Abstract
Introduction:
Dichloromethane (DCM) and trichloroethylene (TCE) are both common halohydrocarbons which are widely used in industry. The most common exposure route of DCM or TCE is inhalation, but intramuscular injection of DCM and TCE is very rarely.
Case presentation:
A 47-year-old male presented ecchymosis, pain, and swelling of the left limb following accident injection of ABS glue (main ingredients were DCM and TCE). The swelling of his left limb was lasted until day 6 and WBC, NEUT, CK, CK-MB, LDH, AST and ALT were elevated until day 9. He was received intravenous fluids, analgesic, antibiotics, and other supportive measures and finally discharged from the hospital on day 16.
Discussion:
Both DCM and TCE could cause local irritation and systemic toxicity involving central nervous system, liver, kidney and other organs. In this case, because the small dose of toxicant and injection being the route of exposure, the patient mainly suffered from local toxicity resulting in painful soft tissue swelling with slight hemocytology and biochemistry disorder. Although there are no specific antidotes for DCM and TCE poisoning, supportive measures may also provide satisfactory therapeutic results.
Conclusions:
DCM and TCE poisoning following intramuscular injection could present mainly with local and system poisoning symptoms and successfully treated with supportive measures.
Introduction
Dichloromethane (DCM; CAS No.75-09-2) and trichloroethylene (TCE; CAS No.79-01-6) are both common halohydrocarbons which are widely used in industry as detergents, solvents, or extraction agents. DCM and TCE poisoning can result from inhalation of the vapor, ingestion, or absorption through the skin.1,2 However, to the best of our knowledge, intramuscular injection of DCM and TCE is very uncommon. We describe the case of a male patient who developed local and systemic symptoms following accidental intramuscular injection of DCM and TCE.
Case
A 47-year-old male presented to our Poison Control Center (PCC) with pain and swelling in the left lower limb following accidental injection of “ABS glue” (~5 mL) into the inner left thigh for 6 h. The patient alleged that a syringe with a needle, filled with about 5 mL of acrylonitrile butadiene styrene (ABS) glue, accidentally fell and stabbed into his left thigh at work. Due to the stabbing pain, he folded his legs subconsciously and injected nearly all of the ABS glue into his left thigh with his right leg. The patient felt severe pain at the injection site and was sent to the nearest local clinic by his colleagues. He was immediately diagnosed with an unknown chemical injection injury and the local doctor administered oxygen and parenteral dexamethasone and pethidine. The patient was referred to our PCC for further treatment 6 h later.
The patient was conscious, cooperative with no neurological symptoms. He only complained of left leg pain. Upon physical examination, he had a normal body temperature with a heart rate of 84/min, a blood pressure of 136/84 mmHg, and a respiratory rate of 20/min. Local examination revealed a red puncture lesion over the inner aspect of the left thigh and exhibited an erythematous reaction (Figure 1(a)). The left lower limb exhibited swelling and tenderness (Figure 1(b)); the maximum perimeters were 48 cm (left thigh) versus 46 cm (right thigh) and 35 cm (left calf) versus 32.5 cm (right calf), respectively. Local temperature, sensation, arteriopalmus of the dorsalis pedis artery, and other neurological, thoracic, and abdominal examinations were normal.
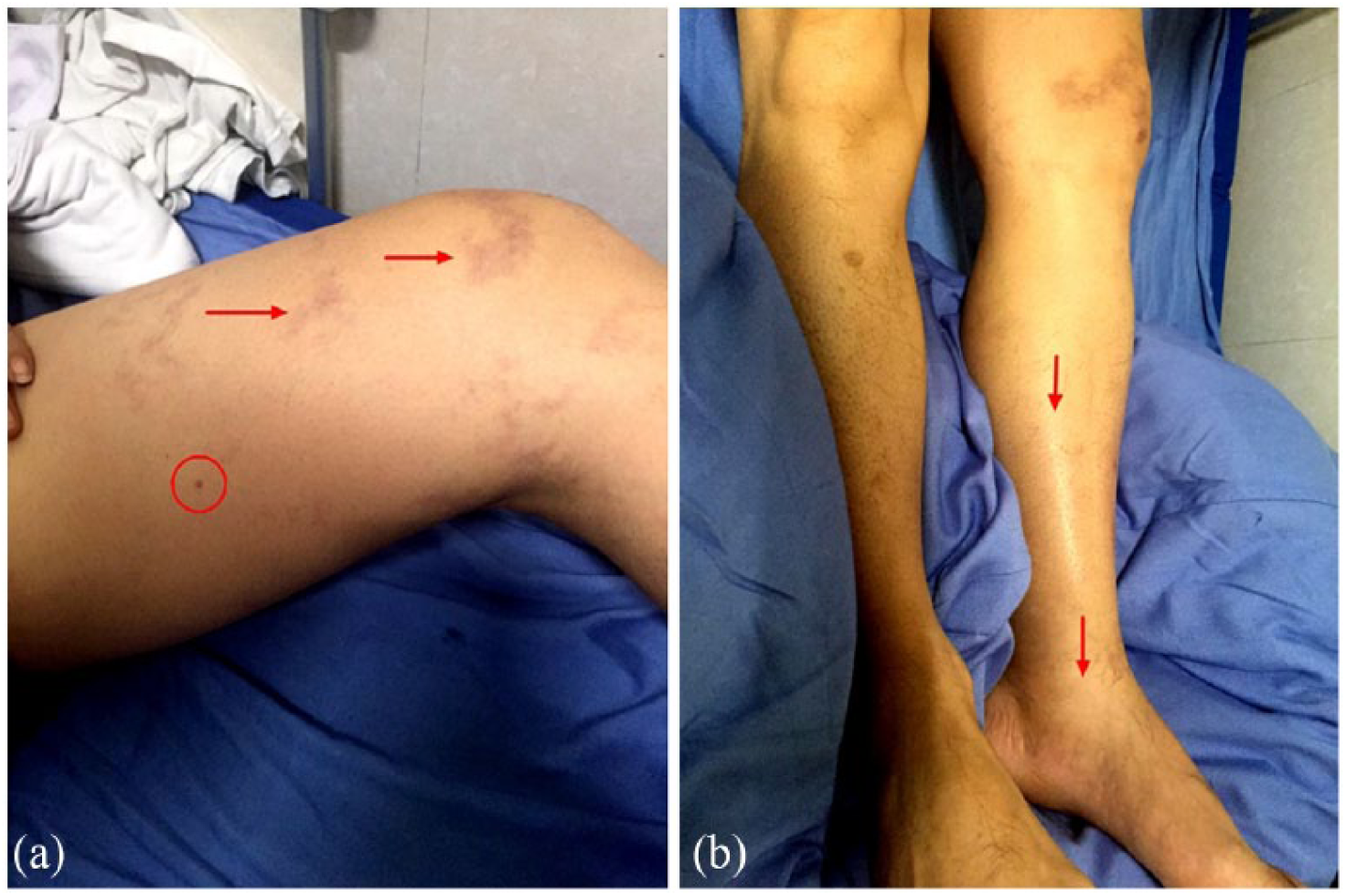
Photos of the patient’s lower limbs after accidental intramuscular injection of ABS glue: (a) red cycle shows the puncture lesion and red arrow shows the ecchymosis; (b) red arrows show swelling of the left lower limb versus right lower limb.
Routine blood tests, myocardial enzymes, liver function, and renal function are shown in Tables 1 and 2. Serum electrolytes, coagulation functions, and blood gas analysis showed normal values. Computed tomography (CT) scans showed mild left lower limb soft tissue swelling, especially over crus dorsal, but no foreign body was found. Ultra-sonic examination of lower limb vessels showed that the left anterior, posterior tibial, and the left fibular vein were obscured by soft tissue swelling. Other lower limb veins and arteries were normal. He was treated with intravenous fluid and 10 mg i.v. furosemide was given intravenously to enhance the elimination of poisons and for edema control. Intramuscular analgesic (dezocine 5 mg once a day) was continued for 2 days, anti-inflammatory therapy (i.v. dexamethasone 10 mg once a day) was continued for 3 days, and Latamoxef sodium (i.v. 1.0 g bid) was administered empirically for local wound infection. The Chinese traditional medicines named “Hu Li San” (containing prepared kusnezoff monkshood root, pseudo-ginseng, and others, external use) and “Salviae Miltiorrhizae Liguspyragine Hydrochloride and Glucose Injection” were applied for improving local microcirculation. Pain in the patient’s left lower limb improved after day 1. Meanwhile, the left lower limb swelling progressed. On day 2, laboratory analysis with gas chromatography–mass spectrometry (GC-MS) on the “ABS glue” collected from the patient’s workshop showed that it contained 76.287% DCM and 23.713% TCE. Blood gas analysis shown carboxyhemoglobin (COHb) was 1.8% (normal reference: 0~2%). On day 4, maximum perimeters of the patient’s lower limbs became 52 cm (left thigh) versus 46 cm (right thigh) and 37 cm (left calf) versus 32 cm (right calf), and tension vesicles were observed on the inner aspect of the left leg. On day 6, tension vesicles were broken and the swelling started resolving. However, white blood cell (WBC), neutrophil (NEUT), creatine kinase (CK), creatine kinase-MB isoenzyme (CK-MB), lactate dehydrogenase (LDH), aspartate aminotransferase (AST), and alanine aminotransferase (ALT) increased to peak levels before gradually declining to normal levels on day 9 (Tables 1 and 2). The patient was finally discharged from the hospital on day 16.
Routine blood tests of the patient.
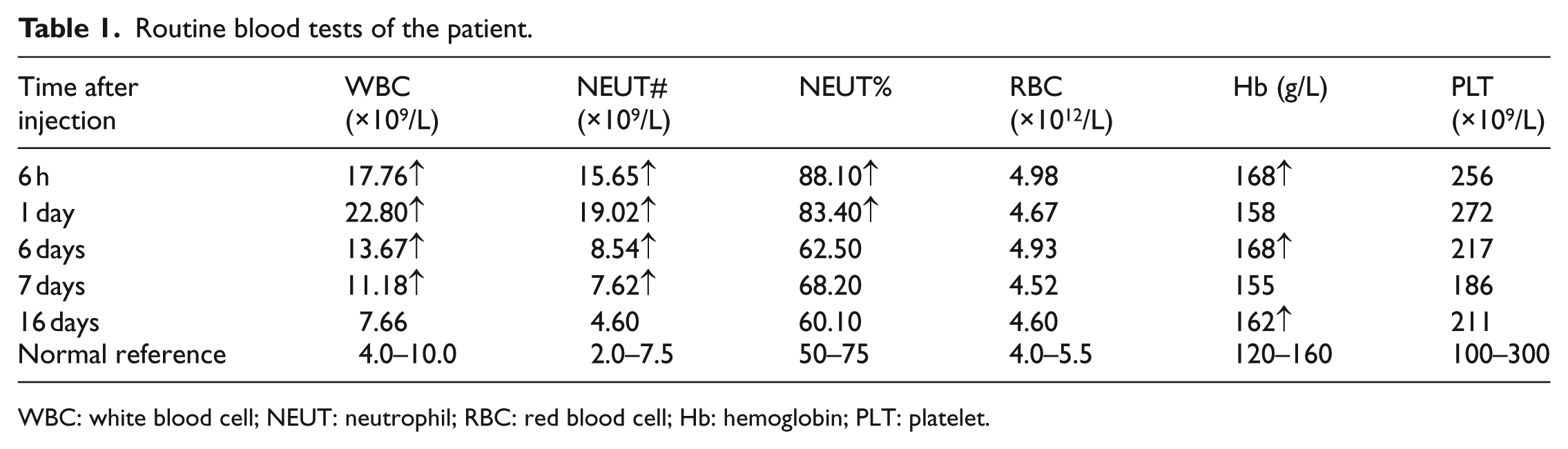
WBC: white blood cell; NEUT: neutrophil; RBC: red blood cell; Hb: hemoglobin; PLT: platelet.
Myocardial enzymes, liver function, and renal function test of the patient.
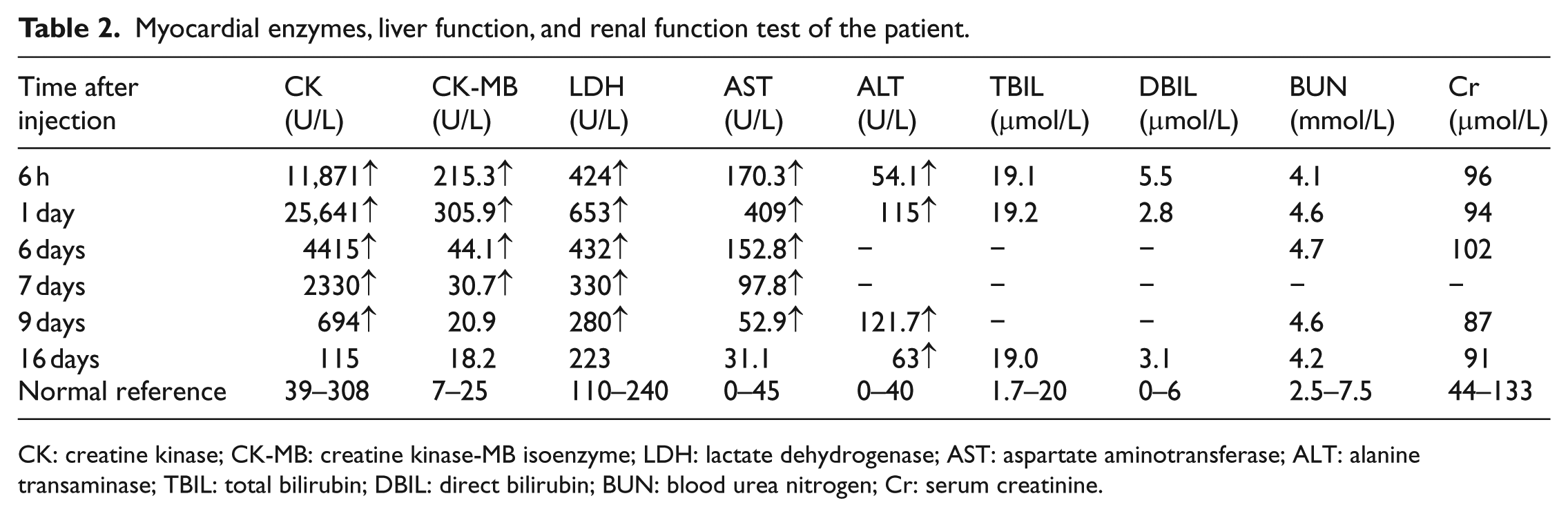
CK: creatine kinase; CK-MB: creatine kinase-MB isoenzyme; LDH: lactate dehydrogenase; AST: aspartate aminotransferase; ALT: alanine transaminase; TBIL: total bilirubin; DBIL: direct bilirubin; BUN: blood urea nitrogen; Cr: serum creatinine.
Discussion
ABS glue is a commonly used adhesive for binding ABS resin or other plastics. It often contains acetone, chloroform, or other organic solutions, such as DCM and TCE in this case.
DCM, also known as methylene chloride, is an organic solvent. It is a highly volatile and colorless liquid with a chloroform-like smell. DCM is used in various industrial applications as a degreaser, an extraction medium, a lacquer remover, a paint stripper, an aerosol propellant, and a flammability depressant. The most common route of absorption is inhalation, but poisoning through both the skin and ingestion are also possibilities. 3 Airway exposure to DCM can result in poisoning caused by either the direct effects of DCM or by its metabolite, carbon monoxide (CO). Central nervous system (CNS) effects of DCM exposure include impairment of vision, 4 hearing, 5 psychomotor performance, and, in more severe cases, unconsciousness and death. 6 Caustic esophageal and gastric mucosal injuries, acute toxic hepatitis, and renal failure after ingestion of DCM were also reported.3,7
TCE is a colorless liquid with sweet, chloroform-like odor. It is also a volatile, chlorinated, organic chemical often used as a degreaser for metal parts or as a general-purpose solvent for lipophilic compounds. It is often used as an ingredient in paint removers, typewriter correction fluids, rug-cleaning fluids, and spot removers. TCE can be absorbed through the digestive tract or respiration and distributes throughout the body, preferentially partitioning into fat. TCE is metabolized primarily by the liver to its active forms, trichloroacetic acid, and free trichloroethanol, and is eliminated through the kidneys and lungs. 8 Inhalation affects the human CNS, resulting in dizziness, headache, confusion, euphoria, facial numbness, and weakness. 9 In addition, lung, 10 liver, 11 kidney, 12 cardiac, immunological, 13 and carcinogenic effects have been reported in humans. 14
In our case, the patient mainly suffered from local toxicity resulting in painful soft tissue swelling that spread down from the injection site over his left thigh to his left leg with tension blisters which took around 2 weeks to resolve. There was no CNS symptom observed in our case and his COHb was also within the normal level. This may be explained by the small dose of DCM and injection being the route of exposure; furthermore, immediate oxygen therapy (within 30 min after the accident) may have helped in the prevention of carboxyhemoglobinemia. Local tissue irritation or even necrosis was the main clinical manifestation of poisoning in the patient together with leukocytosis, raised aminotransferase levels, and raised CK-MB. Surgical debridement, incision, and drainage have been carried out to eliminate poisons in other chemical injection reports; 15 however, for our patient, DCM and TCE were absorbed in a short period of time by injection and radiographic inspection found no foreign agent near the injection point, which is why this therapeutic intervention was not used in our case. Although there are no specific antidotes for DCM and TCE poisoning, supportive measures may also provide satisfactory therapeutic results.
Conclusions
In summary, we have reported a case of DCM and TCE poisoning following intramuscular injection—a novel route of administration—presented mainly with local poisoning symptoms and successfully treated with supportive measures only.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (No. 81302460) and Research Project of Shenzhen Health and Family Planning Commission (No. SZFZ2017026).
Availability of data and materials
The clinical documentation of the presented case cannot be made public due to the detailed identifiable information of the patient.
Ethical approval
This research was approved by Research Project Ethics Committee of Shenzhen Prevention and Treatment Center of Occupational Disease on 12 December 2016.
Informed consent
The participant had given informed written consent to report the individual patient data.
