Abstract
Objective:
To evaluate the safety and effectiveness of olanzapine compared to midazolam for the pre-hospital management of excited delirium syndrome.
Methods:
A retrospective review of patients who were treated by ambulance crew for excited delirium syndrome from 2016 to 2019 was performed. The drug of choice was given per Fire Service Department’s paramedic protocol (intramuscular midazolam January 2016 to October 2018 and intramuscular olanzapine October 2018 to December 2019). The primary outcome was the proportion of patients experienced at least one adverse event. The secondary outcome was the successful sedation rate.
Results:
A total of 201 patients were included in the study. Ninety-nine patients had pre-hospital midazolam and 102 received olanzapine. Thirty-one patients experienced adverse event after arrival to A&E, including a drop of Glasgow Coma Scale score to below 8 (90%), hypotension (6%), and airway obstruction (3%): 17 (17% patients; 1 airway obstruction and 16 Glasgow Coma Scale score below 8) in midazolam group and 14 (14% patients; 2 hypotension and 12 Glasgow Coma Scale score below 8) in olanzapine group (p = 0.41). Sixty-eight patients (69%) in midazolam group achieved successful sedation, compared with 79 (77%) in olanzapine group (p = 0.20). More adverse events were seen with midazolam than olanzapine in alcohol intoxication subgroup with statistical significance (12 vs 2, p = 0.03).
Conclusion:
Both midazolam and olanzapine can achieve a satisfactory success rate of sedation. More adverse events are associated with midazolam use in patients with acute alcohol intoxication, especially over-sedation and airway compromise. Olanzapine would be a safer choice in the pre-hospital setting where the resource is limited, and differentiation of underlying causes is not always possible.
Introduction
Excited delirium syndrome (ExDS), also named as agitated delirium or acute behavioral disorder, is a clinical emergency in both emergency departments (EDs) and pre-hospital setting.1,2 It can lead to complications, patient and staff injuries, as well as property damage if the incident is not safely and effectively managed. 3 Ambulance personnel are at the forefront of managing this challenging condition. To facilitate timely medical assessment, treatment, and safe transport to hospital, parenteral sedation is mandated if verbal de-escalation fails.4,5
The choice of parenteral agent for ExDS often comes down to two drug classes, benzodiazepines and antipsychotics, either alone or in combination. 6 Benzodiazepines act rapidly, but adverse events (AEs) and requirement for additional sedation are common.7–9 Antipsychotics, such as droperidol, olanzapine, and haloperidol, are considered to be as effective as benzodiazepines, being safer, and with a lower requirement for additional sedation.7,9–11
In Hong Kong, benzodiazepines and antipsychotics are frequently used as monotherapy for the management of ExDS in the ED. 12 Recently, olanzapine has been gaining popularity among emergency physicians for use in treating agitation because of its high efficacy and good safety profile. 13 In the pre-hospital setting in Hong Kong, the drug of choice was intramuscular midazolam from January 2016 to October 2018 and intramuscular olanzapine from October 2018 to December 2019, according to the paramedic protocol. The majority of the studies in this area are in the ED setting, while pre-hospital studies of benzodiazepines and antipsychotics for ExDS are uncommon.14–16 Moreover, direct comparison between parenteral midazolam and parenteral olanzapine in the pre-hospital setting has not previously been reported in the literature. Therefore, we conduct a study to determine the safety and effectiveness of olanzapine compared to midazolam for the management of acute behavioral disturbance in pre-hospital setting. The results will give insight to the choice of sedatives for pre-hospital management of ExDS.
Methods
Study design and data extraction
A retrospective review of patients who were treated by ambulance crew for ExDS was performed from 2016 to 2019. Data were retrieved from the Paramedic Services Quality Assurance System (PSQAS) of Fire Service Department (FSD) by QA team of Paramedic Training Centre of Fire and Ambulance Services Academy. Patient’s clinical data and their outcomes were then matched by using the Accident and Emergency Information System (AEIS) and the Clinical Management System (CMS) of the Hospital Authority (HA) by two investigators and entered into a spreadsheet. The drug of choice was given per FSD’s paramedic protocol, in which intramuscular midazolam 5 mg was given from January 2016 to October 2018 while intramuscular olanzapine 10 mg was given from October 2018 to December 2019. The reason for switching the drug from midazolam to olanzapine was due to administrative and logistic issues. Patient demographics, prescriptions for olanzapine, reason for administration, AEs, additional sedatives used, comorbidities, clinical diagnoses, and patient discharge destination were recorded. The study was approved by FSD and the HA Research Ethics Committee (HA REC).
Pre-hospital protocol for ExDS
Ambulance personnel were authorized to administer sedatives (intramuscular midazolam 5 mg or intramuscular olanzapine 10 mg) when the patient is agitated and aggressive with a tendency to harm oneself or other people, and at the same time verbal de-escalation fails. The protocol would not be executed if the patient had history of allergic reaction to the drug and previous history of neuroleptic malignant syndrome.
Inclusion and exclusion criteria
Patients of age 12 years or older having received parenteral midazolam or olanzapine in the pre-hospital setting for ExDS from 1 January 2016 to 30 September 2019 were eligible for inclusion in the study. Those electronic ambulance journey reports with the corresponding ED records missing were excluded.
Outcomes
The primary outcome was the proportion of patients experienced at least one AE. AE included any of the following: airway obstruction requiring airway maneuver or intervention, oxygen saturation < 90% on room air, respiratory rate < 10 per min, systolic blood pressure < 90 mmHg, GCS score below 8, dystonic reactions (for olanzapine, an antipsychotic drug), and allergic reactions.
The secondary outcome was the successful sedation rate, which was defined as the proportion of patients who did not develop AE and at the same time did not require additional sedation within 1 h after arrival to ED. Other outcome measurements include ED additional sedation defined as any additional sedation administered during the ED stay, additional sedation administered within 1 h of initial sedation, number of additional sedation administrations, time of pre-hospital sedation to the first additional sedation in the ED, time of ED stay, hospital admission and the admitting specialty, number of injuries to patients or staffs, and death during the ED stay and within 30 days after the index ED attendance.
Statistical analysis
The sample size for the study is based on the estimated AE rates of midazolam and droperidol (an antipsychotic drug) taken from a previous pre-hospital study. 14 For midazolam this was 23% and for droperidol 7%. Using a β of 0.2 (power 80%) and α of 0.05, it was calculated that 154 (77 each arm) was the sample size required for the study. The primary and secondary outcomes were reported using descriptive statistics including median, interquartile range (IQR), and ranges for continuous data. Mann–Whitney U test was employed for numerical data analysis while Fisher’s exact test was used to analyze categorical data. A p value of < 0.05 was considered statistically significant. IBM SPSS (version 26; IBM Corp., Armonk, NY) was used for all analyses. Subgroup analysis was performed to find out the association between the AE rate in different etiologies of ExDS and sedatives of choice.
Results
There were 217 patients aged 12 years or older with ExDS received pre-hospital intramuscular midazolam or olanzapine during the study period, in which 110 patients received midazolam and 107 patients received olanzapine. Of these, 11 midazolam and 5 olanzapine patients were excluded because of missing data, leaving 201 patients (142 males (71%) median age 34 years; range: 12–88 years), of which 99 patients received midazolam and 102 patients received olanzapine, that were included for analysis (Figure 1). Baseline characteristics and vital signs are compared for both groups and are similar (Table 1).
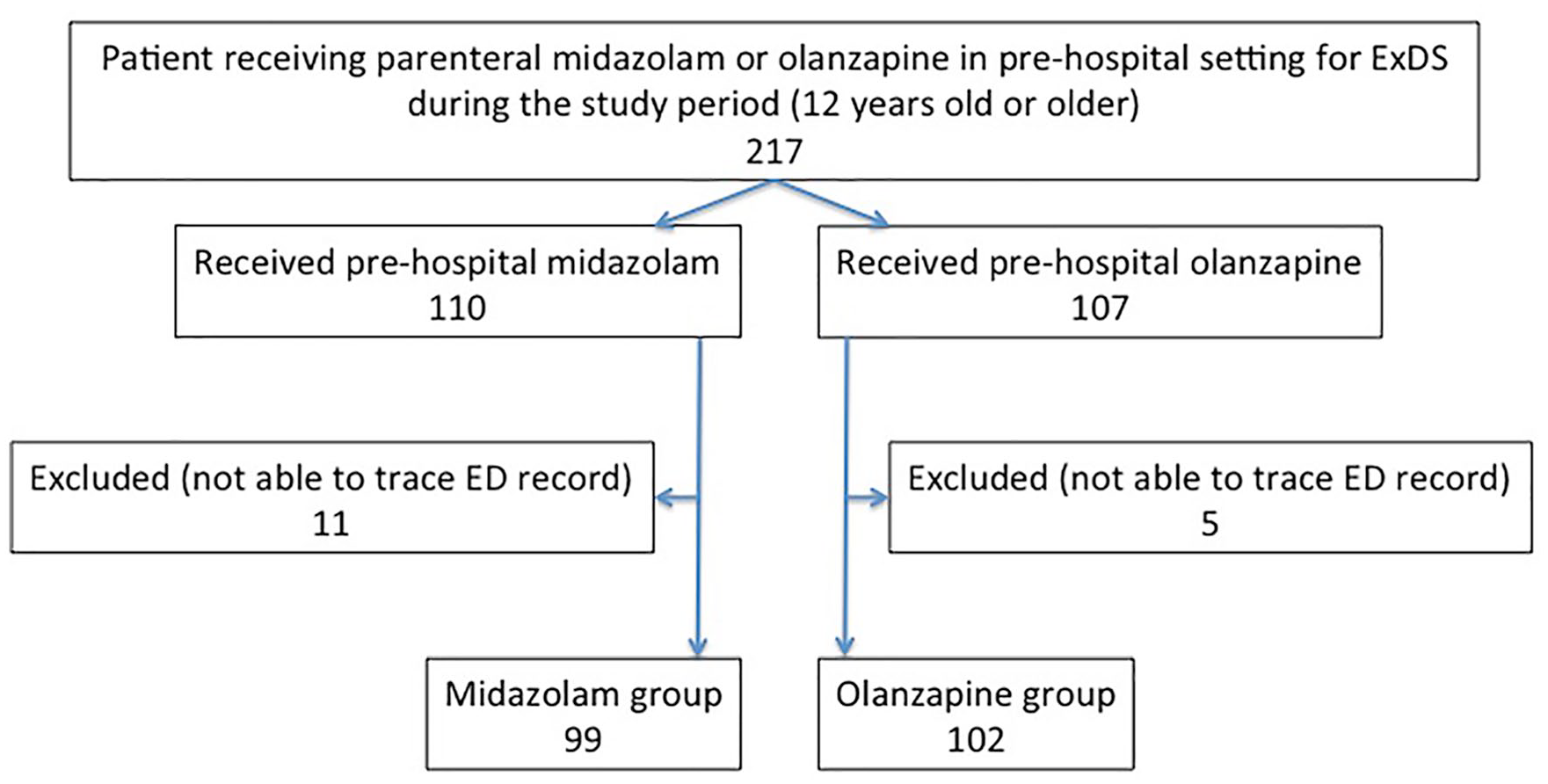
Flow chart of all patients with acute behavioral disturbance during the study period.
Baseline characteristics of the two groups.
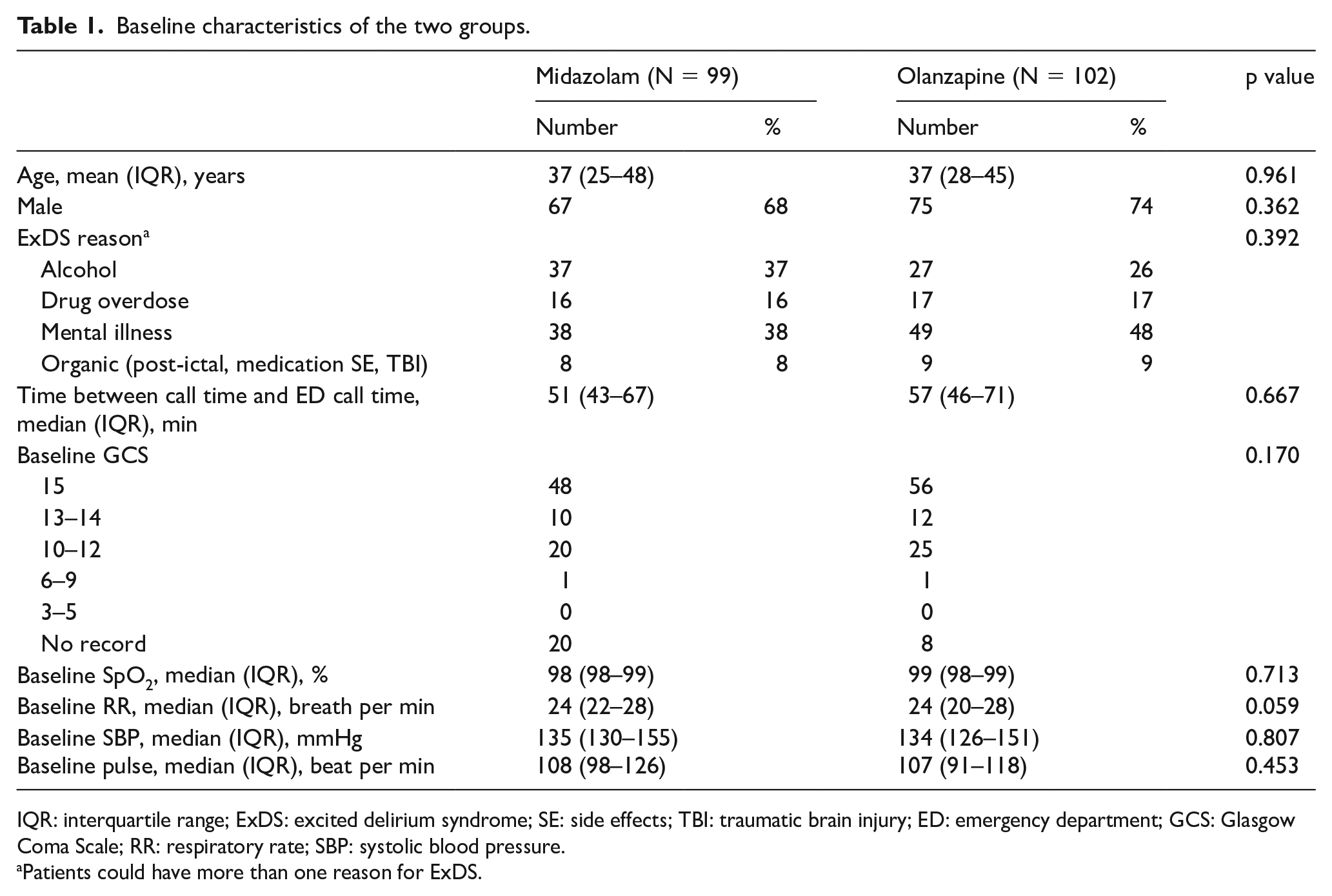
IQR: interquartile range; ExDS: excited delirium syndrome; SE: side effects; TBI: traumatic brain injury; ED: emergency department; GCS: Glasgow Coma Scale; RR: respiratory rate; SBP: systolic blood pressure.
Patients could have more than one reason for ExDS.
There were 17 AEs in 17 patients (17/99 (17%)) in the midazolam group compared to 14 AEs in 14 patients (14/102 (14%)) in the olanzapine group (p = 0.42) (Table 2). In midazolam group, 16 (16%) patients dropped in GCS to below 8, while 1 (1%) patient had airway obstruction requiring nasopharyngeal airway. In olanzapine group, AEs were 2 (2%) episodes of hypotension and 12 (12%) patients dropped in GCS to below 8.
Adverse events.
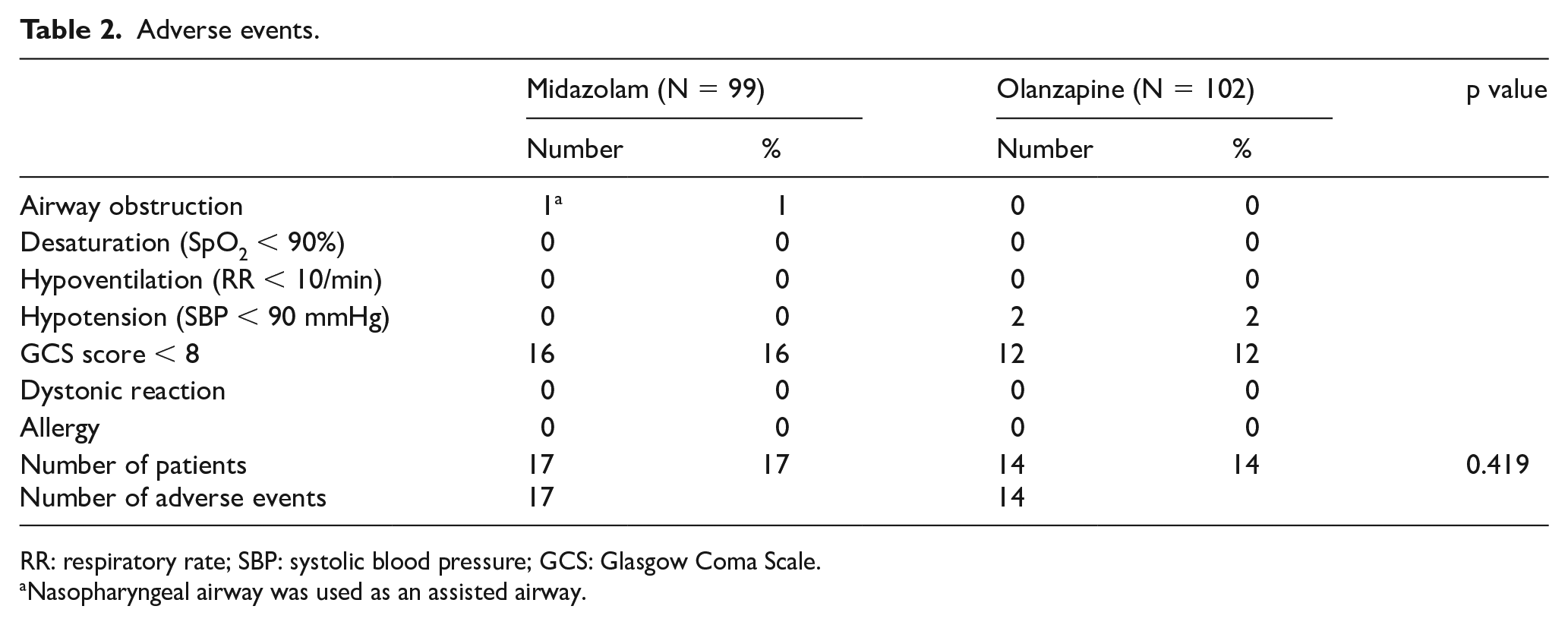
RR: respiratory rate; SBP: systolic blood pressure; GCS: Glasgow Coma Scale.
Nasopharyngeal airway was used as an assisted airway.
Secondary outcomes are presented in Table 3. We defined successful sedation as no additional sedation in the ED required and no AE encountered. Overall, there were 68 (69%) patients in the midazolam group and 79 (77%) patients in the olanzapine group who achieved successful sedation (p = 0.20) (Figure 2). Additional sedation after arrival to the ED was required for 26 patients (26/99 (25%)) who received midazolam compared to 19 patients (19/102 (19%)) who received olanzapine, in which additional sedation within 1 h was required for 19 patients (19%) who received midazolam compared to 13 patients (13%) who received olanzapine. The median number of additional sedation administration in the midazolam group was one (range, 1–5) and in the olanzapine group was one (range, 1–3). The median time between pre-hospital sedation and first additional sedation in the ED was 32 (IQR: 29–54) min in midazolam group compared to 45 (IQR: 38–76) min in olanzapine group (p = 0.04). The median time of ED stay was 128 (IQR: 61–264) min in midazolam group compared to 151 (64–353) min in olanzapine group. There were 71 (72%) hospital admissions in midazolam group compared to 75 (74%) in olanzapine group. The most common specialty of hospital admission in both groups were Emergency Medicine (midazolam 32; olanzapine 27), Psychiatry (midazolam 16; olanzapine 21), and Internal Medicine (midazolam 15; olanzapine 20). There was no injury in both groups to patients, ambulance personnel, and ED staff. No death during the ED stay was reported in both groups. Two patients (2%; one sepsis and one sympathomimetic drug overdose with cardiac arrest) died within 30 days of ED attendance in midazolam group compared to zero in olanzapine group. No non-compliance was noted by the Quality Assurance team of FSD during the study period.
Secondary outcomes in the two groups.
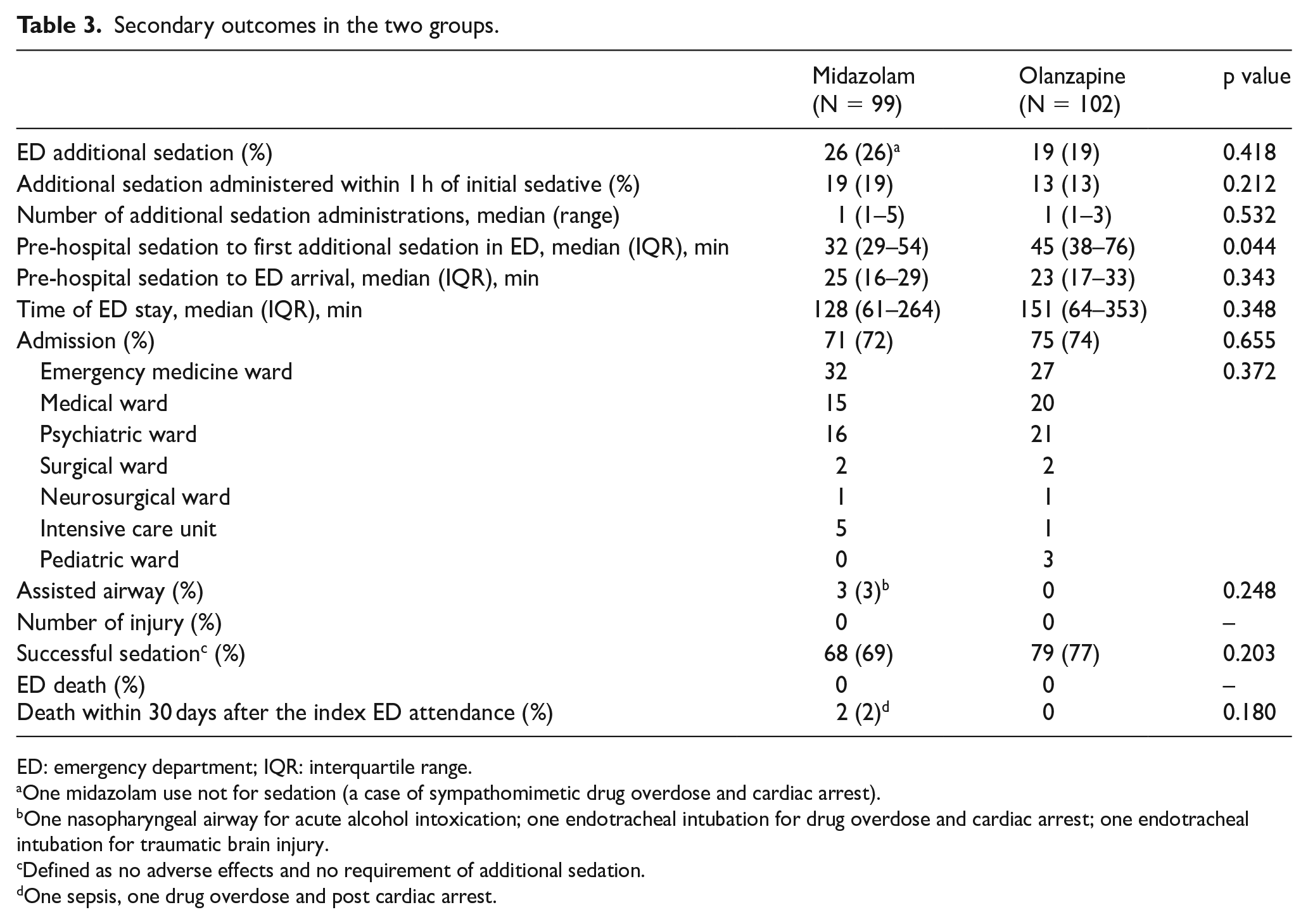
ED: emergency department; IQR: interquartile range.
One midazolam use not for sedation (a case of sympathomimetic drug overdose and cardiac arrest).
One nasopharyngeal airway for acute alcohol intoxication; one endotracheal intubation for drug overdose and cardiac arrest; one endotracheal intubation for traumatic brain injury.
Defined as no adverse effects and no requirement of additional sedation.
One sepsis, one drug overdose and post cardiac arrest.
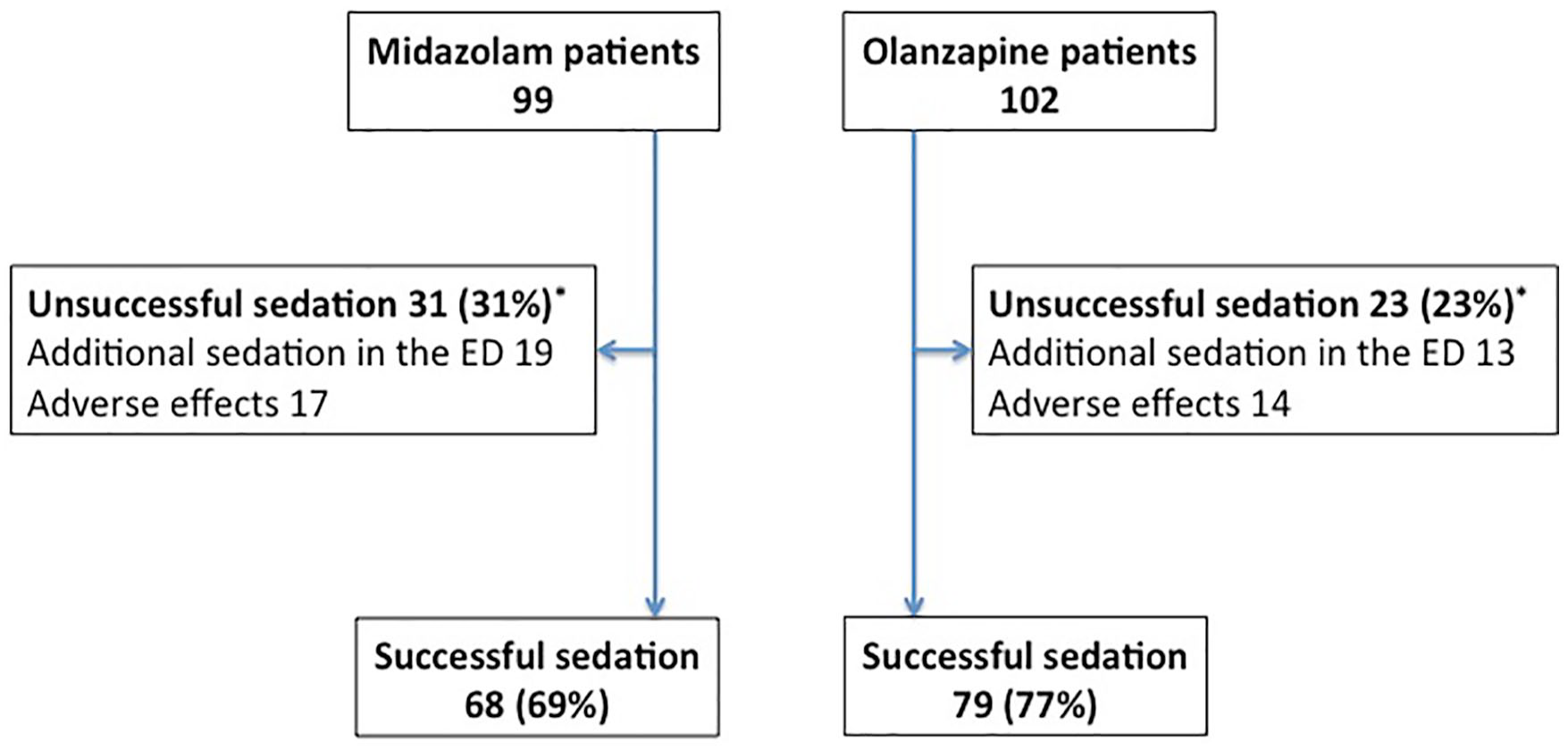
Patients successfully sedated.
Subgroup analysis
We divided the patients into four subgroups according to the cause of ExDS: acute alcohol intoxication (total 64 patients (64/201: 32%); midazolam 37 patients (58%); olanzapine 27 patients (42%)), drug overdose (total 33 patients (33/201: 16%); midazolam 16 patients (48%); olanzapine 17 patients (52%)), mental illness (total 87 patients (87/201: 43%); midazolam 38 patients (44%); olanzapine 49 patients (56%)), and organic cause (total 17 patients (17/201: 8%); midazolam 8 patients (47%); olanzapine 9 patients (53%)). Baseline characteristics are similar between midazolam and olanzapine in all subgroups. The majority of drug overdose was caused by stimulants (69% in midazolam group; 76% in olanzapine group).
Primary outcomes of the subgroups are listed in Table 4. Most AEs were associated with acute alcohol intoxication (57%), in which 11 (11/37 (30%)) patients who received midazolam had a GCS below 8 and one (1/37 (3%)) had airway obstruction requiring nasopharyngeal airway, compared to two AEs (GCS below 8) in olanzapine arm (p = 0.03; odds ratio (OR) = 8.4, 95% confidence interval (CI) = 1.9–37.9). This difference is statistically significant.
Adverse events in the subgroups.
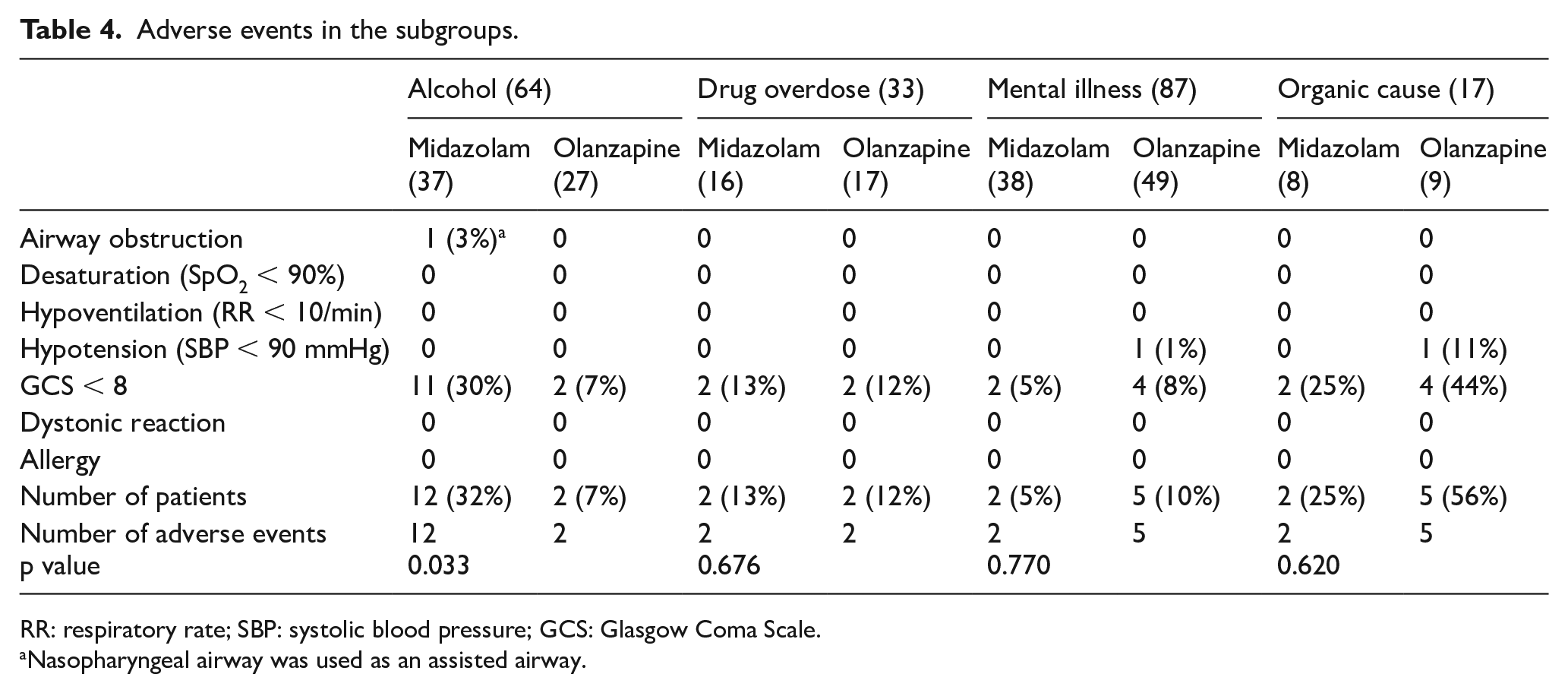
RR: respiratory rate; SBP: systolic blood pressure; GCS: Glasgow Coma Scale.
Nasopharyngeal airway was used as an assisted airway.
There is no statistical difference between pre-hospital midazolam and olanzapine in patients with ExDS of all four causes in the rate of successful sedation.
Discussion
To our knowledge, this is the first study, though not a randomized control trial, to compare midazolam with olanzapine for management of undifferentiated ExDS in pre-hospital setting. We found that 73% (147/201) of all patients achieved adequate sedation for clinical management and did not require additional sedation. No death was reported during the stay in ED. The overall AE rate was 15% (31/201). We defined over-sedation with GCS dropped to below 8 as one of the AEs because these patients are often prone to have more complications, for example, aspiration and airway problems, and they may need longer observation in ED before discharge. A GCS score less than 8 is an indication for intubation for airway protection according to the Advanced Trauma Life Support (ATLS) protocol. Previous study on the relationship between GCS and airway protective reflexes stated that reduced GCS is significantly related to gag and cough reflexes. 17 No staff or patient injury was reported in both groups. Two deaths were reported within 30 days of admission in midazolam group but the causes were not directly related to the sedation. One died of severe sepsis while the other died of sympathomimetic overdose in which additional doses of benzodiazepine were given. There were three patients requiring airway interventions in which two of them had endotracheal intubation, but both cases were not purely a sequel of the medication itself. Sedation can lead to hypoxia and respiratory depression necessitating airway intervention, particularly in patients intoxicated from alcohol, but intubation may also be due to the consequence of underlying pathologies causing the altered mental status and agitation. 18
Atypical antipsychotics appear to have a risk of QTc interval prolongation, though these agents have only been implicated in the development of Torsades de Pointes (TdP) in rare case reports and FDA AE reports.19,20 However, olanzapine was found to have low risk in association with QTc prolongation and TdP. 21 In the recent randomized controlled trial, no statistical difference of QTc prolongation was found among midazolam, olanzapine, and haloperidol groups. 22 Since electrocardiogram (ECG) was not done in all cases in this retrospective study, data collection on QTc interval was not performed. In subgroup analysis, midazolam used in patients with alcoholic intoxication was associated with more AE that is, over-sedation and airway obstruction while olanzapine did not have any AE. Different hospitals have different management protocols and ethanol level was not routinely checked for ExDs so ethanol level was not available in this retrospective study. Compared to the recent randomized controlled trial, 22 no significant differences in AE rates were found between midazolam and olanzapine as an initial pharmacological treatment for undifferentiated acute agitation. However, the perceived alcohol intoxication in this study is lower than this study (23.9% vs 32%). Other previous studies in both pre-hospital13,14 and ED setting7,8,10,11 also found higher rate of AE when midazolam was used, though subgroup analysis to different causes of ExDS was not performed in these studies. This may have clinical implication when managing ExDS as a significant portion of them was caused by alcohol intoxication (32%) as shown in this study. Olanzapine would be a better choice in this setting especially when the underlying cause leading to ExDS is often unknown to ambulance personnel. Moreover, midazolam, being one of the on-list dangerous drugs, requires special storage and handling. Using olanzapine instead would save the tedious steps of signing in and out when taking and returning the drugs.
Additional sedation was used to measure the effectiveness outcome in this study. One may argue that this may not be an accurate measurement of clinical efficacy of a sedative and may subject to many confounding factors, for example, local guidelines, physician’s preference, and need for imaging. However, additional sedation is considered to be a meaningful outcome in agitation research and also in clinical practice because rapid and effective sedation achieved with a single injection has a number of advantages. 23 Uncontrolled agitation and delirium may impose danger of physical injuries to patient and ambulance crew while repeated medication administration increases the risks of needle stick injuries. As a result, rapid sedation achieved after a single administration of medication may help shorten the duration of acute behavioral disturbance and prevent physical injuries and metabolic complications related to the prolonged agitated state. Furthermore, effective sedation in a timely manner can also expedite a full evaluation of the patient to rule out other causes of altered mental status that may require additional diagnostics and interventions.18,24 Previous studies in the pre-hospital and ED settings showed greater requirement for additional sedation with midazolam over antipsychotics.7,9,10,14,25 However, our study found no statistical difference in additional sedation between midazolam and olanzapine (19% vs 13%; p = 0.21). Direct comparison of additional sedation rate among the studies is difficult as the definitions of additional sedation and protocols vary among studies. In an ED study, it was defined as with 6 h 7 while in another study, 1 h was the limit. 11 In a pre-hospital study, 14 it meant any sedation given after successful sedation in the ambulance or in the first hour after arrival to the ED. In our study, it included any sedation given after the initial sedation on ambulance and during the ED stay.
The median time between pre-hospital sedation and first additional sedation in the ED for olanzapine group was significantly longer than midazolam group (45 min IQR 38–76 vs 32 min IQR 29–54; p = 0.04). This could be explained by the longer duration of action of olanzapine. A longer time before the first additional sedation means more time is available for managing the patient in the ED. The duration of ED stay, however, did not differ significantly between two groups (olanzapine 151 min IQR 64–353 vs midazolam 128 min IQR 61–264; p = 0.35). A longer ED stay is subject to many factors, including the need for in-patient consultation, admission block, turnover rate of lab results, and availability of imaging.
This study has several limitations, including those related to its retrospective design. There is a possibility of ambiguous, missing, and unknown data in data collection from the medical records. The sample size is small in this study though the data-missing rate was not high (7.37%). Since this retrospective study looked into two different periods in which two different sedatives were used, one may argue that the findings could be attributed to measurement bias over time. However, the measurement mainly involved clinical parameters including GCS and vital signs as well as complication detection. Those are the core skills of paramedics in their daily practice and would not be expected to change dramatically over time. In addition, the findings of this study may not be generalizable to other populations with different patient demographics, emergency medicine service system, ED settings, and in-patient services. Another potential limitation was utilization of additional sedation as our effectiveness outcome. We decided to use this outcome in this study given the retrospective nature, as it is objective and reliably determined. Chan et al. 22 found that midazolam resulted in faster sedation in patients with undifferentiated agitation in the ED setting compared to olanzapine. We did not attempt to measure the onset of action and duration of sedation of different sedatives in this retrospective study. Since other methods of assessing sedation are available, these performance parameters would better be evaluated by prospective studies in pre-hospital setting in future.14,23,26 Also, ethanol level may affect the GCS and subsequent AE rate after sedation. Future prospective study should include this as a parameter for further analysis.
Conclusion
Both midazolam and olanzapine can achieve a satisfactory success rate of sedation. More AEs are associated with midazolam use in patients with acute alcohol intoxication, especially over-sedation and airway compromise. Olanzapine would be a safer choice in the pre-hospital setting where the resource is limited, and differentiation of underlying causes is not always possible.
Footnotes
Acknowledgements
We would like to thank QA team of Paramedic Training Centre of Fire and Ambulance Services Academy, Fire Services Department of Hong Kong for data collection and retrieval of records.
Author contributions
W.L.L. and K.L.M. contributed to study design. All authors contributed to acquisition of data. W.L.L. and K.L.M. analyzed the data. W.L.L. drafted the manuscript. K.L.M. contributed to critical revision for important intellectual content.
Data availability statement
Raw data were generated from the Paramedic Services Quality Assurance System (PSQAS) of Fire Services Department and, the Accident and Emergency Information System (AEIS) and the Clinical Management System (CMS) of the Hospital Authority. Derived data supporting the findings of this study are available from the corresponding author W.L.L. on request.
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the Hospital Authority Research Ethics Committee (REC Ref. No.: HKECREC-2019-101) and the Fire Service Department. The requirement for patient consent was waived by the ethics board.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
