Abstract
Objective:
The aim of this study was to explore the effects and safety of low-dose hydrocortisone in patients with septic shock.
Methods:
The PubMed, EMBASE, and Cochrane Central Register of Controlled Trials were searched from database inception until 1 August 2018. Two reviewers performed literature selection, data extraction, and quality evaluation independently.
Results:
Twelve randomized controlled trials were included in this meta-analysis. The combined results showed that low-dose hydrocortisone use had no survival benefit in patients with septic shock (relative risk = 1.09; 95% confidence interval = 0.88–1.05; P = 0.37). But low-dose hydrocortisone use was useful for shock reverse (relative risk = 1.09; 95% confidence interval = 1.00–1.19; P = 0.04) and in shortening the time of vasopressor support (weighted mean difference = −1.79, 95% confidence interval = −2.05 to −1.52; P < 0.00001). In addition, use of low-dose hydrocortisone was associated with a higher risk of hyperglycemia (relative risk = 1.21; 95% confidence interval = 1.04–1.40; P = 0.01) and hypernatremia (relative risk = 6.34; 95% confidence interval = 1.19–33.81; P = 0.03). There was no significant improvement of intensive care unit mortality (relative risk = 1.11; 95% confidence interval = 0.93–1.33; P = 0.23) or hospital mortality (relative risk = 1.08; 95% confidence interval = 0.94–1.24; P = 0.29), length of intensive care unit (weighted mean difference = −1.84; 95% confidence interval = −5.80 to 2.11; P = 0.36) or length of hospital (weighted mean difference = 0.11; 95% confidence interval = −2.06 to 2.29; P = 0.98), and time of mechanical support (weighted mean difference = −0.69; 95% confidence interval = −1.76 to −0.38; P = 0.20) with the use of low-dose hydrocortisone. There was no significant difference in secondary infection (relative risk = 1.04; 95% confidence interval = 0.91–1.18; P = 0.57), recurrence of shock (relative risk = 1.47; 95% confidence interval = 0.64–3.39; P = 0.36), and gastrointestinal bleeding (relative risk = 1.41; 95% confidence interval = 0.89–2.22; P = 0.14) with the use of low-dose hydrocortisone.
Conclusion:
Although there was no effect of low-dose hydrocortisone on survival of patients with septic shock, it is associated with a higher rate of shock reversal and shortening duration of vasopressor support; thus, low-dose hydrocortisone may be an alternative drug in septic shock patients who are refractory to fluid resuscitation and vasopressors.
Background
Septic shock is the most common diagnosis associated with poor outcome. 1 Nevertheless, besides early fluid resuscitation and antibiotics use, no consensus reached by physicians on other pharmacologic treatments. Glucocorticoid is one of hot topics. High-dose glucocorticoids used in septic shock patients were reported to be associated with worse outcome. 2 In 2004, Annane et al. 3 found that low-dose glucocorticoid could decrease the 28-day mortality rate. Hydrocortisone is the most reported drug. But, the positive effects of low-dose glucocorticoid were not confirmed by following studies. 4 The aim of this study was to explore the effects and safety of low-dose hydrocortisone alone in patients with septic shock by meta-analysis.
Methods
Registration and protocol
The present meta-analysis was registered in PROSPERO (https://www.crd.york.ac.uk/PROSPERO/), and registration number is CRD42018090792. The conduct and reporting of the study follow the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.
Eligibility criteria
Patients: The patients are septic shock patients. Septic shock was defined according to the 1992 American College of Chest Physicians/Society of Critical Care Medicine (ACCP/SCCM) Consensus (before 2016), 5 or the Third International Consensus Definitions for Sepsis and Septic Shock. 6
Intervention: Low-dose hydrocortisone, a daily dose of hydrocortisone ≤300 mg. 7
Comparisons: Placebo or standard treatment irrespective of dose, duration, intravenous infusion method, or co-intervention.
Outcomes: The primary endpoint was 28-day mortality; The secondary endpoints included long-term mortality, reversal of shock, length of stay, renal replacement therapy, mechanical ventilation, duration of vasopressor support, and related adverse events. Reversal of shock was defined as the maintenance of a systolic blood pressure of at least 90 mm Hg without vasopressor support for at least 24 h or as described in each included study. 7
Study type: Randomized clinical trials or quasi-randomized controlled trials.
Data source
PubMed, EMBASE, and Cochrane library were searched for eligible studies from its inception until 1 August 2018. The combination of Medical subject headings (MeSH) and text word search was used in this study.8–10 Detailed literature search strategy was in Appendix 1. Ongoing and unpublished clinical trials were searched via http://www.isrctn.com, ClinicalTrials.gov, and the World Health Organization trial registry. In addition, the references of eligible papers were screened to obtain additional articles.
Literature selection and data extraction
Two reviewers performed literature selection through three steps: (1) excluding duplicates, (2) excluding clearly non-relevant studies by reading titles and abstracts, and (3) selecting eligible studies for meta-analysis by reading the remaining studies in full text. Two reviewers performed data extraction using a pre-defined sheet and the characteristics of each study, the baseline information of included patients, items necessary for methodological evaluation, and the primary and secondary endpoints. Any disagreement was solved by discussion or consulting with the third reviewer.
Quality evaluation
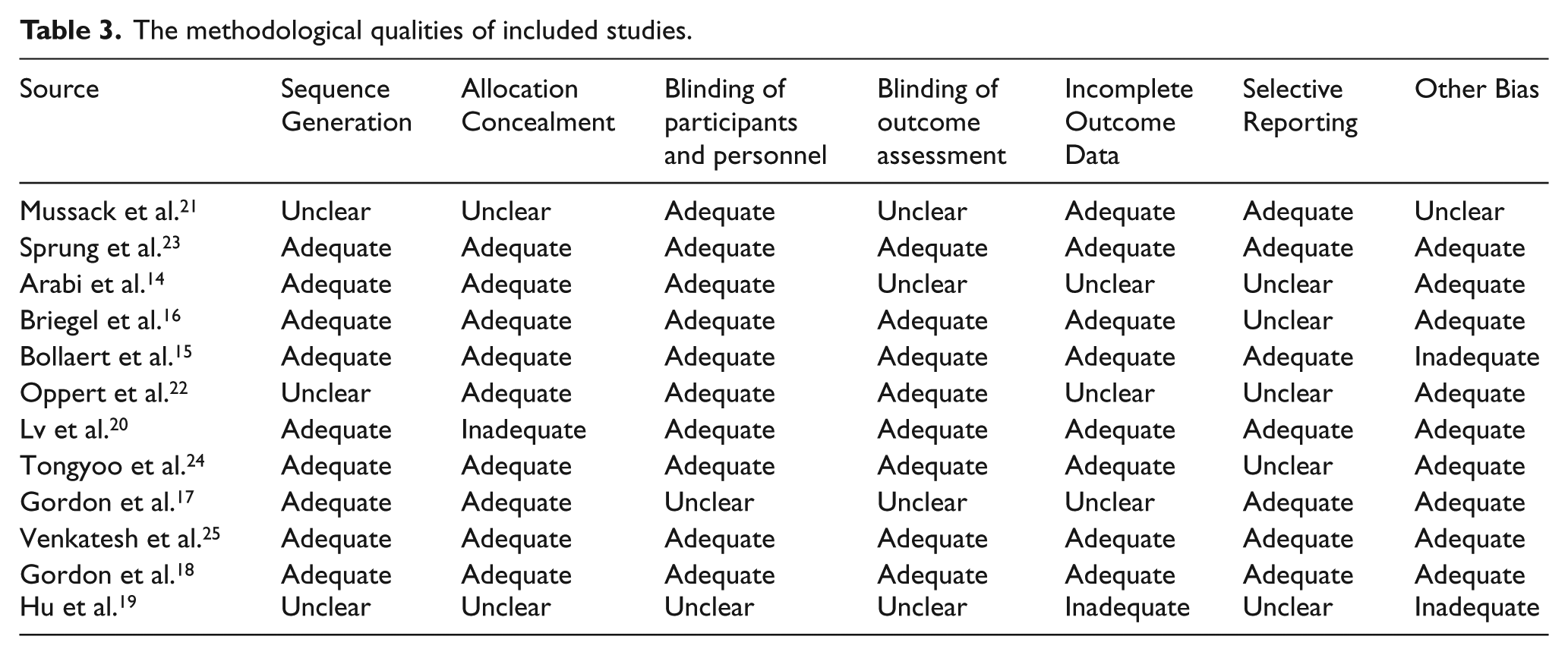
Two reviewers evaluated the quality of included studies independently using the Cochrane Risk Assessment Tool. 11 The following domains were evaluated: sequence generation, allocation concealment, blinding of patients and personnel, blinding of outcome assessors, incomplete outcome data, and selective reporting. Sequence generation will be considered adequate if the study explicitly described an appropriate randomization procedure to generate an unpredictable sequence of allocation, including computerized randomization, use of random number tables, and coin tossing. Concealment of allocation will be considered adequate if specific methods to protect allocation were documented and implemented. Performance bias will be considered low if a study reported participant, caregiver, and researcher blinding. Blinding of outcome assessment will be considered adequate if outcome assessors and adjudicators were blinded. And each domain was assessed as adequate, inadequate, or unclear.
Meta-analysis
The risk ratio was used for categorical variables and mean deviation was used for continuous variables. Heterogeneity between studies was explored using the I2 test and chi-square test. I2 > 50 or P < 0.1 indicates significant heterogeneity and random-effect model would be applied. Otherwise, fix-effect model was applied. Publication bias was evaluated by funnel plot and Egger’s test. In some studies, only median was reported, and we used the approach that proposed by Wan et al. 12 and Luo et al. 13 to estimate the mean and standard deviation. One pre-specified subgroup analysis was performed based on adrenal responsiveness (corticotropin stimulation test responders vs. non-responders).
Results
A total of 1777 citations were obtained. Finally, 12 studies with 5400 patients met eligibility criteria.14–25 Figure 1 shows the process of literature search and reasons for exclusion of studies. The reason of excluded literature was in Appendix 2. Among them, five trials were multicenter.15,17,18,23,25 In most studies, low-dose hydrocortisone treatment was compared with placebo. And in the study by Hu et al., 19 low-dose hydrocortisone treatment was compared with standard treatment. Ten trials14,15,17,18,20–25 reported 28-day mortality, four trials16–18,23 reported intensive care unit (ICU) mortality, and five trials14,17–19,23 reported hospital mortality. Table 1 shows an overview of the detailed information of the included studies. Table 2 shows the excluded reason of these excluded studies. Table 3 shows the summary of risk of bias assessments for all included studies.
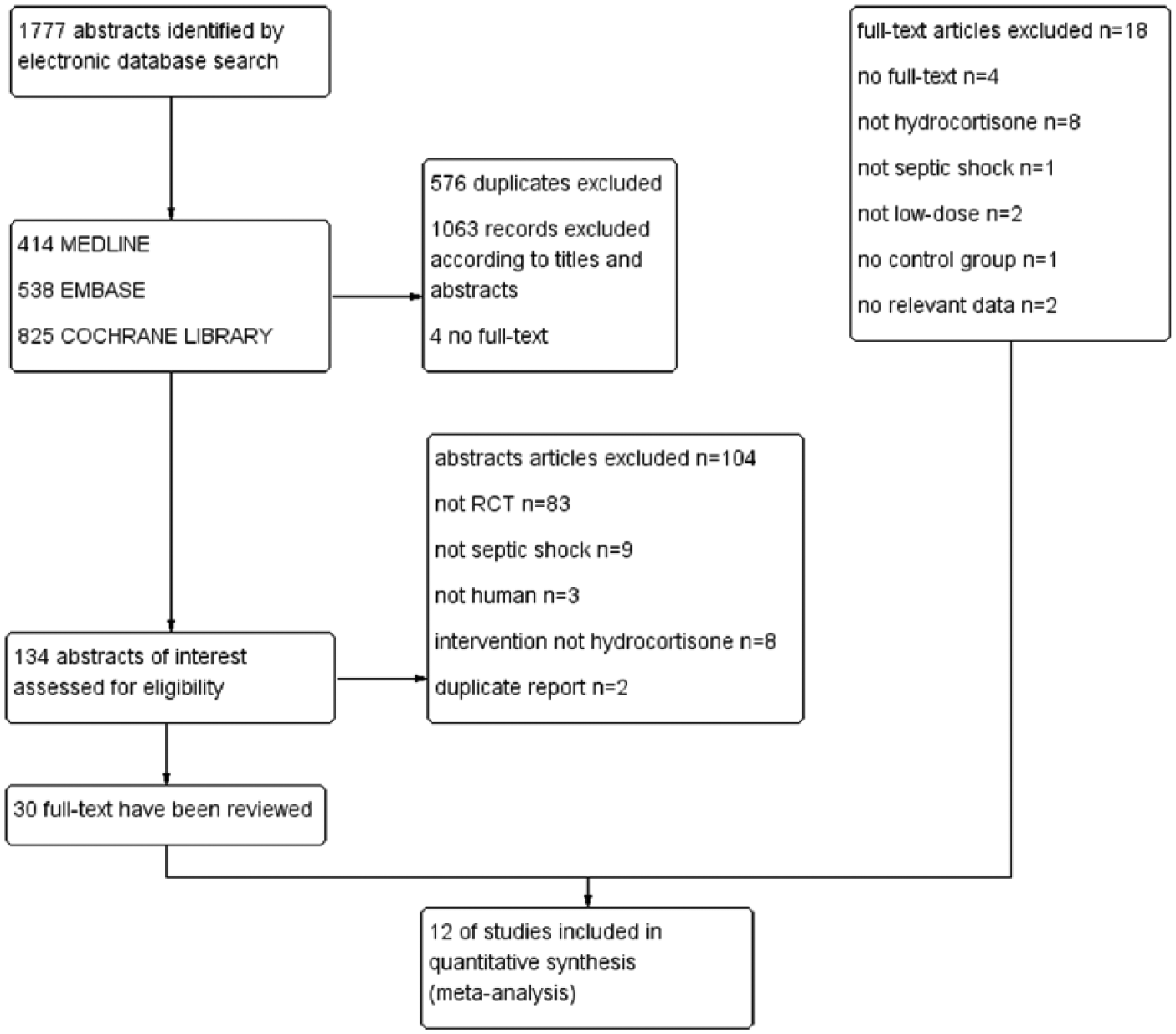
Flow diagram of the literature search.
Excluded literature.
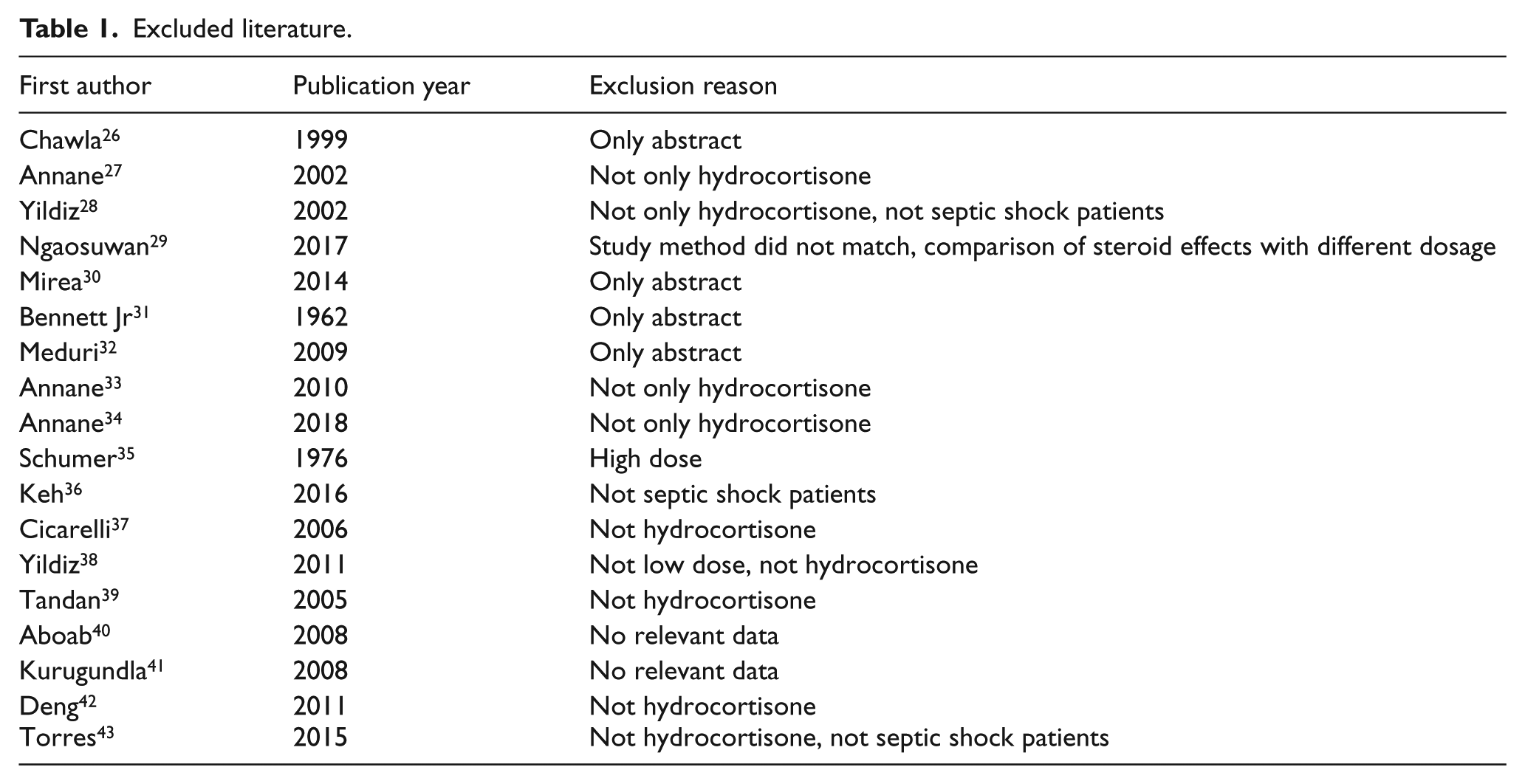
Characteristics of the included trials.
No: number; min: minutes; kg: kilogram; h: hours; q: quaque; ivi: intravenous.
The methodological qualities of included studies.
Primary outcome
28-day mortality
Ten studies14,15,17,18,20–25 and 5283 patients eligible for analysis. During the 28-day follow-up, 662 (25.1%) patients in the low-dose hydrocortisone group died and 689 (38.5%) patients in the control group died. The pooled relative risk (RR) was 0.96 (95% confidence interval (CI) = 0.88–1.05; P = 0.37) with no significant heterogeneity (I2 = 21% by fixed-effects model) (Figure 2).
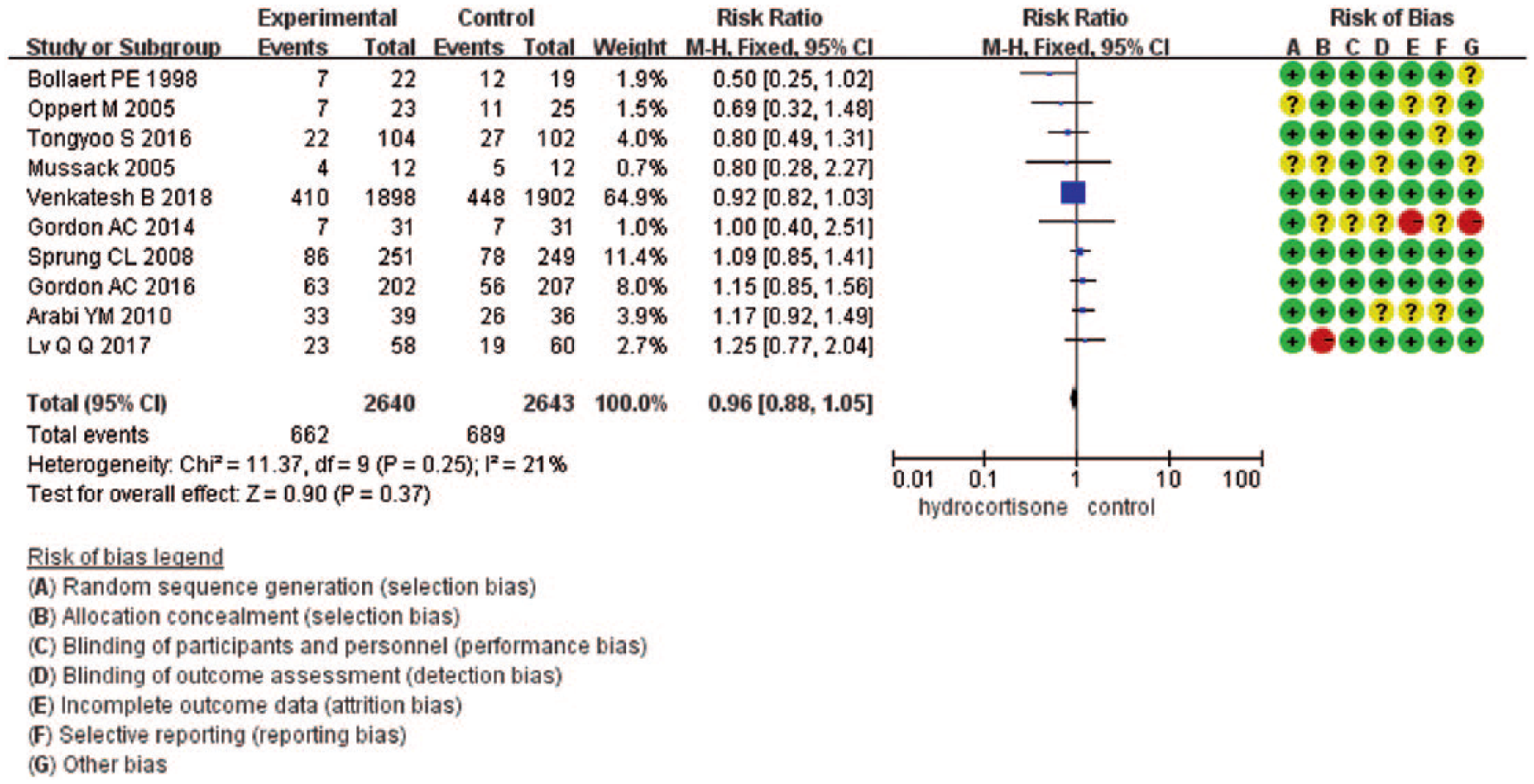
Forrest plot showing the effect of low-dose hydrocortisone compared to placebo or control on 28-day mortality in adult septic shock patient.
Sensitive analyses
The result of 28-day mortality was driven by the study by Venkatesh et al., 25 which carried 64.9% of the weight. A sensitivity analysis was performed by excluding the study by Venkatesh et al. And it did not significantly change the result (RR = 1.05; 95% CI = 0.91–1.21; P = 0.49; I2 = 8%) (Appendix 2).
Subgroup analyses
In two trials,15,23 the authors explored the different effects of hydrocortisone according to the responsiveness of patients. In the group of patients who were responsive to hydrocortisone, the combined RR was 0.80 (95% CI = 0.43–1.47), and in the non-responders, the combined RR was 0.86 (95% CI = 0.60–1.22) (Appendix 2).
Secondary outcomes
ICU mortality
Four trials16–18,23 were eligible for this analysis. The ICU mortality in the intervention and control group was 171 of 504 (33.9%) patients versus 154 of 507 (30.4%) patients. The combined RR was 1.11 (95% CI = 0.93–1.33; P = 0.23; I2 = 0%) (Table 3).
One-year mortality
Two trials16,23 (n = 540) reported 1-year mortality. One-year mortality for treated versus control patients was 142 of 271 (52.4%) versus 133 of 269 (49.4%) (RR = 1.06; 95% CI = 0.90–1.25; P = 0.46; I2 = 0%) (Table 3).
Shock reversal
Two trials15,22 (n = 89) reported the shock reversal rate within 7 days, and five trials14,16,20,21,23 (n = 757) reported the shock reversal rate in entire treatment period. Shock reversal by day 7 for treated versus control patients was 27 of 45 (60.0%) versus 22 of 44 (50.0%) (RR = 1.46; 95% CI = 0.31–6.96; P = 0.63; I2 = 89%). Shock reversal in entire treatment period for treated versus control patients was 291 of 380 (76.6%) versus 265 of 377 (70.3%) (RR = 1.09; 95% CI = 1.00–1.19; P = 0.04; I2 = 6%) in the fixed-effects model (Figure 3); and RR = 1.09; 95% CI = 0.99–1.19; P = 0.07; I2 = 6% in the randomized-effects model. Shock reversal in the whole seven trials for treated versus control patients was 318 of 425 (74.8%) versus 287 of 421 (68.2%) (RR = 1.11; 95% CI = 0.94–1.31; P = 0.23; I2 = 53%) (Appendix 2).
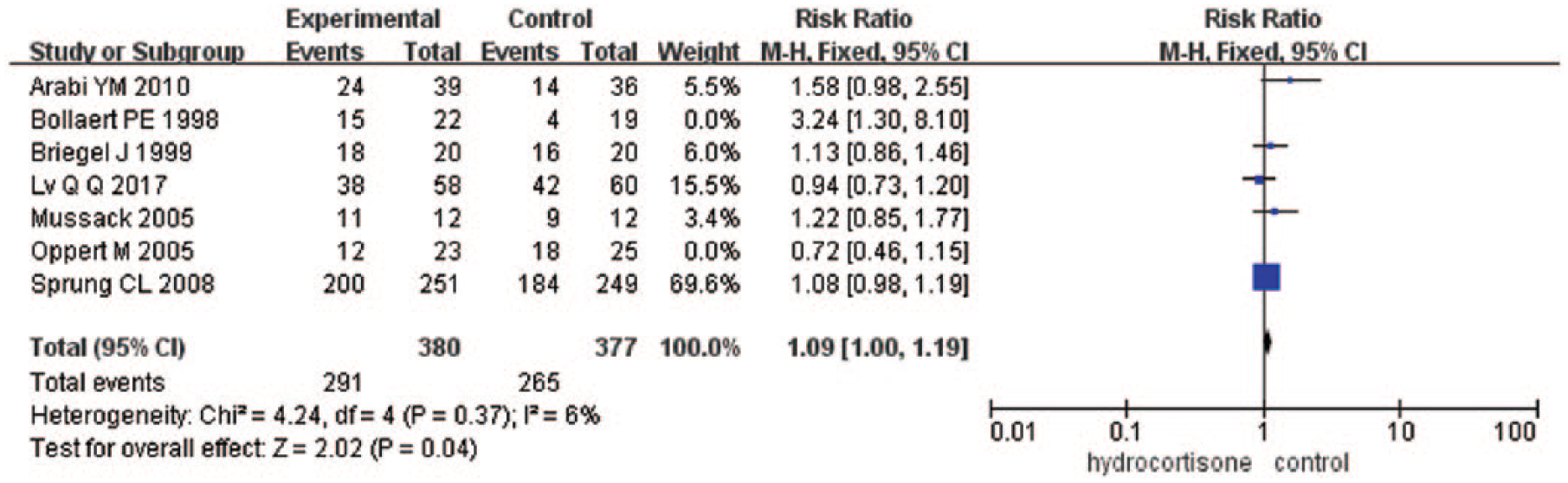
Forrest plot showing the effect of low-dose hydrocortisone compared to placebo or control on shock reversal in five trials.
Time to resolution of shock
Three trials16,23,25 (n = 4340) reported the time to resolution of shock. The combined weighted mean difference (WMD) was −0.80 days (95% CI = −1.48 to −0.11; P = 0.02; I2 = 91%) (Table 3). The source of heterogeneity might be due to the trial of Sprung et al. 23 A sensitivity analysis was performed by excluding the study by Sprung et al., and the results did not change the overall conclusion (WMD −0.25 days, 95% CI = −0.34 to −0.15; P < 0.001; I2 = 0%).
Duration of vasopressor support
Five trials15,19,21,22,24 (n = 395) reported the duration of vasopressor support of two groups. Among them, the mean and standard deviation were reported in two studies,19,24 and mean and quartile were reported in three studies.15,21,22 The combined WMD was −1.78 days (95% CI = −2.04 to −1.52; P < 0.001; I2 = 0%) (Figure 4). Two trials19,24 reported average time of vasopressor also showed hydrocortisone can reduce the time of vasopressor support (WMD = −1.79, 95% CI = −2.05 to −1.52; P < 0.001; I2 = 0%) (Appendix 2).
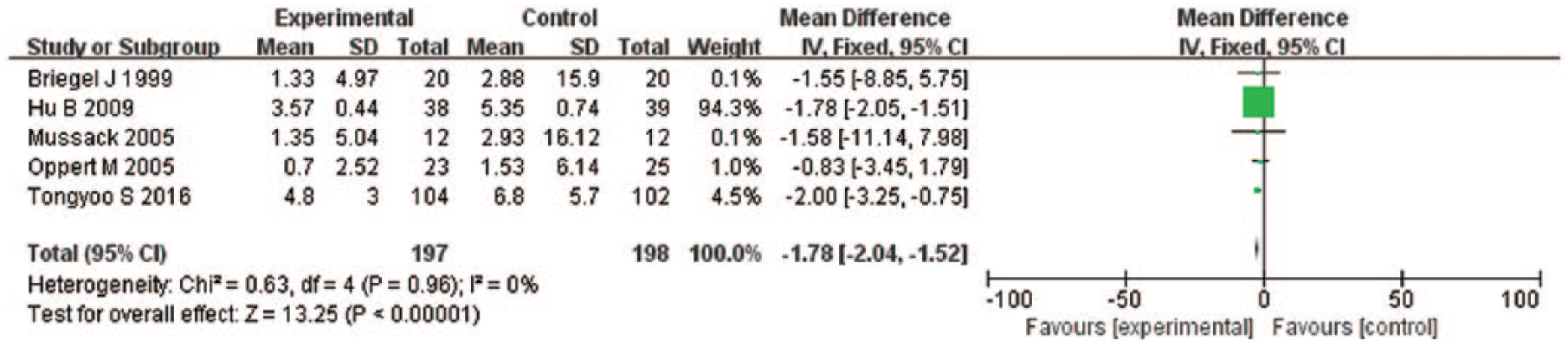
Forrest plot showing the effect of low-dose hydrocortisone compared to placebo or control on time of vasopressor support in adult septic shock patient.
Renal replacement therapy
Five trials16,18,21,24,25 reported the number of patients requiring renal replacement therapy. The reported renal replacement therapy rates for treated versus control patients were 652 of 2236 (29.2%) patients versus 714 of 2243 (31.8%) patients, and the pooled RR was 0.92 (95% CI = 0.84–1.00; P = 0.05; I2 = 0%) (Table 3). And the outcome was driven by the study by Venkatesh et al., 25 which carried 85.9% of the weight. A sensitivity analysis was performed after removing the study by Venkatesh et al. (RR = 0.82; 95% CI = 064–1.04; P = 0.10; I2 = 0%) (Appendix 2).
Serious adverse events
Recurrence of shock was reported in two trials14,25 (n = 3875 patients), and the pooled RR was 1.47 (95% CI = 0.64–3.39; P = 0.36; I2 = 72%) (Table 4). Gastrointestinal bleeding was reported in six trials14–16,23–25 (n = 4662 patients), and the pooled RR was 1.41 (95% CI = 0.89–2.22; P = 0.14; I2 = 5%) (Table 3). Secondary infections were reported in five trials14,15,23–25 and the pooled RR was 1.04 (95% CI = 0.91–1.18; P = 0.57; I2 = 0%) (Table 4). Hyperglycemia was reported in three trials,14,24,25 and the pooled RR was 1.21 (95% CI = 1.04–1.40; P = 0.01; I2 = 0%) (Table 4). Hypernatremia was reported in two studies,16,25 and the pooled RR was 6.34 (95% CI = 1.19–33.81; P = 0.03; I2 = 0%) (Table 4).
The effect of hydrocortisone on tertiary outcomes in patients with septic shock.
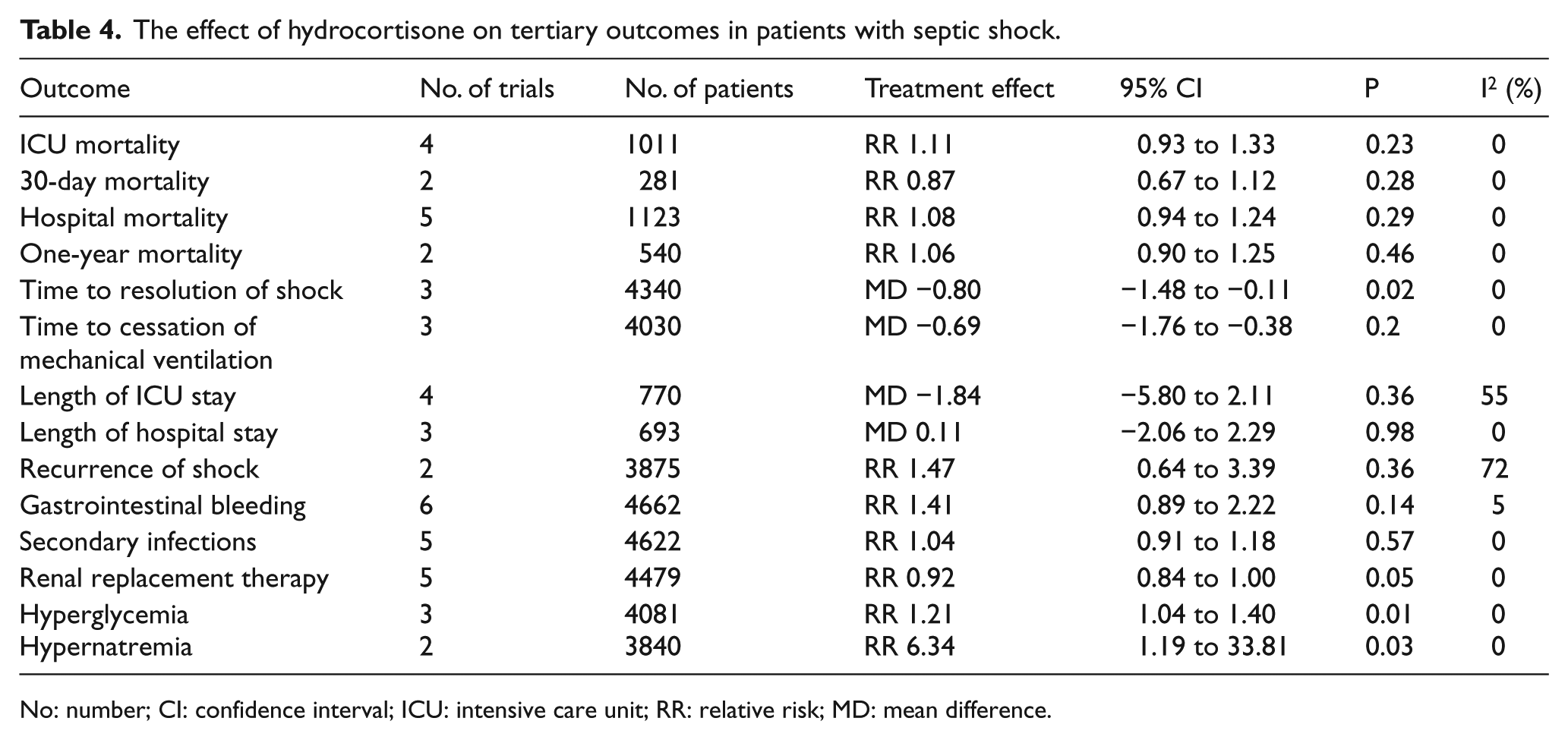
No: number; CI: confidence interval; ICU: intensive care unit; RR: relative risk; MD: mean difference.
Discussion
The results of the present study showed that low-dose hydrocortisone has no significant effects on survival in patients with septic shock. However, use of low-dose hydrocortisone could shorten the duration of vasopressor support and be associated with a higher rate of shock reversal. Meanwhile, we found that low-dose hydrocortisone could reduce the occurrence of renal replacement therapy. This may be related to less use of vasopressor in the intervention group. In addition, hydrocortisone was associated with an increased risk of developing hyperglycemia and hypernatremia. There were no significant effects of low-dose hydrocortisone treatment on ICU/hospital length of stay, gastrointestinal bleeding, and secondary infection.
The pathophysiology of septic shock involves complicated interactions between infectious organisms and host that can result in multi-system organ failure and death. 44 Corticosteroids may be beneficial in patients with septic shock due to its multi-effect.27,45–47 However, in view of the current evidence, the effects of corticosteroids on these patients remain unclear.25,34 In 1951, Hahn et al. 48 first reported the therapeutic effect of hydrocortisone on severe infection. From the 1950s to 1980s, high-dose corticosteroids like methylprednisolone (30 mg/kg) or dexamethasone (3–6 mg/kg) for 1 to 2 days were accepted in adult septic shock patients. 49 Although Schumer et al. 35 found that high-dose dexamethasone and methylprednisolone (corticosteroid replacement therapy) could significantly reduce the mortality of patients with septic shock in 1976, their conclusion did not corroborate in the following studies. And high-dose corticosteroids even might increase the mortality rate.2,50,51 Then corticosteroid replacement therapy stopped. In the late 1990s, Bollaert et al. 15 reported low-dose hydrocortisone could significantly improve the hemodynamic status and facilitate shock reversal in septic shock patients. And their results were confirmed in several following studies (usually 200–300 mg of hydrocortisone per day for 5–7 days).27,49,52,53 So, low-dose hydrocortisone was recommended in septic shock patients by the 2004 Surviving Sepsis Campaign guidelines. 54 But following studies reported inconsistent results of low-dose hydrocortisone on mortality.23,25,27,34 Our study showed low-dose hydrocortisone could shorten the duration of vasopressor use and was beneficial for shock reversal. But there was no statistical effect of low-dose hydrocortisone on mortality. Previous meta-analyses focusing on corticosteroids in septic shock patients also reached inconsistent conclusion.7,52,55,56 Rochwerg et al. 56 in 2018 and Annane et al. 7 in 2009 in their meta-analysis reported that corticosteroids were associated with a higher survival rate when compared with patients in the control group. Different inclusion criteria may be responsible for this inconsistent result. In these two studies, the authors did not focus on patients with septic shock. However, in the present study, only septic shock patients were eligible for meta-analysis. Then, in the Rochwerg 56 study, besides hydrocortisone, other corticosteroids were also analyzed in combination. And in the present study, only the effects of hydrocortisone were analyzed to minimize the heterogeneity.
Our results are similar to those of Rygard et al. 45 who demonstrated that corticosteroid therapy does not reduce mortality rates but does appear to consistently reduce the time to shock reversal. Eleven of these studies met our inclusion criteria.14–18,20–25 It may be the reason that our results are similar to the study of Rygard, there was a little different in length of ICU stay between our study with the study of Rygard et al., it may be associated with the different inclusion criteria, our study only includes the trials of hydrocortisone alone, and specific length of ICU stay for each of these subpopulations was not reported.
The physiologic action of fludrocortisone is similar to that of hydrocortisone. However, the former has considerably heightened and prolonged effects on electrolyte balance and carbohydrate metabolism. Two large randomized controlled trials (RCTs) by Annane et al.27,34 in 2002 and 2018 demonstrated the mortality beneficial in septic shock adults with the use of hydrocortisone combined with fludrocortisone. However, another large RCT named COIITSS study, also was launched by Annane et al., 33 included 509 patients with septic shock adults, and found that the use of fludrocortisone in conjunction with hydrocortisone was associated with an increased risk of infection. The different outcomes may associate with the more severe degree of the septic shock in COIITSS study or the time to use corticosteroids. A meta-analysis by Rygard 45 reported a survival benefit of hydrocortisone plus fludrocortisone therapy in a subgroup of three trials. But their result was driven by the study by Annane, 34 and more studies are required.
As for the optimal time to use hydrocortisone, the Surviving Sepsis Campaign guidelines 57 suggest consideration of corticosteroids therapy for patients with septic shock who were refractory to fluids and vasopressors, based on the results of several studies.7,25 In the study by Annane et al. 27 (50 mg intravenous bolus every 6 h and fludrocortisone 50 µg once daily), they enrolled patients within 8 h from onset of septic shock to randomization and they found mortality beneficial. In the APROCCHSS trial 34 (50 mg intravenous bolus every 6 h and fludrocortisone 50 µg once daily for 7 days), 1241 patients were randomized within less than 24 h from onset of septic shock, and they also reported mortality beneficial. In the CORTICUS study 23 (50 mg of intravenous hydrocortisone every 6 h for 5 days) and ADRENAL trial 25 (200 mg hydrocortisone per day for 7 days), patients were randomized 24 to 72 h and 20 to 90 h after the onset of septic shock and they did not found mortality beneficial. A retrospective study by Park et al. 58 (≤300 mg/day of hydrocortisone or equivalent) in 2012 showed that septic shock patients in the early therapy group (within 6 h after the onset of septic shock) had a lower mortality rate than those in the late therapy group. Another prospective study by Katsenos 59 (daily administration of 50 mg q 6 h for 7 days) in 2014 also reported early therapy with hydrocortisone within 9 h after the onset of shock was associated with a lower mortality rate than those received hydrocortisone more than 9 h after the onset of shock. It seems that there is a trend of decreasing mortality rate of early use of hydrocortisone. But high-quality RCTs focusing on early use versus late use of hydrocortisone are scarce and more evidence are needed. Keh et al. 36 explored the effects of hydrocortisone on the development of septic shock in patients with severe sepsis. And they found hydrocortisone therapy in patients with severe sepsis can not reduce the risk of development of septic shock within 14 days. This evidence does not support the preventive use of hydrocortisone in sepsis patients. As for the dosing strategy of hydrocortisone, common regimens for corticosteroids include hydrocortisone 100 mg intravenous bolus every 8 h or 50 mg intravenous bolus every 6 h. Another option for providers is 100 mg intravenous bolus followed by an infusion of 0.18 mg/kg/h. Lots of studies reported there was no mortality beneficial between continuous infusion and bolus infusion, but the glycemic controls well when hydrocortisone is administered as a continuous infusion as compared with intermittent bolus. 44
The strength and limitation of this meta-analysis
In order to minimize potential bias, only randomized controlled trials evaluating low-dose hydrocortisone in septic shock patients were eligible. And trials focusing on other types of corticosteroids were excluded, and observational studies were also excluded. Several limitations of this meta-analysis should be concerned. First, different septic shock diagnostic criteria, different administration route, and different hydrocortisone dose may bias the results of this meta-analysis. But due to the limited sample size, we cannot perform further subgroup analysis. Second, the interaction between corticosteroids and other drugs like vasopressin is not evaluated in the present study. It has been reported that corticosteroids and vasopressin used together might improve the prognosis of patients with septic shock. 53
The implications for clinical practice and further studies
Based on the results of this study, low-dose hydrocortisone is beneficial for hemodynamic status in septic shock patients, although there is no association between use of low-dose hydrocortisone and survival rate. Meanwhile, use of low-dose hydrocortisone did not result in a higher risk of serious adverse events. Thus, if there are no obvious contraindications, including myopathy with neuromuscular blocking agents, diabetic ketoacidosis, human immunodeficiency virus infection, etc, 59 low-dose hydrocortisone can be considered as an adjunctive treatment in patients with septic shock. In future studies, the septic shock should be confirmed using the newest criteria. Based on the results of this study, more studies are required on economic impact of utilizing hydrocortisone in patients with septic shock. It is essential to standardize the timing, method, discontinuation of hydrocortisone administration, and adjunctive therapy in septic shock patients. In addition, long-term endpoints and functional rehabilitation should be the primary outcome.
Conclusion
The results of this study show no survival benefit of low-dose hydrocortisone in patients with septic shock, but it is associated with a higher rate of shock reversal and shortening duration of vasopressor support. Thus, low-dose hydrocortisone may be an alternative drug in septic shock patients who are refractory to fluid resuscitation and vasopressors. Further studies focusing on precision medicine of hydrocortisone in septic shock patients are needed.
Footnotes
Appendix 1
Appendix 2
Acknowledgements
My deepest gratitude goes first and foremost to Professor Ma, my supervisor, for his constant encouragement and guidance. Second, I would like to express my heartfelt gratitude to my senior fellow, who gave me a lot of help. I am also greatly indebted to my alma mater—Zhejiang University. Finally, our deepest gratitude go to the anonymous reviewers for their careful work and thoughtful suggestions that have helped improve this paper substantially.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
