Abstract
Background:
On initial treatment in the emergency room, trauma patients should be assessed using simple clinical indicators that can be measured quickly.
Objectives:
The purpose of this study is to investigate the relationship between the injury severity score and blood test parameters measured on emergency room arrival in trauma patients.
Methods:
Trauma patients transferred to Gunma University Hospital between May 2013 and April 2014 were evaluated in this prospective, observational study. Blood samples were collected immediately on their arrival at our emergency room and their hematocrit, platelet, international normalized ratio of prothrombin time, activated partial thromboplastin time, fibrin/fibrinogen degradation products, and D-dimer were measured. We evaluated the correlations between the injury severity score and those biomarkers, and examined whether the correlation varied according to the injury severity score value. We also evaluated the correlations between the biomarkers and the abbreviated injury scale values of six regions.
Results:
We analyzed 371 patients. Fibrin/fibrinogen degradation products and D-dimer showed the greatest coefficients of correlation with injury severity score (0.556 and 0.543, respectively). The area under the curve of the receiver operating characteristic was larger in patients with injury severity score ⩾ 9 than in those with injury severity score ⩾ 4; however, patients with injury severity score ⩾ 9 or ⩾16 showed no significant differences. The area under the curve of fibrin/fibrinogen degradation products was larger than that of D-dimer at all injury severity score values. The chest abbreviated injury scale had the strongest relationship with fibrin/fibrinogen degradation products.
Conclusion:
Fibrin/fibrinogen degradation products and D-dimer were positively correlated with injury severity score, and the relationships varied according to trauma severity. Chest trauma contributed most strongly to fibrin/fibrinogen degradation product elevation.
Introduction
Caring for injured patients remains a mainstay of emergency medical practice, and emergency physicians play a vital role in the stabilization and diagnostic phases of trauma care. 1 Trauma patients accounted for 23.1% of the patients who were transported to emergency departments by ambulance in Japan in 2015. 2 Death due to trauma is classified as “accidental death” in Japan. “Accidental death” was the sixth most common cause of death and accounted for a high percentage of the deaths in Japanese individuals of 5–19 years of age in 2015. 3 Trauma has a remarkable effect on both health and a country’s socioeconomics.
When a trauma patient is transferred to the emergency room, the immediate recognition of the severity of trauma is essential for determining the optimal treatment strategy. The injury severity score (ISS), 4 which has been validated in numerous studies, is one of the most frequently used anatomical scoring systems for trauma. The ISS is one of the most widely applied scoring systems for describing patient injuries in studies on trauma outcomes. 5 The ISS is used to quantify the risk of an outcome after trauma for both clinical and research purposes. 6 The ISS of trauma patients can be determined quickly, which makes it clinically useful. However, it is impossible to determine the ISS immediately after arrival at the emergency room, and the accurate evaluation of injuries based on X-ray examinations, sonography, or computed tomography is necessary. We previously demonstrated that the ISS could be easily predicted based on routine examinations in the emergency setting, and focused our attention on the blood coagulation parameters of trauma patients. We found that the blood levels of fibrin/fibrinogen degradation products (FDP) and D-dimer on arrival at the hospital were directly associated with the ISS. 7 We also reported a model for predicting the ISS based on these findings. 8 However, our previous studies were retrospective and included only a small number of patients. This study was prospective in nature and included a relatively large study population. We prospectively examined the association between the ISS and the results of ordinary blood tests. In addition, based on these results, we attempted to answer the following questions:
Does the association between the ISS and blood tests vary according to the level of ISS?
Does the association between the abbreviated injury scale (AIS) and the blood test parameters differ in each of the six anatomical regions (head and neck, face, chest, abdomen, extremities and pelvis, and dermis)?
Patients and methods
The protocol of this study was approved by the research ethics board of Gunma University Hospital; the need for informed consent was waived (No. 12-75). This prospective, observational study was conducted in a single institution between May 2013 and April 2014. Trauma patients, who were transferred to Gunma University Hospital by ambulance and whom trauma team (trauma team in our hospital basically respond to every trauma patient from slight to severe cases) initially treated during this time period, were analyzed. The exclusion criteria were as follows: age < 18 years, burns, conditions, and/or diseases resulting in coagulation abnormalities, advanced malignant tumors, pregnancy, severe hepatic dysfunction, anticoagulant use, renal failure patients on hemodialysis, and refusal to participate.
Blood samples were collected from patients immediately on their arrival at our emergency room. Their hematocrit (Ht) levels and platelet (Plt) counts were measured in our central laboratory by fluorescence flow cytometry using an XE-5000 (Sysmex Corporation, Kobe, Japan). The international normalized ratio of prothrombin time (PT-INR), activated partial thromboplastin time (APTT), FDP level, and D-dimer level were measured in our central laboratory by coagulating methods, synthesized substrate assays, and immunoturbidimetry using a CS-5100 hemostasis system (Sysmex Corporation).
The ISS was calculated from the medical records by two investigators. Those two investigators were blinded to the laboratory results. We evaluated the correlations between the ISS and Ht, Plt, PT-INR, APTT, Fib, FDP, and D-dimer. We then chose the biomarkers for which the correlation coefficient with ISS was >0.5 and examined whether the correlation varied according to the ISS value. We set three ISS thresholds: ISS = 4 (mild injury or not, Analysis A), 9 (moderate injury or not, Analysis B), and 16 (severe trauma9,10 or not, Analysis C). We also evaluated whether the correlations between the biomarkers and the AIS values of six regions (head and neck (Head), face, chest, abdomen, extremities and pelvis (Ext.), and dermis) were different and whether the presence of brain injury (Head AIS ⩾ 3) influenced the results.
Statistical analysis
Age is shown as mean (±standard deviation). The other parameters are shown as median (interquartile range (IQR)). Correlations between the ISS and the other parameters were analyzed by Spearman’s rank correlation. Receiver operating characteristic (ROC) curves were used to evaluate the efficacy in predicting trauma severity and to determine the cut-off Youden index value. The optimal cut-off point was the one that maximized the sum of the sensitivity and specificity when employing the Youden index approach. Correlations between the AIS and parameters that were found to be correlated with the ISS were analyzed by multiple linear regression analysis. Parameters that were correlated with the ISS were set as the dependent variables; each AIS score was set as an objective variable. Multicollinearity was evaluated by the variance inflation factor, and autocorrelation was evaluated using the Durbin–Watson ratio. IBM SPSS Statistics 22 (IBM Corporation, Endicott, NY, USA) was used to perform the statistical analyses. P values of <0.05 were considered to indicate statistical significance.
Results
Between May 2013 and April 2014, we had 486 trauma patients. In those 486 patients, 79 patients were excluded based on the exclusion criteria described in section “Patients and methods.” In addition, 36 patients in whom we could not obtain sufficient data regarding coagulofibrinolysis markers were also excluded. Finally, 115 patients were excluded.
We analyzed 371 patients (male, n = 220; female, n = 151; mean age, 58.1 ± 21.6 (18–99) years). The causes of trauma included traffic accident (n = 189), fall (n = 145), sports-related injury (n = 12), assault (n = 5), self-injury (n = 8), and others (n = 12). The median ISS was 4 (IQR: 8). A total of 12 patients had penetrating trauma; the other 359 had blunt trauma. There were eight self-injured patients (penetrating trauma, n = 6; blunt trauma, n = 2). A total of 360 patients survived, while 11 patients died. The distribution of the ISS values and the number of patients are shown in Figure 1. As shown in Figure 2, 167 patients (45.0%) had an ISS value of <4.
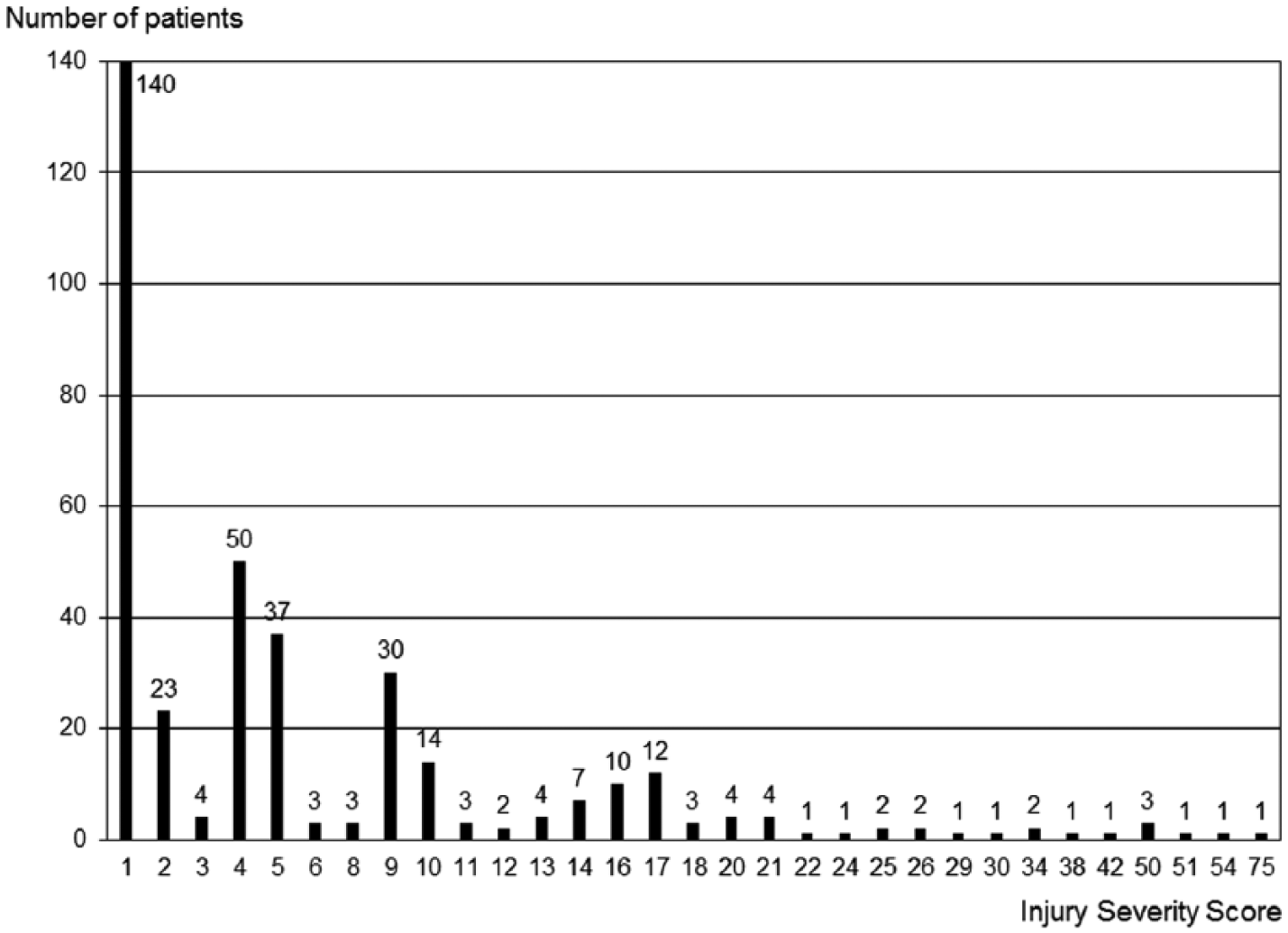
Distribution of the injury severity score (ISS).
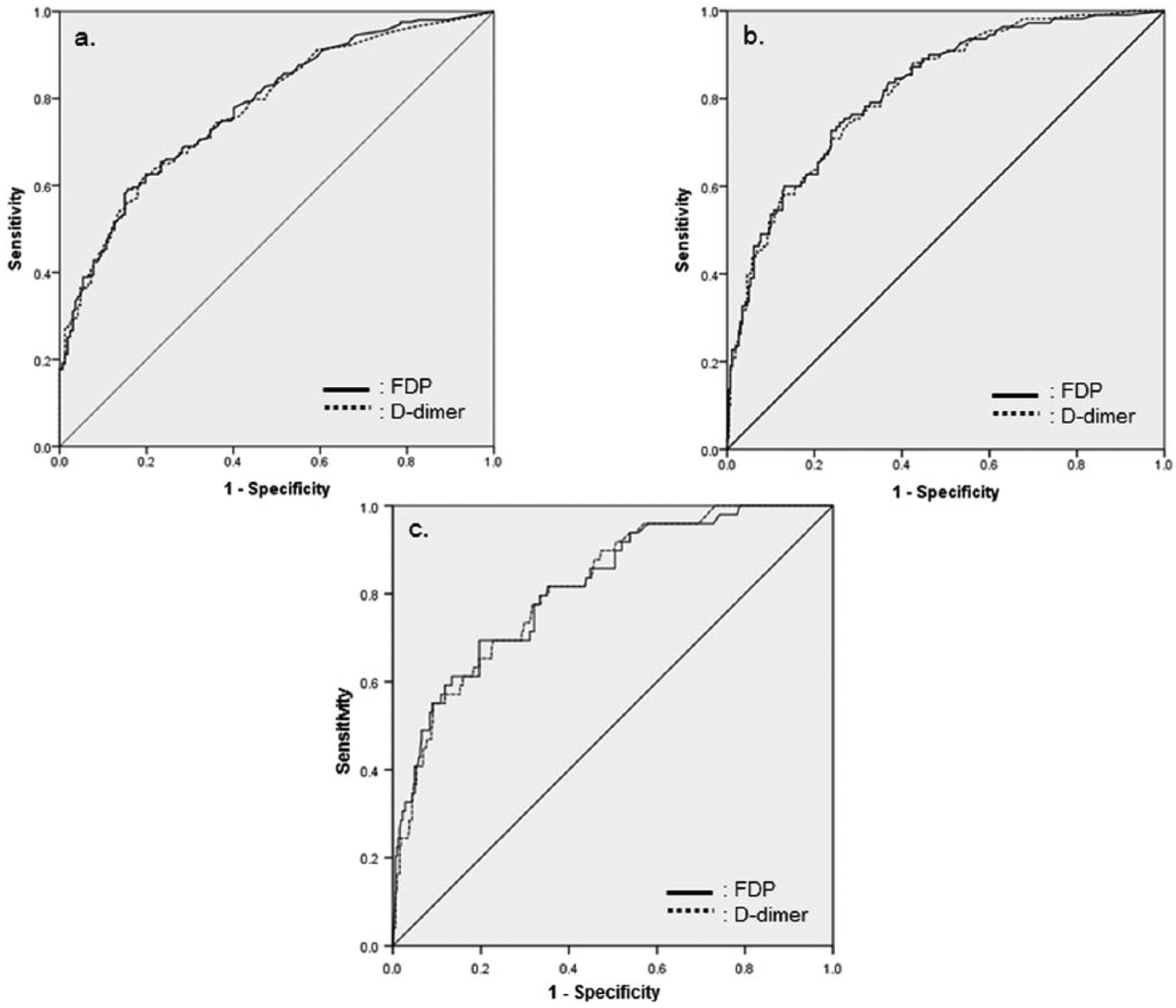
Receiver operating characteristic curves: (a) Analysis A (threshold ISS: 4), (b) Analysis B (threshold ISS: 9), and (c) Analysis C (threshold ISS: 16).
The median values for each parameter and the correlation coefficients for the ISS are shown in Table 1. From the table, it can be seen that Ht, Plt, PT-INR, APTT, and Fib were poorly correlated with the ISS. The correlation coefficients of FDP and D-dimer were >0.5 (FDP: 0.556; D-dimer: 0.543) and higher than those of the other parameters. We suspected that FDP and D-dimer would have moderate or greater correlations with the ISS. Based on these results, we examined whether the correlations between ISS and FDP or D-dimer varied according to the ISS.
Coefficient of correlation with ISS.
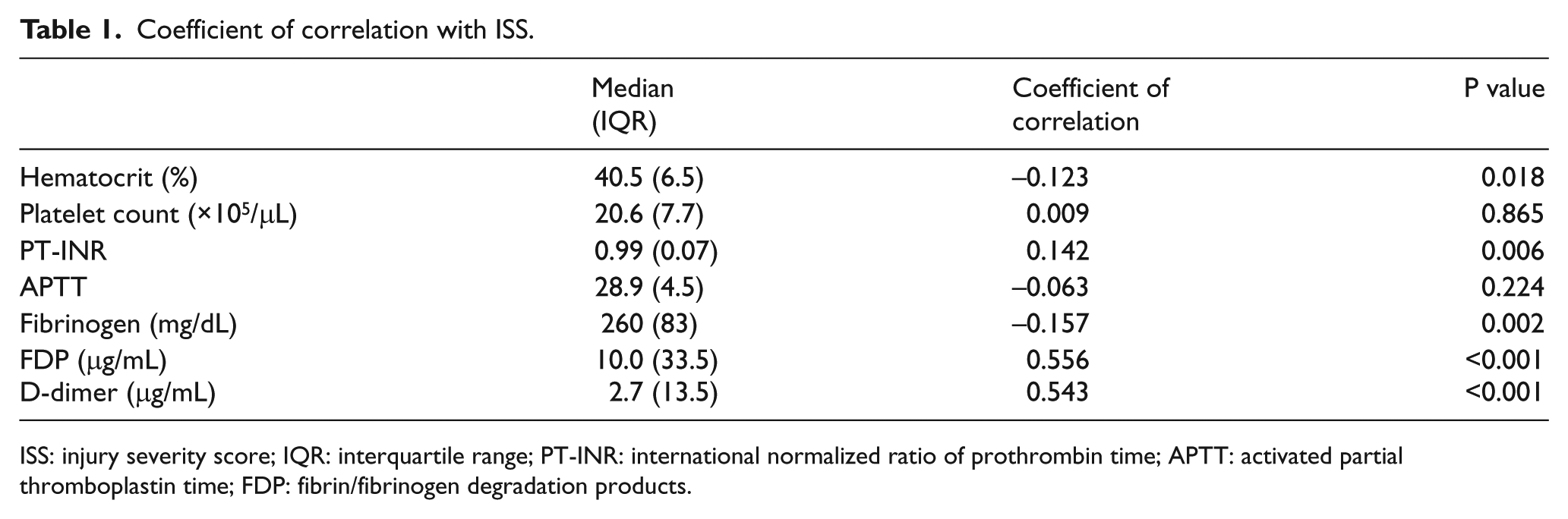
ISS: injury severity score; IQR: interquartile range; PT-INR: international normalized ratio of prothrombin time; APTT: activated partial thromboplastin time; FDP: fibrin/fibrinogen degradation products.
The ROC curves for detecting ISS using FDP and D-dimer are shown in Figure 2. Figure 2(a) shows the ROC curve for a threshold ISS of 4 (ISS ⩾ 4, n = 204; ISS < 4, n = 167; Analysis A), Figure 2(b) shows the ROC curve for a threshold ISS of 9 (ISS ⩾ 9, n = 111; ISS < 9, n = 260; Analysis B), and Figure 2(c) shows the ROC curve with a threshold ISS of 16 (ISS ⩾ 16, n = 50; ISS < 16, n = 321; Analysis C). The area under the curve (AUC) of the ROC curves, the cut-off FDP and D-dimer levels, sensitivity, and specificity of each ISS score group are shown in Table 2. The AUC was >0.7 in all cases and larger in Analysis B than in Analysis A. There were no remarkable differences between Analyses B and C. Analysis B was the only analysis in which the sensitivity and specificity were both >70%. The AUC of FDP was larger than that of D-dimer in all analyses.
Results of ROC curves showing the relation among FDP, D-dimer, and ISS.
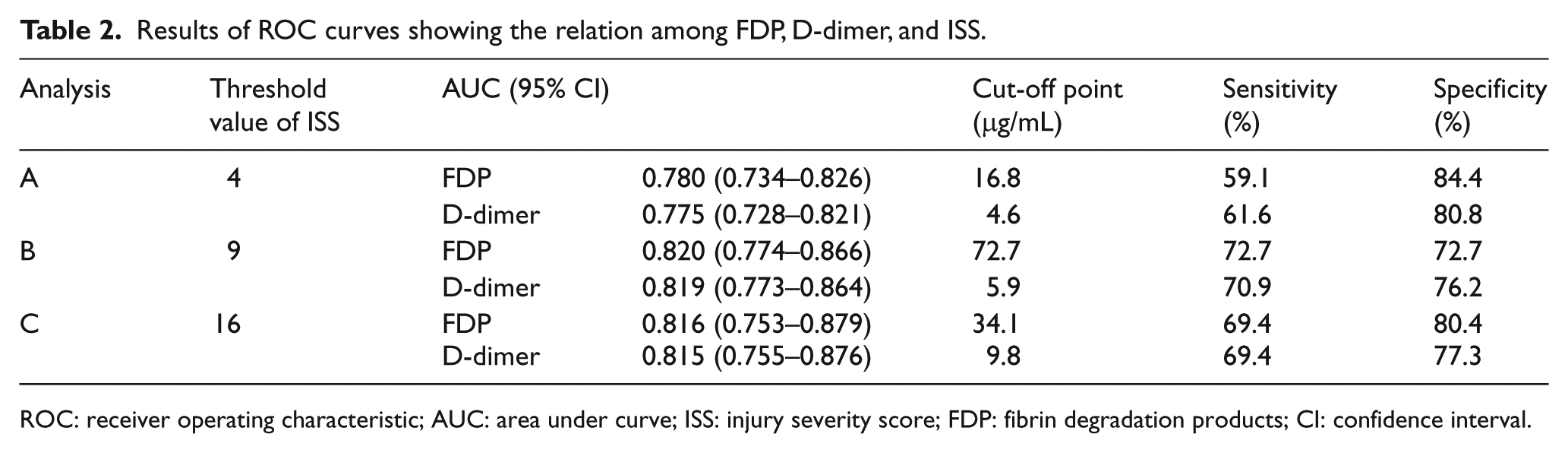
ROC: receiver operating characteristic; AUC: area under curve; ISS: injury severity score; FDP: fibrin degradation products; CI: confidence interval.
The distribution of the AIS values for each region and the numbers of patients were as follows: Head AIS ⩾ 3 and other AIS < 3 (mainly brain injury alone; n = 39; 10.5%), and Head AIS ⩾ 3 and other AIS ⩾ 3 (multiple injuries with brain injury; n = 9; 2.4%) (Table 3). We investigated whether the correlation between the AIS and FDP values differed in each of the six body regions. The multiple linear regression analysis revealed that the standardized partial regression coefficient (SPRC) was the highest for chest AIS; the SPRCs of Ext. AIS and Head AIS were the second and the third highest, respectively (Table 4). The results of a multiple linear regression analysis for patients with Head AIS ⩾ 3 (n = 48) revealed that the SPRC of the Head AIS was the highest (Table 5). In contrast, the results of the multiple linear regression analysis of patients with Head AIS < 3 (n = 323) revealed that the SPRCs of the chest AIS and Ext. AIS were the highest and second highest, respectively (Table 6). The Durbin–Watson ratio was ~2 in all multiple linear regression analyses; thus, autocorrelation was not suspected.
Distribution of AIS.
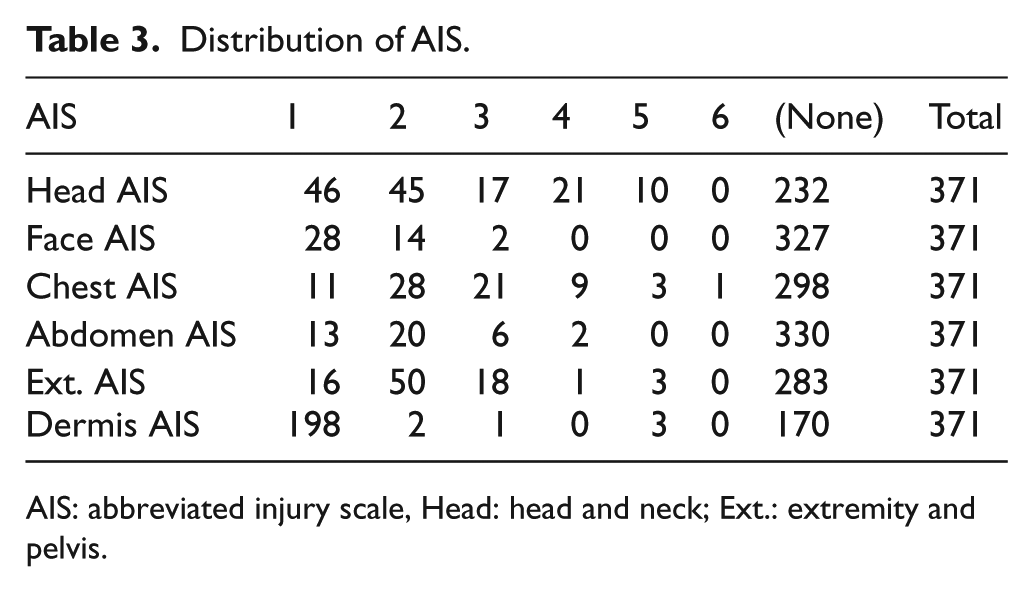
AIS: abbreviated injury scale, Head: head and neck; Ext.: extremity and pelvis.
Results of multiple liner regression analysis showing the correlation between the AIS and FDP values that differed in each of the six body regions.
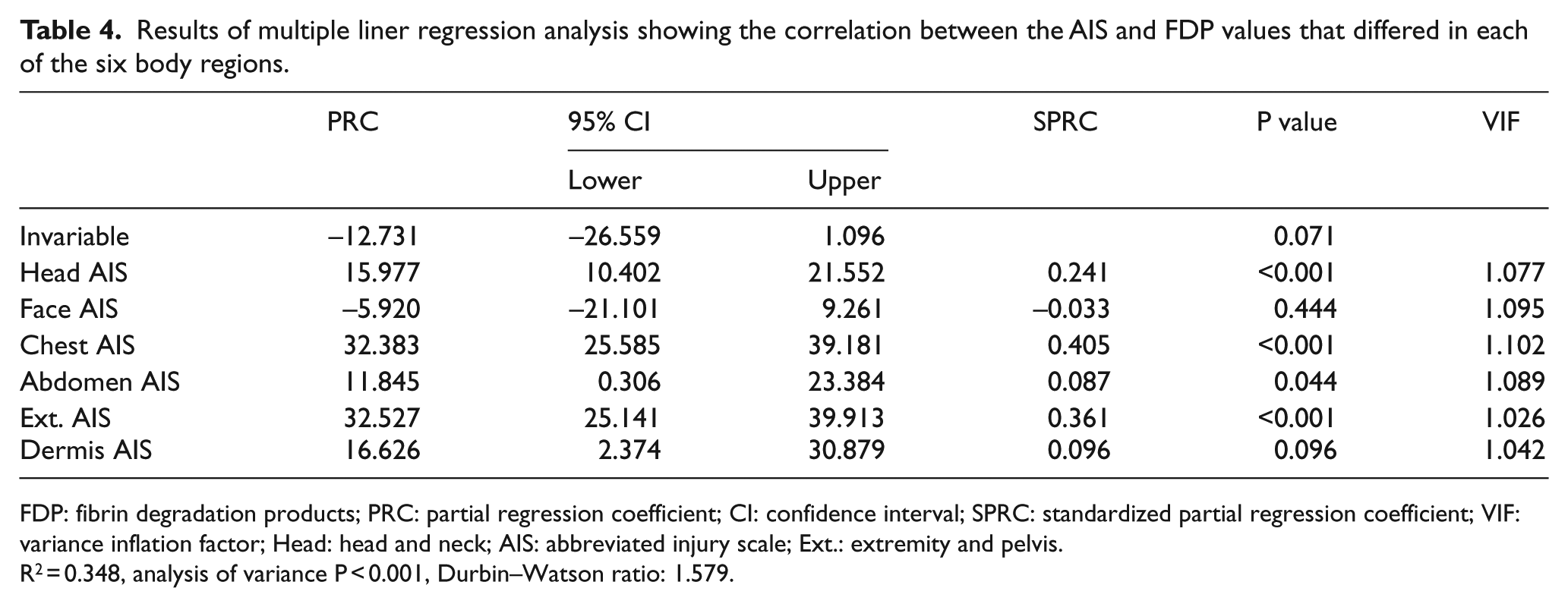
FDP: fibrin degradation products; PRC: partial regression coefficient; CI: confidence interval; SPRC: standardized partial regression coefficient; VIF: variance inflation factor; Head: head and neck; AIS: abbreviated injury scale; Ext.: extremity and pelvis.
R2 = 0.348, analysis of variance P < 0.001, Durbin–Watson ratio: 1.579.
Results of multiple linear regression analysis showing the correlation between the AIS and FDP values that differed in each of the six body regions (patients with Head AIS ≧ 3, n = 48).
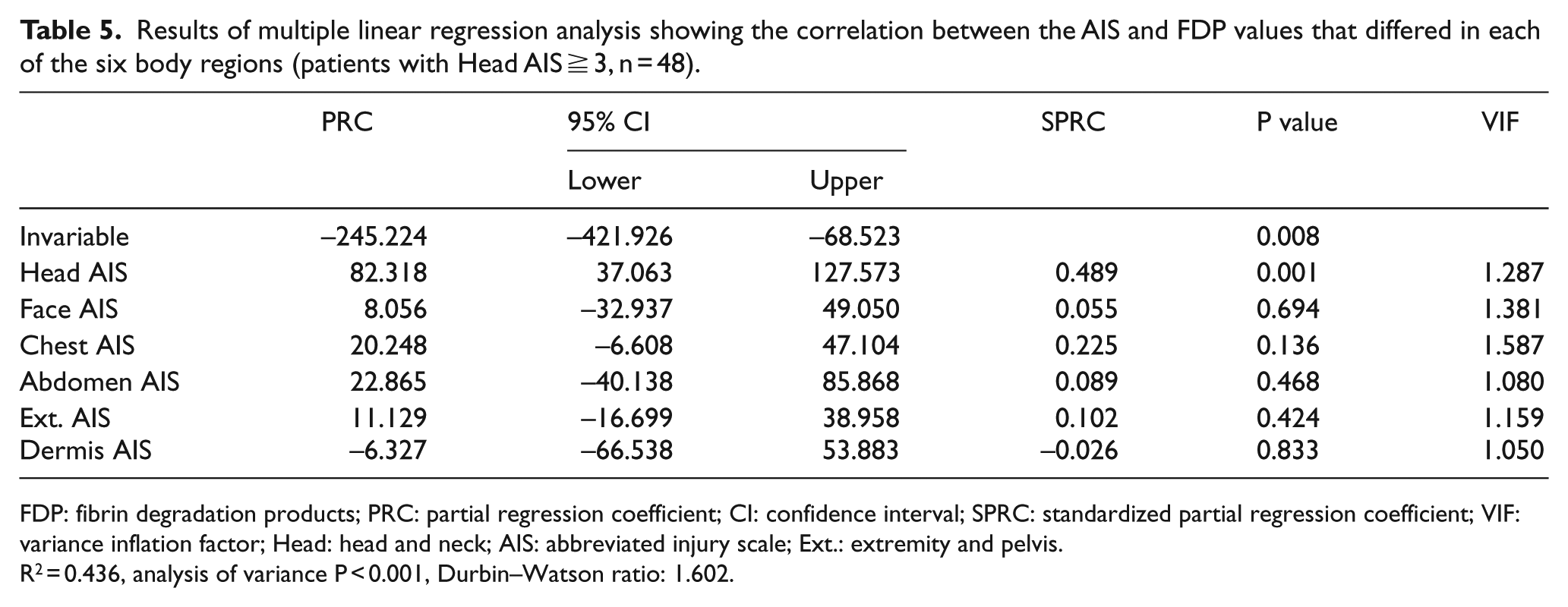
FDP: fibrin degradation products; PRC: partial regression coefficient; CI: confidence interval; SPRC: standardized partial regression coefficient; VIF: variance inflation factor; Head: head and neck; AIS: abbreviated injury scale; Ext.: extremity and pelvis.
R2 = 0.436, analysis of variance P < 0.001, Durbin–Watson ratio: 1.602.
Results of multiple linear regression analysis showing the correlation between the AIS and FDP values that differed in each of the six body regions (patients with Head AIS < 3, n = 323).
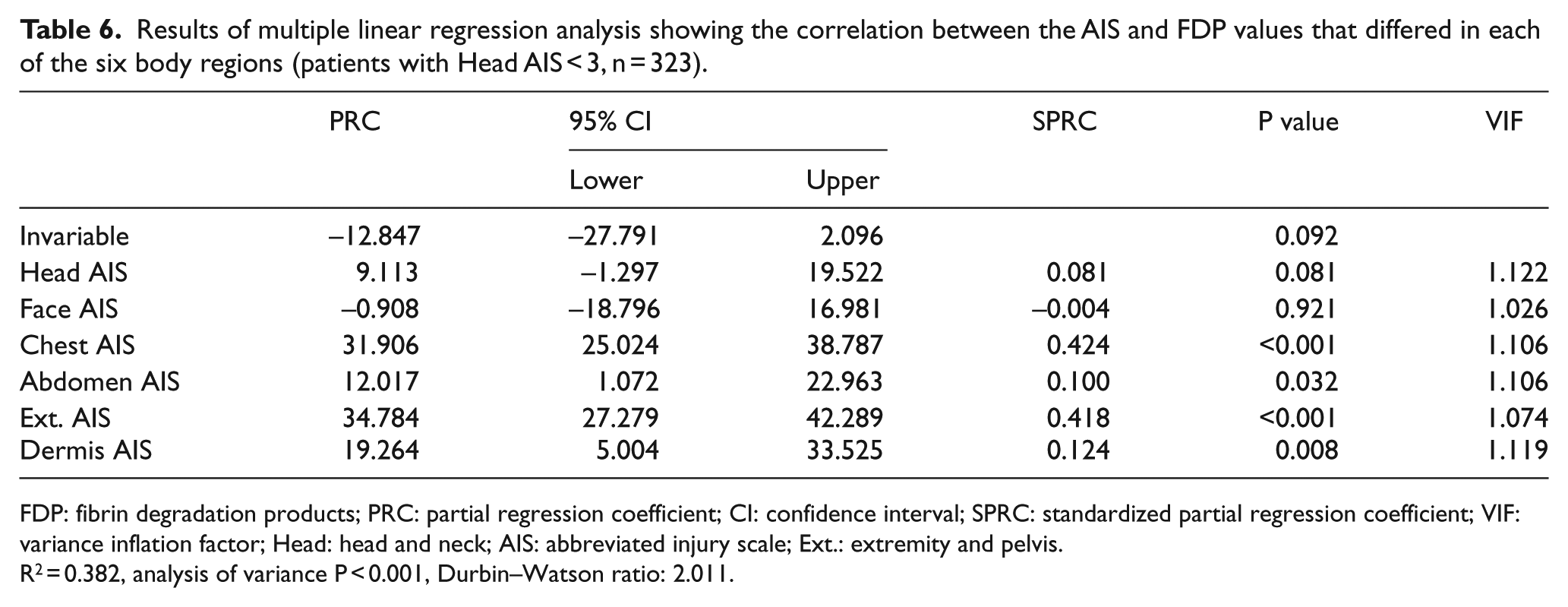
FDP: fibrin degradation products; PRC: partial regression coefficient; CI: confidence interval; SPRC: standardized partial regression coefficient; VIF: variance inflation factor; Head: head and neck; AIS: abbreviated injury scale; Ext.: extremity and pelvis.
R2 = 0.382, analysis of variance P < 0.001, Durbin–Watson ratio: 2.011.
Discussion
This study showed that the FDP and D-dimer levels were more strongly correlated with the ISS than other ordinary blood test values, and that the correlation in moderately or more severely injured patients tended to be stronger than that in mildly injured patients. In addition, chest AIS had a greater influence on the of FDP level than the AIS values of the five other regions in all patients. Head AIS had the strongest effect on the FDP level in patients with Head AIS ⩾ 3. In contrast, chest AIS had the greatest influence on the FDP level in patients with Head AIS < 3. Many of the patients in this study were slightly injured because patients with ISS < 4 accounted for 45.0% of all the trauma patients.
The clinical and imaging findings are important in the medical examination of trauma patients because the details of their injuries (e.g. site and severity) can be quickly understood. These findings are also necessary to obtain the AIS and ISS, which represent important information. The ISS is a well-known scoring system that is widely used to assist in clinical decision-making and to aid physicians in the initial evaluation of trauma. 11 The ISS is an anatomical score, and an independent predictor of mortality, which is mostly used for patients with multiple injuries. 12 However, it is impossible to determine the ISS immediately after a patient’s arrival at the emergency room.
The FDP and D-dimer levels had moderate or greater correlations with the ISS and were more strongly correlated than the other routinely obtained blood parameters in this study. This was in line with the findings of a previous retrospective report. 7 FDP is a product of the degradation of fibrin and fibrinogen, while D-dimer is a specific cross-linked fibrin derivative that is a product of the endogenous fibrinolytic degradation of fibrin. The FDP and D-dimer levels are easily measured and are commonly considered to be useful laboratory markers of coagulation activation; they have also been found to be correlated with the extent of thrombosis.13,14 It has already been pointed out that coagulation biomarkers are associated with the prognosis of trauma cases. In 1992, Kearney et al. 15 reported that coagulopathy may be correlated with poor survival in patients with head trauma. Recently, some investigators have reported the possibility that blood coagulation biomarkers may be associated with the prognosis of brain trauma patients.16–18 Other reports mention that a high FDP level was associated with uncontrollable bleeding19,20 and high mortality 19 in trauma patients. Furthermore, Hayakawa et al. 21 reported that D-dimer elevation was associated with the severity of tissue damage in trauma patients and an indicator of hyperfibrinolysis during the early phase of trauma, and suggested that a high D-dimer level on arrival at the emergency department was a strong predictor of early death or the need for massive transfusion in patients with severe trauma. The prognosis of trauma patients has been reported to be associated with coagulation biomarkers such as FDP and D-dimer; however, few reports have mentioned a correlation between the ISS and coagulation biomarker levels.
In this study, the correlation between ISS and the FDP and D-dimer levels varied according to the severity of trauma; the correlation tended to become greater as the trauma became more severe. However, the AUC values in Analyses B and C were almost the same. In addition, the AUC of FDP was greater than that of D-dimer at all ISS values. Raza et al. 22 suggested that fibrinolytic activation occurs in the majority of trauma patients and that the degree of fibrinolytic activation is associated with significantly worse outcomes in terms of mortality and morbidity. We think that FDP is more strongly associated with the ISS than the D-dimer level, and that the correlation between FDP and ISS might become stronger in patients with moderate and severe trauma.
There were differences in the associations between the AIS values of each of the six regions and FDP. Among patients with Head AIS ⩾ 3, the head region had the strongest association with FDP. In contrast, the chest region had the strongest association with FDP among patients with head AIS < 3. Finally, the chest region had the strongest association with FDP in all patients. Brain tissue contains a rich tissue plasminogen activator, and it is recognized that the FDP level becomes elevated as a result of fibrinolysis by tissue plasminogen activator, which is released when brain tissue is damaged. 23 FDP has been reported to be a useful biochemical marker for predicting the deterioration of hematoma, the severity of trauma, and mortality in patients with head injuries.24–26 In contrast, we could not find any published reports regarding the correlation between FDP and chest injury or between FDP and the AIS of the six body regions. To the best of our knowledge, this is the first report to mention the differences in the correlations between the AIS values of the six body regions and FDP. Yanagawa et al. 23 explained that the FDP level might be a useful biochemical parameter for the initial evaluation of the severity of trauma and mortality, even in blunt trauma patients without head injury. Actually, the chest and Ext. AIS values were strongly associated with FDP in patients with Head AIS < 3 in this study. Tissue plasminogen activator is secreted from vascular endothelial cells. 27 It is hypothesized that the associations between FDP and the chest and Ext. AIS values—where blood vessels are abundant—became stronger in patients with Head AIS < 3.
Limitations
This study is associated with some limitations. The study was performed at a single institution, and the study population was relatively small. In addition, many of the patients were considered to be only slightly injured because 45.0% of the trauma patients were classified as ISS < 4. This might therefore have influenced our results. Furthermore, the timing of blood sampling after arrival at the emergency room was not considered, and timing of blood examination from time of injury will carry significant effect on the correlation with the ISS score. Finally, trauma of the neck was included in Head AIS, and we only evaluated the clinical data and did not investigate the detailed mechanisms underlying our results. A multi-center study should thus be performed in the future to clarify these points.
Conclusion
The FDP and D-dimer levels were positively correlated with the ISS; these correlations varied depending on the severity of trauma. The FDP level was more strongly correlated with the AIS of the chest, head and neck, and extremities and pelvis, in comparison to the AIS of the other three regions.
Footnotes
Acknowledgements
All the authors have read the manuscript and approved this submission. All authors made substantial contribution to the concept or design of the work and acquisition, analysis, or interpretation of data; drafted the article or revised it critically for important intellectual content; and approved the version to be published. Each author has participated sufficiently in the work to take public responsibility for appropriate portions of the content.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
Our data in this manuscript were obtained from trauma patients transferred to Gunma University Hospital between May 2013 and April 2014. All data regarding this manuscript are available upon request.
Informed consent
The need for informed consent was waived.
Ethical approval
The protocol of this study was approved by the research ethics board of Gunma University Hospital.
