Abstract
Introduction:
Acute carbon monoxide (CO) poisoning could lead to headache, dizziness, myocardial injury, neurological sequela, and death. Sudden hearing loss is a rare symptom of acute CO poisoning.
Case Presentation:
Here, we report a case of a 42-year-old woman who suffered from acute hearing loss after exposure to a suicidal environment of high concentration of CO. Partial recovery of hearing was demonstrated after a combination of corticosteroid and hyperbaric oxygen therapy was given.
Discussion:
The mechanism of sudden hearing loss caused by acute CO poisoning is not well-established. It is believed to be related with the hypoxic damage to the cochlea. The characteristic of sudden hearing loss caused by acute CO poisoning is that it often affects in high frequency bilaterally.
Conclusion:
This report would prompt the clinician in early recognition of this sudden hearing loss of uncommon etiology.
Introduction
Carbon monoxide (CO) is a colorless, odorless gas produced as a by-product of incomplete combustion of carbon-containing compounds. The presentation of CO poisoning can be classified into accidental and intentional. The symptoms of accidental CO poisoning are often non-specific including impaired concentration, sleep disturbance, lethargy, fatigue, dizziness, nausea/vomiting, blindness, and chest pain.1,2 CO poisoning is an uncommon cause of hearing loss, especially after acute poisoning. 3 Delayed diagnosis of sudden hearing loss may decrease the effectiveness of treatment. If there is no severe conscious disturbance and clear exposure history of CO, CO poisoning would be misdiagnosed easily. Here, we report an unusual clinical case of sudden hearing loss following acute CO poisoning to bring into the notice of clinicians the emergent pitfall.
Case report
A 42-year-old woman came to our emergency department with symptoms of hearing loss along with dizziness, nausea, and chest tightness for few hours. She complained of hearing loss in the left ear and tinnitus in the right ear. She had a history of smoking over 10 years with daily consumption of 10 cigarettes. She was apparently healthy with no history of hearing loss in the past. Initially, Meniere’s disease was diagnosed. However, after a thorough history session, she reported that her husband was found unconscious from carbon monoxide poisoning associated with home fuel-burning heating equipment when she came home in the morning. She started to feel discomfort after she had completed the admission program for her husband. Her vital signs at admission were as follows: pulse rate: 100 beats/min; respiratory rate: 15/min; blood pressure: 115/80 mmHg; and temperature: 37.3°C. Her Glasgow Coma Scale was 15. The neurological examination was unremarkable.
Arterial blood gas showed a pH of 7.4, a partial pressure of carbon dioxide (pCO2) of 33.2 mmHg, a pO2 of 95.5 mmHg, an HCO3 of 26.0 mmol/L, and arterial oxygen saturation (sO2) of 100%. Laboratory test results showed elevated white blood cell count of 11.3 x 109/L. We also checked the level of cardiac enzyme and carboxyhemoglobin (COHb) due to chest pain and exposure to CO. Elevated levels of troponin I of 0.5 μg/L (normal range, 0~0.3μg/L), creatine kinase-MB fraction of 60 μg/L (normal range, 0~20 μg/L), and COHb of 12% were noted after termination of exposure without oxygen supplement for about four hours. Chest X-ray imaging showed a small focal consolidation on the lower right lobe.
Due to the elevated COHb level and sudden hearing loss on the admission day, the patient was referred to the hyperbaric therapeutic unit and underwent a session of oxygen therapy at 3 atmosphere absolutes (ATA) and 90-min duration. Thereafter, an audiometric test was performed, which showed bilateral asymmetrical mid- to high-frequency hearing loss (Figure 1(a)). For the right ear, findings indicated hearing loss at frequencies of 500 Hz; 1000 Hz; 2000 Hz; and 8000 Hz. For the left ear, findings indicated hearing loss at 500 Hz, 1000 Hz, 2000 Hz, 4000 Hz, and 8000 Hz. Bone conduction thresholds interweaved with air conduction thresholds indicating the impairment to be sensorineural in origin bilaterally. A tapered course of intravenous corticoid treatment was prescribed (methylprednisolone at a dose of 1 mg/kg/day) for 2 weeks. And the patient was subjected to a total of 10 sessions of hyperbaric oxygen (HBO) therapy at 3 ATA of 90 min duration each. About 2 weeks later, when the HBO therapy was finished, the patient reported improvement in hearing loss and subsiding of tinnitus. We repeated the audiometric test, which revealed a partial recovery of hearing loss (Figure 1(b)).
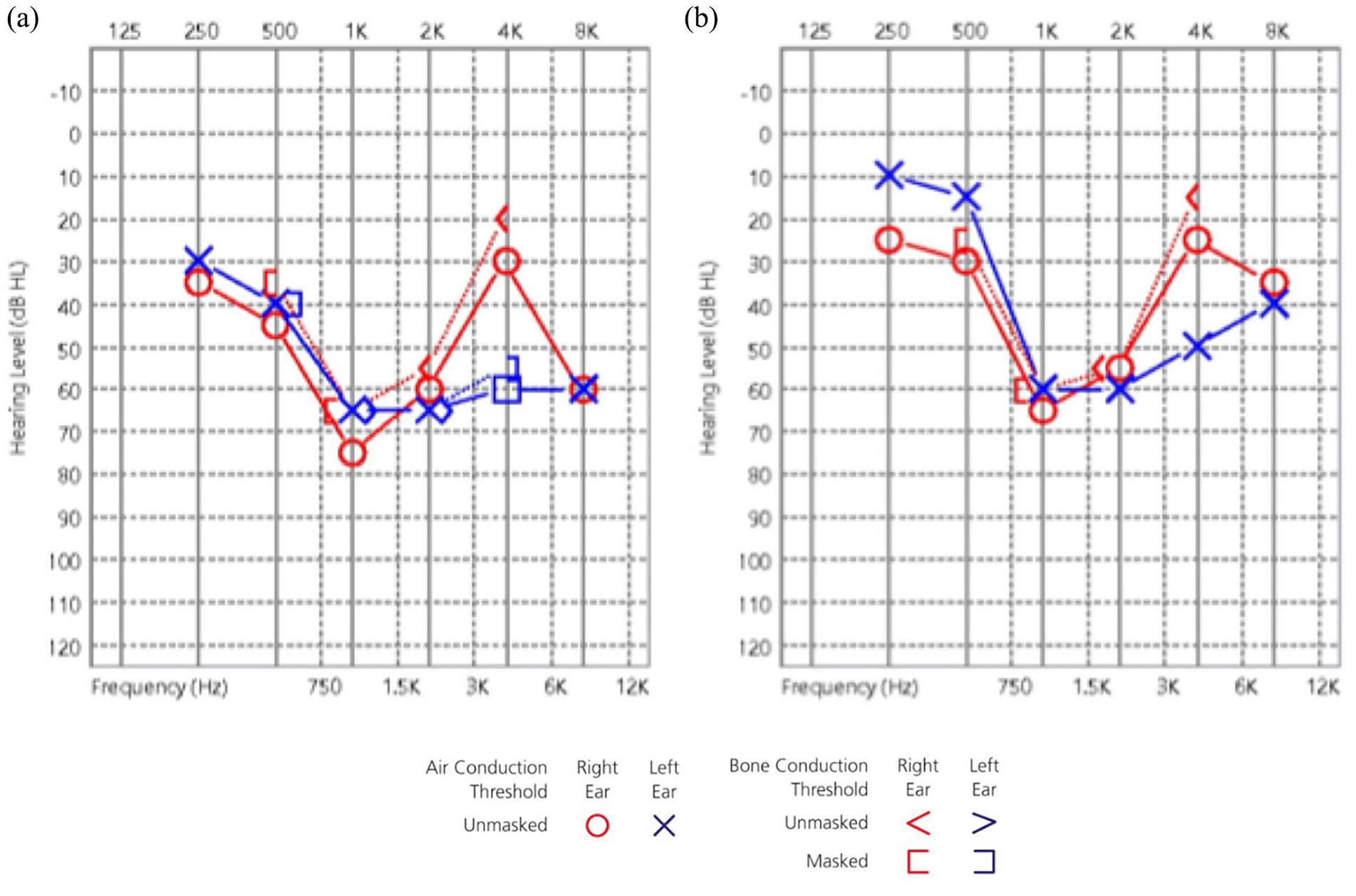
Patient’s audiometric test (a) on the day of admission and (b) after 10 sessions of hyperbaric oxygen (HBO) therapy.
Discussion
CO poisoning is a major health issue accounting to 40,000 emergency department visits and up to about 6000 deaths annually in the United States alone. However, sudden hearing loss caused by acute CO poisoning is uncommon and easily misdiagnosed.4,5
CO poisoning is ideally diagnosed by a clinical triad: symptoms consistent with CO poisoning, history of recent CO exposure, and elevated COHb levels. However, these criteria are not strict and the most important one is the exposure history. COHb levels of 2% or greater in non-smokers and 10% or greater in smokers are considered abnormal and may produce symptoms. 2 CO poisoning depends on the amount of time of exposure, concentration of CO in the environment, and minute alveolar ventilation. 6 CO binds to the hemoglobin molecule in red blood cells with an affinity that is 300 times that of oxygen. The half-life of the CO in room air is about 300 min, which could be reduced to as low as 30 min under hyperbaric oxygen conditions, 100% oxygen at 3 ATA. 6
Lumio 1 presented 78% incidence of sudden hearing loss among 700 cases of chronic CO poisoning in 1948. However, the first study of sudden hearing loss caused by acute CO poisoning was not reported until 1969. 7 Since then, a few reports have been published about the sudden hearing loss following acute CO poisoning (Table 1).3–11 One study reported that dichloromethane (DCM or methylene chloride), which can metabolize to CO, and this can also result in hearing impairment when intoxicated. 5 Regarding sudden hearing loss, we need early identification of the etiology, such as infectious diseases, neurological disorders, ototoxic agents, immunologic causes, head injury, or neoplasms. 12 However, we also need to emphasize the role of some ototoxic substances found at the workplace and home environment, such as toluene, styrene, lead, trichloroethylene, mercury, carbon disulfide, and CO. 13 The correlation between the severity of hearing impairment and duration and/or concentration of exposure to CO is not well established. In addition, the mechanism of hearing damage caused by CO is still in debate. One hypothesis is that the binding of carbon monoxide with hemoglobin to form COHb reduces the oxygen’s transport ability leading to tissue hypoxia. Under hypoxic environments, the base of the cochlea cannot perform aerobic metabolism due to the lack of blood flow and oxygen to the brain, which causes damage to cells at the base of the cochlea. 14
Summary of case reports of sudden hearing loss following acute CO poisoning.
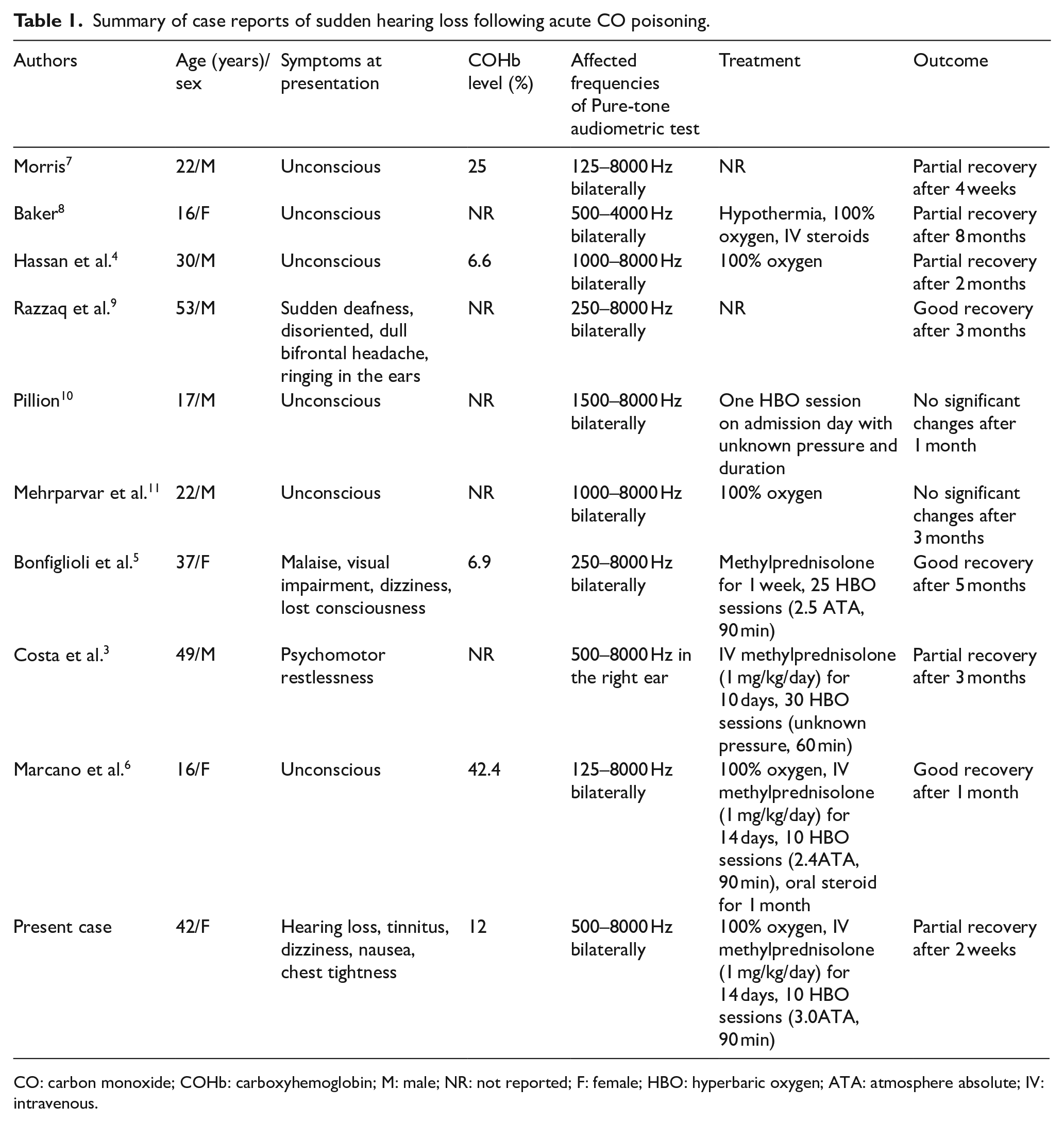
CO: carbon monoxide; COHb: carboxyhemoglobin; M: male; NR: not reported; F: female; HBO: hyperbaric oxygen; ATA: atmosphere absolute; IV: intravenous.
The characteristics of CO-induced hearing loss can be concluded in three points: high-frequency deficit (due to hypoxic damage to the base of the cochlea), bilateral, and reversible as presentation of our case.3,4,6,10 Nevertheless, although rare, a low-frequency deficit, unilateral hearing loss, and an irreversible condition have also been reported,3,10, 11 audiometry of which may present a “U-shaped” curve, also called a “cookie-bite.” 8
If a patient is diagnosed with hearing loss caused by acute CO poisoning, the subsequent management we suggested should be done as below: (1) termination of exposure of CO; (2) monitoring for any cardiac, neurological, and pulmonary complications; (3) corticosteroid therapy;3,6 and (4) HBO therapy. Chronic exposure to CO could lead to a poor prognosis. Therefore, close monitoring for cardiac, neurological, and pulmonary dysfunction may aid in minimizing the lethal complications. Corticosteroid therapy, including a tapered course of intravenous corticosteroid administration (methylprednisolone at a dose of 1 mg/kg/day) for 10 to 14 days and/or intratympanic steroid injections as a salvage therapy are recommended. 15 HBO therapy should be performed for the patients with neurological deficits, cardiac ischemia, loss of consciousness, metabolic acidosis, or COHb values > 25%. 16 However, because COHb levels do not always correlate with clinical symptoms, some authors suggested treatment with HBO in the patients having clinical symptoms of CO intoxication, until the COHb concentration reduces to less than 3% or clinical symptoms resolve completely. 17 Other beneficial effects of HBO therapy are possible reduction in occurrence of cerebral edema and prevention of delayed neurological syndrome. 6
If there is no common etiology in patients with sudden hearing loss, ototoxic substances, especially CO poisoning in the working/home environment should be considered as a diagnostic possibility. As evident from the summary of current literature available (Table 1), it is difficult to establish associations between outcome, age, symptoms, COHb level, and treatment. Therefore, more studies are needed to determine the best clinical guidelines for CO poisoning. HBO and corticosteroid therapy are treatment options for sudden hearing loss following acute CO poisoning. Nevertheless, the treatment efficacy was not proven by controlled studies. Our report highlights that early recognition and subsequent management may mitigate the morbidity and help in boosting the recovery process in hearing loss.
Footnotes
Acknowledgements
The authors would like to thank our department colleagues and the devotion of this patient.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Availability of data and materials
All patient information is available from her case note in our hospital record unit and cannot be legally or ethically uploaded here.
Informed consent
Written informed consent for publication was taken from the patient.
Ethical approval
No ethical approval was requested for this case. However, to maintain patient confidentiality, our case report did not include any data or picture that would have enabled identification of the patient.
Human rights
This case report was prepared according to the World Medical Association Declaration of Helsinki.
