Abstract
Background and objectives:
No previous studies have investigated the relationship between the optic nerve sheath diameter and raised intracranial pressure in pediatric patients or have evaluated the usefulness of optic nerve sheath diameter in ocular ultrasound and brain computed tomography/magnetic resonance imaging. This study aimed to meta-analyze the diagnostic performance of optic nerve sheath diameter for the diagnosis of raised intracranial pressure in pediatric patients.
Methods:
A database search of PubMed and EMBASE was performed to identify relevant studies. Bivariate modeling and hierarchical summary receiver operating characteristics modeling were performed to evaluate diagnostic performance. A pooled diagnostic odds ratio with a 95% confidence interval, not including 1, was considered informative. Subgroup analysis was performed according to the modality (ocular ultrasound vs brain computed tomography/magnetic resonance imaging). We performed meta-regression analyses for heterogeneity exploration.
Results:
Eleven studies involving 546 patients were included. According to pooled diagnostic odds ratios, optic nerve sheath diameter was informative for the evaluation of raised intracranial pressure (diagnostic odds ratio, 47; 95% confidence interval, 11–206). Optic nerve sheath diameter showed a pooled sensitivity of 0.88 (95% confidence interval, 0.79–0.94), a pooled specificity of 0.86 (95% confidence interval, 0.70–0.95), and an area under the hierarchical summary receiver operating characteristics curve of 0.93 (95% confidence interval, 0.91–0.95) for the diagnosis of raised intracranial pressure. According to the subgroup analysis, ocular ultrasound (sensitivity, 0.91 (95% confidence interval, 0.81–0.96); specificity, 0.86 (95% confidence interval, 0.65–0.96)) showed higher sensitivity and comparable specificity than optic nerve sheath diameter measured on brain computed tomography/magnetic resonance imaging (sensitivity, 0.75 (95% confidence interval, 0.51–0.99); specificity, 0.91 (95% confidence interval, 0.74–1.00)). On meta-regression analysis, the study design, number of patients, and reference standard were the sources of heterogeneity.
Conclusion:
Optic nerve sheath diameter may be a useful method for predicting raised intracranial pressure in pediatric patients. We recommend that the measurement of optic nerve sheath diameter should be performed using ocular ultrasound for a more accurate diagnosis of raised intracranial pressure in pediatric patients.
Introduction
In adults, raised intracranial pressure (ICP) in the presence of acute neurological disease is an important cause of secondary brain injury and may lead to fatal complications.1,2 In pediatrics, although anatomical differences such as cranial rigidity and subdural space are present, this condition is just as detrimental because the resulting impact on intracranial compliance is unpredictable. 3 Ultimately, as in adults, raised ICP causes secondary ischemic injury in children via decreased cerebral blood flow which results in limited delivery of cerebral oxygen.4,5
The gold standard for raised ICP diagnosis is direct ICP monitoring via an intracranial catheter. However, this procedure is invasive and carries a risk of serious complications, such as infection.6,7 Alternatively, the presence of papilledema on fundoscopy has been used as an indirect tool for detecting raised ICP; however, it has low sensitivity, especially in children under 8 years. 8 Furthermore, there are many reports on brain computed tomography (CT), which has been traditionally used for indirect measurement of raised ICP, and the measurement of midline shifts > 5 mm and basal cistern and sulcal effacement is considered unreliable for the prediction of raised ICP.9,10
Recently, optic nerve sheath diameter (ONSD) on ocular ultrasonography (US) and brain CT/magnetic resonance imaging (MRI) has been introduced as an indirect tool for raised ICP measurement, and its reliability and usefulness have been demonstrated.3,11,12 Because the optic nerve connects to the dura mater that surrounds the brain and the cerebrospinal fluid (CSF) fills the cavity between the optic nerve and optic nerve sheath, raised ICP and increased CSF between the optic nerve and optic nerve sheath result in an enlarged ONSD. 13
Few meta-analyses14–17 have demonstrated the relationship between ONSD on US and raised ICP. Furthermore, these studies focus only on adults14,15,17 or mixed populations, 16 and only evaluate the usefulness of ONSD on ocular US. They did not evaluate the relationship between ONSD and raised ICP in pediatric patients or evaluate the usefulness of ONSD on ocular US and brain CT/MRI. Therefore, we believe that the diagnostic performance of ONSD for predicting raised ICP in pediatric patients needs further exploration, and high-level evidence needs to be presented via quantitative synthesis of data from the existing studies. In addition, the pooling of results will be interesting because published studies have used different modalities (ocular US vs brain CT/MRI) and different cutoff values.
This meta-analysis aimed to evaluate the diagnostic performance of ONSD for the prediction of raised ICP in pediatric patients. In addition, we performed a subgroup analysis to evaluate the performance of different modalities for the diagnosis of ONSD.
Methods
This meta-analysis followed the revised guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses of Diagnostic Accuracy Studies (PRISMA-DTA) statement. 18 Institutional review board approval and informed consent were waived because the nature of this study was a systemic review and meta-analysis.
Data sources
The PubMed and EMBASE databases were searched up to 1 March 2019 to identify English-language studies on the use of ONSD for predicting raised ICP in pediatric patients. The search terms “optic nerve sheath diameter,” “intracranial pressure,” or “pediatric” were combined with “diagnosis,” “sensitivity,” “specificity,” or “receiver operating characteristic” as follows: ([“optic nerve sheath diameter”] or [“ONSD”]) AND ([“intracranial pressure”] OR [“ICP”]) AND ([“child”] or [“children”] or [“pediatric”] or [“pediatric”] or [“adolescent”]) AND ([“diagnosis”] OR [“sensitivity”] OR [“specificity”] OR [“receiver operating characteristic”] OR [“ROC curve”]). The bibliographies of the identified articles were screened to identify additional relevant studies. Two investigators screened the titles and abstracts for potential eligibility, and any disagreements were resolved through discussion.
Study selection
We included the following: (1) studies on pediatric patients (under 18 years), (2) studies on the use of mean ONSD (average ONSD of left and right eyes) on ocular US or brain CT/MRI as the index test, (3) studies on the use of invasive or noninvasive ICP monitoring as the reference standard, (4) studies with sufficient information to reconstruct 2 × 2 contingency tables regarding sensitivity and specificity, and (5) studies that were original research articles.
We excluded the following: (1) studies that were case reports or case series; (2) studies that were review articles, guidelines, consensus statements, letters, editorials, clinical trials, and conference abstracts; (3) studies not pertaining to the field of interest; (4) studies involving repetitive ONSD measurements; and (5) studies with insufficient information to reconstruct 2 × 2 contingency tables.
Quality assessment and data extraction
Two investigators independently extracted data on patient and study characteristics. The same investigators evaluated methodological quality using the Quality Assessment of Diagnostic Accuracy Studies-2 (QUADAS-2) tool. 19 Any disagreement between the reviewers was resolved through discussion.
A standardized form was used to extract data on: (1) patient characteristics, including patient numbers, percentage of raised ICP, clinical features, mean age, age range, and sex; (2) study characteristics, including study origin, publication year, study design, study period, reference standard, and blinding to reference standard; and (3) interpretative characteristics, including modality, interpreter, ONSD measurement method, and ONSD cutoff value.
We extracted the study outcomes to construct 2 × 2 tables (i.e. true-positive, true-negative, false-positive, and false-negative results). We calculated the 2 × 2 tables using the Bayesian method if only sensitivity and specificity were presented in an eligible study. For the overall diagnostic performance of ONSD, the result with the highest accuracy was extracted. If two or more reviewers independently assessed the diagnostic accuracy, the result with the highest accuracy was extracted.
Data synthesis and analysis of the diagnostic performance
Patient demographic characteristics and extracted covariates were summarized using standard descriptive statistics. Continuous variables were expressed as means and 95% confidence intervals (CIs). Categorical variables were expressed as frequencies or percentages, unless otherwise stated.
We used a bivariate random-effects model for analyzing and pooling the diagnostic performance (sensitivity and specificity) across studies. To derive summary estimates of diagnostic performance, we plotted estimates of the observed sensitivities and specificities for each test in forest plots and hierarchical summary receiver operating characteristic (HSROC) curves derived from individual study results.20–22 These results were plotted using HSROC curves with 95% confidence and prediction regions. In addition, pooled sensitivities, specificities, diagnostic odds ratios (DORs), areas under the curve, and positive and negative likelihood ratios were calculated. Features showing a pooled DOR with a 95% CI not including 1 were considered to be informative.
Heterogeneity was determined using Cochran’s Q test (p < 0.05 indicated the presence of heterogeneity) and the I2 test (0%–40%, possibly no heterogeneity; 30%–60%, moderate heterogeneity; 50%–90%, substantial heterogeneity; and 75%–100%, considerable heterogeneity). 23 When heterogeneity was noted, heterogeneity according to a “threshold effect” was analyzed by visual assessment of the coupled forest plots of sensitivity and specificity. A meta-analysis of diagnostic test accuracy studies simultaneously evaluates a pair of outcomes (i.e. sensitivity and specificity).20–22 Sensitivity and specificity are commonly inversely correlated and influenced by the threshold (cutoff) value. In addition, Spearman’s correlation coefficient between the sensitivity and false-positive rate was calculated to determine any threshold effect, and a coefficient of >0.6 indicated a considerable threshold effect. 24 We omitted Deeks’ funnel plot 25 of individual studies to test for publication bias according to the PRISMA-DTA.
Subgroup analysis
For detailed evaluation of the diagnostic performance of ONSD for predicting raised ICP according to modality, we performed an additional subgroup analysis according to ocular US and brain CT/MRI. In addition, we compared the diagnostic performance of ONSD for the prediction of raised ICP using ocular US and brain CT/MRI.
Meta-regression analyses
Meta-regression analyses were performed using several covariates to investigate the potential causes of heterogeneity: (1) study design (prospective vs retrospective), (2) total patients (⩾30 vs <30), (3) proportion of raised ICP (⩾50% vs <50%), (4) study population (only children vs children and adolescents), (5) reference standard (only invasive monitoring vs invasive monitoring with the inclusion of noninvasive methods), (6) blinding (blinding vs not reported), (7) interpreter (only radiologists vs include non-radiologists), and (8) ONSD cutoff value (⩾5 mm vs <5 mm).
All statistical analyses were performed by one author with 5 years’ experience in performing systematic reviews and meta-analyses. Statistical analyses were performed using the “midas” and “metandi” modules in Stata software (version 10.0; StataCorp LP, College Station, TX, USA) and the “mada” package in R software (version 3.4.1; R Foundation for Statistical Computing, Vienna, Austria). A p-value of <0.05 was considered statistically significant.
Results
Literature search
Figure 1 shows a flow diagram summarizing the literature search. During the initial search, 168 studies were identified. After removing 36 duplicates, we reviewed 132 titles and abstracts and excluded 117 studies. These studies were case reports, letters, editorials, and conference abstracts (n = 48), review articles, guidelines, and consensus statements (n = 26), or not in the field of interest (n = 43). After reviewing the full text of 15 eligible articles, we excluded four studies as they involved repetitive ONSD measurements (n = 1) 26 or lacked sufficient information to reconstruct 2 × 2 contingency tables (n = 3).27–29 Ultimately, 11 original research articles,3,11–13,30–36 including a total of 546 patients, were included in the meta-analysis.
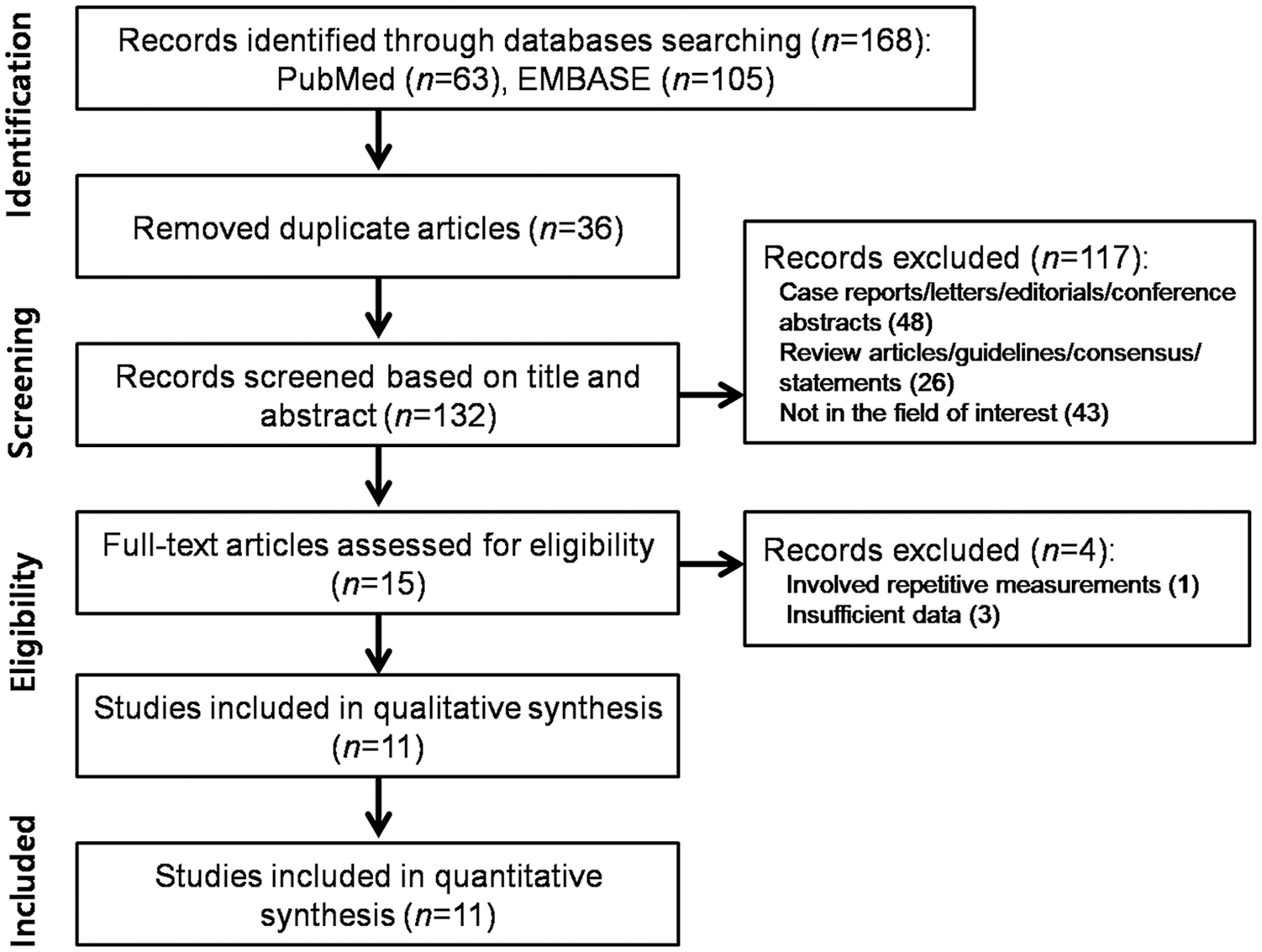
Flow diagram showing the study selection process for the meta-analysis.
Study characteristics
Patient characteristics are summarized in Table 1. Patient numbers ranged from 11 to 174 (mean age, 3–14.1 years). Study and interpretive characteristics are summarized in Tables 2 and 3, respectively. All studies were single-center studies with consecutive patient recruitment. Nine studies12,13,30–36 from 11 articles used ocular US, one 3 study used brain CT, and one 11 used brain MRI. All studies measured ONSD 3 mm behind the globe/optic disk/papilla. All studies used the average ONSD (ONSD of right eye + ONSD of left eye/2) for evaluation of diagnostic performance of ONSD. Six studies3,12,31,33,34,36 from 11 articles used only invasive ICP measurements via an ICP monitoring catheter as the reference standard, four studies11,13,30,32 used invasive or noninvasive ICP measurements (such as CT or fundoscopy) as the reference standard, and one study 35 used only noninvasive ICP measurements (CT) as the reference standard.
The included patients’ demographic characteristics.
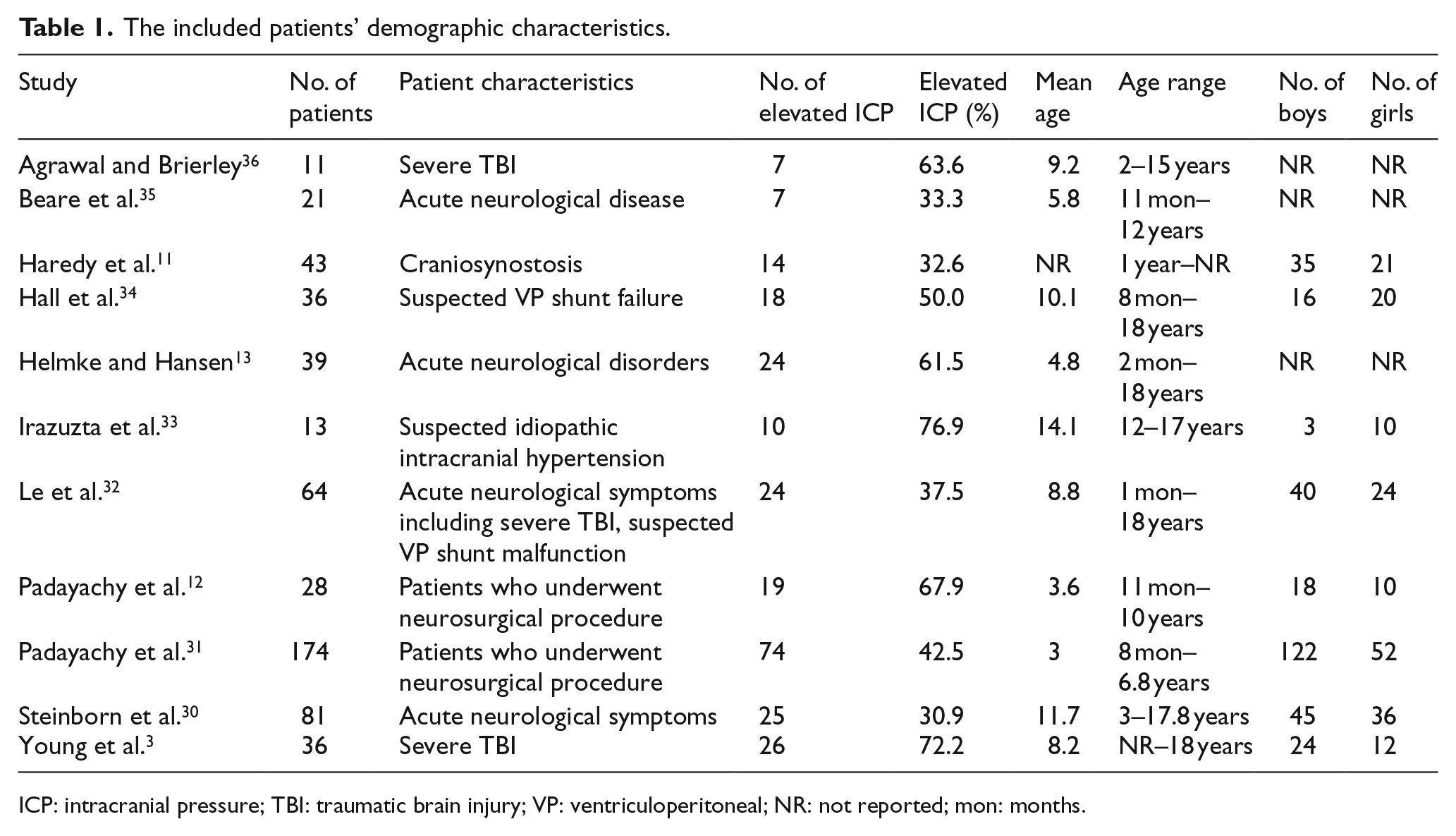
ICP: intracranial pressure; TBI: traumatic brain injury; VP: ventriculoperitoneal; NR: not reported; mon: months.
Characteristics of the included studies.
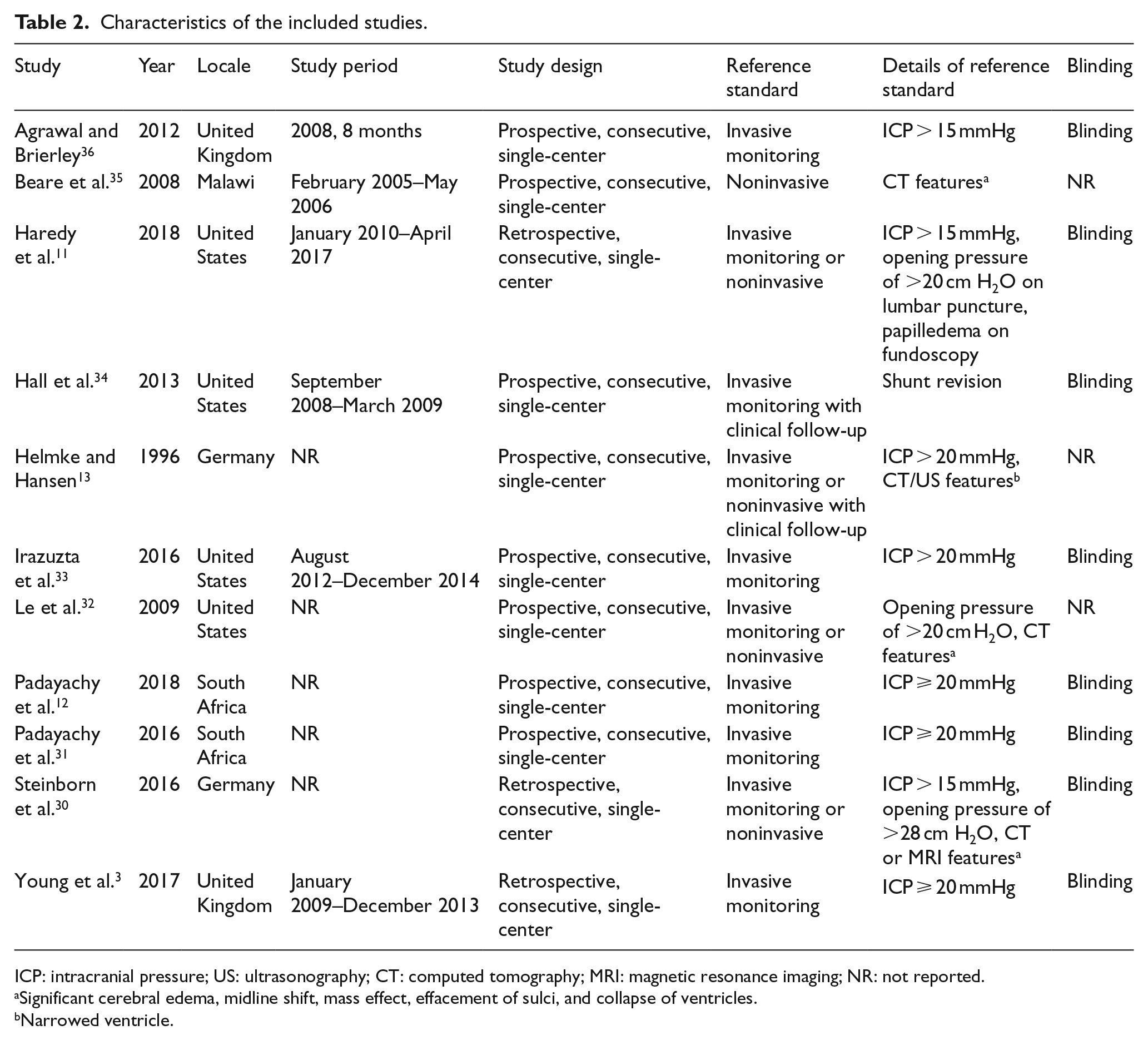
ICP: intracranial pressure; US: ultrasonography; CT: computed tomography; MRI: magnetic resonance imaging; NR: not reported.
Significant cerebral edema, midline shift, mass effect, effacement of sulci, and collapse of ventricles.
Narrowed ventricle.
Interpretative characteristics of the included studies.
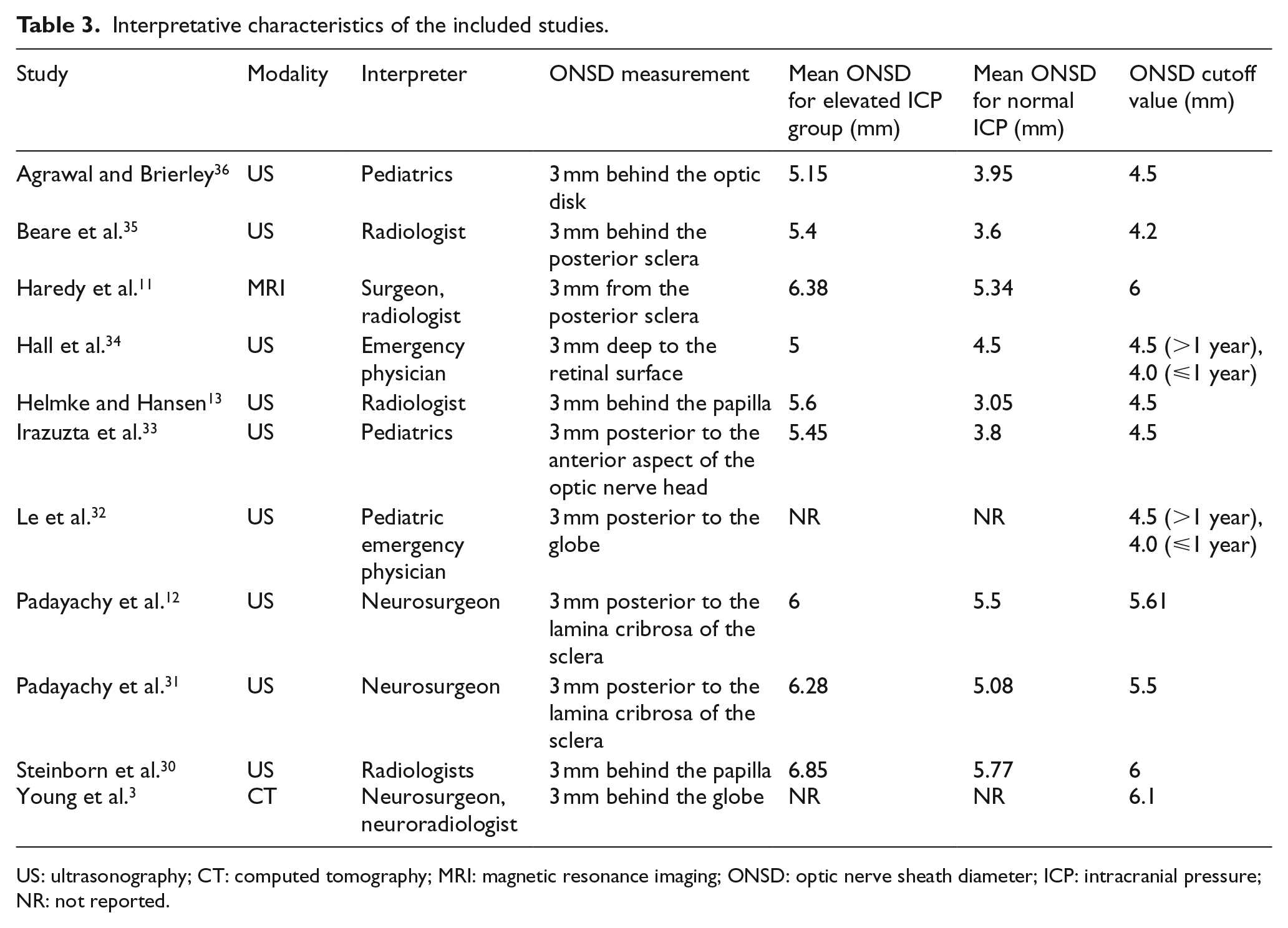
US: ultrasonography; CT: computed tomography; MRI: magnetic resonance imaging; ONSD: optic nerve sheath diameter; ICP: intracranial pressure; NR: not reported.
Quality assessment
Supplemental Figure S1 shows the risk of bias and applicability concerns for the 11 included studies. No studies were considered to be seriously flawed according to the QUADAS-2 tool. All studies satisfied ⩾4 of the seven items.
Regarding the patient selection domain, three studies3,32,36 were considered to have a high risk of bias because they included only severe traumatic brain injury (TBI) patients. Regarding the index test domain, four studies32–34,36 were considered to have a high risk of bias because they used a pre-designated ONSD cutoff value before study processing. Regarding the reference standard domain, all studies were considered to have a low risk of bias. Regarding the flow and timing domains, all studies showed an unclear risk of bias because the mean interval between ONSD measurement and the reference standard was not reported. All studies exhibited low concerns in regard to applicability to our research question with regard to patient selection, index testing, and reference standard domains.
Diagnostic performance of ONSD for prediction of raised ICP
The pooled sensitivity and specificity were 0.88 (95% CI, 0.79–0.94) and 0.86 (95% CI, 0.70–0.95), respectively. According to the pooled DORs with 95% CIs, ONSD was informative for the evaluation of the raised ICP (DOR, 47; 95% CI, 11–206). The pooled positive and negative likelihood ratios were 6.5 (95% CI, 2.6–16.3) and 0.14 (95% CI, 0.07–0.27), respectively. The Q test revealed significant heterogeneity (Q = 6.393, p = 0.02). Sensitivity (I2 = 74.74%) and specificity (I2 = 85.46%) indicated substantial and considerable heterogeneity, respectively. A threshold effect was shown by visual analysis of the coupled forest plot of sensitivity and specificity (Figure 2) as well as a corresponding correlation coefficient of 0.286 (95% CI, −0.177 to 0.722) between sensitivity and the false-positive rate. The area under the HSROC curve was 0.93 (95% CI, 0.91–0.95; Figure 3).
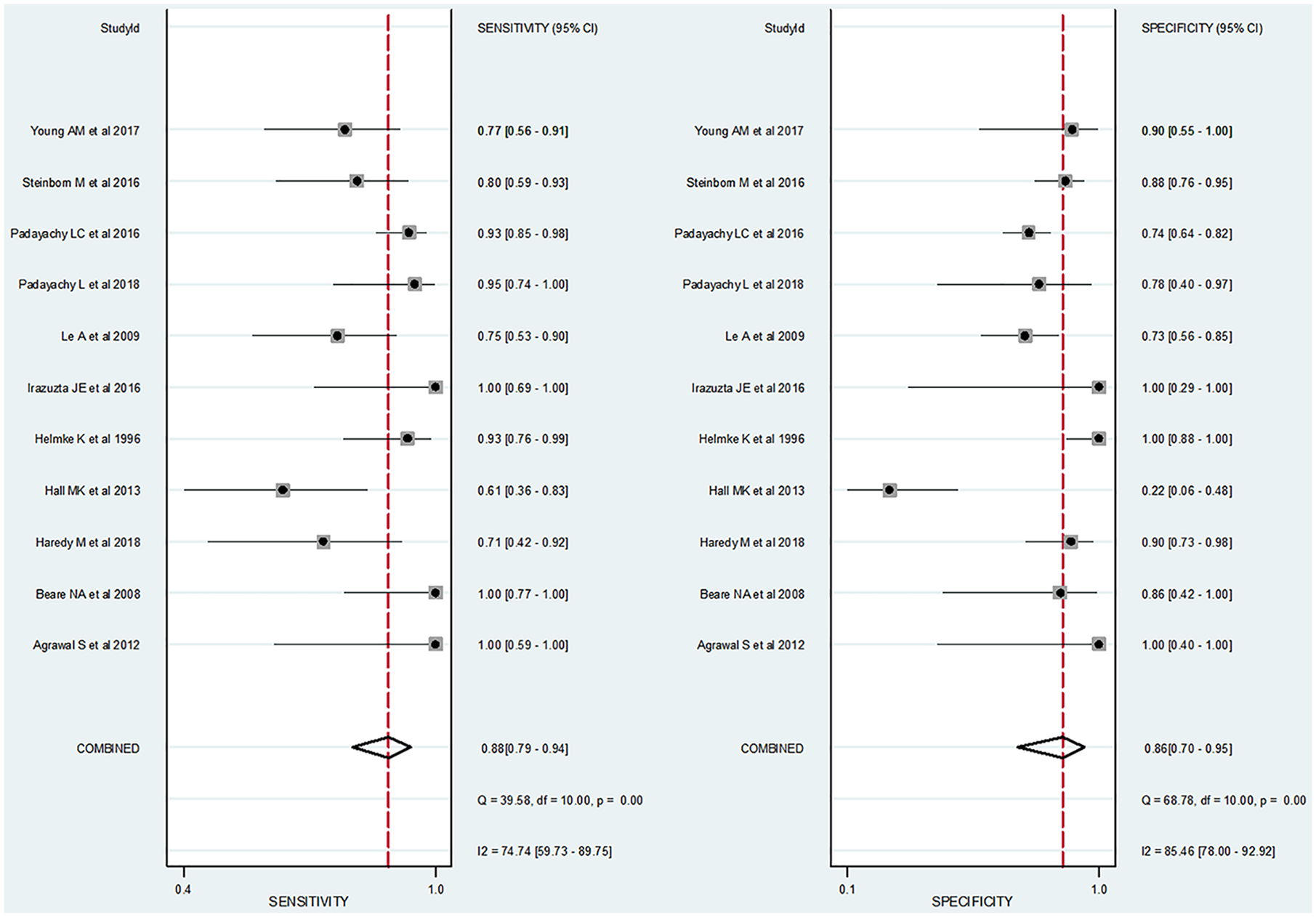
Coupled forest plots of pooled sensitivity and specificity of the optic nerve sheath diameter measurement for diagnosis of raised intracranial pressure in pediatric patients. Numbers are pooled estimates with 95% CIs in parentheses. Dots in squares represent sensitivity and specificity. Horizontal lines represent the 95% CI for each included study. The combined estimate (“Combined”) is based on the random-effects model and is indicated using diamonds. Corresponding heterogeneities (I)2 with 95% CIs are provided in the bottom right corners: I2 = 100% × (Q − df)/Q, where Q is Cochran’s heterogeneity statistic and df is the degrees of freedom.
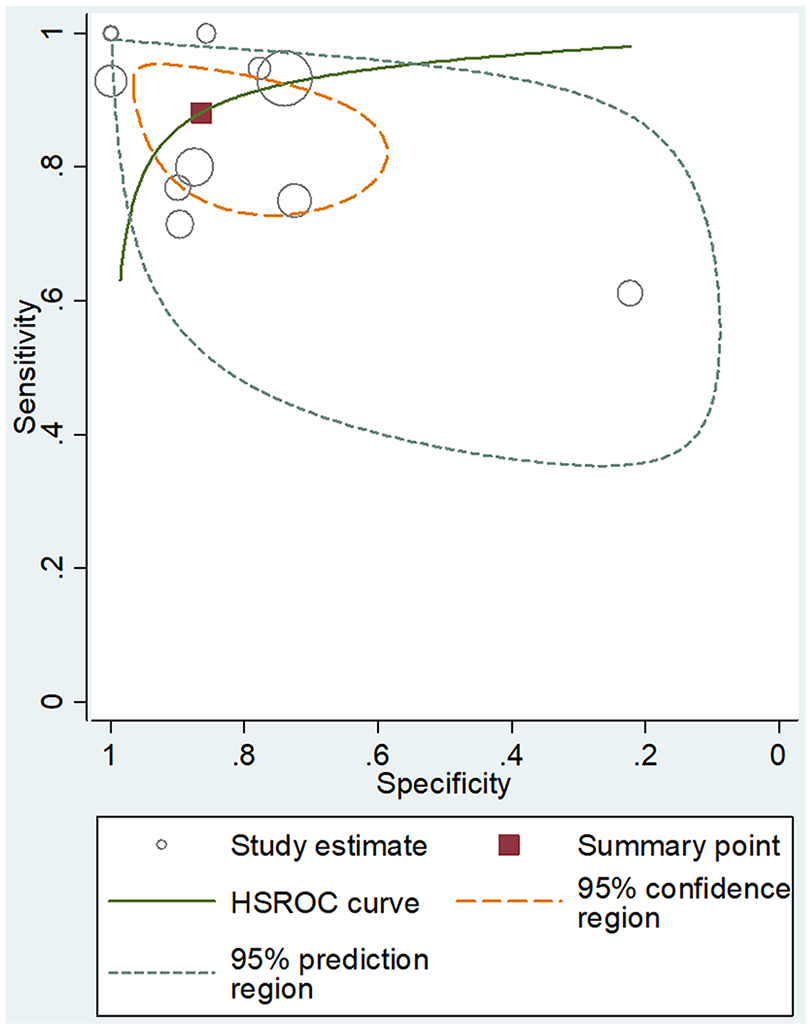
Hierarchical summary receiver operating characteristic curve for the performance of optic nerve sheath diameter measurement for diagnosis of raised intracranial pressure in pediatric patients. The summary point (red box) indicates that the summary sensitivity was 0.88 and the summary specificity was 0.86. The 95% confidence region represents the 95% CIs of summary sensitivity and specificity, and the 95% prediction region represents the 95% CIs of sensitivity and specificity for each included study. The study estimates indicate the sensitivity and specificity estimated using the data from each study. The size of the marker is scaled according to the total number of patients in each study.
Ocular US vs brain CT/MRI
For ocular US, the pooled sensitivity and specificity were 0.91 (95% CI, 0.81–0.96) and 0.86 (95% CI, 0.65–0.96), respectively. For brain CT/MRI, the pooled sensitivity and specificity were 0.75 (95% CI, 0.51–0.99) and 0.91 (95% CI, 0.74–1.00), respectively. The sensitivity was significantly higher for ocular US compared to brain CT/MRI (p = 0.02). However, the specificity was not significantly different (p = 0.84).
Meta-regression analysis results
The results of the meta-regression analyses (Supplemental Table S1) showed that the significant sources of heterogeneity in sensitivity were study design (p = 0.02) and total number of patients (p = 0.03), with higher sensitivity reported in prospective studies with a relatively small number of patients (<30) compared to those with a relatively large number of patients (⩾30). The only significant source of heterogeneity in specificity was the reference standard (p = 0.04), with lower specificity reported in studies with only invasive ICP monitoring as the reference standard compared to studies with mixed or only noninvasive ICP methods. Other factors, including proportion of raised ICP, study population, blinding, interpreter, and ONSD cutoff value, were not significantly different (sensitivity, p = 0.12–0.90; specificity, p = 0.13–0.85).
Discussion
According to the present meta-analysis, ONSD measurement was an informative method (DOR, 47) with excellent performance for diagnosis of raised ICP in pediatric patients (sensitivity, 88%; specificity, 86%). In the subgroup analysis, ONSD measured on ocular US (sensitivity, 91%; specificity, 86%) showed higher sensitivity and comparable specificity than ONSD measured on brain CT/MRI (sensitivity, 75%; specificity, 91%).
The mechanism of ONSD enlargement in raised ICP is well-known. The optic nerve and optic nerve sheath are cylindrical structures that directly connect the eyeball to the cranium, running posterior-centrally and slightly upward toward the optic chiasm. The ONSD is surrounded by the optic nerve sheath which is filled with CSF, and it communicates directly with the subarachnoid space and reflects raised ICP due to increased pressure that causes enlargement of the ONSD.37,38
Our results have important clinical implications because neurocritical care management of pediatric patients focuses on prevention and prompt treatment of secondary insults. 2 In adults, enlarged ONSD is a predictive factor of mortality and poor prognosis.39–41 In fact, patients with enlarged ONSD show a 2.0–22.7-fold greater risk of mortality compared to those with normal ONSD.39,40 Thus, early detection of raised ICP is crucial. ONSD measurements on ocular US or brain CT have many advantages as they are simple, effective, and can be measured in real-time. Moreover, ONSD measurement may help in active surveillance by allowing the assessment of ICP changes during follow-up. 42
Four previous meta-analyses14–17 evaluated the diagnostic performance of ONSD, although these studies had several limitations. First, they only evaluated the usefulness of ultrasonographic ONSD and did not include brain CT or MRI. In addition, no studies have compared the diagnostic performance of ONSD measured on ocular US and brain CT/MRI. Second, these studies did not perform a thorough analysis of the potential sources of heterogeneity, as they did not distinguish between sensitivity and specificity for the effects of covariates, precluding any recommendations regarding methods to improve the diagnostic performance of ONSD. Finally, two studies14,16 did not use hierarchical models such as bivariate or HSROC models, which are recommended statistical tools for the meta-analysis of studies regarding diagnostic accuracy.20,21
In terms of cutoff values, the proposed ONSD showed variation. Most studies (n = 10) used 4.5 mm or more as the ONSD cutoff value for differentiating raised and normal ICP. In studies with children younger than 1 year, ONSD cutoff values for differentiating raised and normal ICP were decreased and 4.0 mm was used as the cutoff value. It has been clearly demonstrated that an ONSD < 4.5 mm for children over 1 year and <4.0 mm for children younger than 1 year indicates no significant ONSD enlargement. Since we did not have raw data from the studies, a single ONSD cutoff value could not be calculated based on this meta-analysis.
Our subgroup analysis revealed that ocular US was more useful than brain CT for diagnosis of raised ICP. In particular, the pooled sensitivity of the ONSD on ocular US was significantly higher than that on brain CT. For this reason, we speculated that brain CT was obtained parallel to the tuberculum sellae–occipital protuberance line, and not parallel to the optic nerve. Consequently, brain CT might demonstrate an oblique image rather than a horizontal one, or not visualize the whole optic nerve sheath. In contrast, ocular US was obtained perpendicular to the optic nerve. Thus, the real diameter of the optic nerve sheath might be more accurate on ocular US. Thus, we recommend that ONSD measurement is performed using ocular US to diagnose raised ICP with greater accuracy.
Our meta-regression analysis revealed that the proportion of total patients, study design, and reference standard were sources of heterogeneity. In particular, the pooled sensitivity of the ONSD was higher in prospective studies with a relatively small number of patients (<30) than in retrospective studies with a relatively large number of patients (⩾30). In addition, pooled specificity of the ONSD was higher in studies with mixed or only noninvasive ICP methods as the reference standard than those with only invasive ICP monitoring. Further prospective studies with larger sample sizes and invasive ICP monitoring as the reference standard are needed.
This study had several limitations. The first limitation was the relatively small number of included studies. Nevertheless, we were able to draw several important conclusions regarding the diagnostic performance of ONSD and related factors (modality), which we believe provides a useful overview because we used broad search terms and only included easily accessible studies (published in English and available in the PubMed and EMBASE databases). The second limitation was that all included studies revealed positive results and that fact could be attributed to publication bias, which is impossible to quantify. Although we omitted Deeks’ funnel plots according to the PRISMA-DTA guidelines, we observed a low probability of publication bias (p = 0.52), which suggests that this factor did not undermine our results. The third limitation was the methodological differences between the included studies, and the extensive meta-regression analysis revealed that these variables were also significant sources of heterogeneity. This methodological diversity might affect the pooled estimates, especially as the symptom onset, time to examination, and pre-hospital management were not assessed in the meta-regression analysis because not all studies reported these factors. Further prospective studies with larger sample sizes and standardization of patient management are needed to establish the optimal parameters and cutoff value for ONSD.
Conclusion
In conclusion, the present meta-analysis revealed that ONSD measurement may be a useful method for predicting raised ICP in pediatric patients. We recommend that ONSD measurement should be performed using ocular US to more accurately diagnose raised ICP in pediatric patients. The use of routine ocular US, which is less time-consuming and easily accessible without radiation hazard, may be helpful for the detection of raised ICP in pediatric patients.
Supplemental Material
Supplemental_Figure_S1 – Supplemental material for Optic nerve sheath diameter measurement for predicting raised intracranial pressure in pediatric patients: A systematic review and meta-analysis
Supplemental material, Supplemental_Figure_S1 for Optic nerve sheath diameter measurement for predicting raised intracranial pressure in pediatric patients: A systematic review and meta-analysis by Sun Hwa Lee, Seong Jong Yun and Dong Hyeon Kim in Hong Kong Journal of Emergency Medicine
Supplemental Material
Supplemental_materials_190628 – Supplemental material for Optic nerve sheath diameter measurement for predicting raised intracranial pressure in pediatric patients: A systematic review and meta-analysis
Supplemental material, Supplemental_materials_190628 for Optic nerve sheath diameter measurement for predicting raised intracranial pressure in pediatric patients: A systematic review and meta-analysis by Sun Hwa Lee, Seong Jong Yun and Dong Hyeon Kim in Hong Kong Journal of Emergency Medicine
Footnotes
Author contributions
S.H.L. and D.H.K. designed the study and the literature search; S.H.L. and S.J.Y. analyzed data; S.H.L. and D.H.K. wrote the manuscript; S.H.L., S.J.Y., and D.H.K. made the critical revision of this manuscript. All the authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
All data generated or analyzed during this study are included in this published article. The materials described in the manuscript will be freely available to any scientist wishing to use them for non-commercial purposes, without breaching participant confidentiality.
Informed consent
Since the study was a meta-analysis, informed consent was not obtained. Written informed consent was not necessary because no patient data have been included in the manuscript.
Ethical approval
This study was approved by the local ethics committee. To maintain patient confidentiality, the forms did not include any data that would have enabled identification of any patients. The procedures performed in this study followed the ethical standards in the 1964 Declaration of Helsinki, as revised in 2008, as well as the national law.
Human rights
This study was conducted according to the World Medical Association Declaration of Helsinki. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
