Abstract
Introduction:
Chlorfenapyr is a pyrrole-based pro-insecticide. The main activity of this agent is the inhibition of adenosine triphosphate synthesis, and it is used in agriculture to control several insects and mites resistant to carbamate, organophosphate and pyrethroid insecticide. The World Health Organization classifies this agent as a class II toxin (moderately hazardous).
Case presentation:
All previously reported cases of chlorfenapyr poisoning in humans occurred following oral ingestion except one case involving exposure to chlorfenapyr vapour.
Discussion:
In this report, we describe a case of chlorfenapyr intoxication after skin exposure in a 49-year-old patient who died 5 days after exposure.
Keywords
Introduction
Chlorfenapyr (trade names: Pirate, Stalker and Alert) is a pyrrole-based pro-insecticide used for the control of various insects and mites on cotton, ornamentals and a number of vegetable crops. 1 It has been accepted for use in several countries including Brazil, Australia, Mexico, the United States, Japan and South Korea. 2
Acute toxicity after ingestion of technical-grade chlorfenapyr is categorized as Tox category I in mice and II in rats. 3 In humans, intoxication with this pesticide is fatal, with distinctive clinical and neuroradiological features.4 –11 In previous reports, the exposure routes were mostly oral, with only one case 11 reporting vapour form of chlorfenapyr as the cause. To the best of our knowledge, chlorfenapyr intoxication after dermal exposure in humans has not yet been reported in the literature. We present the first fatal case of chlorfenapyr intoxication after skin exposure.
Case report
A 49-year-old male was referred to our emergency department (ED) with binocular visual disturbances, diaphoresis and thirst. His wife reported that he had slipped and fallen on a basket containing a 10% chlorfenapyr solution while preparing pesticide spray, 5 days before admission. His arm, anterior chest and abdomen were exposed to the pesticide for several hours because he continued to work without removing the wet working clothes. On the following day, he complained of visual disturbance and voiding difficulty and intermittently said strange things without notable skin lesions. Over the next 3 days, he remained without treatment. On the fourth day, as his symptoms worsened, he presented to a nearby hospital, where he underwent a brain magnetic resonance imaging (MRI) scan. The brain MRI showed diffuse bilateral high signal intensities in the internal capsule, brain stem and cerebellar peduncle on diffusion-weighted images and low signal intensities in the same area on an apparent diffusion coefficient map (Figure 1). He was transferred to our ED for further management. On admission, he complained of thirst, blurred vision and difficult urination. His past medical history was unremarkable except for untreated hypertension. On initial physical examination, he was alert and neurologically intact. His vital signs were as follows: blood pressure 140/90 mmHg, pulse rate 92/min, respiratory rate 17/min and body temperature 36.5°C. His oxygen saturation was 98% on room air. Sweating on the face and body was noted. Laboratory testing revealed the following: serum lactate = 1.0 mmol/L, aspartate aminotransferase (AST) = 170 IU/L, alanine aminotransferase (ALT) = 56 IU/L, lipase = 169.9 U/L, CK-MB = 29.64 µg/L, creatine kinase = 4484 U/L and myoglobin = 857.5 ng/mL. Blood gas analysis while undergoing oxygen therapy at flow rate of 2 L/min showed pH = 7.446, pO2 = 108.4 mmHg, pCO2 = 29.1 mmHg and HCO3 = 20.2 mmol/L. A chest radiograph showed no abnormalities. An ophthalmology consult for his blurred vision was made and the results were unremarkable.
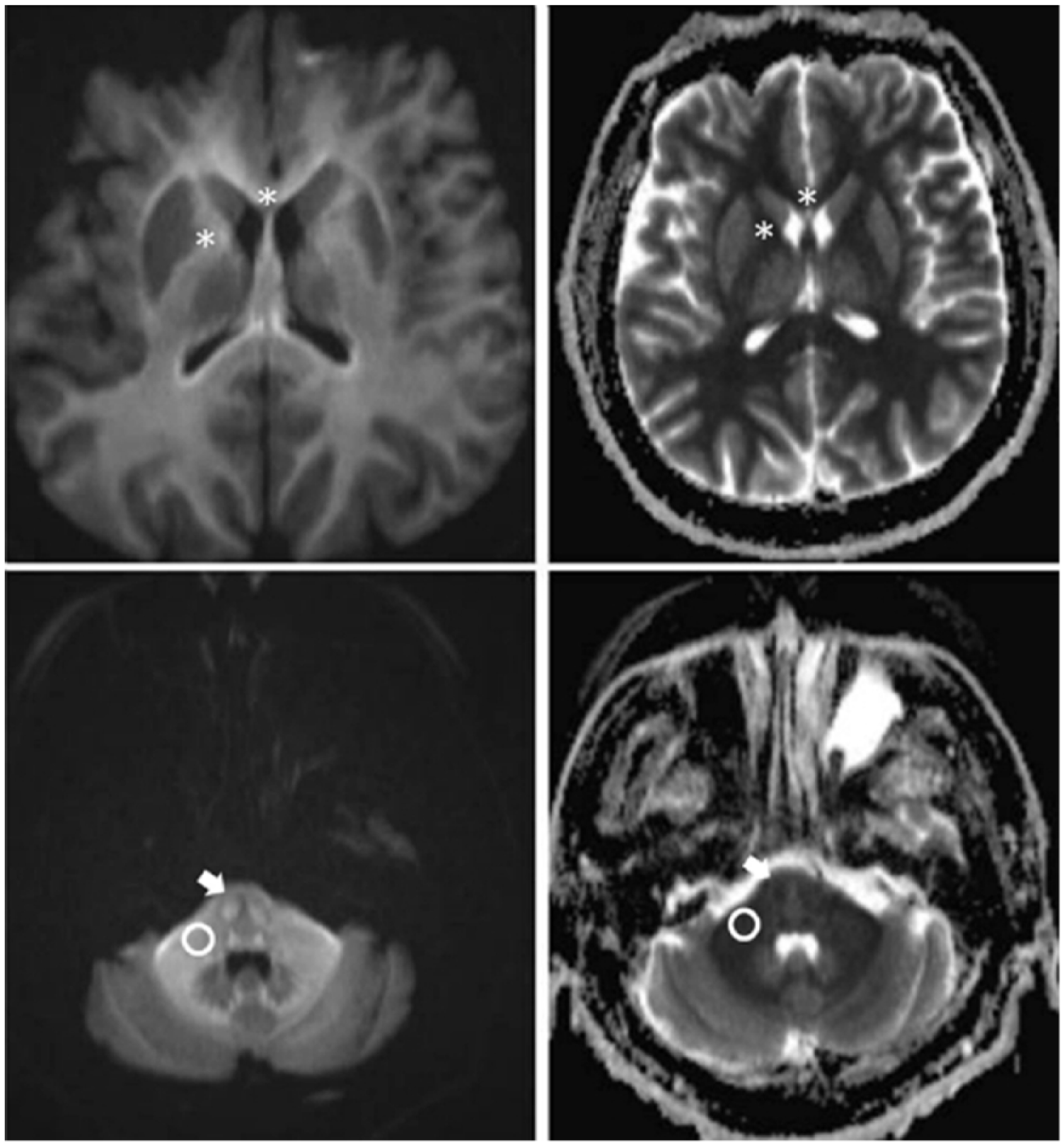
Diffusion-weighted MRI (Top left and bottom left) shows diffuse bilateral high signal intensities in the internal capsule and genu of corpus callosum (asterisk), pons (white arrow) and cerebellar peduncle (circle). Apparent diffusion coefficient map (Top right and bottom right) shows low signal intensities in the same areas.
He was admitted to the intensive care unit for close monitoring. After 2 h, he became confused, and his respiratory rate and body temperature increased to 23 cycles/min and 38.5°C, respectively. He was intubated for airway protection. Continuous infusion of remifentanil, intravenous administration of paracetamol and tepid sponging were performed; 40 min after intubation, tachypnoea was still observed and the body temperature increased to 41.5°C. Arterial blood pressure monitoring showed systolic blood pressure of 250 mmHg. Soon after, he developed cardiac arrest and cardiopulmonary resuscitation was initiated. However, the cardiac arrest persisted, and he died 11 h after admission.
Discussion
To date, eight case reports of chlorfenapyr intoxication in humans have been reported. Excluding one case involving chlorfenapyr vapour, seven cases of chlorfenapyr poisoning occurred following oral ingestion. In these previous reports, high fever, severe diaphoresis, tachypnoea, thirst, blurred vision rhabdomyolysis and progressively worsening neurologic symptoms were observed. The clinical manifestations in our patient were similar to those previously reported. These characteristic clinical features can be explained by the toxic mechanisms of chlorfenapyr. The N-ethoxymethyl group from chlorfenapyr is removed by mixed functions of oxidases to form the toxic metabolite CL 303268. This compound uncouples oxidative phosphorylation in the mitochondria, resulting in a disruption of adenosine triphosphate production, cellular death and ultimately death of the organism. 3 The effects of the uncoupling of oxidative phosphorylation are expressed as clinical symptoms, more apparent in cells with high energy demands such as cardiac muscle, skeletal muscle, retina and the central nervous system (CNS). 12
Involvement of the entire white matter tract was a characteristic radiologic finding in this case, which was similar to that observed in previous reports.6,7,10 United States Environmental Protection Agency 13 reported that rats with chlorfenapyr intoxication at 52 weeks revealed vacuolar myelinopathy and myelin sheath swelling on neurohistopathological examination. Similar pathological changes in myelin and white matter necrosis were observed during the autopsy of patients with toxic leukoencephalopathy. 14 Given these findings, chlorfenapyr may injure the white matter of the CNS, which manifests as neurological symptoms such as paraplegia, seizure and changes in mental state.
In previous reports, chlorfenapyr intoxication was fatal. Endo et al. 4 reported that 8 out of 24 patients with chlorfenapyr poisoning died. According to recent reports, 5 patients4 –8,11 expired, 1 patient 10 was discharged with paraplegia and 1 patient 9 was discharged without any sequelae. One fatal case occurred due to exposure to a small amount (estimated 20 mL) of chlorfenapyr. 8 In our patient, a large area of skin was accidentally exposed to the substance and the patient worked while wearing the contaminated clothes for several hours, which might have resulted in enhanced absorption of chlorfenapyr and fatal outcome. We presume that the patient shrugged off his exposure to chlorfenapyr because he had used this pesticide every year without any clinical problems and the substance was not irritable to his skin. According to the animal studies, chlorfenapyr does not irritate the skin or eye of rabbits and is of low toxicity after dermal exposure in rabbits (LD50 > 2000 mg/kg body weight). 3 Given its low toxicity with dermal exposure, removal of contaminated clothes and rinsing exposed skin thoroughly with water immediately after exposure could minimize the absorption of this agent and prevent death.
Of note, our patient and two patients5,11 from previous case reports developed toxic symptoms on the day following the exposure. But other patients6 –8,10 were asymptomatic for 7 to 14 days before the development of symptoms of neurotoxicity, and this asymptomatic period has been described as the latent period. Although it remains unclear why the clinical courses vary between patients, there is a need for prolonged in-hospital observation of patients with chlorfenapyr intoxication.
Currently, in comparison with the fatality of chlorfenapyr intoxication, specific treatments in individuals exposed to this agent have not yet been established. All patients, in previous reports, were treated conservatively and one patient underwent haemodialysis for the treatment of rhabdomyolysis. Renal replacement therapy (RRT) might be attempted because the molecular weight of chlorfenapyr is small (407.6 Da) and can readily cross the membrane. However, lack of data about other physiochemical properties of chlorfenapyr in humans such as protein binding, volume of distribution and lipid solubility prevent justification for the use of RRT. Therefore, removing contaminated clothing and washing the exposed area are most important life-saving procedures following dermal exposure to chlorfenapyr.
In conclusion, emergency physicians must keep in mind that all patients poisoned with chlorfenapyr, even in case of dermal exposure, have a potential for an inexorable course and mortality. The latent period should also be considered in the management of patients poisoned with chlorfenapyr. This is the first reported case, to the best of our knowledge, of fatal chlorfenapyr poisoning following dermal exposure.
Footnotes
Acknowledgements
The authors thank the anonymous reviewers for many helpful comments and suggestions on the manuscript. In addition, they thank Mi-Sun Sin for assistance with art work.
Authorship
Each author certifies that they (1) made a substantial contribution to the concept or design of the work or acquisition, analysis or interpretation of data; (2) drafted the article or revised it critically for important intellectual content and (3) approved finally the version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Availability of data and materials
The authors declare that the data supporting the findings of this report are included within the article.
Informed consent
The institutional review board approval was obtained and informed consent was waived for this retrospective case report (approval no. 1805-003-066).
Ethical approval
We obtained ethical approval from the institutional review board (approval no. 1805-003-066).
Human rights
This case report was performed in accordance with Ethical Principles for Medical Research Involving Human Subjects, as outlined in the Helsinki Declaration of 1975, as revised in 2000. In this article, patient’s name, initial, hospital number, date of birth or other protected healthcare information were not disclosed.
