; 95% CI = −20.008 to −7.817) than for the ketorolac group (−4.888 mm; 95% CI = −10.983 to 1.208), with the treatment effect (−9.025 mm; 95% CI = −17.656 to −0.393; p = 0.041) demonstrating both non-inferiority (upper 95% CI = <13 mm) and superiority (upper 95% CI = < 0 mm) of methoxyflurane versus ketorolac. The treatment effect at 15 and 30 min also demonstrated non-inferiority of methoxyflurane versus ketorolac. However, the result at 60 min was inconclusive and failed to show non-inferiority of methoxyflurane as the 95% CI of treatment effect crossed both zero and the non-inferior margin.
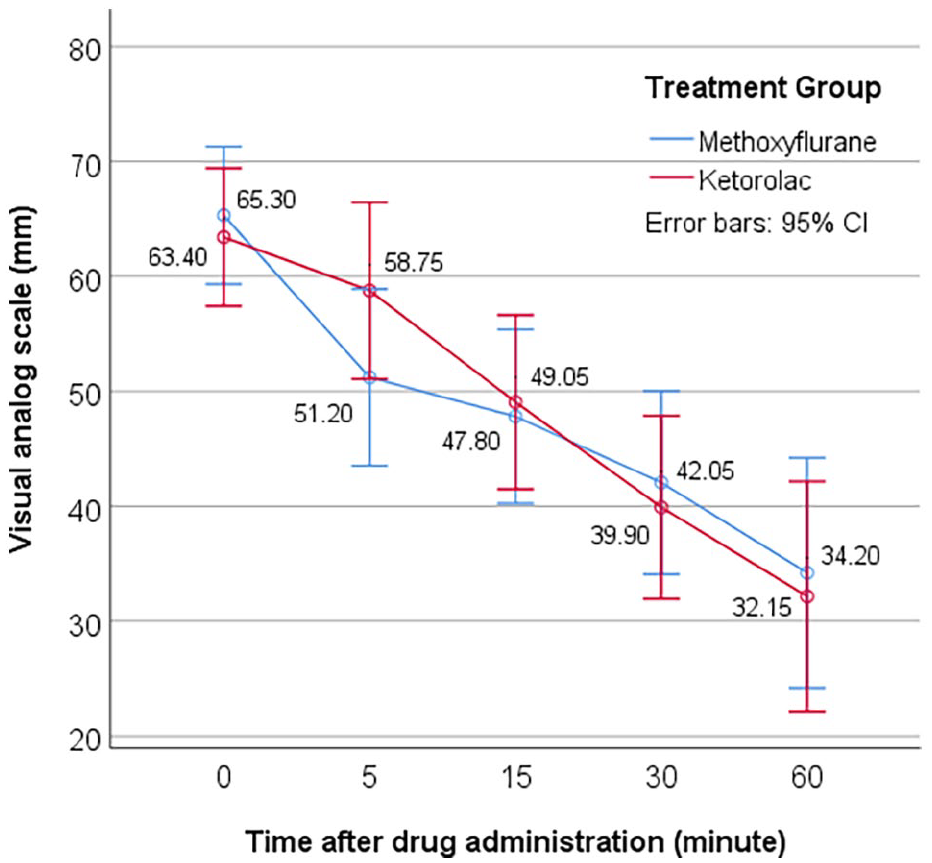
Mean VAS pain scores from baseline over 60 min after drug administration (intention-to-treat population).
Repeated-measures analysis of change from baseline in VAS pain score over 60 min after drug administration (intention-to-treat population).
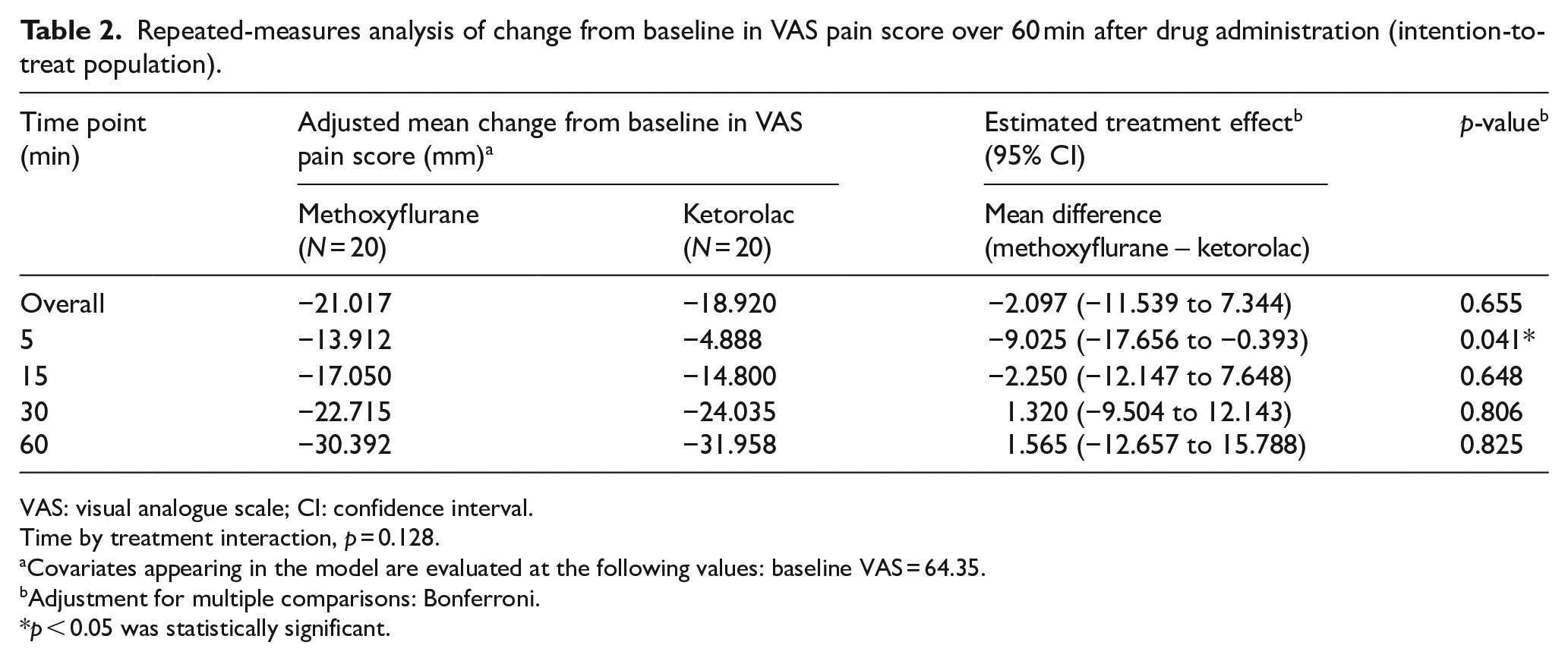
VAS: visual analogue scale; CI: confidence interval.
Time by treatment interaction, p = 0.128.
Covariates appearing in the model are evaluated at the following values: baseline VAS = 64.35.
Adjustment for multiple comparisons: Bonferroni.
p < 0.05 was statistically significant.
Vital signs and RSS score were evaluated before and 5, 15, 30, and 60 min after the start of treatment. There were no significant differences between the two treatment groups in mean changes in vital sign parameters (Table 3). Mean changes from baseline in the methoxyflurane group and the ketorolac group over 60 min were, respectively, −5.548 mm Hg versus −1.252 mm Hg for systolic blood pressure, −5.188 mm Hg versus −0.049 mm Hg for diastolic blood pressure, −1.83 bpm versus −2.215 bpm for pulse rate, and 0.100% versus 0.113% for oxygen saturation. The sedation level in terms of the worst RSS score was not significantly different between the two treatment groups (p = 0.487). Most patients had an RSS score of 2 (cooperative, oriented, and tranquil) at all time points. Only one patient had a score of 3 (responsive to commands only) at 30 min and one patient persisted at a score of 3 after 15 min in the methoxyflurane group.
Repeated-measures analysis of change from baseline in vital sign parameters over 60 min after drug administration (intention-to-treat population).
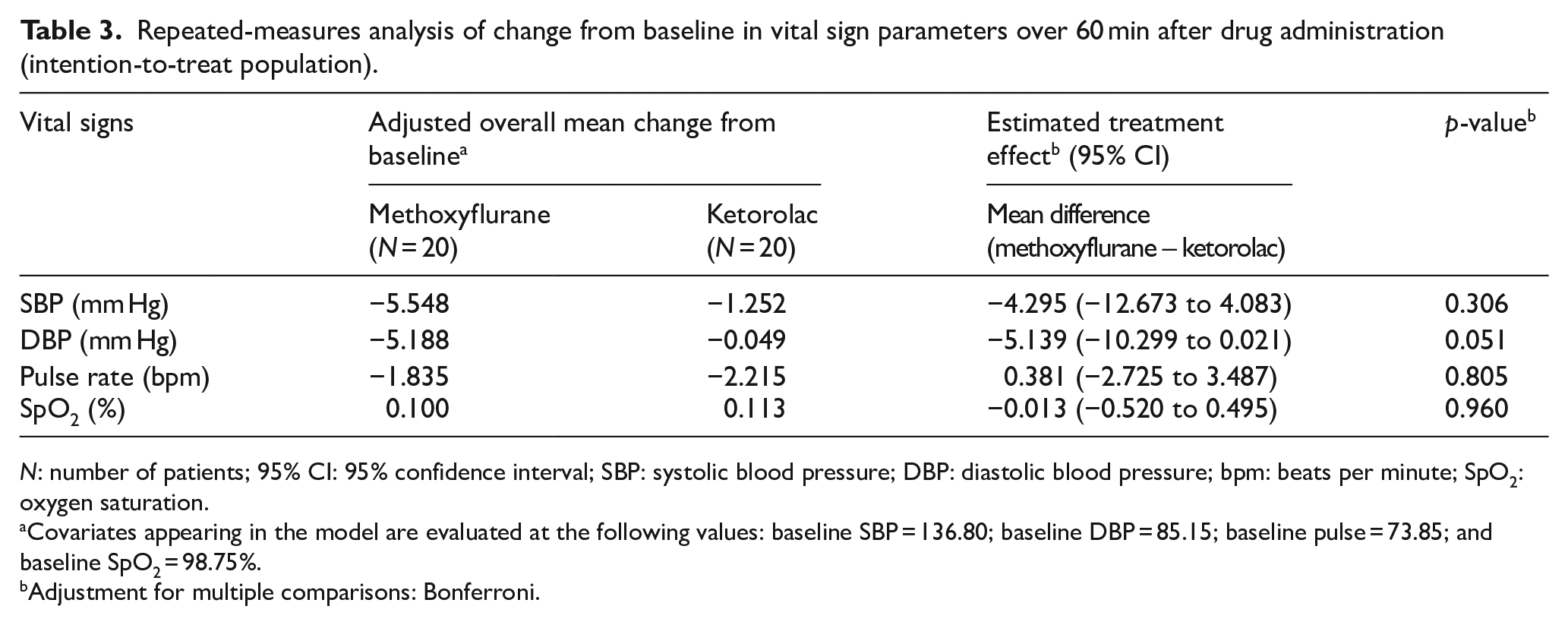
N: number of patients; 95% CI: 95% confidence interval; SBP: systolic blood pressure; DBP: diastolic blood pressure; bpm: beats per minute; SpO2: oxygen saturation.
Covariates appearing in the model are evaluated at the following values: baseline SBP = 136.80; baseline DBP = 85.15; baseline pulse = 73.85; and baseline SpO2 = 98.75%.
Adjustment for multiple comparisons: Bonferroni.
The incidence of TRAEs is summarized in Table 4. TRAEs were reported more frequently in the methoxyflurane group than the ketorolac group (p = 0.008). A total of seven patients (35%) in the methoxyflurane group reported nine TRAEs (six dizziness, two drowsiness, and one cough). None of the patients in the ketorolac group reported TRAEs. There were no serious adverse reactions and the effects were transient. No patients required admission or discontinued the study because of TRAEs. Only one patient in the methoxyflurane requested rescue medication after the completion of VAS assessment. There was no significant difference in the use of rescue medication between the two treatment groups (p = 1.000).
Numbers of patients with treatment-related adverse events. a .
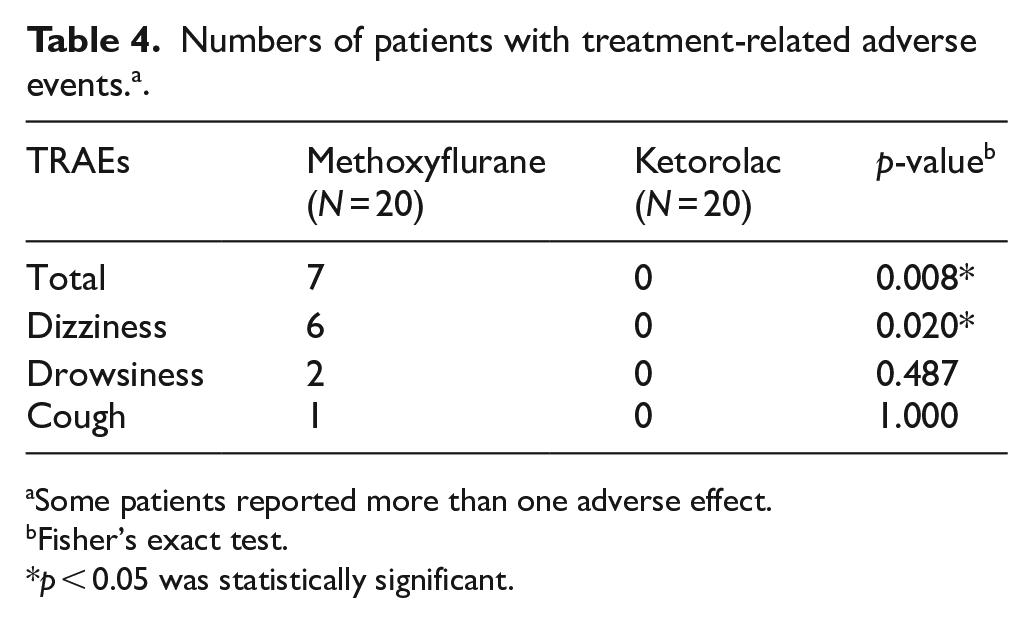
Some patients reported more than one adverse effect.
Fisher’s exact test.
p < 0.05 was statistically significant.
Global medication performance was assessed by patients 60 min after drug administration. A greater proportion of patients in the methoxyflurane group had their satisfaction with pain control rated as “very satisfied” or “satisfied” compared to the ketorolac group (60% vs 55%; p = 0.665; Figure 2(a)). Similarly, more patients rated their overall acceptance of treatment, based on treatment effect, adverse effect, and method of drug administration, as “highly acceptable” or “acceptable” for methoxyflurane than ketorolac (65% vs 60%; p = 0.651; Figure 2(b)).
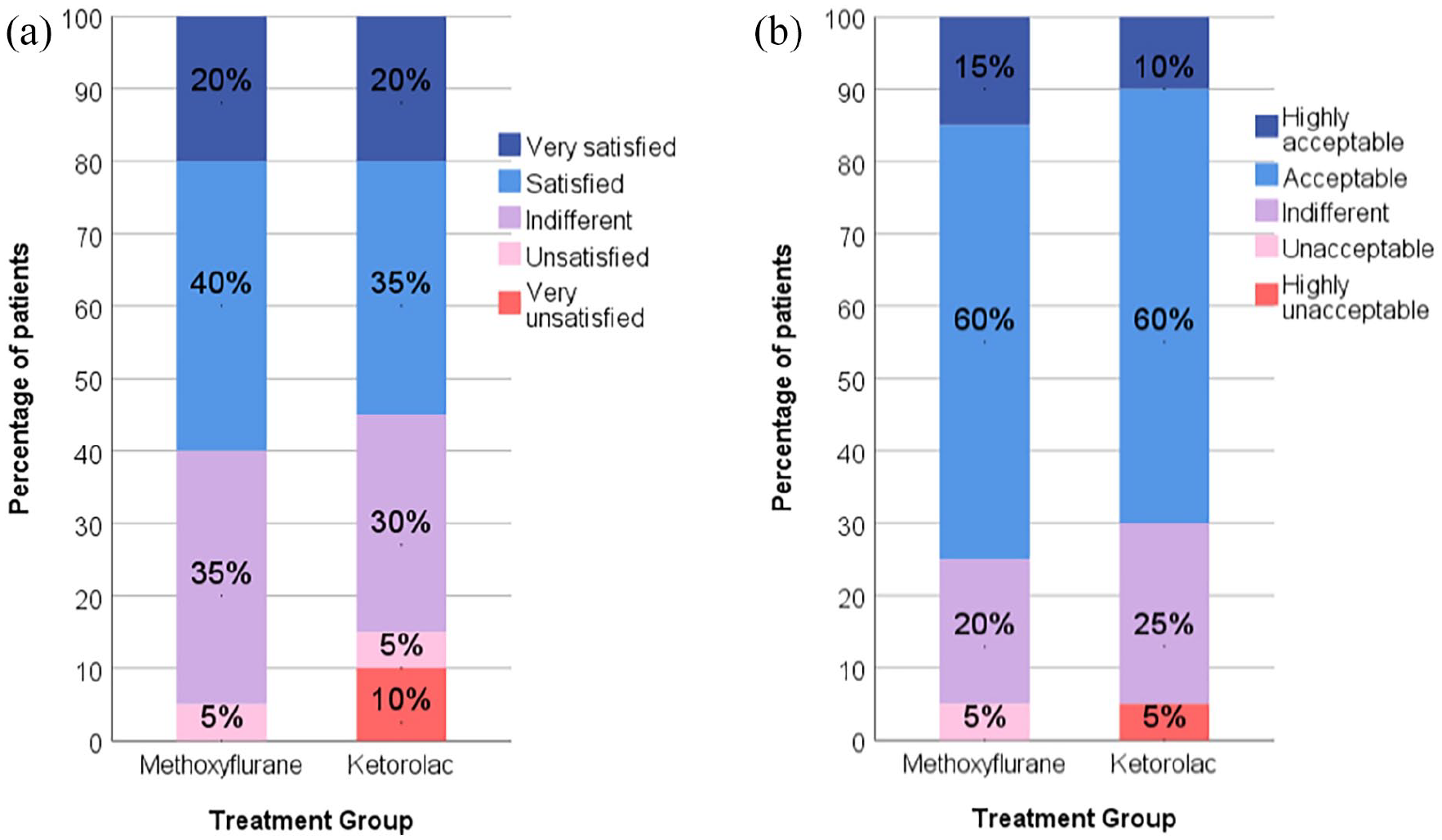
Global level of satisfaction with pain control and overall acceptance rated by patients using 5-point Likert-type scale (intention-to-treat population). a) Satisfaction with pain control b) Overall acceptance.
Discussion
Despite the extensive use of methoxyflurane in Australia and New Zealand for 40 years, only a few studies18–21 have tested methoxyflurane with active comparators for acute pain management in patients with trauma. In the prehospital settings, a large retrospective study showed the analgesic effect of methoxyflurane was inferior to IV morphine and intranasal fentanyl, 18 whereas one randomized controlled trial versus intramuscular tramadol showed that methoxyflurane resulted in a significantly greater and faster reduction in pain score. 19 Two multi-center, open-label, randomized controlled trials carried out in the EDs compared methoxyflurane with standard analgesic treatment (SAT) chosen by the attending emergency physicians according to local protocols on pain management.20,21 The InMEDIATE (Inhaled Methoxyflurane: Pain relief in adult trauma patients in Spain) trial 20 enrolled 156 patients in the methoxyflurane group and 149 patients in the SAT group who were given NSAIDs, metamizole, or paracetamol as the first-step analgesic, with some patients received second-step opioid and diazepam as coanalgesic. The overall reduction in pain score on the NRS in the first 20 min was 2.47 with methoxyflurane versus 1.39 with SAT (treatment effect = 1.00; 95% = CI 0.84 to 1.32). The MEDITA (Methoxyflurane in Emergency Department in Italy) trial 21 enrolled 135 patients in the methoxyflurane group and 135 patients in the SAT group who were given IV paracetamol or ketoprofen for moderate pain (NRS = 4–6) and IV morphine for severe pain (NRS = ⩾7). The overall change in VAS pain intensity in the first 10 min was significantly greater in the methoxyflurane group than the SAT group (–14.73 mm vs −8.78 mm; treatment difference = −5.94 mm; 95% CI = −8.83 to −3.06 mm). Both studies showed that methoxyflurane provided superior pain relief with significant reductions in pain score. However, given the heterogeneity of agents used in SAT pain management, additional studies that compare methoxyflurane with individual analgesic agents are required to fully investigate specific treatment differences.
This study is the first head-to-head prospective trial comparing methoxyflurane with ketorolac as the active comparator, reflecting the current practice in EDs in Hong Kong as ketorolac is a standard therapeutic option commonly used for moderate pain in patients with acute trauma. Our results showed that methoxyflurane provided non-inferior analgesia in the first 30 min and superior analgesia at 5 min with a mean within-group reduction in VAS pain score of 13.912 mm (i.e. >20% improvement from baseline VAS of 64.35 mm) which represented a clinically relevant improvement of pain. Studies by Todd et al. 16 and Gallagher et al. 17 suggested a change of approximately 13 mm on a 100-mm VAS to be the minimum clinically important difference for acute pain, while Cepeda et al. 22 suggested an approximately 20% reduction in NRS pain score corresponds to “minimal improvement” in patients with acute pain. This is consistent with previous studies that reported clinically significant improvement in pain score for the methoxyflurane group at the first assessment time point, ranging from 3 to 5 min.10,20 Comparing with the ketorolac group, which only achieved a VAS pain score reduction of −4.888 mm at 5 min and −14.800 mm at 15 min, methoxyflurane had faster onset to reach clinically relevant improvement of pain. This is not unexpected as the onset of action in inhalation delivery can be as fast as IV. Following inhalation through the mouthpiece, the vaporized methoxyflurane reaches the pulmonary alveoli that provide a large surface area for rapid absorption of drugs into the systemic circulation, thus enabling a rapid onset of analgesic action occurring within 6–10 inhalation. 10 In contrast, at 60 min, methoxyflurane failed to show non-inferiority. One 3 mL methoxyflurane inhaler provides analgesia that lasts for 25–30 min if used continuously or up to an hour with intermittent use. 10 Since patients in the methoxyflurane group were given only one single inhaler, some of them might have the initial vials exhausted before the 60 min study time point. A second inhaler can be administered (the maximum permitted dose is 6 mL in any 24 h) 8 to extend the analgesic effect.
Nevertheless, methoxyflurane resulted in a significant reduction of VAS pain score of 30.392 mm over 60 min, although the magnitude of decline in pain intensity achieved was less than that in the randomized controlled trials conducted in the European countries,20,21 which demonstrated similar degrees of pain score reduction (32.46 mm VAS in the MEDITA trial; 3.19 NRS in the InMEDIATE trial) over a shorter period of 20 min in the methoxyflurane group. This can be explained by the difference in race and ethnicity in the major study populations. Asians were found to have higher pain sensitivity,23,24 higher pain scores, lower pain thresholds, and lower tolerance when compared with Caucasians.25,26 Therefore, the efficacy results in these studies may not be generalizable to the Chinese population in Hong Kong.
Similar to previous clinical trials,10,19–21 more TRAEs were reported in the methoxyflurane group, but none of them were severe and patient-reported ratings indicated high patient acceptance of methoxyflurane. The TRAEs of methoxyflurane observed were consistent with its known safety profile. The most frequently occurring TRAEs, dizziness and drowsiness, were related to the sedative effect of methoxyflurane. However, no safety concerns of excessive sedation were raised as blood pressure and oxygenation of patients remained stable during the study. Previous studies also reported no significant effects of methoxyflurane on vital sign parameters.10,27
The strength of this study lies in its randomized controlled design, allocation concealment, and comparison with a viable alternative that reflects the current practice in Hong Kong. The recruited adult patients with acute trauma pain due to a broad range of injuries commonly encountered in EDs also reflect a real-world emergency setting. Moreover, in contrast to previous studies with shorter assessment endpoints up to 20–30 min, the extra endpoint at 60 min in this study provided additional information about the duration of analgesic effect of methoxyflurane. Nevertheless, there are several limitations to this study. First, the sample size was small and patients were recruited in a single center. Although power calculation was performed to determine the sample size, the analysis was based on the primary outcomes of pain scores. This calculated sample size may not be large enough to prevent a potential type II error for secondary outcomes. The interpretation of these outcomes must be made with caution. Further large-scale multi-center studies are needed to increase the power and generalizability. Second, randomization of patients by drawing from equal preset numbers of envelopes could cause selection bias, but the demographic and baseline characteristics of the patients in both groups were still comparable. Third, both patients and investigators were not blinded to the allocated treatment due to practical differences in the route of administration. A double-blind study using a double-dummy design is preferred to reduce patient and investigator bias, yet it may raise ethical issues as it could result in delayed administration and cause unnecessary pain for patients who received intramuscular placebos. Finally, laboratory safety tests and follow-up were not arranged, but previous clinical studies have identified no concerns of hepatotoxicity and nephrotoxicity from clinical laboratory evaluations performed at baseline and follow-up on day 14.10,20
Conclusion
Methoxyflurane provided non-inferior analgesia in the first 30 min with a faster onset of action when compared with ketorolac in moderate traumatic pain. Although more patients in the methoxyflurane group experienced adverse reactions, this is expected given its pharmacological action, and all those reactions were mild and transient. Therefore, methoxyflurane can be considered as a rapid-acting, effective, and safe first-line alternative to the currently available analgesic treatment for traumatic pain in emergency settings. With its advantages of rapid onset of action, easy-to-use, and non-invasive drug delivery, methoxyflurane can also be used to bridge other analgesics with slower onset or until venous access is available for IV analgesics to be administered. Given the limitations in this study, further large-scale research is needed to confirm our preliminary results and explore the treatment effect in patient subgroups of different demographic characteristics and types of injury.
Supplemental Material
sj-docx-1-hkj-10.1177_10249079211040695 – Supplemental material for A randomized non-inferiority pilot study on the use of methoxyflurane (Penthrox®) for pain control in the emergency department
Supplemental material, sj-docx-1-hkj-10.1177_10249079211040695 for A randomized non-inferiority pilot study on the use of methoxyflurane (Penthrox®) for pain control in the emergency department by Ka Ying Wong, James Siu Ki Lau, Axel Yuet Chung Siu and Pui Gay Kan in Hong Kong Journal of Emergency Medicine
Footnotes
Acknowledgements
The authors thank all the staff of the Accident and Emergency Department, Ruttonjee Hospital who contributed to this study, especially Advanced Practice Nurse Ms Mi-yi Ng for her assistance and Dr Yau-ming Chau and Dr Tak-kuen Chan for their active participation in patient recruitment.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
The data that support the findings of this study are available from the corresponding author, Ka Ying Wong, upon reasonable request.
Informed consent
Written informed consent was obtained from the research subjects for their participation and their anonymized information to be published in this article.
Ethical approval
Ethics approval was obtained from the Hong Kong East Cluster Research Ethics Committee on 22 October 2020 (reference no. HKECREC-2020-064). The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee.
Human rights statement
The author declared that the human rights and safety of research subjects were protected through adherence to local laws, Declaration of Helsinki, institutional policies and whenever applicable, the ICH-GCP.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
