Abstract
Introduction:
Kyushin® is a widely used herbal medicine in East Asia for heart failure. Toad venom in present in Kyushin®, which has positive inotropic effect as digitoxin.
Case presentation:
An 81-year-old, female patient presented with decreased mental status after overdose of Kyushin®. The first electrocardiogram showed junctional tachycardia with 142/min, suddenly dropped to 27/min and followed by ventricular fibrillation. After one cycle of CPR, spontaneous circulation returned but junctional bradycardia, tachycardia, and ventricular fibrillation appeared. After six times of defibrillation, spontaneous circulation returned, and mechanical ventilator and transcutaneous pacing were applied. Plasma toxicology test revealed digitoxin 66.90 ng/mL by cloned enzyme donor immunoassay and digoxin 0.76 ng/mL by kinetic interaction of microparticles in solution immunoassay. After 8 h from presentation, the patient’s mental status came to be alert, and then transcutaneous pacing was removed.
Discussion:
Cloned enzyme donor immunoassay has been reported to be highly cross-reactive with digoxin-like substances, which strongly supports bufotoxin to be responsible for arrhythmia of the patient.
Conclusion:
Poisoning of bufotoxin in Kyushin® can cause cardiac arrhythmia, even arrest, but without digoxin-specific Fab, conventional therapy could be successful.
Introduction
Kyushin® (Chinese, meaning “saving heart”), manufactured by Boryung pharmaceuticals, Republic of Korea, is a tiny, dark-brown herbal medicine pill with unique scent and taste. It is widely used in East Asia for heart failure and can be purchased without a prescription. Its ingredients are toad venom 0.83 mg (as bufosteroid 48 μg), musk 0.167 mg, oriental bezoar bovis 0.5 mg, white ginseng 4.167 mg, swine gall bladder 1.33 mg, pearl 1.25 mg, dried swine liver powder 1.0 mg, and borneolum 0.45 mg in a pill. 1
Toad venom, found in secretion from granular glands of the skin, parotid glands, liver, ovary, and eggs of some species of toads (the genus bufo in the family bufonidae, Bufo gargarizans, Bufo stejnegeri in East Asia) contains several toxins and bioactive alkaloids, such as bufadienolides (bufogenins like bufalin, cinobufagin, cinobufotalin, and bufotenine), which are cardioactive steroids like cardenolides, cardiac glycosides from toxic foxglove plants. They have several lactone rings in chemical structure, potent inhibitory effect of Na+-K+-ATPase activity in myocardial cell membrane and positive inotropic effect as digoxin and digitoxin.2–5
Dried extracts of the secretion, Chan Su in Chinese, Senso in Japanese, have been used in traditional medicines in East Asia for inflammation and heart failure. But when overdosed, it can cause gastrointestinal manifestations such as nausea and vomiting, cardiac arrhythmia, symptoms of central nervous system such as confusion, hallucination, and coma, similar to those of digoxin intoxication.2,6
We report a case of Bufotoxin intoxication of an 81-year-old female patient who showed signs of acute digitalis overdose and recovered from tachy-brady arrhythmia and cardiac arrest.
Case report
An 81-year old, female patient presented in the emergency department with symptoms of decreased mental status, vomiting, and fluctuation of heart rate. About 2 h 30 min before ED presentation, a medical crew in the nursing facility found the patient in decreased mentality and an empty 100-tablet bottle of Kyushin® next to the patient, but it was unclear how many tablets had been in the bottle.
Her Glasgow coma scale score was 8 points without any focal neurologic deficits, systolic blood pressure (SBP) was 104 mm Hg, heart rate 142/min with IV dopamine infusion from the facility, respiratory rate 24/min and body temperature 36.0°C. Capillary blood glucose level was 15.7 mmol/L, arterial blood gas analysis under oxygen supply of 15 L/min via non-rebreathing mask showed pH 7.408, PCO2 29 mm Hg, PO2 206.9 mm Hg, HCO3– 18.5 mmol/L, O2 saturation 99.5% and lactate 4.5 mmol/L. The patient’s breathing sound on auscultation was clear and heart sound was normal without murmur.
She had medical histories of dementia, major depressive disorder for 30 years and several episodes of intentional drug overdose. But during recent 5 years, she has not been taking any medications for her problems.
The first 12-lead electrocardiogram (ECG) on patient’s arrival showed junctional tachycardia with heart rate of 142/min with right bundle branch block, corrected QT interval 429 ms (Figure 1). After 3 min, suddenly the heart rate dropped to 27/min, ECG showed atrial fibrillation with slow ventricular response, and IV atropine 1 mg bolus, IV dobutamine infusion started (Figure 2).
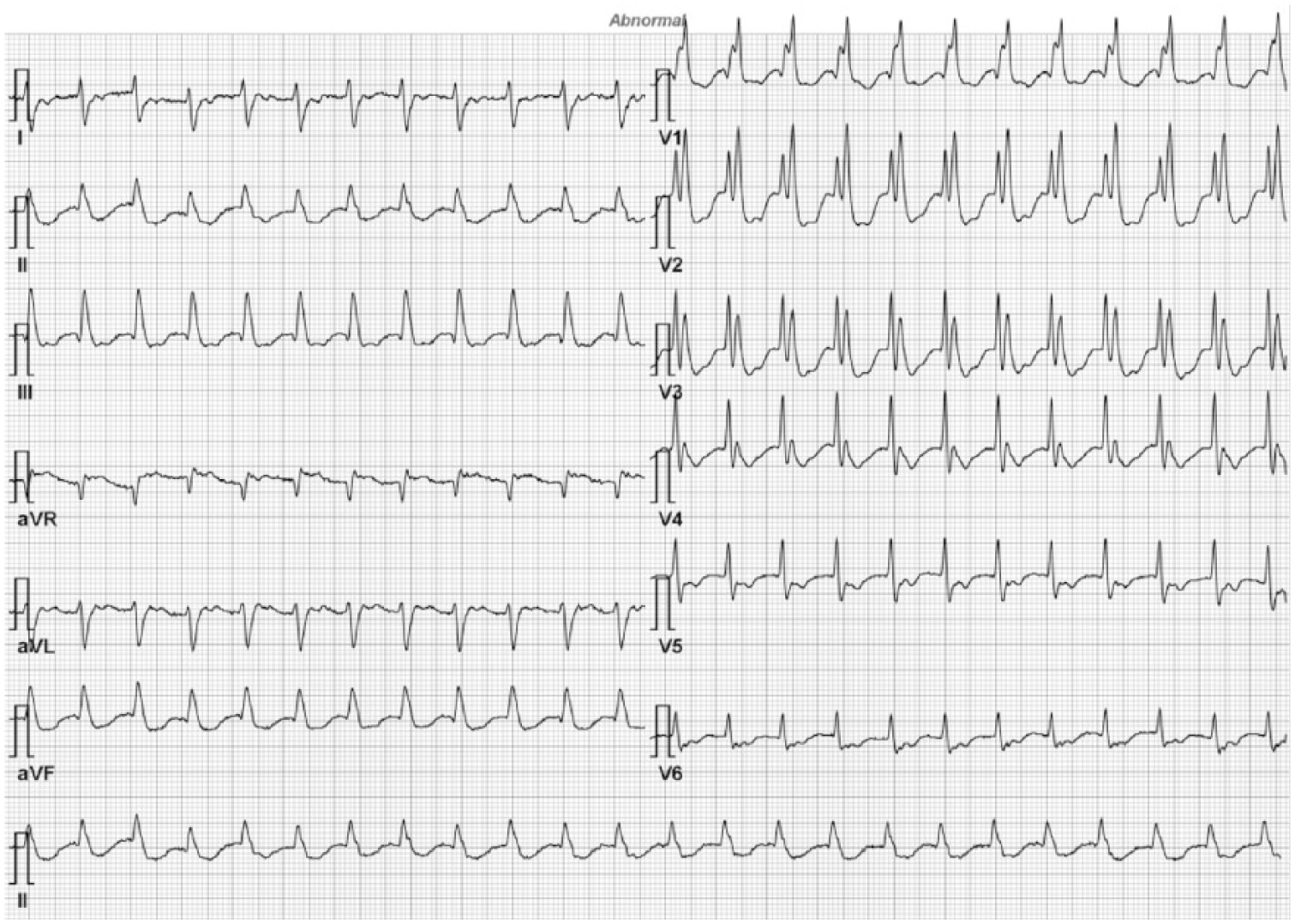
The initial ECG of the patient showed junctional tachycardia with right bundle branch block.
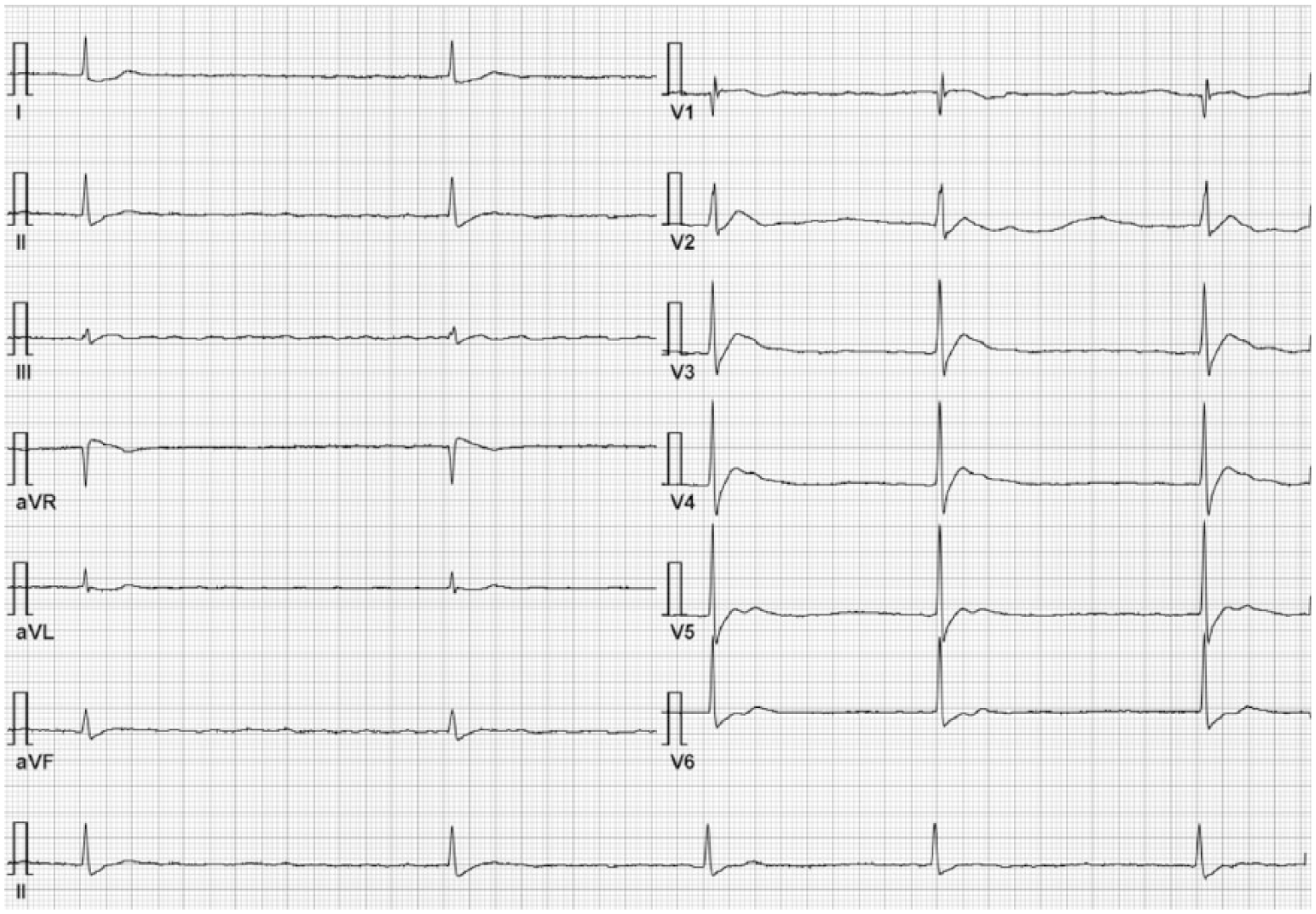
Atrial fibrillation with slow ventricular response suddenly occurred after tachycardia.
After 15 min, ventricular fibrillation on ECG monitor was detected, cardiopulmonary resuscitation started, and defibrillation with energy of 200 J was delivered. After one cycle of CPR, patient’s spontaneous circulation returned, but junctional bradycardia showed for a moment, junctional tachycardia above the rate of 150/min and ventricular fibrillation appeared in order (Figure 3).
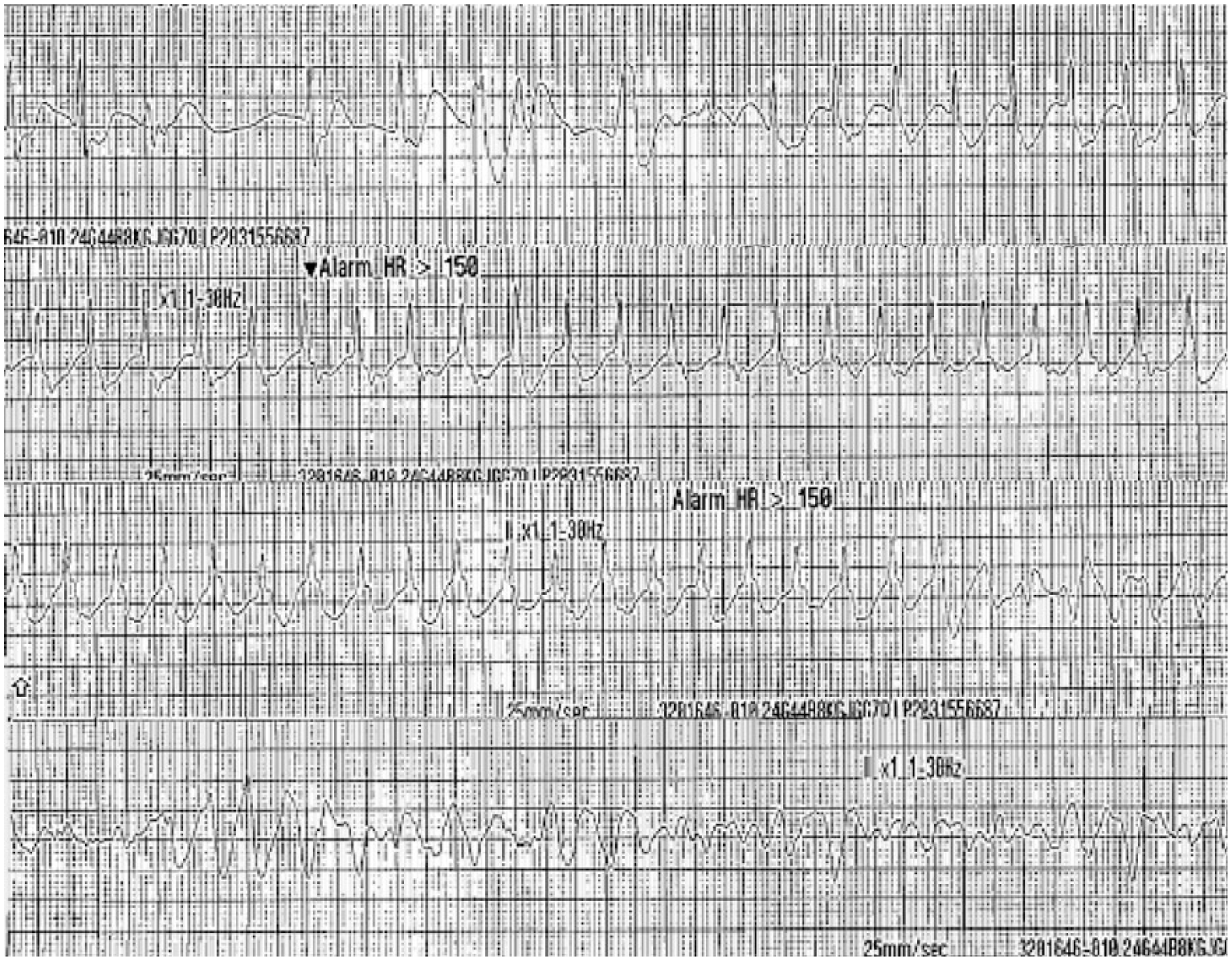
Junctional bradycardia turned into junctional tachycardia, then deteriorated to ventricular fibrillation.
During CPR, six times of defibrillation were delivered, IV magnesium 2 g, and amiodarone 300 and 150 mg were injected. After return of patient’s spontaneous circulation, SBP was 134 mm Hg, ECG showed junctional bradycardia with rate of 43/min. Then, external cardiac transcutaneous pacing with the current of 130 mA and rate of 60/min, mechanical ventilator and activated charcoal of 50 g via nasogastric tube were applied.
Initial chest radiography showed mild cardiomegaly, but there is a limitation to read because it is AP view, white blood cell count was 28.82 × 109/L, hemoglobin 12.6 g/dL, platelet count 263 × 109/L, glucose 12.3 mmol/L, aspartate transaminase 27 IU/L, alanine transaminase 11 IU/L, blood urea nitrogen 5.5 mmol/L, creatinine 79.58 μmol/L, sodium 142.7 mEq/L, potassium 4.4 mEq/L, chloride 102.1 mEq/L, creatine kinase 87 U/L, lactate dehydrogenase 386 U/L, troponin-T 0.132 ng/ML, and creatine kinase-MB 3.9 mg/mL in laboratory tests.
Plasma toxicology test from the blood sample obtained 5 h after ingestion revealed that the serum concentration of digitoxin was 66.90 ng/mL, measured by cloned enzyme donor immunoassay (CEDIA, AU680, Beckerman Coulter, Mitsubishi chemical medians, Tokyo, Japan) and digoxin 0.76 ng/mL, by kinetic interaction of microparticles in solution immunoassay (KIMS, Cobas Integra 800, Roche, Seoul clinical laboratories, Yongin, Republic of Korea).
After 8 h from ED presentation, cardiac transcutaneous pacing was removed, SBP was 115 mm Hg and mental status came to be alert. ECG showed sinus bradycardia and first degree atrioventricular block with rate of 53/min, PR interval 238 ms and corrected QT interval 497 ms. After 10 h from presentation, the patient was weaned from mechanical ventilator and endotracheal tube was extubated.
With the consent of guardians, the patient was transferred to the nursing facility that she was hospitalized before ED presentation. One week later, the patient’s recovery from arrhythmias without any serious complication was confirmed by telephone.
Discussion
Musk, one of the ingredients in Kyushin®, an active substance in muscone which is an organic compound as oil from ventral preputial glandular secretion of male musk deer, has been well known to be anti-inflammatory and synergistic cardiotonic agent in East Asia. The potentiation of β-adrenergic effect via activation of Ca2+-dependent protein kinase with enhanced uptake of Ca2+ into cardiac muscle is presumed to be the mechanism of action of inotropic effect on myocardium. 7 But, there is no documented case or experimental report suggesting that musk and β-agonist, like dobutamine we used for bradycardia, causes arrhythmia or cardiac arrest in human or animals. Although the study for verification is required, it is very unlikely that musk actually played some role in tachy-brady arrhythmia of the patient.
The recommended daily dose for Kyushin® is two tablets per dose, three times a day. 1 Commonly accepted oral dose of toad venom for adult is 3–5 mg per day, the maximum dose cannot exceed 135 mg per day, and the oral LD50 of toad venom has been known to be 0.36 mg/kg. 8 The patient was estimated to have intoxicated up to 83 mg of toad venom in maximum, which did not exceed the maximum daily dose but for the LD50, it seemed to be enough dose to show toxic effects of toad venom.
The plasma level of digoxin was 0.76 ng/mL in therapeutic range (KIMS immunoassay, therapeutic range of the laboratory is between 0.8 and 2.0 ng/mL) but that of digitoxin was 66.9 ng/mL over the normal range (CEDIA, normal range between 10 and 30 ng/mL).
Although immunoassays for the level of digitoxin such as CEDIA and fluorescence polarization immunoassay (FRIA) are known to be highly cross-reactive with endogenous digoxin-like immunoreactive substances (DLIS), drugs such as spironolactone, herbal substances like Chan Su and oleander because of the use of polyclonal antibody in the test,3,9–12 KIMS, and competitive chemiluminescence immunoassay (CLIA) are reported to hardly cross-react with those due to the use of newly developed monoclonal antibody.12,13 The cross reactivity of the Cobas system for digoxin level is documented to be 7.7% with cardenolide, 1.2% with digitoxigenin, and 6.2% with digoxigenin. 14 Moreover, the intoxicated patient did not have any medical history of liver disease that could interfere the metabolism of digitoxin and bufotoxin. Based on the above facts, the toxicology test results strongly support that bufotoxin was responsible for arrhythmia and cardiac arrest of the patient.
Since RB Jeffrey et al. 3 first reported two cases of the patients survived from toad venom poisoning following treatment with digoxin-specific Fab fragments, this antibody has been suggested to be one of the effective remedies for bufotoxin poisoning. In acute toad venom poisoning, anti-digoxin antibody of 400 mg (10 vials) has been administered to humans with acute toad venom poisoning. It has been described that if there is no clinical response in 30 min to 1 h, 190–380 mg should be repeated.3,15
There was suspicion that bufotoxin may had caused clinical manifestation of the patient, but we could not use anti-digoxin antibody because there was not near local poison control center with the antidote. So, we had no choice but to do conventional therapy, based on the Advanced Cardiovascular Life Support (ACLS) guidelines for tachy-brady arrhythmia.
Despite the government-led, national-wide project for providing toxin-specific antidotes for intoxication patients since 2010, it is true that the supply of some antidotes, like anti-digoxin antibody, is often inadequate. Now, the second project from the year of 2016 is expected to overcome the lack of supply.
Conclusion
Poisoning of bufotoxin in Chan Su, one of the ingredients in Kyushin®, can cause cardiac arrhythmia, even arrest as acute digitalis intoxication, so digoxin-specific Fab fragment will be the treatment of choice for acute intoxication, but in the circumstance without the antidote, conventional therapy could be successful as well.
Footnotes
Authorship
Dr Cha and Dr So had first managed the patient and ensured all the medical records to be sorted in chronological order without omission.
Dr Cha rearranged the patient’s treatment and test result records according to the causal relationship and timeline, and examined and reviewed the relationship between the toxicological test results and drug components.
Dr Jeong pointed to the possibility that other herbal components might have been involved in causing arrhythmias in the patient and suggested that muscone might have played some roles after reviewing a number of documents.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The case report was evaluated and approved by the Catholic University Institutional Review Board (Number VC17ZESI0117).
Informed consent
All efforts were made to obtain informed consent from the patient or her legal representative but could not be received. We tried to contact the nursing facility, home and caregivers by telephone and mail, but could not reach anyone, so we anonymized all information of the patient as much as possible.
