Abstract
Background:
To manage and reduce painful procedures in the emergency wards, procedural sedation as well as analgesia can be used. Propofol combinations are the most common short-acting intravenous sedative drugs in the field of emergency medicine.
Objectives:
This research aimed to compare the effectiveness, safety, and complications of intravenous infusion of ketofol with fentofol in painful emergency procedures.
Methods:
This study was a randomized, double-blind clinical trial. Sampling was simple and used random assignment; 196 participants were randomly assigned to two groups of 98 patients each. In each group, injection was done with fentanyl–propofol or ketamine–propofol. For data gathering, pre-procedure, and procedure, modified Aldrete’s scoring checklist was used. In order to determine the levels of sedation, we used the Ramsay Sedation Scale. Data analysis was performed via SPSS Version 20 using statistical tests such as mean ± standard division, t-test, chi-square test, and analysis of variance.
Results:
Among the participants in this study, shoulder procedure was performed more than other procedures (56.6%). The mean of recovery time was significantly different in the two groups (ketofol: 5.65 ± 0.35 vs fentofol: 9.33 ± 0.78); the recovery time in group B (fentofol) was longer than that in group A (ketofol) (p = 0.001). Drug complications were statistically significant in the two groups, and complications by ketofol were less than those by fentofol (p = 0.001). “Hypotension and bradycardia” were observed with fentofol and “tachycardia” was observed with ketofol. The performance of these two drugs was not the same when subject to various procedures.
Conclusion:
Results of this study revealed that ketofol provided better analgesia and sedation, faster recovery time, lesser complications and adverse events, and reduced bradycardia and hypotension.
Introductions
One of the techniques that can be used to cause analgesia is sedation. In this technique, the prescription of tranquilizers does not cause any analgesic effects. 1 Procedural sedation and analgesia causes minimally decreased levels of consciousness that maintain the patient’s ability for airway protection. They are used in emergency wards to manage and reduce painful procedures. 2 Propofol is one of the most common short-acting intravenous sedative drugs in the field of emergency medicine, which is used in children and adults. Also, for intubated or mechanically ventilated patients in intensive care units (ICUs) concerning induction and maintenance of general anesthesia, and in gastrointestinal endoscopic procedure propofol can be used. It is suggested that ketofol combinations induce effective sedation in patients of all age groups when used in procedures such as spinal anesthesia or in gynecological, ophthalmological, and cardiovascular procedures. This drug is highly desirable because of its favorable pharmacology. As it provides no analgesic effect, it should be used with other opioids. Ketamine and fentanyl are two opioids whose combinations with propofol provide analgesic effect. It was in the 1990s that the use of ketamine–propofol (also called ketofol) began. Its application was observed and is in line with decreased emergence reactions following the use of propofol before ketamine administration. 3 This combination has been used in different settings during burn dressing change, interventional radiology procedures, and procedural sedation in emergency departments (EDs).4–6 It is important to mention that vomiting due to ketamine will be minimal; a shorter recovery time and agitation are the potential benefits of ketofol to ketamine in procedural sedation and analgesia.7,8 Fentanyl is beneficial to pains which are intense and short in duration, and it can manage them effectively. 9 Among the opioid drugs, fentanyl can lead to a reduction in the dosage of propofol and decrease its complications, 10 but there is a lack of enough evidence investigating the use of fentanyl and propofol combinations (also called fentofol) during emergency procedures. Although some studies have investigated the propofol combination, it seems that there is a need for comprehensive evidence about various procedures in emergency wards. Therefore, this research was conducted to compare the effectiveness, safety, and complications of intravenous infusion of ketofol with fentofol in painful emergency procedures. On the premise of the available literature, we assume that the infusion of ketofol is more reasonable and has more favorable hemodynamics and provides better recovery in comparison with fentofol.
Methods
This randomized, double-blind clinical trial was conducted from September 2017 to December 2018 in patients referred to the emergency ward of Shahid Bahonar Hospital at Kerman University of Medical Sciences. Bahonar is a 300-bed educational hospital and is a level III trauma center with 51,000 admissions annually, of which 80% of them are adults. The ED is managed by 12 full-time emergency medicine specialists having emergency medicine board certification. In this study, all emergency physicians were trained, and we included patients who required procedure sedation and analgesia. In addition, five nurses and respiratory therapists were trained in the study protocol.
Eligible patients were chosen to enter the study by any trained emergency physician who just happened to be on that particular shift and was responsible for the clinical management of these patients. We initiated the study after receiving approval from the ethics committee of Kerman University of Medical Sciences (Code Number: IR.KMU.REC.1395.220). Also, we obtained an Iranian clinical trial number: IRCT2016050615941N4. The inclusion criteria were patients with the American Society of Anesthesiologists (ASA) physical status 1 and patients who needed a painful procedure in the ED. Patients with clinically significant cardiovascular/hepatic diseases, epileptic disease, respiratory disease, O2 saturation less than 92%, systolic blood pressure less than 100 mmHg, and Glasgow Coma Scale score less than 15 were excluded. Besides, a procedure longer than 1 h and hypersensitivity to the drugs were the other two exclusion criteria. We informed the participants and their guardians (in the case of children) regarding the anesthetic technique, and written consent was also obtained from them. Sampling was simple and 196 participants were randomly assigned to two groups of 98 patients each by performing computerized randomization schedule through random allocation software (http://mahmoodsaghaei.tripod.com/Softwares/randalloc.html) to generate the treatment sequences for two groups of equal sizes. In each group, injection was performed with fentanyl–propofol or ketamine–propofol. A pharmacist was consulted for medication use. Study drugs were prepared in the emergency pharmacy in pre-filled and pre-drawn syringes by the study pharmacist. Syringes were covered with opaque layer and labeled as A (ketofol) or B (fentofol). Concerning the blindness of the procedures, in order to prepare syringes with the same volume, drugs were diluted in group A to provide sufficient volume. Syringes were prepared at the beginning of the day and then were placed at the middle of the pharmacy refrigerator, and if they were not used the same day, they were discarded after 24 h.
The pharmacist was not aware of patients’ recruitment as well as the procedural protocol. After accessing the computerized randomization schedule, the nurse obtained informed consent from patients, allocated a number and letter to patients, and then informed it to the emergency pharmacy. This nurse was not involved in drug preparation, injection or drug treatment, sedation procedure, or subject recruitment. Then patients were transferred to the emergency room. After examination and preparation of patients, procedures were carried out in the emergency room with cardiopulmonary resuscitation equipment, an airway and resuscitation cart. Drugs that were covered with opaque layer were transferred to the emergency room. Procedural sedation and analgesia was performed with an emergency physician who was not aware of the manner of drug allocation to group A or B.
After controlling vital signs (blood pressure, temperature, pulse rate, level of consciousness, and respiratory rate), two intravenous lines were inserted. All patients were under continuous pulse oximetry, cardiac monitoring, and capnography. No preoxygenation was provided. The nurse and the emergency physician conducted procedural sedation and analgesia. The emergency physician had the responsibility to conduct and administer the drugs based on the study protocol.
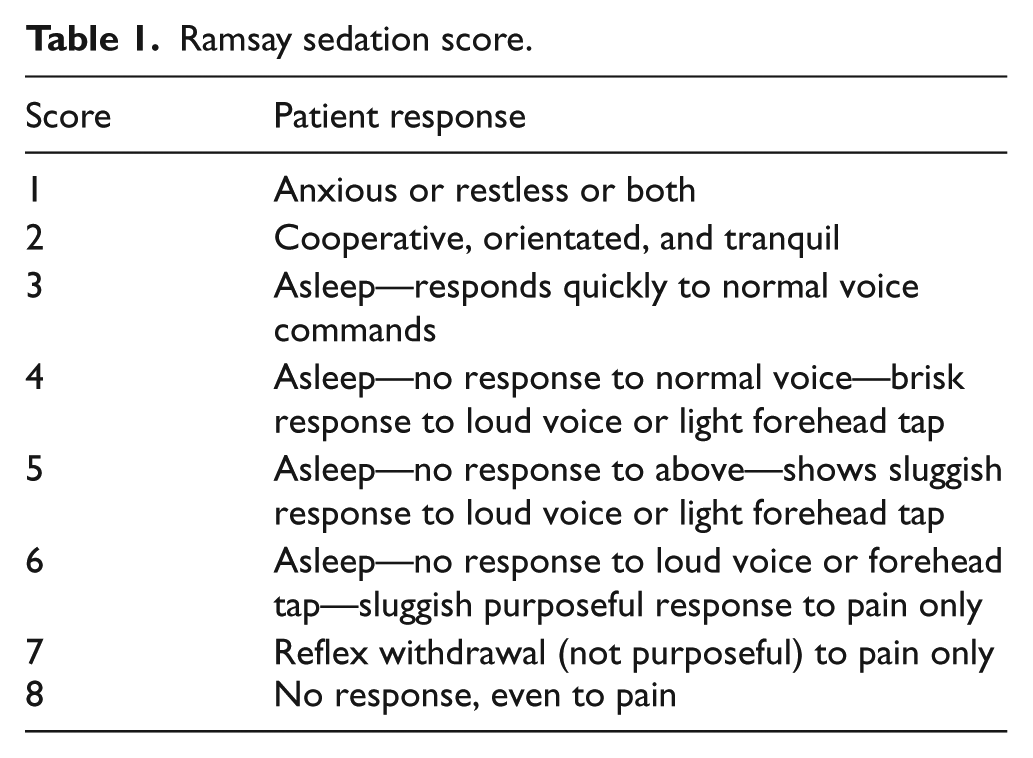
Patients received an initial dose of the study drugs including ketofol (0.5 mg/kg each of ketamine and 2 min later 1 mg/kg of propofol that was defined as group A) or fentofol (1 mg/kg of fentanyl and 2 min later 1 mg/kg of propofol that was defined as group B) administered during 15–30 s. The emergency physician used the Ramsay Sedation Scale 11 (Table 1) to assess the level of sedation for each patient, 1 min after the initial dose and every minute thereafter. Upon the decision of the emergency physician concerning the Ramsay sedation score (RSS) <5, more medication based on the study protocol was administered at the same dose (as indicated above) during 15–30 s. If the patient achieved deep sedation (RSS ⩾5), the procedure was stopped.
Ramsay sedation score.
Data were collected on separate data sheets and clinical record on other sheets. Before the sedation, demographic data including age, gender, mechanism of injury, ASA class, drug and past medical history, study sequence number, and pain severity on a scale of 1–10 were recorded carefully. While performing the procedure, the study drug (number of milligrams) and RSS for each patient (every 1 min) as well as the time of sedation were recorded by the emergency physician. The start and end times of the procedure were recorded by the emergency physician. Blood pressure measurements were taken and recorded every 4 min with an automated noninvasive cuff. We electronically recorded pulse oximetry, continuous capnography, pulse rate, and respiratory rate.
During the procedure and recovery period, the emergency physician recorded any adverse respiratory event, including apnea, cyanosis, and bradypnea according to explicit criteria. Using the Modified Aldrete Scale 11 (Table 2) patient recovery was assessed every 2 min up to 16 min by a registered nurse. The recovery times were recorded on a sheet by the emergency physician. After recovery, we asked patients about their sleep and its quality as well as any remembrance of the procedure conducted based on a 10-point Likert-type scale ranging from 1 = “not at all effective” to 10 = “extremely effective.”
Modified Aldrete Scale. a
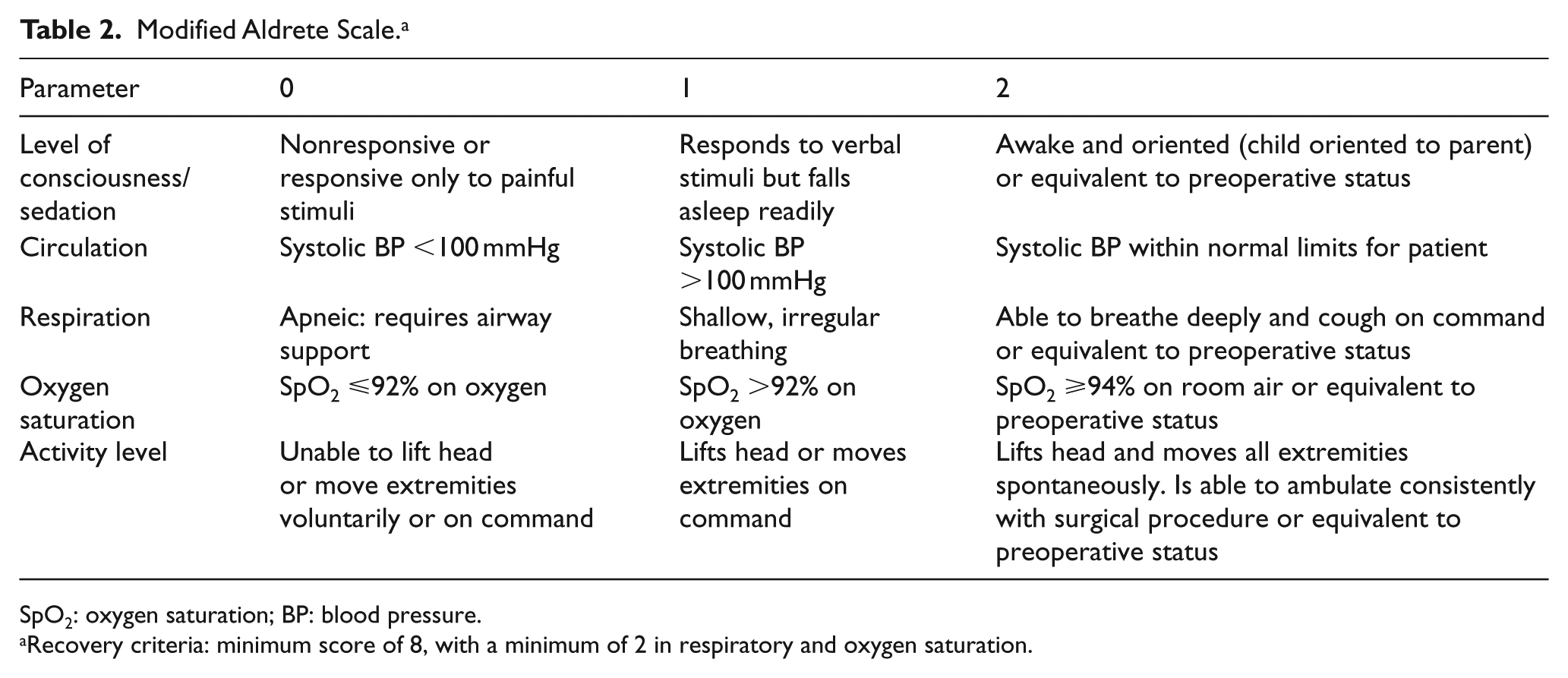
SpO2: oxygen saturation; BP: blood pressure.
Recovery criteria: minimum score of 8, with a minimum of 2 in respiratory and oxygen saturation.
Any events of body rigidity or agitation were recorded by the emergency physician during the procedure.
We also recorded recovery agitation (confusion, unpleasant dreams, restlessness, and dysphoria), nausea, vomiting, and headache during the recovery phase.
Patients’ satisfaction regarding the procedure adopted was assessed using a 10-point Likert-type scale (ranging from 1 = “not satisfied” to 10 = “extremely satisfied”). After completion of the procedures, to analyze data, data sheets were gathered and entered in Statistical Package for the Social Sciences (SPSS) (IBM Corp. Released 2011. IBM SPSS Statistics for windows, Version 20.0. Armonk, NY: IBM Corp.) by an assistant who was not involved in the study and its processes.
The early or primary outcome was evaluated as the number of respiratory adverse event defined by oxygen saturation less than 90%, central apnea, clinically apparent pulmonary aspiration, partial and complete upper airway obstruction, and laryngospasm or bronchospasm. Secondary outcomes were sedation time, sedation stability, sedation efficacy, total medication dosage, induction time, recovery time, procedure time, and the incidence of adverse events. Sedation stability was defined based on the number and proportion of patients having a score of 5 or greater (RSS) during the procedure or not needing additional medicine for the completion of the procedure.
Based on the following criteria, sedation was considered effective: (1) the patient did not have an annoying recall of the performed procedure, (2) experienced a sedation-related adverse event causing the stoppage of the procedure, (3) any adverse events which resulted in unplanned prolonged ED observation (3 h or more, as defined by the regional ED sedation guideline), and (4) unplanned admission finalized by the treating physician. The interval from the first dose of the administered medication until a score of 5 was obtained (based on the RSS) or the procedure initiated was considered as the induction time. The duration from the first administration of medicine to procedure completion was defined as the sedation time. We asked the treating physician to determine the start and end times of the procedure. By the same token, a respiratory therapist recorded the times. The intervening time between the last time of medication use and when the criteria of discharge were met was considered as the recovery time. This time was calculated by Aldrete’s score.
Complications and adverse events were bradycardia (pulse rate less than 50 beats/min with hypotension or chest compressions), vomiting, hypotension (decreased systolic blood pressure requiring administration of fluid bolus or vasopressors), rash, procedural agitation (patient aggression and reactivity to painful manipulation), and recovery agitation (restlessness, confusion, dysphoria, and unpleasant dreams). Treatment for recovery agitation was recorded and defined as either physical restraint or the use of sedative medication such as opium or benzodiazepines. Muscular rigidity was another recorded adverse event. The patients with Aldrete’s score ⩾12 were candidates for discharge. This study investigated the complications of these two drugs and compared the degree of their effectiveness and safety.
Statistical analyses were performed by a person unaware of the study groups. Data analysis was done via SPSS software Version 20 using descriptive statistical tests such as mean ± standard division, frequency, and percentage. Independent t-test, chi-square test, one-way analysis of variance (ANOVA), and repeated-measures ANOVA were used to compare the recovery rate and the time required for recovery.
Results
The flowchart of study cases is shown in Figure 1 which presents patients with sedation during the study.
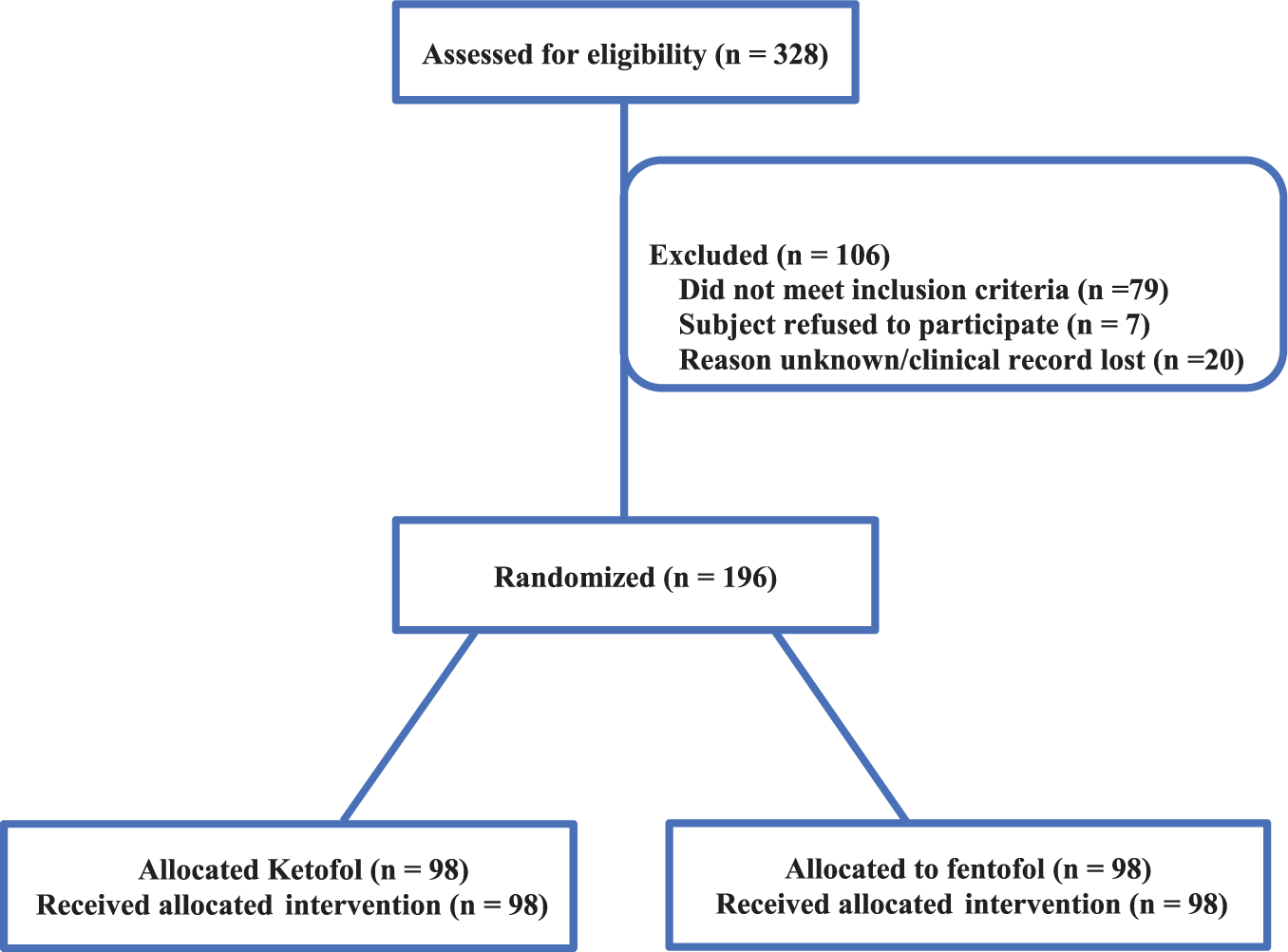
Flowchart of study cases.
This study was performed on 196 participants, 141 males and 55 females. The mean of participant’s age was 31.42 ± 8.54 years, with a range of 3–52 years. Demographic characteristics such as age, sex, repeated drug dose, and painful procedures are shown in Table 3. The most common procedure was orthopedics. The emergency physician who performed the procedures approved them as successful implementation. There was no hypoxic event before the initiation of a procedure or after recovery from sedation. There was no unplanned prolonged (2 h or more) ED stay or unplanned admission to the hospital. Blinding assessment results were similar between the two groups, and no evidence suggested unblinding.
Characteristics of patients receiving intravenous ketofol or fentofol.
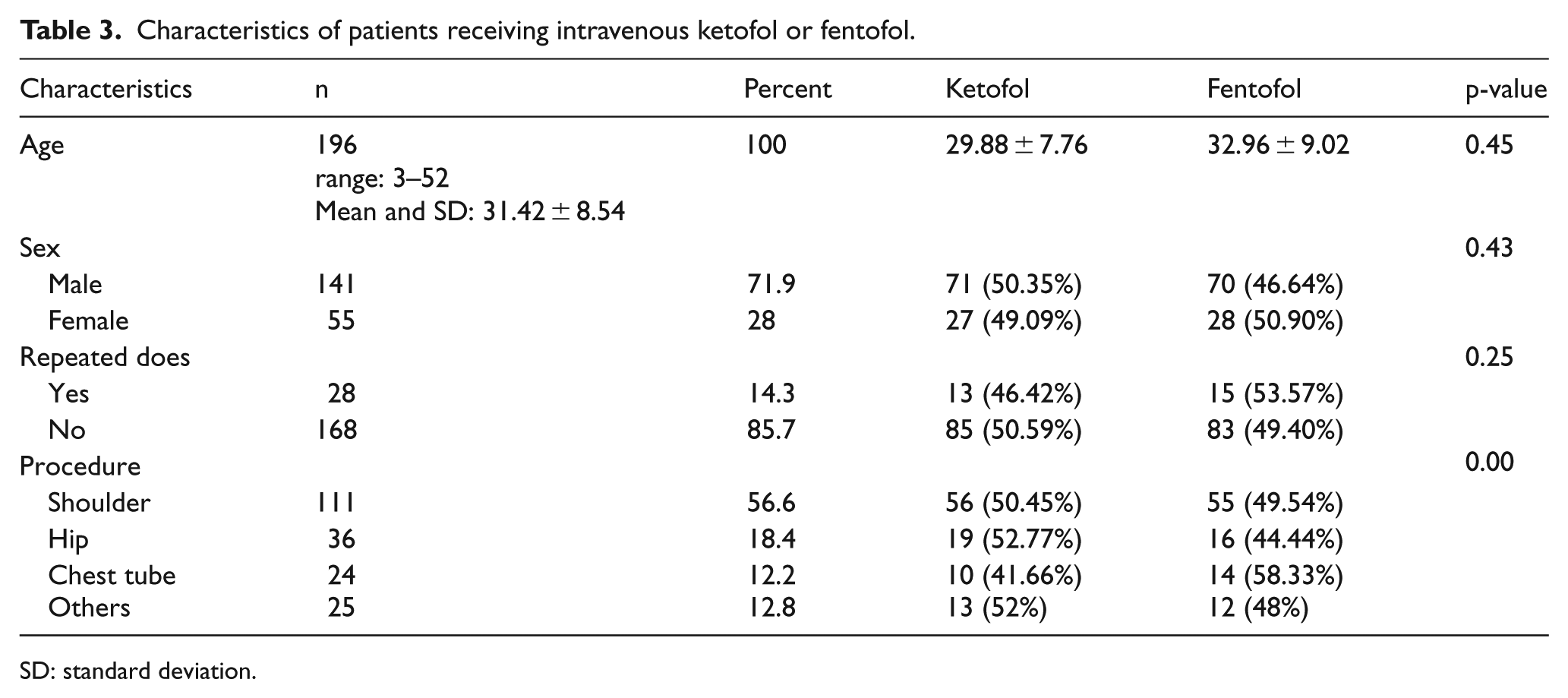
SD: standard deviation.
There were no respiratory events and intervention in both groups. Most of the participants (85.7%) did not need to repeat the drug dose. Among the patients who participated in this study, shoulder procedure was performed more than other procedures (56.6%), and about 42.9% of participants (most of them) did not show any drug complications. Table 4 compares the two groups with regard to recovery time, sedation stability, induction time, and drug complications. The mean of recovery time was different in the two groups (ketofol: 5.65 ± 0.35 min vs fentofol: 9.33 ± 0.78 min); the recovery time in group B (fentofol) was longer than that in group A (ketofol). This difference was statistically significant (p = 0.001). Investigating the duration of recovery time with respect to gender did not show a statistically significant relationship between males and females (p = 0.4). In addition, the mean of recovery time in group A with ketofol sedation (p = 0.63) and in group B with fentofol sedation (p = 0.20) was not significant with regard to the effect of sex. Our data presented in Table 4 show that drug complications were statistically significant in the two groups (p = 0.001). Drug complications with ketofol were less than those with fentofol: 51.2% of participant with ketofol sedation had no complication versus 48.8% with fentofol. Complications such as “hypotension and bradycardia” were observed with fentofol and “tachycardia” was observed with ketofol. Minor complications such as vertigo, nausea, headache, and weakness are shown in Tables 3 and 4.
Recovery time and drug complications with ketofol or fentofol.
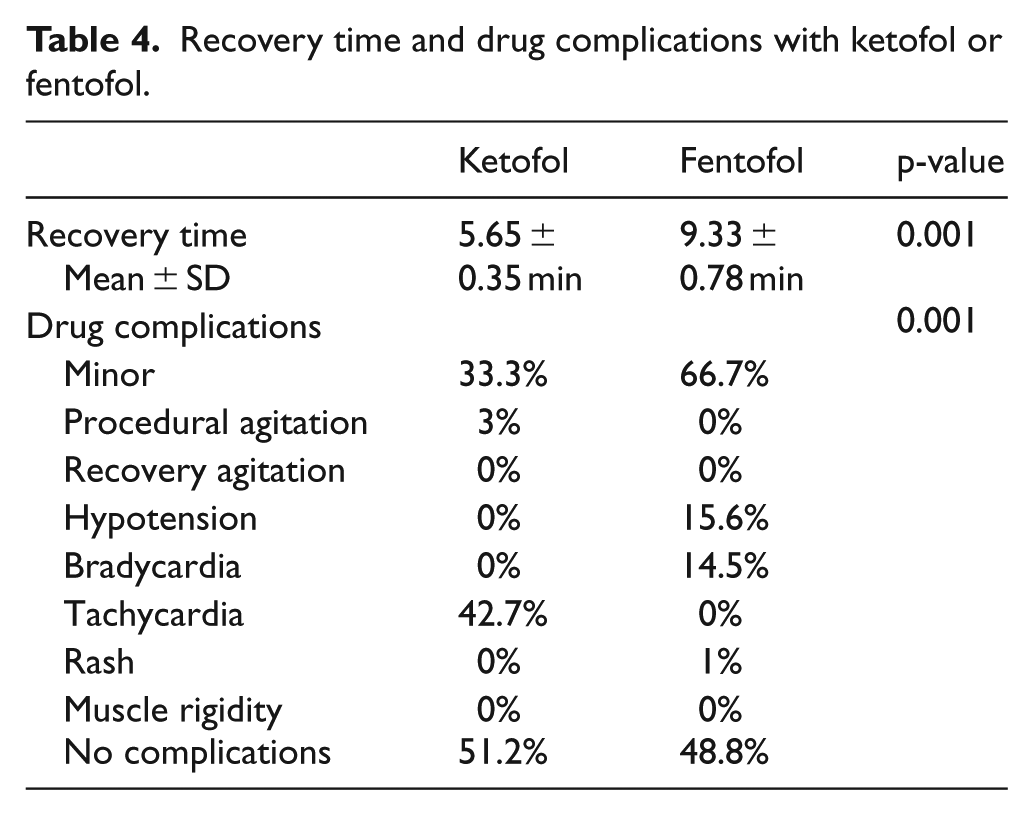
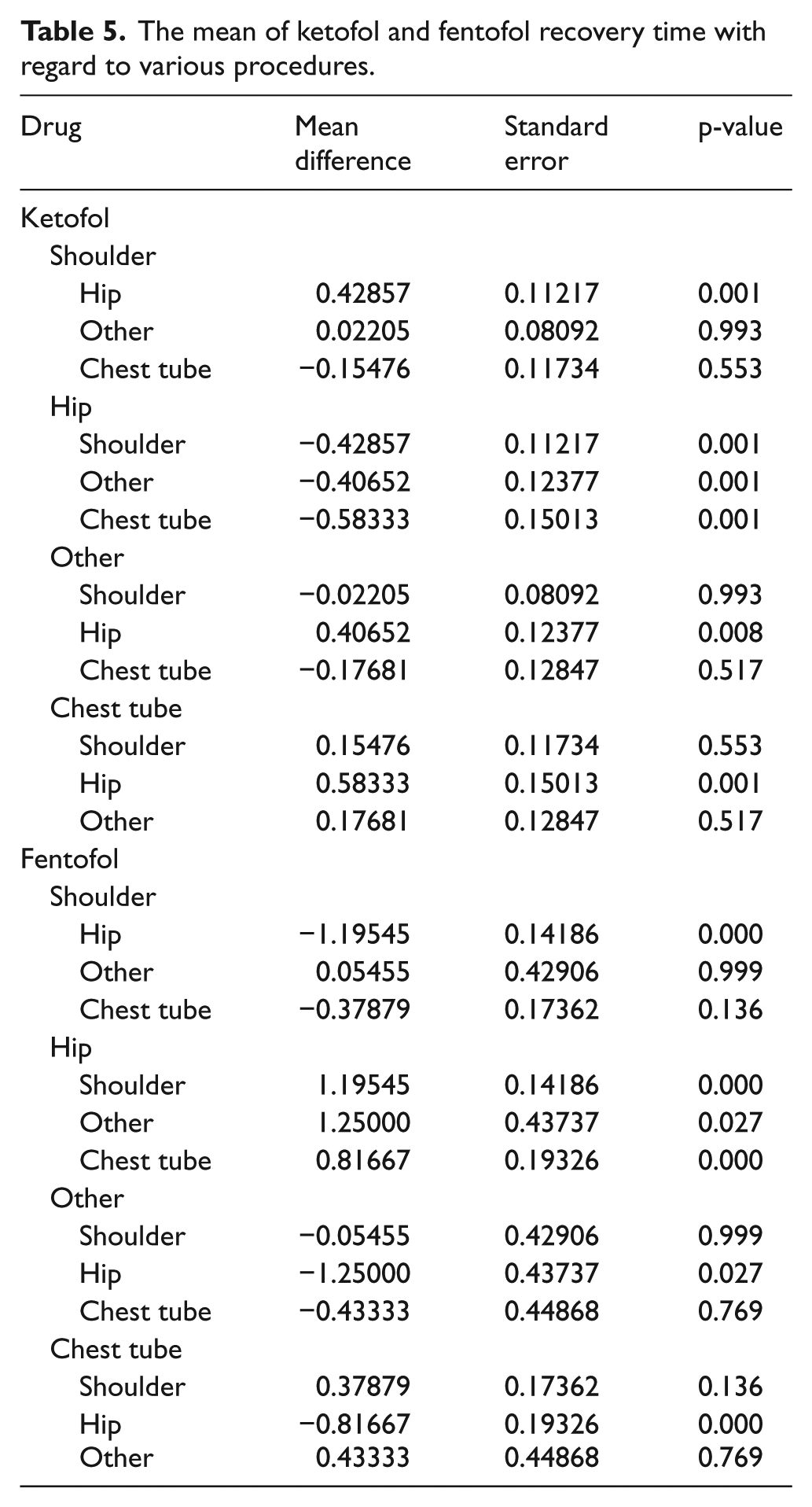
Based on ANOVA test, evaluating the mean of recovery time with regard to drug separation in patients with various painful procedures showed that the performance of these two drugs is not the same when subject to various procedures. For instance, patients with shoulder procedure who were under sedation by fentofol recovered faster than those with hip procedures. Similarly, patients with chest tube procedure recovered faster than those with hip procedure (Table 5). Conversely, the recovery time for patients under sedation by ketofol during hip procedure was more significant than other procedures and they recovered faster (p = 0.001).
The mean of ketofol and fentofol recovery time with regard to various procedures.
Discussion
In this study, we investigated the effectiveness and safety of ketofol and fentofol. By the same token, we compared them in painful emergency procedures. Results illustrated that both ketofol and fentofol provide effective and safe sedation, but ketofol had better effectiveness with shorter recovery time than fentofol (Table 4).
Evidence shows that ketofol sedation has more benefits of cardiovascular stability and airway preservation in procedural sedation with a counterbalance of the effects of each drug. 12 Based on the study conducted by Tosun et al., 4 we observed that mean heart rates and blood pressure were at baseline during sedation and recovery time in most patients was short. We identified ketofol as an effective sedative in emergency procedures, and only tachycardia (42.7%) complication was observed in patients with ketofol sedation. Ketamine is an intravenous induction agent producing a unique dissociative anesthetic state characterized by dissociation between the thalamocortical and limbic systems and causes a significant increase in heart rate and arterial blood pressure via central and peripheral mechanisms; hence, it is contraindicated in patients with raised intracranial pressure, hypertension, cardiac disease, or coronary artery disease. Ketamine-induced cardiac stimulations could be advantageous in the management of severely ill or septic shock patients both as an anesthetic and as a sedative agent due to its ability to elevate hemodynamic parameters and the suggested tendency to exert a protective anti-inflammatory effect against the sepsis process itself.
Conversely, hypotension and bradycardia were observed in patients with fentofol sedation. Hypotension and bradycardia occurred in 15.6% and 14.5% of patients in the fentofol group, respectively, compared with 0% in the ketofol group. It seems that ketofol acts by deactivating its side effects and preserves the sedative effect. In the study by Khutia et al., 13 hypotension occurred in 38.63% of patients in the propofol–fentanyl group compared with 14.58% in the propofol–ketamine group. Sharma et al. 14 reported that respiratory depression, hypotension, and bradycardia and delayed recovery were associated with the fentofol group too. In a research done by Manickam and Mohanasundaram 15 findings showed that analgesia and sedation were better provided by ketofol. Besides, in comparison with fentofol among patients admitted in the ED, ketofol had a major role in reducing post-induction hypotension as well as causing less adverse respiratory events. Tosun et al. 4 compared propofol–ketamine with propofol-fentanyl combinations for deep sedation and analgesia. They concluded that the comfort of anesthesia was better with a propofol–ketamine combination. In line with our findings, the study undertaken by Messenger et al. 16 showed that under the breathing room air, ketamine was safer than fentanyl when used as an adjunct analgesic to ED procedure with titrated propofol. When compared to fentanyl–propofol, ketamine–propofol caused substantially fewer intra-sedation events at all levels of severity while having similar efficacy and recovery profile. 16 Guit et al. 17 reported that the combination of fentanyl with propofol depressed hemodynamics (e.g. hypotension, tachycardia, and systematic vascular resistance), but the combination of ketamine with propofol resulted in stable hemodynamics. This finding is consistent with our results.
Seol et al.
18
reported that propofol–ketamine and propofol–remifentanil combinations were effective for sedation and to achieve analgesia in pediatric patients. In other study, Godambe et al.
19
observed desaturation in 31 cases of pediatric patients receiving propofol–fentanyl combination. In the emergency ward due to the rapid flow of patients and limitation of beds, recovery time is very important. Reduction in the duration of recovery time is critically important in procedural sedation and analgesia medications. In this study, the recovery time in the ketofol group was shorter than that in the fentofol group. This finding is consistent with the studies of Erden et al.
5
and Akin et al.
20
Evidence shows that propofol and low doses of ketamine are more effective and safe sedation regime than propofol–fentanyl in respect to hemodynamic stability and apnea.
13
We believe that due to the synergistic effect of fentanyl and propofol (both sedative), recovery time in the fentofol group is larger than that in the ketofol group, and sedation is deeper than ketofol. The study by Akhondzadeh et al.
21
concerning the comparison of propofol–fentanyl with propofol–ketamine in patients undergoing the endoscopic retrograde cholangiopancreatography (ERCP) procedure showed that both groups were similar in terms of the hemodynamic and sedation criteria due to the lower amount of pain and apnea in the ketofol group. In this study, time to RSS 5 and sedation time in both groups were the same—3 (2–4) min and 7 (4–9) min, respectively. Conversely, in the study by Kurdi and Deval,
ketofol group achieved desired sedation levels 5–6 on RSS faster as compared to that in the fentofol group. Sedation scores were higher and better maintained in patients in group B (ketofol 1:2) who received higher amount of propofol intra-operatively compared to the patients in the other groups.
22
In their study, the mean total sedation time was comparable in both ketofol groups, and it was prolonged in the fentanyl–propofol group similar to studies by Abdellatif 23 and Timm et al. 24 In our study, the mean total sedation time with ketofol and fentofol was the same—7 (4–9) min—but the recovery time with ketofol was faster than that with fentofol. In line with our study, Kb et al. 25 indicated that “propofol-ketamine was superior to propofol-fentanyl in terms of hemodynamic stability, absence of respiratory depression, post-operative analgesia and recovery.” Chandar et al. 26 realized that pain caused by propofol injection in the fentanyl group was more than in the ketamine group, and the depth of anesthesia in the propofol–ketamine group was more than in the propofol–fentanyl group. Also, Takzare et al. 27 indicated that both combinations had good sedation in patients who underwent upper gastrointestinal endoscopy (UGIE). Conversely, more side effects were observed with the combination of propofol–ketamine. This combination even resulted in stable hemodynamics and deeper sedation.
Findings revealed that the performance of ketofol and fentofol in terms of duration of recovery time with various procedures (e.g. hip, shoulder, and chest tube) is not the same. Therefore, we propose more studies to be designed to investigate the performance of these two drugs and their effects on painful procedures in EDs.
Limitations
Even if every attempt was made to conceal drug allocation, physician blinding may not have been complete. Sedation bias may have occurred because of stopping limitation. Small sample size is another limitation, and we suggest other studies with more participants.
Conclusion
Research findings show that “ketamine provides analgesia during propofol sedation with fewer adverse airway events compared with fentanyl or alfentanil.”5,16,28 Evidence highlights that ketamine is considered as an analgesic drug during procedural sedation. 29 Our findings reveal that ketofol has the advantages of better analgesia and sedation, faster recovery time, lesser complications and adverse events, and reduced bradycardia and hypotension. Therefore, we conclude that both ketofol and fentofol are logical choices for sedation and analgesia during painful emergency procedures, but ketofol is superior to fentofol due to better analgesia and sedation, lesser complications, and faster recovery time. Owing to the more complications caused by fentofol, ketofol seems to be a more suitable and reasonable choice in this regard.
Footnotes
Author contributions
S. T. and M. M. contributed to conception and design, acquisition of data or analysis, and interpretation of data; drafting the article or revising it critically; and final approval of the version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
The datasets generated and/or analyzed during the current study are available from corresponding author on request.
Informed consent
Participants and their guardians (in case of any children) were informed about the procedure (the anesthetic technique) and written informed consent was obtained from them. We made efforts not to publish the names of the patients, their initials, and identity. Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Ethical approval
This study was approved by the ethics committee of Kerman University of Medical Sciences with Code Number: IR.KMU.REC.1395.220. Also, we obtained an Iranian clinical trial number: IRCT2016050615941N4.
