Abstract
Background:
Tramadol is a common analgesia used in Emergency Department for trauma patients. However, it causes multiple side effects, most notably nausea and vomiting.
Objectives:
The aim of this study is to determine whether routine administration of prophylactic metoclopramide in patients receiving intravenous tramadol for injuries of extremities is beneficial.
Method:
A randomised, double-blinded, placebo-controlled trial was carried out on 200 trauma patients requiring tramadol for acute pain in Emergency & Trauma Department of Sarawak General Hospital. Patients were randomised into two groups, group one received metoclopramide 10 mg intravenously while group two received placebo. Nausea severity (measured on a visual analogue scale) before and after tramadol administration, number of episodes of vomiting and patients’ demographic data were recorded.
Results:
A total of 191 patients were recruited, in which the metoclopramide group had 96 patients while the placebo group had 95 patients. In total, 5 out of 191 patients had significant worsening of nausea severity and all were from the placebo group (5.26%). The difference was statistically significant (p = 0.029, Fisher’s exact test). Only 1 out of 191 patients vomited and was from the placebo group. No adverse reaction was reported in both groups of patients.
Conclusion:
Routine administration of intravenous metoclopramide may be beneficial in musculoskeletal trauma patients receiving tramadol.
Introduction
Pain is one of the most common presenting complaints in emergency departments (EDs). 1 Acute pain relief is an important aspect in emergency care, alleviating not only patients’ discomfort but also interrupting the pain cycle which releases stress hormones. Trauma is one of the common causes of acute pain, and opioids are commonly used as primary analgesics in emergency setting. However, opioids come with adverse effects, most notably gastrointestinal effects of nausea, vomiting and constipation, central nervous effects such as sedation, and cardio-respiratory depression. Many strategies have been postulated to minimise opioid-induced adverse effects. 2
Opioid-induced nausea and vomiting are one of the common side effects. It has been presumed that routine administration of prophylactic antiemetic is beneficial in patients receiving opioids in order to prevent opioid-induced nausea and vomiting. 3 According to one Australian study, 23% of ED patients were given prophylactic metoclopramide after opioid administration. However, the efficacy of prophylactic metoclopramide was poorly established, causing further doubt in routine prophylactic metoclopramide. 4 Some literature reviews mentioned that prophylactic antiemetics generally are not necessary before administration of opioid. 2
This study aims to determine if there is benefit in routine administration of an antiemetic (metoclopramide) to patients receiving intravenous (IV) opioid (tramadol) for injuries of extremities.
Research methodology
Study design and setting
This study was a randomised, double-blinded, placebo-controlled trial conducted in the Emergency & Trauma Department (ETD) of Sarawak General Hospital. The conduct of the study was approved by the Medical Research and Ethics Committee (MREC) Malaysia and was registered with the National Medical Research Register (NMRR-16-1688-32638). The trial ran over approximately 6 months.
We calculated that 180 patients would need to be recruited to give this study 80% power at 5% significance level. This was based on assumption that 20% of patients receiving tramadol would have worsening nausea or vomited, as literature review showed that incidence of nausea and vomiting for patients treated with therapeutic doses of opioids were 8%–40% and 15%–40%, respectively. 5
Selection of participants
Patients presenting to ETD of Sarawak General Hospital with traumatic injuries of extremities requiring tramadol for pain relief were recruited. Patients were not recruited consecutively. They were recruited at unplanned intervals during the study period, subject to availability of the investigator and patient consent. Informed consent was obtained from all participating patients (Appendix 1). Patients were eligible for this study if they were aged 18 years or older and sustained from acute injuries of extremities during their presentation to ETD. Patients were excluded should they have any of the following:
Known allergy to metoclopramide;
Injuries to other organ systems (brain, chest, abdomen and pelvis);
Concurrently taking medication with antiemetic effect, including antihistamines, phenothiazines and dopamine antagonists;
A history of vomiting since time of injury;
Patients who have received tramadol or metoclopramide 8 h prior to arrival at ETD;
Below age of 18 years on day of presentation or patients who could not give consent to the study;
Any alteration in level of consciousness;
Hemodynamic instability or primary diagnosis requiring time critical intervention;
Pregnancy or lactation;
History or known case of vertiginous disorder;
Currently undergoing chemotherapy or radiotherapy.
If there were no exclusion criteria, written informed consent was obtained and baseline information, including details of the injuries sustained, was recorded. All patients recruited were given IV tramadol 50 mg. If pain relief was not achieved during the study period of 1 h, patients were given additional doses of tramadol to a maximum of total dose of 100 mg. Patients who required further doses of tramadol beyond 100 mg or other analgesic agents within the study period were withdrawn from the study. This is to avoid unnecessarily increased incidence of nausea and vomiting due to tramadol overdose as the recommended dose of IV tramadol by British National Formulary is 50–100 mg and also side effects from other types of analgesics.
Interventions
The study drugs were prepared under sterile conditions by a pharmacist independent to the study and kept in packs. Each study pack contained two 3-mL syringes, each containing identically appearing clear fluid. These were either (1) one 3-mL syringe containing 1 mL of tramadol 50 mg and one 3-mL syringe containing 2 mL of metoclopramide 10 mg or (2) one 3-mL syringe containing 1 mL of tramadol 50 mg and one 3-mL syringe containing 2 mL of sodium chloride 0.9%. The patients recruited were randomised to receive one of the study packs. A computer-generated random number sequence would be used to assign treatment allocations and numbering of the study packs. The allocation list was kept by the pharmacist, who could be contacted in the event of serious adverse event. The pharmacist monitored the pack numbers and prepared the study packs in batches of four. All study packs were stored in ETD drug refrigerator and kept for a maximum of 24 h.
After enrolment and recording of baseline information, the medication in the study pack was administered. Tramadol was administered over 2–3 min, as per British National Formulary. Treatment for patients’ presenting illness was performed at the same time. All patients recruited were then followed up until 60 min. Nausea severity ratings before and 60 min after administration of medication, number of episodes of vomiting and any other adverse events were obtained. The need for use of antiemetic rescue medication (IV ondansetron 4 mg) was determined on discussion between patient and the physician.
To maintain blinding, treatment allocations were revealed only after study completion, when all outcome measurements had been performed and recorded by the investigator in the study database. The treatment packs were unblinded prior to statistical analysis.
Methods of measurement
Nausea severity was rated on a visual analogue scale (VAS) on enrolment (before administration of study drugs) and 60 min after administration of the study drugs. VAS is a standard 100 mm line marked ‘no nausea’ at the left end and ‘worst nausea imaginable’ at the right end. Use of VAS for measurement of nausea severity and change has been validated, and the minimum clinically significant difference defined for this study is 20 mm. 6 Number of vomiting episodes during the 60-min study period was self-reported by the patient.
Outcome measures
The primary outcome was mean change in severity rating on the VAS 60 min after administration of the study drugs. Secondary outcomes were number of vomiting episodes, need for rescue medication and adverse events.
Data entry and analysis
The data were recorded into several variables as below:
Demographic variables: age, gender, marital status, race, level of education and co-morbid(s);
Data peculiar to the injury sustained: date and time of presentation to ETD, date and time of injury, type of injuries (fracture, laceration wound or soft tissue injury) and site of injury;
Randomisation information: date of randomisation, time of administration of medication in study pack, additional dose of tramadol if needed for further pain relief during the study period;
Outcome information: VAS of nausea severity in mm at 0 min (before administration of study pack) and 60 min after administration of study pack, number of episodes of vomiting during the 1-h study period, use of rescue medication (IV ondansetron 4 mg), other adverse reactions from medication given.
Data were then entered and analysed using Statistical Package for Social Science (SPSS) for Windows, version 23.0. Baseline data are presented as mean, number and percentage. For the primary outcome, change in rating is reported as mean. Comparison of mean change between groups uses independent t-test. Significant increment of nausea severity on VAS (20 mm or more) is compared between groups with either Chi-square test or Fisher’s exact test, whichever appropriate.
Results
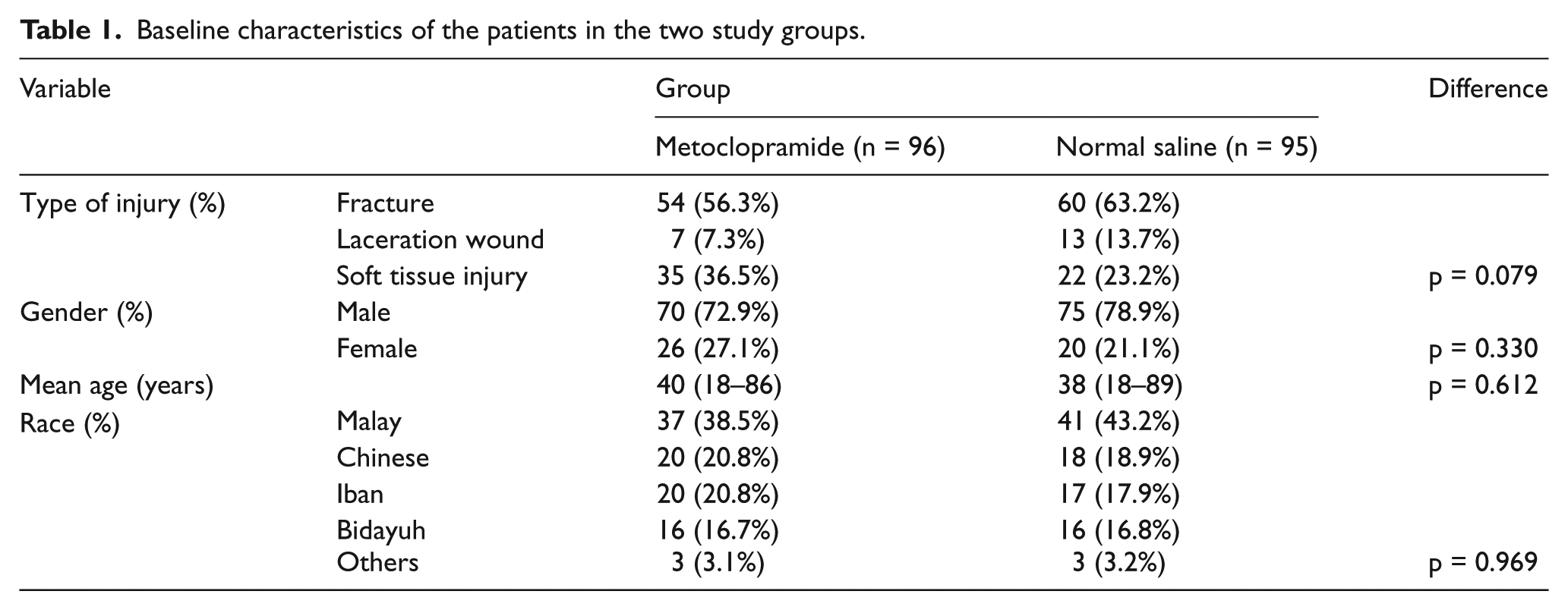
A total of 200 patients were recruited over the study period of 6 months, of which 191 of them were randomised (Figure 1). Analysis at the end of the study showed that 96 patients received metoclopramide and 95 patients received placebo (normal saline). Characteristics of the patients were demonstrated in Table 1. The two groups were evenly matched for type of injury, gender, age and race (Table 1).
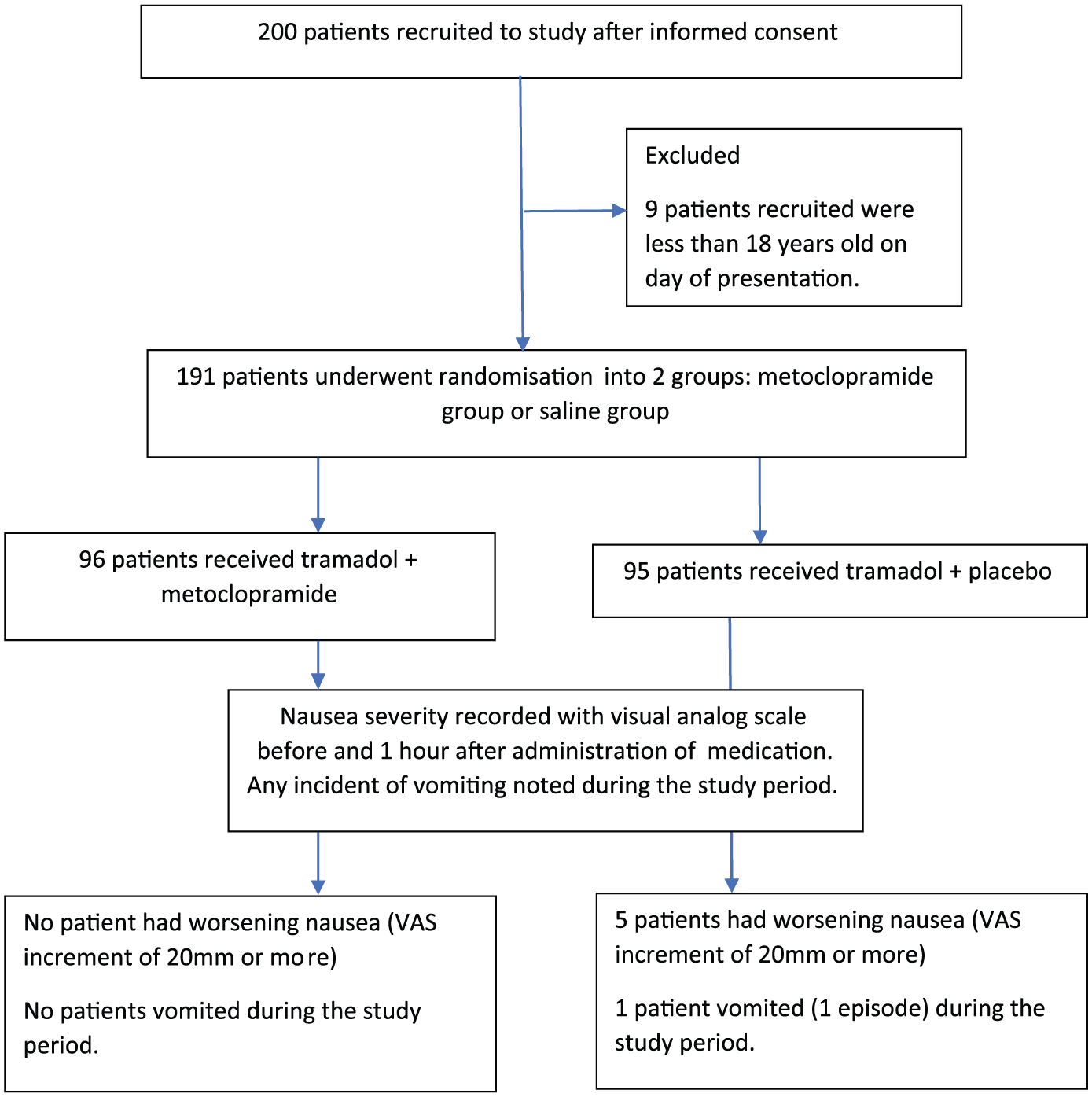
Study flowchart.
Baseline characteristics of the patients in the two study groups.
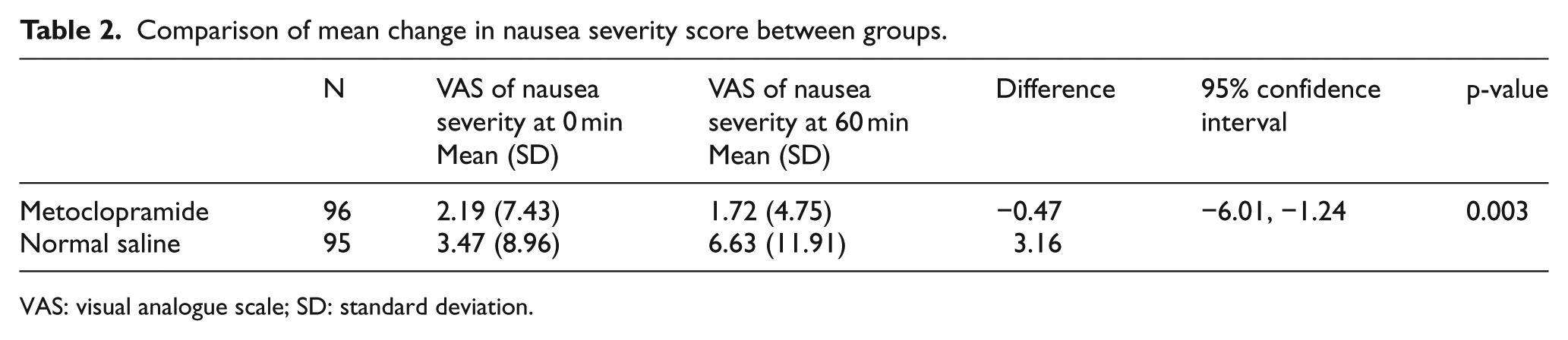
Severity of nausea was measured using VAS before administrating the treatment pack, which contained tramadol 50 mg and either metoclopramide 10 mg or placebo, and 1 h after administration of medication. Additional dose of tramadol could be given during the study period to a maximum total dose of 100 mg. There was a mild drop in the mean VAS score in patients who were given metoclopramide, while in patients who were given placebo there was an increase in the mean VAS score of nausea severity (Table 2), and the difference was significant (p = 0.003).
Comparison of mean change in nausea severity score between groups.
VAS: visual analogue scale; SD: standard deviation.
During the 1-h study period, 5 patients out of 191 (2.62%) had significant worsening of nausea, as measured by increment of more than 20 mm on VAS. All 5 patients were in the group receiving placebo (5/95, 5.26%), making the difference significant (p = 0.029, Fisher’s exact test), as shown in Table 3. Only 1 patient out of 191 (0.52%) vomited, and was in the group receiving placebo. One patient in the metoclopramide group received additional 50 mg of tramadol, and did not have significant worsening of nausea and vomiting. None of the patients in the placebo group received additional dose of tramadol.
Increment of nausea severity visual analogue scale of more than 20 in the study groups.

VAS: visual analogue scale.
None of the patient required rescue medication (IV ondansetron 4 mg) for the relief of severe nausea and vomiting. There were no other adverse effects noted in any of the study patients. Measurement bias was minimised because of patients’ self-reporting of outcomes, and the use of VAS as a measure in this setting has been validated.
Discussion
Pain relief is one of the priorities of emergency medicine. In our centre, tramadol has been the IV opioid of choice for analgesic in trauma patients. All opioids have side effects, including tramadol. 7 The most common side effects caused by opioids are nausea and vomiting. Prophylactic antiemetics have been used together with opioids to counter their emetic side effects. In the United Kingdom, prophylactic antiemetic is routinely administered to all patients receiving opioids. 1 Reports showed that incidence of nausea and vomiting for patients treated with therapeutic doses of opioids were 8%–40% and 15%–40%, respectively.5,8 These studies included medical and surgical patients as well as patients who were on regular doses of tramadol. When used in post-operative pain management, it was found that tramadol has relatively high incidence of nausea and vomiting, with up to 20% of patients complained of nausea. 9 Opioids induce nausea and vomiting through multiple mechanisms. These may include direct stimulation of chemoreceptor trigger zone (CTZ), decreased gastrointestinal motility and increased vestibular sensitivity. 10 Multiple antiemetics have been used to treat opioid-induced emesis, including antipsychotics, dopamine receptor antagonists, serotonin antagonists, antihistamines and corticosteroids. 11 According to a review by Canadian Agency for Drugs and Technologies in Health in 2014, there is no proven clinical benefit of one antiemetic over another in treating opioid-induced nausea and vomiting. 12
D2 (dopamine receptor subtype 2), 5-HT3 (serotonin type 3) and NK1 (neurokinin 1) neuroreceptors can be found in the CTZ. Dopamine receptor antagonists work in this neural pathway to treat opioid-induced nausea and vomiting. 13 Metoclopramide is a dopamine (D2) receptor antagonist with short half-life and mixed 5-HT3 receptor antagonist and 5-HT4 (serotonin type 4) receptor agonist. In addition to its antidopaminergic action, metoclopramide has a prokinetic activity on the upper digestive tract. Its combined action on CTZ and intestinal motility contribute to its powerful antiemetic effect. However, like most dopamine antagonists, metoclopramide has no selectivity for the dopamine receptors in the CTZ; therefore, it may cause hyperprolactinaemia and extrapyramidal motor disturbance, by acting on dopaminergic systems in other parts of the brain. 14 Therefore, use of metoclopramide is not without risk. Up to 20% of patients who are given therapeutic dose of metoclopramide experience side effects, most notably drowsiness and lassitude 15 and approximately 1% of patients experience true dystonic reaction. 16 Its usage has therefore been restricted by European Medicines Agency. 17
There is currently no evidence in the literature to show that nausea and vomiting caused by tramadol administration can be significantly reduced or prevented by prophylactic antiemetic. We have come across similar studies in the literature, but those studies involved morphine instead of tramadol.
In this study, we intended to limit recruitment to patients who sustained traumatic injury of the extremities in order to minimise other factors which also result in nausea and vomiting. This is a prospective, randomised, double-blinded, placebo-controlled study. The randomisation process involved the supply of prefilled, numbered syringes containing either metoclopramide or normal saline. These syringes were prepared by an independent pharmacist. After administration of the medications, the patient was observed for 1 h and nausea severity and number of episode of vomiting were recorded.
Our study showed that the incidence of nausea and vomiting is low (2.62% and 0.52%, respectively) when tramadol is used in treating acute pain in patients with traumatic limb injuries. However, all of those who experienced nausea and vomiting were from the placebo group. There was statistically significant difference between the two groups in severity of nausea, as measured by increment of 20 mm on VAS. However, none of the patients required rescue medication, ondansetron, which is a serotonin 5-HT3 receptor antagonist.
This study was designed to determine if there was benefit in the routine administration of an antiemetic to patients receiving IV tramadol in the ED. The group selected was confined to trauma of the extremities. The study suggested that prophylactic antiemetic may be beneficial. Although the incidence of nausea and vomiting was low in patients receiving tramadol, statistically there was significant difference in the nausea severity ratings.
Target dose is a key determinant in tramadol tolerability. The rate of adverse reactions may differ depending on different dosages of tramadol. Gana et al. conducted a study to determine the relationship between tramadol dose and discontinuation rate due to adverse reactions. It demonstrated that the discontinuation rate was significantly higher in subjects receiving higher dose of tramadol. 18 Another study also showed higher dose of tramadol (2 mg/kg) was associated with higher rate of adverse effects, despite having better analgesic effect. The rate of side effects was as high as 66.1% when higher dose of tramadol was administered. 19 Further studies may be carried out to determine the role of prophylactic antiemetic with different dosages of tramadol.
Besides, in this study, sample size recruited was based on the assumption of 20% of patients having worsening nausea after IV tramadol administration. This calculation was based on previous literature review. In this study, the incidence of nausea was much lower. Therefore, future study with higher number of sample size based on lower incidence of tramadol-induced nausea and vomiting is needed to confirm the findings.
In addition, the results of this study are only applicable to IV tramadol and cannot be applied to other routes of delivery (e.g. intramuscular administration) and other types of opioid as different opioids have varying emetic potential.
Conclusion
Prophylactic antiemetic metoclopramide may be beneficial in reducing severity of nausea due to IV tramadol administration. However, the incidence of tramadol-induced vomiting is low, only one patient vomited in this trial. Therefore, evaluation of prophylactic metoclopramide in larger trials of patients seems warranted. Further study may include patients who present with other illnesses besides trauma and also include different routes of tramadol administration.
Footnotes
Appendix 1
Acknowledgements
The authors want to acknowledge and thank all the supervisors, Associate Prof Dr R.A.L.M., Dr K.P.Y.Y. and Dr V.A.P.K. for their time and effort spent on guiding me to complete this research. The authors express foremost gratitude to Dr Mohamad Adam Bujang, statistician of Clinical Research Centre of Sarawak General Hospital, and Miss Mary Chen Siew Ying, pharmacist of Emergency & Trauma Department of Sarawak General Hospital, for helping.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Conflict of interest was declared under Medical Research and Ethics Committee (MREC) Malaysia on 25 September 2016.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Availability of data and materials
All the data and patient information will be kept private. All study data and personal information will be kept for 5 years at Emergency & Trauma Department Record Office of Sarawak General Hospital after the study is completed and will be destroyed after the period of storage.
Ethical approval
Ethical approval was obtained from the Medical Research and Ethics Committee (MREC) Malaysia and was registered with the National Medical Research Register (NMRR-16-1688-32638).
Human rights
Patients can always withdraw from the study at any time during the study period. All the emergency care will be given even if they are not part of the study anymore. If patients are injured as a result of being in this study, they can contact the study doctor. In the event of a bodily injury or illness directly resulting from the study product, reasonable and necessary treatment will be paid for.
